In the previous article in this series, The Earth’s Energy Budget – Part Two we looked at outgoing longwave radiation (OLR) and energy imbalance. At the end of the article I promised that we would look at problems of measuring things and albedo but much time has passed, promises have been forgotten and the fascinating subject of how the earth really radiates energy needs to be looked at.
If you are new to the idea of incoming (absorbed) solar radiation being balanced by OLR, or wonder how the solar “constant” of 1367W/m2 can be balanced by the earth’s OLR of 239W/m2 then take a look at Part One and Part Two.
Introduction
If you’ve read more in depth discussions about energy balance or CO2 “saturation” you might have read statements like:
More absorption by CO2 causes emission of radiation to move to higher, colder layers of the atmosphere
If these kind of comments confuse you, sound plain wrong, or cause you to furrow your brow because “it sounds like it’s probably right but what does it actually mean?” – well, hopefully some enlightenment can be found.
Effective Radiation
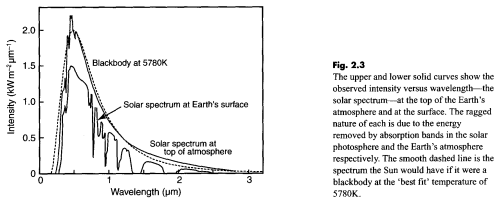
The sun’s core temperature is millions of degrees but we see a radiation from the sun that matches 5780K – its surface temperature:
In this figure there are two spectra: the top one is how the sun’s radiation looks before it reaches the top of the earth’s atmosphere – contrasted with the dotted line of a “blackbody” – or perfect radiator – at 5780K (5507°C for people new to Kelvin or absolute temperature).
The bottom one – of less interest for this article – is how the sun’s radiation looks at the earth’s surface after the atmosphere has absorbed at various wavelengths.
Why don’t we see a radiation spectrum from the sun that matches millions of degrees?
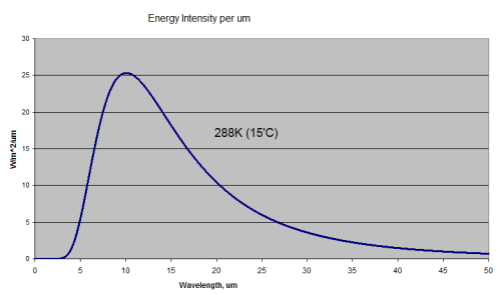
If we measure the upward longwave radiation from the earth’s surface at 15°C we see an effective “blackbody” radiator of 288K (15°C). But why don’t we see a radiation spectrum of 5000K – the temperature somewhere near the core?
The answer to both questions is that radiation from the hotter inner areas of these bodies gets completely absorbed by outer layers, which in turn heat up and radiate at lower temperatures. In the case of the sun, the radiation spectrum includes hotter areas below the surface that are not absorbed at some wavelengths, as well as the surface itself.
In the case of the earth it’s really the top skin layer that emits longwave radiation.
So when we measure the radiation from the earth with a surface temperature of 15°C (288K) we know we will see a longwave radiation that matches this 288K. This will be a total energy radiated of 390W/m2 with the peak wavelength of 10.1μm. The temperature below the surface is irrelevant.
(Well, it’s not really irrelevant. The hotter layers below warm up the layers above – through conduction and radiation).
This is what the radiation looks like:
This assumes an emissivity of 1. The emissivity of the surface of the earth varies slightly but is close to 1, typically around 0.98. Watch out for a dull post on emissivity at some stage..
At the top of atmosphere, as many know, the OLR is around 239W/m2. For those confused by how it can be 390W/m2 at the surface and 239W/m2 at the top, the answer is due to absorption and re-radiation of longwave radiation by trace gases – the “greenhouse” effect. See the CO2 – An Insignificant Trace Gas? series, and especially Part Six – Visualization and CO2 Can’t Have that Effect Because.. if you don’t understand or agree with these well-proven ideas.
If the earth’s atmosphere was completely transparent to longwave radiation this spectrum would look exactly the same at the earth’s surface and at the top of atmosphere (TOA).
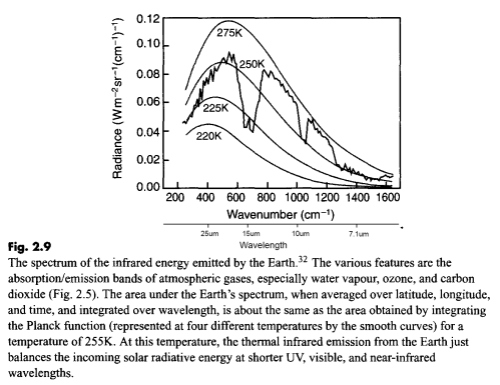
Here’s what it does look like with some typical blackbody radiation curves overlaid:
(Note that the spectrum is shown in wavenumber in cm-1. For convenience I added wavelength in μm under the wavenumber axis. Wavelength in μm = 10,000/wavenumber).
For energy balance – if the earth is not warming up or cooling down – we would expect the earth to radiate out the same amount of energy that it absorbs from the sun. That amount is 239W/m2, which equates to an average temperature of 255K (-18°C).
As the text for this graphic shows, when the energy under the curve is integrated this is what it comes to! But as you can see the actual spectrum is not a “blackbody curve” for 255K. So let’s take a closer look.
Everything Gets Through or Nothing Gets Through – a Few Thought Experiments
Imagine a world where the upwards longwave radiation from the earth’s surface didn’t get absorbed by any gases in the atmosphere.
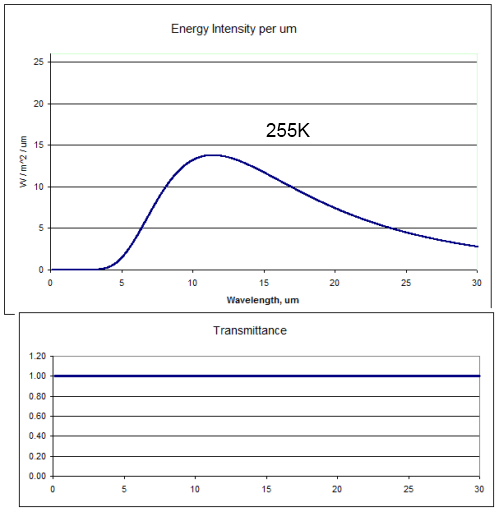
Most people are familiar with that thought experiment – it’s a staple of the most basic radiation model in climate science. The radiation at the top of the atmosphere would look like this (the top graph):
This is the blackbody radiation at 255K (-18°C) with 100% “transmittance” through the atmosphere. The area under the curve, if we extend it out to infinity, is 239W/m2.
And of course, because the radiation hadn’t been absorbed or attenuated in any way, the temperature at the earth’s surface would also be 255K. Chilly.
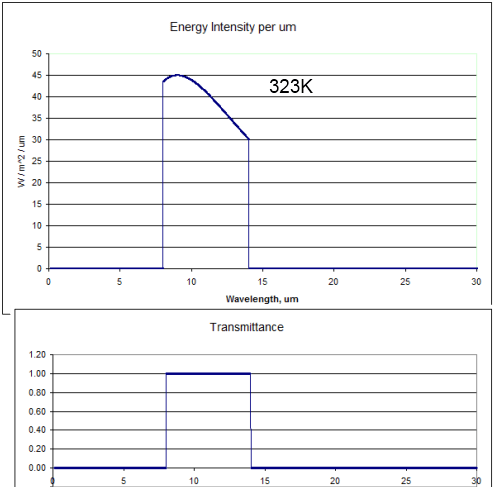
Now let’s think about what would happen if the atmosphere allowed radiation only through the “atmospheric window” and everywhere else the transmittance was zero:
The bottom graph shows how the transmittance of the atmosphere varies with wavelength in this thought experiment.
The top graph in this case is the blackbody radiation from 323K (50°C) only allowed through between 8-14μm. The energy under the curve is 239W/m2. (Note the higher values on the vertical scale compared with the earlier graphs).
So if the atmosphere absorbed all of the surface radiation below 8μm and above 14μm the earth’s surface would heat up until it reached 50°C (323K). Why? Because if the temperature was only 15°C the amount of energy radiated out would only be 141W/m2. More energy coming in than going out = earth heats up. The surface temperature would keep heating up until eventually 239W/m2 made it out through the atmospheric window – which is 50°C.
Closer to The Real World – Illustration of Radiation from Multiple Layers in the Atmosphere
Even in the atmospheric window some radiation is absorbed, i.e. the transmittance is not 1. But let’s assume for sake of argument it is 1. So energy in the 8-14μm band just passes straight through the atmosphere. It’s still a thought experiment.
Lots of gases absorb at lots of wavelengths – which makes thinking about it as a whole very difficult. So we’ll just assume that the rest of the atmosphere outside the atmospheric window all shares the same absorption characteristics – that is, every wavelength is identical in terms of absorption of radiation.
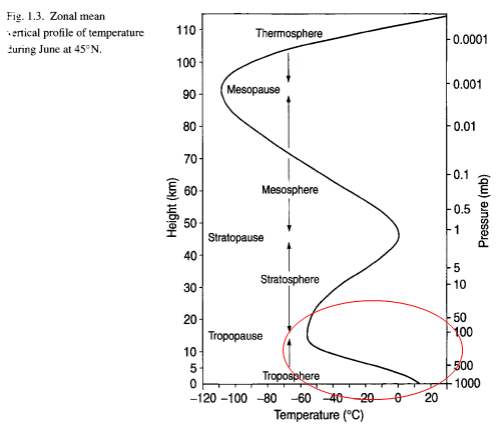
Now let’s try and consider what really happens in the atmosphere. Each “layer” of the atmosphere radiates out energy according to the temperature in that layer. For reference, here is the temperature (and pressure) at different heights:
The highlighted area at the bottom – the troposphere – is the area of interest. This is where most of the atmosphere (by molecules and mass) actually resides.
In our thought experiment radiation from the surface (outside the atmospheric window) gets completely absorbed by the atmosphere, or at least the amount that gets through is very small. Taken to the extreme we would get the result shown a few graphs earlier where the surface temperature rises up to 50’C.
But just because surface radiation doesn’t get out doesn’t mean that radiation from the atmosphere can’t get out.
Each layer of the atmosphere radiates according to its temperature. Even if the atmosphere’s transmittance is zero when considering the entire thickness of the atmosphere, there will be some layer where radiation starts to get through.
This is partly because there is less atmosphere to absorb the closer we get to the “top”. And also because as we get higher in the atmosphere it gets thinner. Less molecules to absorb radiation. Even if some gas is a fantastically good absorber of energy, there must be a point where radiation is hardly absorbed. For example, at the top of the stratosphere, about 50km, the pressure is around 1mbar – 1000x less than at the surface. At the top of the troposphere (the tropopause) the pressure is around 200mbar – 5x less than at the surface.
The challenge in thinking about the atmosphere radiating is that unlike the surface of the earth where all radiation is emitted from the very surface, instead radiation is emitted from lots of different layers:
- Higher up – less absorption, more radiation makes it through
- Lower down – more absorption, less radiation makes it through
But let’s still keep it simple and think about the surface temperature being our standard 15°C (288K) and the atmospheric window letting through everything between 8-14μm. This means 141W/m2 makes it out through this window.
If we have energy balance, the OLR = 239W/m2 in total = 141 (through the atmospheric window) + 98 (radiated from the atmosphere at some height, wavelengths outside 8-14μm).
What temperature equates to this layer in the atmosphere? Well, assuming no absorption above this radiating layer (not really the case), and only radiation outside 8-14μm, the temperature of the atmosphere would have to be 219K, or -54°C. Take a look back at the temperature profile above – this is pretty much the top of the troposphere, around 11km.
Remember that this isn’t exactly how radiation gets radiated out to space – it doesn’t come from one “skin layer”. We might consider that if the transmittance of the atmosphere is 1 at this height, then maybe at 10km the transmittance is 0.8 and at 9km the transmittance is 0.5, and at 8km the transmittance is 0.1..
So each layer is radiating energy, with higher layers being colder but more of their radiation getting through, and lower layers being warmer – so radiating a higher amount – but less of their radiation getting through.
For many people reading, this is a straightforward concept, why so long.. for others it might still seem tough to grasp..
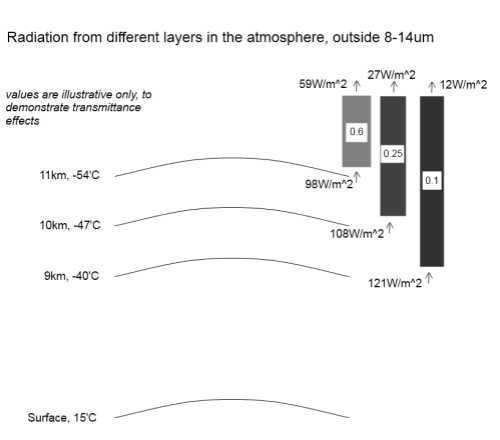
So here is a sample radiation diagram with illustrative values only (and values a little different from above):
What the diagram shows is the radiation outside the 8-14μm band. That’s because in our thought experiment the 8-14μm band doesn’t absorb any radiation (and therefore can’t radiate in this band either).
Take the top layer at 11km. If we calculate the blackbody radiation of 219K (-54°C) and exclude radiation in the 8-14μm band the radiation is 98W/m2. Then the grey block above with “0.6” is the atmosphere above with transmittance of 0.6, so the radiation actually getting through from this layer to the top of atmosphere is 59W/m2. Similarly for the two other layers (with different values).
In total, the energy leaving the top of atmosphere (outside of the atmospheric window) is 98W/m2. (It’s just a coincidence that this is the value of the top layer before any absorption). And inside the atmospheric window was the number we already calculated of 141W/m2, so the total OLR is 239W/m2.
Of course, we all know the real atmosphere is much more complex with lots of different absorption at different wavelengths. But hopefully this “intermediate” example help to explain how the atmosphere radiates out energy.
So finally, onto the real point..
What Happens with More Absorbing Gases?
Remember how this long post started..
If you’ve read more in depth discussions about energy balance or CO2 “saturation” you might have read statements like:
More absorption by CO2 causes emission of radiation to move to higher, colder layers of the atmosphere
Now, maybe this kind of statement will make more sense.
In our model – our thought experiment – above, we had a uniform absorber of radiation outside the atmospheric window. Suppose we increase the amount of this absorber – the skies open and someone pours some more in and stirs it around. Let’s say the amount increases by 10%.
Well, take a look back at the last diagram. See the transmittance values for each layer in the atmosphere – 0.6 at 11km high, 0.25 at 10km high and 0.1 at 9km high.
Regardless of how realistic these actual numbers are, increasing the amount of absorbing gas by 10% will automatically mean that each of the transmittance numbers is reduced by 10%. And so less radiation makes it out to the TOA (top of atmosphere).
Effectively because lower layers are contributing less energy out through TOA the effective radiating height has moved up. It’s not because some directive has been passed down from a higher authority. And it’s not because one layer has stopped and another layer has taken over.
It’s just that lower layers contribute less, so the “average radiating height” is now higher and colder.
(Note: it might look at first sight that the average height is still the same even though the amount of radiation has reduced. This is not really the case, see the note at end).
In our particular example what would happen is that the OLR would reduce from 239W/m2 down to 141+98*0.9=229 W/m2. So the surface would warm up and this would warm up each layer of the atmosphere until eventually a new hotter steady state was reached.
Conclusion
This has been a long post to try and create more of an understanding of how the earth actually radiates energy, and why more of any trace gas increases the “greenhouse” effect.
It does it because more “absorbing gases” reduce the amount of radiation that can make it out from lower layers in the atmosphere. These lower layers are hotter and radiate much more energy. Proportionately more energy will then be radiated from higher layers which are colder, and therefore these radiate less energy.
It’s a not a mystical force that raises the “effective radiating height” in the atmosphere. But the effective radiating height does increase.
Note
In the example above, the three layers together contributed 98W/m2 at TOA. That is an “effective temperature” of 219K – remembering that we are excluding radiation from the 8-14μm window. If we reduce the radiation from these three layers by 10%, we now have 89W/m2 which is about 212K – effectively radiating from a colder level in the atmosphere.