Many people often claim that the atmosphere cannot “heat” the earth’s surface and therefore the idea of the inappropriately-named “greenhouse” effect is scientifically impossible. The famous Second Law of Thermodynamics is invoked in support.
Let’s avoid a semantic argument about the correct or incorrect use of the word “heat”.
I claim that energy from the atmosphere is absorbed by the surface.
This absorbed energy has no magic properties. If the surface loses 100J of energy by other means and gains 100J of energy from the atmosphere then its temperature will stay constant. If the surface hasn’t lost or gained any energy by any other means, this 100J of energy from the atmosphere will increase the surface temperature.
I also claim that because the atmosphere is on average colder than the surface, more energy is transferred from the surface to the atmosphere compared with the reverse situation.
Let’s consider whether this violates the real second law of thermodynamics..
The Conceptual Problem
In Heat Transfer Basics – Part Zero a slightly off-topic discussion about the “greenhouse” effect began. One of our most valiant defenders of the imaginary second law of thermodynamics said:
An irradiated object can never reach a higher temperature than the source causing the radiation
I have demonstrated previously in The First Law of Thermodynamics Meets the Imaginary Second Law that a colder body can increase the temperature of a hotter body (compared with the scenario when the colder body was not there).
In that example, there was more than one source of energy. So, with this recent exchange in Heat Transfer Basics it dawned on me what the conceptual problem was. So this article is written for the many people who find themselves agreeing with the comment above. As a paraphrased restatement by the same commenter:
If the atmosphere is at -30°C then it can’t have any effect on the surface if the surface is above -30°C
Entropy Basics and The Special Case
Entropy is a difficult subject to understand. Heat and temperature are concepts we can understand quite easily. We all know what temperature is (in a non-precise way) and heat, although a little more abstract, is something most people can relate to.
Entropy appears to be an abstract concept with no real meaning – nothing you can get your hands around.
The second law of thermodynamics says:
Entropy of a “closed system” can never reduce
Before defining entropy, here is an important consequence of this second law:
Increasing entropy means that heat flows spontaneously from hotter to colder bodies and never in reverse
This fits everyone’s common experience.
- Ice melts in a glass of water
- A hot pan of water on the stove cools down to room temperature when the heat source is removed
- Put a hold and cold body together and they tend to come to the same temperature, not move apart in temperature
And all of these are easier to visualize than a mathematical formula.
What is entropy? I will keep the maths to an absolute minimum, but we have to introduce a tiny amount of maths just to define entropy.
For a body absorbing a tiny amount of heat, δQ (note that δ is a symbol which means “tiny change”), the change in entropy, δS, is given by:
δS = δQ / T, where T is absolute temperature (see note 1)
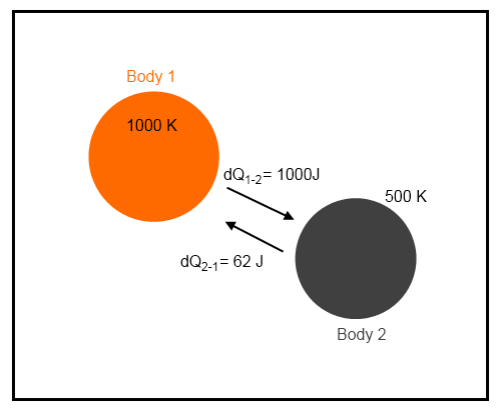
It’s not easy to visualize – but take a look at a simple example. Suppose that a tiny amount of heat, 1000 J, moves from a body at 1000K to a body at 500K: 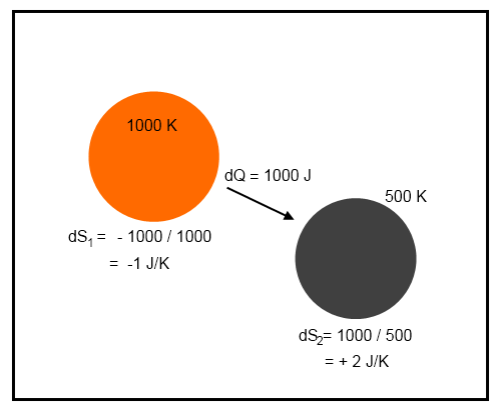
Example 1
The net change in energy in the system is zero because 1000J leaves the first body and is absorbed by the second body. This is the first law of thermodynamics – energy cannot be created or destroyed.
However, there is a change in entropy.
The change in total entropy of the system = δS1 + δS2 = -1 + 2 = 1 J/K.
This strange value called “entropy” has increased.
Notice that if the energy flow of 1000J was from the 500K body to the 1000K body the change in entropy would be -1 J/K. This would be a reduction in entropy – forbidden by the real second law of thermodynamics. This would be a spontaneous flow of heat from the colder body to the hotter body.
Updated note Sep 30th – this example is intended to clarify the absolute basics.
Think of the example above like this – If, for some reason, in a closed system, this was the only movement of energy taking place, we could calculate the entropy change and it has increased.
The example is not meant to be an example of only one half of a radiative energy exchange. Just a very very simply example to show how entropy is calculated. It could be conductive heat transfer through a liquid that is totally opaque to radiation.
The Special Case
The simplest example demonstrating the second law of thermodynamics is with two bodies which are in a closed system.
Let’s say that we have a gas at 273K (Body 1) and a solid (Body 2) surrounded by the gas. The solid starts off much colder.
What is the maximum temperature that can be reached by the solid?
273K
Easy. In fact, depending on the starting temperature of the solid and the respective heat capacities of the gas and solid, the actual temperature that both end up (the same temperature eventually) might be a little lower or a lot lower.
But the temperature reached by the solid can never get to more than 273K. For the solid to get to a temperature higher than 273K the gas would have to cool down below 273K (otherwise energy would have been created). Heat does not spontaneously flow from a colder to a hotter body so this never happens.
This defining example is illuminating but no surprise to anyone.
It is important to note that this special case is not the second law of thermodynamics, it is an example that conforms to the second law of thermodynamics.
The second law of thermodynamics says that the entropy of a system cannot reduce. If we want to find out whether the second law of thermodynamics forbids some situation then we need to calculate the change in entropy – not use “insight” from this super-simple scenario.
So let’s consider some simple examples and see what happens to the entropy.
Simple Examples
What I want to demonstrate is that the standard picture in heat transfer textbooks doesn’t violate the second law of thermodynamics.
What is the standard picture?
This says that two bodies separated in space both emit radiation. And both absorb radiation from the other body (see note 2).
The challenging concept for some is the idea that radiation from the colder body is absorbed by the hotter body.
We start with Example 1 above, but this time we consider an exchange of radiation and see what happens to the entropy of that system.
Example 2
What I have introduced here is thermal radiation from Body 2 incident on Body 1. We will assume all of it is absorbed, and vice-versa.
According to the Stefan-Boltzmann equation, energy radiated is proportional to the 4th power of temperature. Given that Body 2 is half the temperature of Body 1 it will radiate at a factor of 24 = 2 x 2 x 2 x 2 = 16 times less. Therefore, if 1000J from Body 1 reaches Body 2, then 62J (1000/16) will be transmitted in the reverse direction. However, the exact value doesn’t matter for the purposes of this example.
So with our example above, what is the change in entropy?
Body 1 loses energy, which is negative entropy. Body 2 gains energy, which is positive entropy.
δS1 = -(1000-62)/1000 = -938 / 1000 = -0.94 J/k
δS2 =(1000-62)/500 = 938 / 500 = 1.88 J/K
Total entropy change = -0.94 + 1.88 = 0.94 J/K.
So even though energy from the colder body has been absorbed by the hotter body, the entropy of the system has increased. This is because more energy has moved in the opposite direction.
There is no violation of the second law of thermodynamics with this example.
Now let’s consider an example with values closer to what we encounter near the earth’s surface:
Example 3
This isn’t intended to be the complete surface – atmosphere system, just values that are more familiar.
Surface: δS1 = -(390-301)/288 = -89 / 288 = -0.31 J/k
Atmosphere: δS2 = (390-301)/270 = 89 / 270 = 0.33 J/K
Total entropy change = -0.31 + 0.33 = 0.02 J/K.
So even though the temperatures of the two bodies are much closer together, when they exchange energy, total entropy still increases.
Energy from the colder atmosphere has been absorbed by the hotter surface and yet entropy of the system has still increased.
Now, the example above (example 3) is an exchange of a fixed amount of energy (in Joules, J). Suppose this is the amount of energy per second (Watts, W) or the amount of energy per second per square meter (W/m²).
If the atmosphere keeps absorbing more energy than it is emitting it will heat up. If the earth keeps emitting more energy than it absorbs, it will cool down.
If example 3 was the complete system, then the atmosphere would heat up and the earth would cool down until they were in thermal equilibrium. This doesn’t happen because the sun continually provides energy.
The Complete Climate System
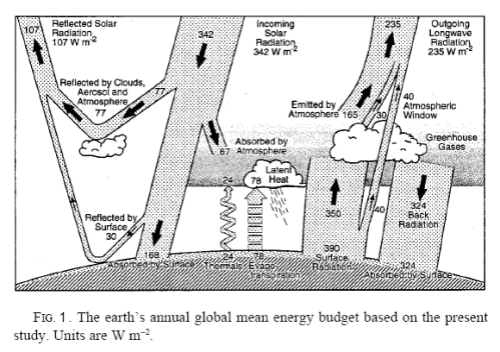
The earth-atmosphere system is very complex. If we analyze a long term average scenario, like that painted by Kiehl and Trenberth there is an immediate problem in calculating the change in entropy:
[Note from Sep 28th – This section is wrong, thanks Nick Stokes for highlighting it and so delicately! Preserved in italics for entertainment value only..] If we consider the surface, for example, it absorbs 492 W/m² (δQ = 492 per second per square meter) and it loses 492 W/m² (δQ = -492 per second per square meter).
Net energy change = 0. Net entropy change = 0.
Why isn’t entropy increasing? We haven’t considered the whole system – the sun is generating all the energy to power the climate system. If we do consider the sun, it is emitting a huge amount of energy and, therefore, losing entropy. But the energy generation inside the sun creates more entropy – that is, unless the second law of thermodynamics is flawed.
[Now the rewritten bit]
Previous sections explained that calculations of entropy “removed” (negative entropy) are based on energy emitted divided by the temperature of the source. And calculations of entropy “produced” are based on energy absorbed divided by the temperature of the absorber. In a closed system we can add these up and we find that entropy always increases.
So the calculation in italics above is incorrect. Change in entropy at the surface is not zero.
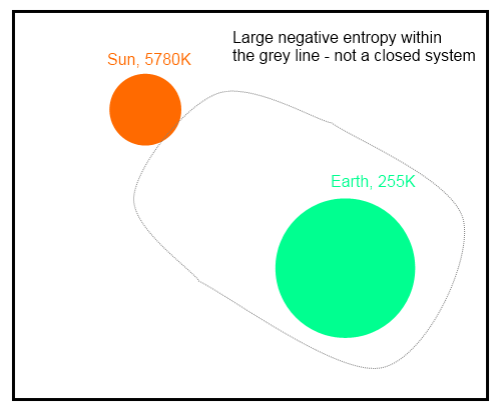
Change in entropy at the surface is a large negative value, because we have to consider the source temperature of the energy.
So as Nick Stokes points out (in a comment below), we can draw a line around the whole climate system, including the emission of radiation by the sun (see example 4 just below). This calculation produces a large negative entropy, because it isn’t a closed system. This is explained by the fact that the production of solar energy creates an even larger amount of positive entropy.
Example 4
The Classic Energy Exchange by Radiation
I was in the university library recently and opened up a number of heat transfer textbooks. All of them had a similar picture to that from Incropera and DeWitt (above). And not a single one said, This doesn’t happen.
In any case, for someone to claim that an energy exchange violates the second law of thermodynamics they need to show there is a reduction in entropy of a closed system.
But one important point did occur to me when thinking about this subject. Let’s reconsider our commenter’s claim:
An irradiated object can never reach a higher temperature than the source causing the radiation
As I pointed out in the The Special Case section – this is true if this is the only source of energy. Yet the surface of the earth receives energy from both the sun and the atmosphere.
If the colder atmosphere cannot transfer energy to a warmer surface, and the second law of thermodynamics is the reason, the actual event that is forbidden is the emission of radiation by the colder atmosphere. When the colder atmosphere radiates energy it loses entropy.
After all, the entropy loss takes place when the atmosphere has given up its energy. Not when another body has absorbed the energy.
Our commenter has frequently agreed that the colder atmosphere does radiate. But he doesn’t believe that the surface can absorb it. He has never been able to explain what happens to the energy when it “reaches” the surface. Or why the surface doesn’t absorb it. Instead we have followed many enjoyable detours into attempts to undermine any of a number of fundamental physics laws in an attempt to defend “the imaginary second law of thermodynamics”.
Conclusion
Entropy is a conceptually difficult subject, but all of us can see the example in “the special case” and agree that the picture is correct.
However, the atmosphere – surface interaction is more complex than that simple case. The surface of the earth receives energy from the sun and the atmosphere.
As we have seen, in simple examples of radiant heat exchange between two bodies, entropy is still positive even when the hotter body absorbs energy from the colder body. This is because more energy flows from the hotter to the colder than the reverse.
To prove that the second law of thermodynamics has been violated someone needs to demonstrate that a system is reducing entropy. So we would expect to see an entropy calculation.
Turgid undergraduate books about heat transfer in university libraries all write that radiation emitted by a colder body is absorbed by a hotter body.
That is because the first law of thermodynamics is still true – energy cannot be created, destroyed, or magically lost.
Other Relevant Articles
The Amazing Case of “Back Radiation” – Part Three
The First Law of Thermodynamics Meets the Imaginary Second Law
Intelligent Materials and the Imaginary Second Law of Thermodynamics
Amazing Things we Find in Textbooks – The Real Second Law of Thermodynamics
Notes
Note 1 – There are more fundamental ways to define entropy, but it won’t help to see this kind of detail. And for the purists, the equation as shown relies on the temperature not changing as a result of the small transfer of energy.
If the temperature did change then the correct formula is to integrate:
ΔS = ∫Cp/T. dT (with the integral from T1 to T2) and the result,
ΔS = Cp log (T2/T1), this is log to the base e.
Note 2 – This assumes there is some “view factor” between the two bodies – that is, some portion of the radiation emitted by one body can “hit” the other. Just pointing out the obvious, just in case..