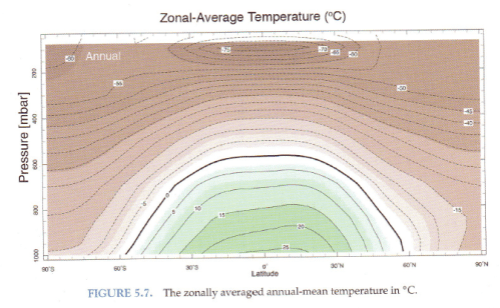
Here is the annual mean temperature as a function of pressure (=height) and latitude:
Figure 1 – Click for a larger image
We see that the equator is warmer than the poles and the surface is warmer than the upper troposphere (“troposphere” = lower atmosphere). No surprises.
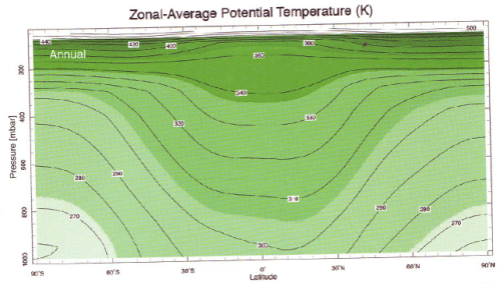
Here is “potential temperature”, whatever that is..
Figure 2 – Click for a larger image
We see that – whatever “potential temperature” is – the equator is warmer than the poles, but this version of temperature increases with height.
Why does temperature decrease with height? What is potential temperature? And why does it increase with height?
The Lapse Rate
Atmospheric pressure decreases with height. This is because as you go higher up there is less air above you, and therefore less downward force due to the weight of this air.
Because pressure decreases – and because air is a compressible fluid – air that rises expands (and air that sinks contracts).
Air that expands does “work” against its surroundings and because of the first law of thermodynamics (conservation of energy) this work needs to be paid for. So internal energy is consumed in expanding the parcel of air outwards against the atmosphere. And a reduction in internal energy means a reduction in temperature.
- Air that rises expands
- Expanding air cools
A little bit more technically.. adiabatic expansion is what we are talking about. An adiabatic process is one where no heat is exchanged with the surroundings. This is a reasonable approximation for typical rising air. It is reasonable because conduction is an extremely slow process (= negligible) in the atmosphere and radiative heat transfer is quite slow.
So if heat can’t be exchanged between a “parcel of air” and its surroundings it is relatively simple to calculate how the temperature changes. An example which contains way too much detail (because it is debunking a “debunking”) at Paradigm Shifts in Convection and Water Vapor?
The essence of the calculation is to equate internal energy changes with work done on the environment.
Textbooks usually start off with the simplest version, the dry adiabatic lapse rate, or DALR. (The “lapse rate” is the change in temperature with height of a parcel of air).
The DALR is for air without any water vapor. Now water vapor is very influential in our climate. The reason for neglecting it and starting off with this simplification is:
- the calculation is easy and everyone (almost) can understand it
- it represents one extreme of the atmosphere (polar climates and upper troposphere)
The result from this simplification:
Change in temperature with height = -g/cp ≈ -10 °C/km, where g = acceleration due to gravity = 9.8 m/s² and cp = heat capacity of air at constant pressure ≈ 1 J/kg.K
So for every km we displace air upwards it cools by about 10°C – so long as we displace it reasonably quickly. Well, this is true if it is dry.
A note on conventions – dry parcels of air moved upwards cool by 10°C per km, but the lapse rate is usually written as a positive number. So a cooling of 10 °C/km = -10 °C/km, but by convention, equals a “lapse rate” of +10 °C/km. This makes it very confusing when people say things like “the environmental lapse rate must be less than the adiabatic lapse rate“. Are we talking about the number with the minus sign in front? Or not?
It’s not easy to think about negative numbers being less than other negative numbers when the “less than” test is applied after they have been made into positive numbers. Not for me anyway. I have to write it down each time.
The Saturated Lapse Rate
If a parcel of air contains water vapor and it cools sufficiently then the water vapor condenses. This releases latent heat.
As a result, moist rising air cools slower than dry rising air
So the saturated adiabatic lapse rate is “less than” the dry adiabatic lapse rate.
E.g. the change in temperature with height of a dry parcel of air ≈ -10 °C/km, while the change in temperature with height of a moist parcel of air in the tropics near the surface ≈ -4 °C/km.
Conventionally we say that the saturated adiabatic lapse rate is less than the dry adiabatic lapse rate. Because we write them as positive numbers.
Now note the caveats around the value for the moist parcel of air rising. I said “..in the tropics near the surface..”, but for the DALR there are no caveats. That’s because once we consider moisture we have to consider how much water vapor and the amount varies hugely depending on temperature (and also on other factors – see Clouds and Water Vapor – Part Three).
The maths is somewhat harder for the saturated adiabatic lapse rate but it’s not conceptually more difficult, there is just an addition of energy (from condensing water vapor) to offset the work done.
Potential Temperature
Potential temperature is usually written with the Greek letter θ.
θ = T.(p0/p)k
where T = (real) temperature, p = pressure, p0 = reference pressure (usually at 1000 mbar) and k = R/cp = 2/7 for our atmosphere (more on this in a later article)
With a bit of tedious maths we can prove that θ stays constant under adiabatic conditions (for dry air).
Let’s look at what that means.
Suppose the surface (1000 mbar) temperature = 288 K (15°C) so also θ = 288K.
Now the air is moved (adiabatically) to 800 mbar, so T = 270 K. That’s what you expect – temperature falls with height. And no change to potential temperature, so θ = 288 K.
Now we move the air to 600 mbar, and T = 249 K. More reduction of temperature. And still θ = 288 K.
So is this a useful parameter – move the air (adiabatically) and the potential temperature stays the same?
The parameter is mathematically sound, but whether it is useful remains to be seen. As an artificial construct no doubt many people will be shaking their heads..
Stability and Potential Temperature Profile
In Density, Stability and Motion in Fluids we saw that for a fluid to be stable, lighter fluid must be above heavier fluid. No surprise to anyone.
And we saw that in mechanical terms equilibrium is different from stability.
An unstable equilibrium can exist, but a slight displacement will turn the instability into motion. Whereas with a stable equilibrium a slight displacement (or a large displacement) will result in a restoring force back to its original position. For the simplest case – an incompressible fluid – this means that the temperature must increase with height.
If you watched the accompanying video of a tank of water being heated from below you would have seen that the instability caused turbulent motion until finally the tank was well-mixed.
We left the more complex case of compressible fluids (like air) until today. What we will find is that with a compressible fluid potential temperature is effectively the same as “real” temperature for an incompressible fluid.
So if potential temperature increases with height the fluid is stable, but if potential temperature decreases with height the fluid is unstable.
Let’s look at two examples:
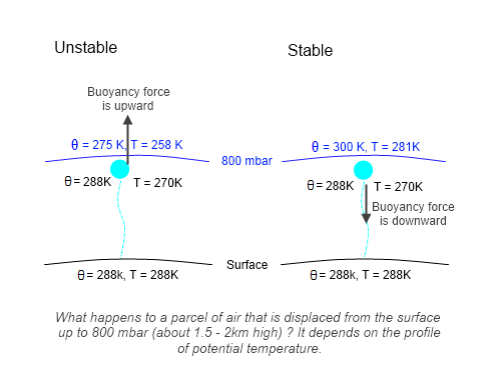
Figure 3
On the left hand side we see an example where potential temperature decreases with height. At the surface, θ = 288 K but at 800 mbar, θ = 275 K. A parcel of air displaced adiabatically from the surface to 800 mbar will keep its potential temperature of 288 K. Now we convert that to real temperatures. The environmental temperature at 800 mbar is 258 K, but the parcel of air cools to only 270 K. This means the displaced parcel is warmer than the surroundings, so it is less dense – and therefore it keeps rising.
This case is unstable – clearly any air that starts rising or falling (perhaps due to atmospheric winds, pressure differentials, etc) will keep rising or falling.
On the right hand side we see potential temperature decreasing with height. The parcel of air displaced from the surface to 800 mbar reaches the same temperature as on the left – 270 K. But here the environmental temperature is 281 K. So the parcel of air is cooler than the surrounding air, so it is more dense – and so it falls.
This case is stable – any air that starts rising or falling experiences a restoring force.
So the potential temperature profile with height tells us whether the atmosphere is stable, neutral or unstable. If potential temperature increases with height the atmosphere is stable, and if potential temperature decreases with height the atmosphere is unstable.
This is exactly the same as comparing the actual temperature change with the lapse rate.
Both answer the same question about atmospheric stability.
Moist Potential Temperature
The previous section slightly over-simplified things because potential temperature is with reference to dry air and yet moisture changes the way in which temperature decreases with height.
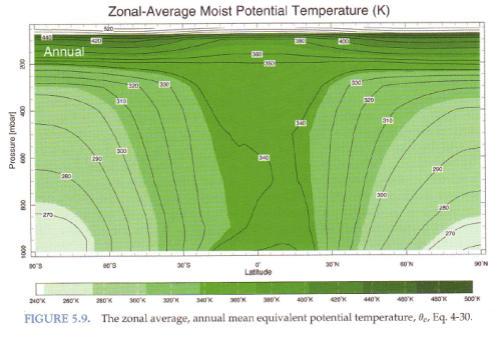
So here is the real deal – moist potential temperature. This is also known as equivalent potential temperature:
Figure 4 – Click for a larger image
Here we see the “real potential temperature” and notice that especially in the tropics moist potential temperature is almost constant with height – up to the tropopause at 200 mbar. This is due to convection creating a well-mixed atmosphere. In the polar regions we see that the atmosphere is still quite stratified, which is due to the lack of convective mixing.
Conclusion
Potential temperature is very useful. It is a method of comparing the temperature of air at two different heights.
And if potential temperature is constant or increasing with height then the atmosphere is stable.
The atmosphere is mostly stable for dry air. If you refer back to figure 2 you see that (dry) potential temperature is quite stratified which means any displaced air experiences a restoring force. So it is moisture in the air that is the enabler for most of the convection that takes place. Figure 4 shows us that the atmosphere is “finely” balanced as far as moist convection is concerned.
(Remember of course that these graphs are annual mean values. It doesn’t mean that dry convection does not occur).
Potential temperature is also a useful metric because the change of potential temperature with height can be used to calculate the strength of the restoring force on displaced air. The result is the buoyancy frequency and the period of internal gravity waves.