One question that has intrigued me for a while – how much of the transmittance (and change in transmittance) from CO2 in the atmosphere is caused by weak lines, and how much is caused by the “far wings” of individual lines.
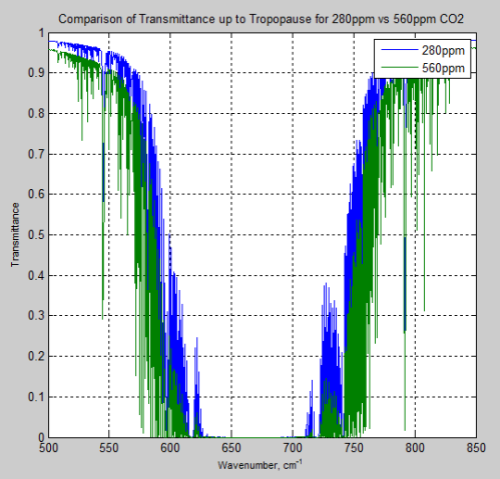
Take a look back at Part Nine. Here is the calculated change in transmittance from the surface to the tropopause for a doubling of CO2 from pre-industrial levels:
Figure 1
We can see that there is significant change outside of the central range of wavenumbers. (For reference, 667 cm-1 = 15 μm).
Here is another of the graphics – showing how an individual line absorbs across a range of wavenumbers:
Figure 2
There are many lines in the HITRAN database for CO2 (over 300,000 lines), many of them weak.
So back to my original question – is it the “wings” = line broadening, of the individual lines, or the weaker lines that have the biggest effect? And how do we quantify it?
The Curve of Growth
The Curve of Growth is about the change in transmittance with an increase in path length (specifically the increase in number of absorbing molecules in the path).
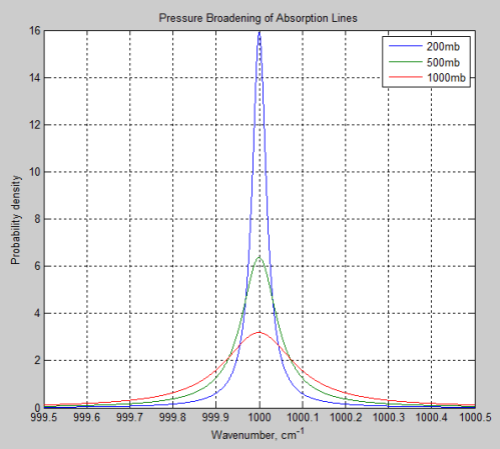
The reason the problem is not so simple is because each line has a line shape that absorbs across a range of wavelengths.
We know that transmittance, t=e-τ – this is the Beer-Lambert law. The spreading out of the line means that the reduction in transmittance as the path increases isn’t as strong as predicted by a simple application of the Beer-Lambert law.
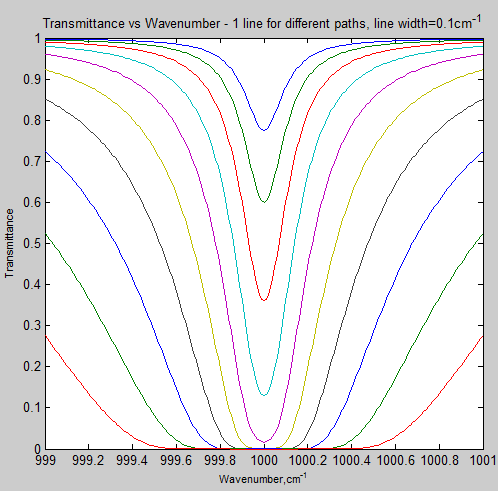
Here is a calculation of this for a typical tropospheric condition (note 1) – for one isolated absorption line with its line center at 1000 cm-1:
Figure 3
Each curve represents a doubling of the number of absorbing molecules in the path. So, if this was at constant density, each curve represents a doubling of the path length.
What does this graph show?
At the line center, an increase in the path length soon causes saturation. But out in the “wings” of the absorption line the reduction in transmittance changes much more slowly.
With some maths we can show that for very small paths (note 2), the absorptance is linearly proportional to the number of absorbers in the path.
And with some more maths we can show that for very large paths (note 2), the absorptance increases as the square root of the number of absorbers in the path.
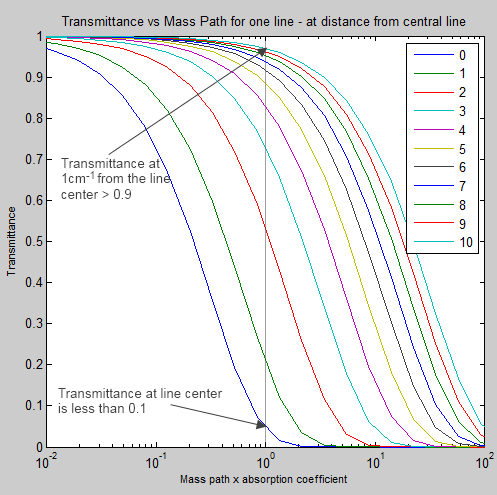
Here is another way to view the “curve of growth”. I haven’t seen it shown this way before:
Figure 4
This is the same scenario, but this time the x-axis (bottom axis) has the effective path length, while each curve represents a different distance from the line center. The legend is showing the distance from the line center compared with the “line width”. So “10” = 1 cm-1 from the line center, while “0” is the line center.
Notice the difference at a mass path x absorption coefficient = 100 (=1) where the line center has a transmittance of 0.05 while at 1 cm-1 distance from the line center the line has a transmittance of 0.97.
What happens when we look at the effect of the whole line?
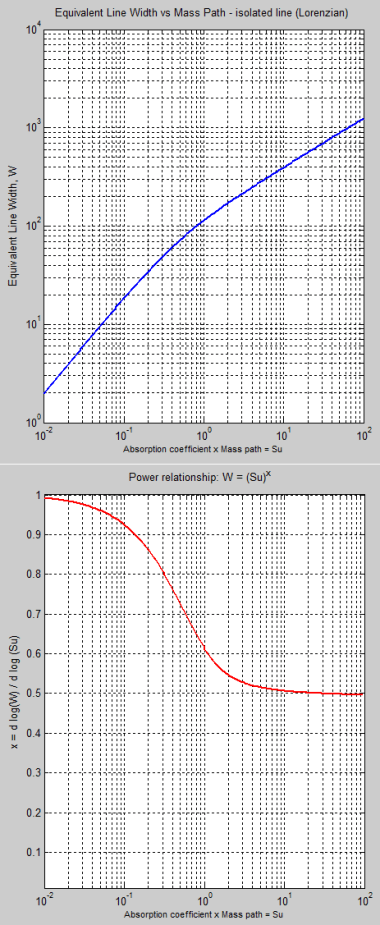
Here is a curve of the equivalent line width, W as the path increases. This equivalent line width is just the total effect across enough bandwidth to take into account the effects of the far wings of the line (note 3):
Figure 5
The 2nd graph is the important one. This shows the power relationship between W and Su, as Su increases – where Su = absorption coefficient x mass of molecules in the path. S is the absorption coefficient, and u is the mass.
For a given line, S is a constant, and so an increase in Su means an increase in the number of molecules in the path.
So if we believe that W = (Su)x, how do we determine x?
We can do it by taking the log of both sides: log(W)=x.log(Su), so by calculating the slope of this log relationship we can see how this value, x, changes.
The 2nd graph shows:
- at very small paths, the line strength is proportional to Su – because x = 1
- at very large paths, the line strength is proportional to √(Su) – because x = 0.5
This is simply backing up by numerical calculation the earlier claim: “With some maths we can show..”
So if you want to understand “the curve of growth” you need to understand that at very small optical thickness the “equivalent line” grows in linear proportion to the mass of molecules in the path, and with very large optical thickness the “equivalent line” grows in proportion to the square root.
And probably the easiest way to see it conceptually is to take another look at Figure 3.
Line Wings in Practice
So back to my original question. Is it the weak lines or the wings of the strong lines that has the effect?
The first step is to calculate the effect of the line wings. I took the MATLAB program developed for Part Nine and made a few changes. The original program had simply applied the equation for line shape out to the edge of the region under consideration. In this version I added a new factor, ca, which “chopped” the line shape. The factor ca limits the line shape for each line to the line center, v0 ± (ca * line width).
The line width is a parameter in the HITRAN database and is a measure of the shape of the line, approximately the value where the strength has fallen to half the peak value.
The original program was run for two values of atmospheric CO2: 280ppm (pre-industrial); and 560ppm (doubling of CO2) – with 15 layers and a calculation every 0.01 cm-1 across the band of 500 cm-1 – 850 cm-1. These are called the “Standard” results. And for these, and all following simulations, we are only considering the main isotopologue of CO2, which accounts for over 98% of atmospheric CO2.
Then the revised program was run for a number of values of ca: 5000, 1000, 100 & 10.
Given that the typical line width is 0.05 – 0.1 cm-1 this means that each line is considered between two extremes: across 250-500 cm-1 down to only 0.5 – 1 cm-1.
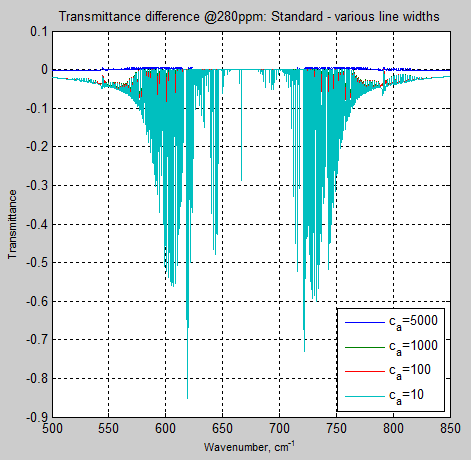
As expected, with ca = 5000, the difference between that and the Standard is almost nil. And as ca is reduced the differences increase:
Figure 6
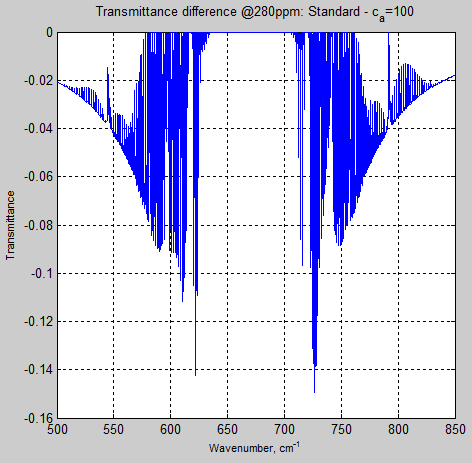
By the time ca = 100, the differences are noticeable, and at ca = 10, the differences in some parts of the band are huge. The above result (figure 6) is dominated by the ca=10 result so here is ca=100 separately:
Figure 7
So this demonstrates that even when the line shape is “limited” to 100x the line width, there is still a noticeable effect in the transmittance calculation for the troposphere.
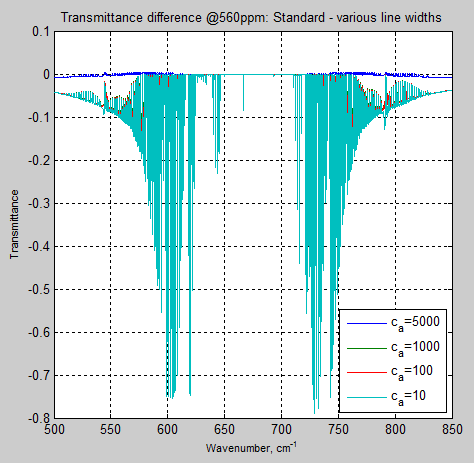
For completeness, here is the same comparison at figure 6, but for 560ppm:
Figure 8
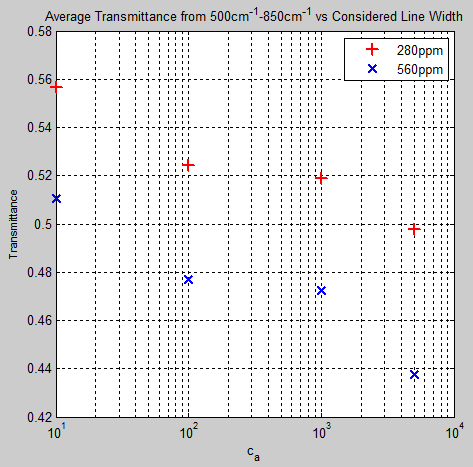
Across this band, the standard case at 280ppm: t = 0.4980, and at 560ppm, t = 0.437.
Here is how the mean transmittance changes as we crop the line shape. Cropping the line shape means that we make the atmosphere more transparent.
Figure 9
We can see from figure 9 that the change in transmittance from 280 – 560 ppm is of a similar magnitude at each artificial “cropping” of the line shape.
Weak Lines
So let’s take a look at the effect of weak lines. For this case I used the same MATLAB program developed for Part Nine but with a user-defined selection of lines. For example, the top 10% of lines by strength.
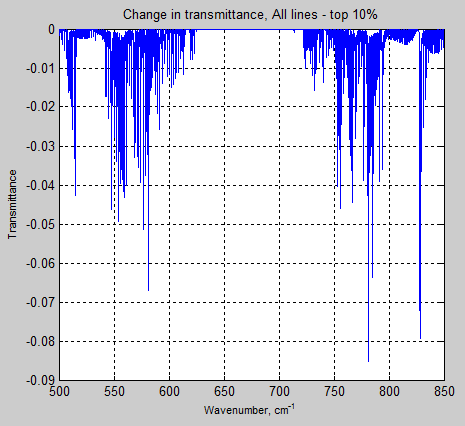
Here is the transmittance change @ 280ppm through the troposphere, for all lines (standard) less the transmittance of the strongest 10% of lines:
Figure 10
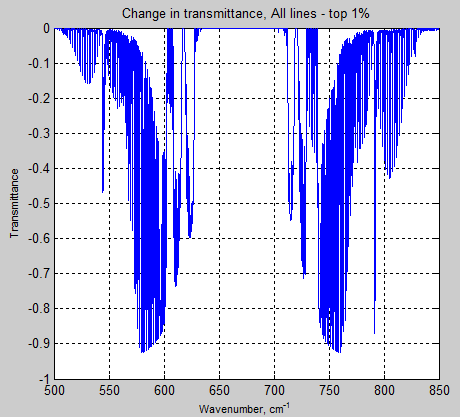
And here is the graph of all lines – the strongest 1%:
Figure 11
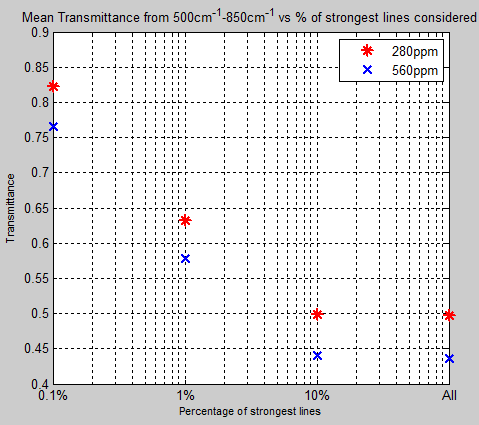
And here is the mean transmittance for both 280ppm and 560ppm as only the strongest lines are considered:
Figure 12
It’s clear that the weakest 90% of lines have virtually no effect on the transmittance of the atmosphere. There are a lot of very weak lines in the HITRAN database.
The calculated transmittance for 100% of the lines at 280ppm = 0.4974 and for the top 10% = 0.4994 – meaning that the top 10% of lines account for 99.6% of the transmittance.
At 560ppm the top 10% of lines account for 99.3%.
Conclusion
Some of this analysis is of curiosity value only.
However, it is very useful to understand the “curve of growth” – and to realize how absorptance increases as the mass in the path increases.
And it’s at least interesting to see how the “far wings” of the individual lines have such an effect on the transmittance through the atmosphere. Even “cropping” the effect at 100x the line width has a significant effect on the atmospheric transmittance.
And for the question posed at the beginning, both the weak lines and the far wings of individual lines have an effect on the total atmospheric transmittance.
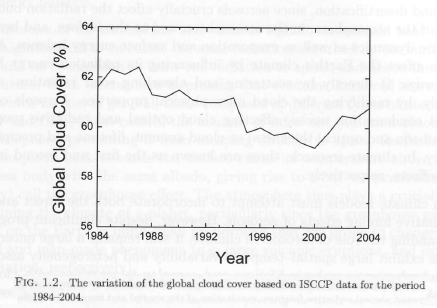
Many people have appreciated the massive absorption at the peak of the CO2 band (around 15 μm). But as we have seen in earlier parts of this series (and as shown in Figure 1), it is towards the “edges” of the band where the largest changes take place as CO2 concentration increases.
Remember as well that total transmittance is not really a complete picture of radiative transfer in the atmosphere. The atmosphere also emits radiation, and so the temperature profile of the atmosphere is just as important for seeing the whole picture.
Other articles in the series:
Part One – a bit of a re-introduction to the subject.
Part Two – introducing a simple model, with molecules pH2O and pCO2 to demonstrate some basic effects in the atmosphere. This part – absorption only.
Part Three – the simple model extended to emission and absorption, showing what a difference an emitting atmosphere makes. Also very easy to see that the “IPCC logarithmic graph” is not at odds with the Beer-Lambert law.
Part Four – the effect of changing lapse rates (atmospheric temperature profile) and of overlapping the pH2O and pCO2 bands. Why surface radiation is not a mirror image of top of atmosphere radiation.
Part Five – a bit of a wrap up so far as well as an explanation of how the stratospheric temperature profile can affect “saturation”
Part Six – The Equations – the equations of radiative transfer including the plane parallel assumption and it’s nothing to do with blackbodies
Part Seven – changing the shape of the pCO2 band to see how it affects “saturation” – the wings of the band pick up the slack, in a manner of speaking
Part Eight – interesting actual absorption values of CO2 in the atmosphere from Grant Petty’s book
Part Nine – calculations of CO2 transmittance vs wavelength in the atmosphere using the 300,000 absorption lines from the HITRAN database
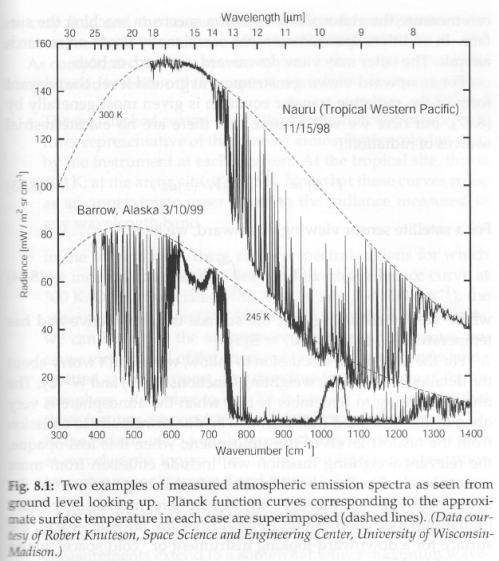
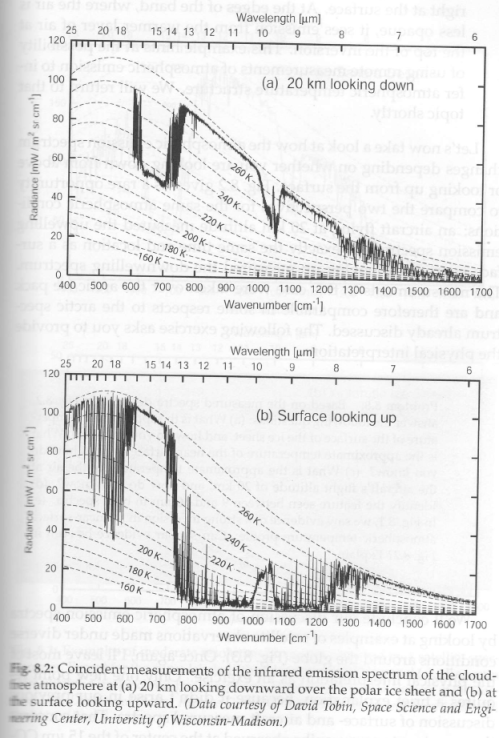
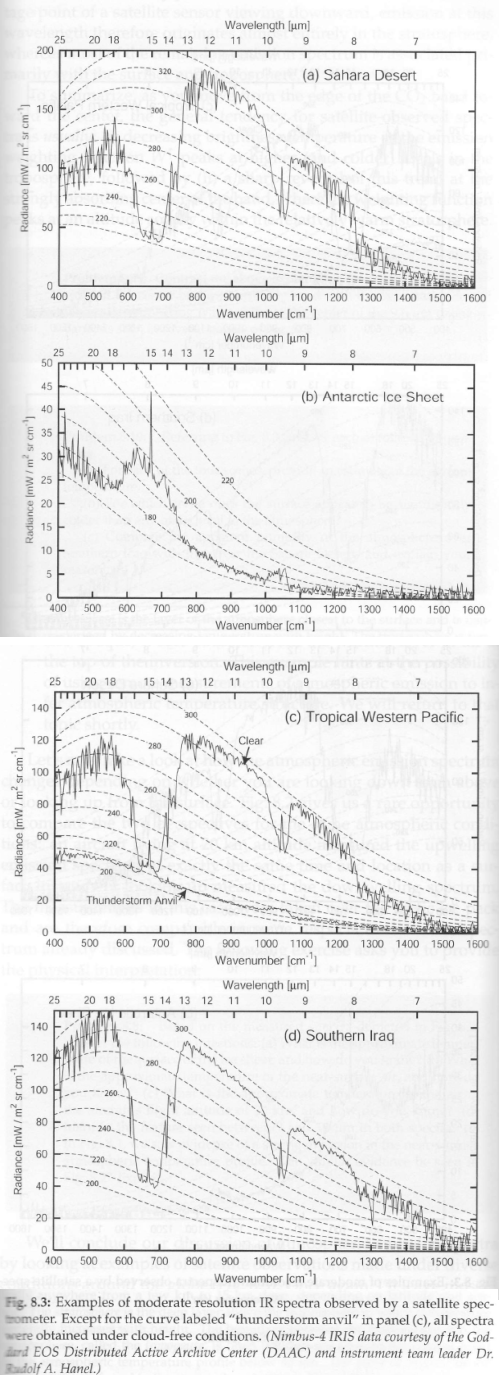
Part Ten – spectral measurements of radiation from the surface looking up, and from 20km up looking down, in a variety of locations, along with explanations of the characteristics
Part Eleven – Heating Rates – the heating and cooling effect of different “greenhouse” gases at different heights in the atmosphere
Part Twelve – The Curve of Growth – how absorptance increases as path length (or mass of molecules in the path) increases, and how much effect is from the “far wings” of the individual CO2 lines compared with the weaker CO2 lines
And Also –
Theory and Experiment – Atmospheric Radiation – real values of total flux and spectra compared with the theory.
References
The HITRAN 2008 molecular spectroscopic database, by L.S. Rothman et al, Journal of Quantitative Spectroscopy & Radiative Transfer (2009)
Notes
Note 1 – As you can see in Part Nine, the line shape differs as the pressure reduces.
Note 2 – Technically speaking, for “very small paths” we are really considering the case where optical thickness, τ <<1 (very much less than 1). And for “very large paths” we are considering the case where optical thickness, τ>>1 (very much greater than 1).
Note 3 – For a line strength = W, if we want to calculate absorptance, a = 1 – t, where t= transmittance, across any given band, Δv, the calculation is very simple:
a = 1-t = W / Δv








The Mystery of Tau – Miskolczi – Part Three – Kinetic Energy
Posted in Basic Science, Commentary, Debunking Flawed "Science" on April 26, 2011| 123 Comments »
In Part One we looked at the calculation of total atmospheric optical thickness.
In Part Two we looked at the claim that the surface and atmosphere exchanged exactly equal amounts of energy by radiation. A thermodynamics revolution if it is true, as the atmosphere is slightly colder than the surface. This claim is not necessary to calculate optical thickness but is a foundation for Miskolczi’s theory about why optical thickness should be constant.
In this article we will look at another part of Miskolczi’s foundational theory from his 2007 paper, Greenhouse Effect in Semi-Transparent Planetary Atmospheres, Quarterly Journal of the Hungarian Meteorological Service.
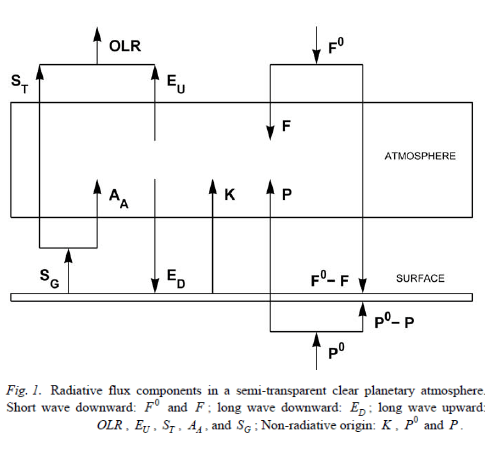
For reference of the terms he uses, the diagram from the 2007 paper:
From Miskolczi (2007)
Figure 1
On pages 6-7, we find this claim:
Many people have puzzled over the introduction of the virial theorem (note 1), which relates total kinetic energy of the atmosphere to total potential energy of the atmosphere. Generally, there is a relationship between potential energy and kinetic energy of an atmosphere so I don’t propose to question it, we will accept it as a given.
By the way, on the diagram SU = SG, i.e. SU = upwards radiation from the surface. And EU = upwards radiation from the atmosphere (cooling to space).
Kinetic Energy of a Gas
For people who don’t like seeing equations, skip to the statement in bold at the end of this section.
Here is the equation of an ideal gas:
pV = nkT (also written as pV = NRT) [1]
where p = pressure, V = volume, n = number of molecules, k = 1.38 x 10-23 J/K = Boltzmann’s constant, T = temperature in K
This equation was worked out via experimental results a long time ago. Our atmosphere is a very close approximation to an ideal gas.
If we now take a thought experiment of some molecules “bouncing around” inside a container we can derive an equation for the pressure on a wall in terms of the velocities of the molecules:
pV = Nm<vx²> [2]
where m = mass of a molecule, <vx²> = average of vx², where vx = velocity in the x direction
Combining [1] and [2] we get:
kT = m<vx²>, or
m<vx²>/2 = kT/2 [3]
The same considerations apply to the y and z direction, so
m<v²>/2 = 3KT/2 [4]
This equation tells us the temperature of a gas is equal to the average kinetic energy of molecules in that gas divided by a constant.
For beginners, the kinetic energy of a body is given by mv²/2 = mass x velocity squared divided by two.
So temperature of a gas is a direct measure of the kinetic energy.
The Kinetic Error
So where on earth does this identity come from?
EU is the upwards radiation from the atmosphere to space.
To calculate this value, you need to solve the radiative transfer equations, shown in Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Six – The Equations. These equations have no “analytic” solution but are readily solvable using numerical methods.
However, there is no doubt at all about this:
EU ≠ 3kTA/2 [5]
where TA = temperature of the atmosphere
that is, EU ≠ kinetic energy of the atmosphere
As an example of the form we might expect, if we had a very opaque atmosphere (in longwave), then EU = σTA4 (the Stefan-Boltzmann equation for thermal radiation). As the emissivity of the atmosphere reduces then the equation won’t stay exactly proportional to the 4th power of temperature. But it can never be linearly proportional to temperature.
A Mystery Equation
Many people have puzzled over the equations in Miskolczi’s 2007 paper.
On p6:
Note that I have added a prefix to the equation numbers to identify they as Miskolczi’s. As previously commented, the P term (geothermal energy) is so small that it is not worth including. We will set it to zero and eliminate it, to make it a little easier to see the problems. Anyone wondering if this can be done – just set F’ = F0 + P0 and replace F0 with F’ in the following equations.
So:
EU = F + K (M5a)
SU − F0 = ED − EU (M6a)
Please review figure 1 for explanation of the terms.
If we accept the premise that AA = ED then these equations are correct (the premise is not correct, as shown in Part Two).
M5a is simple to see. Taking the incorrect premise that surface radiation absorbed in the atmosphere is completely re-emitted to the surface: therefore, the upward radiation from the atmosphere, EU must be supplied by the only other terms shown in the diagram – convective energy plus solar radiation absorbed by the atmosphere.
What about equation M6a? Physically, what is the downward energy emitted by the atmosphere minus the upward energy emitted by the atmosphere? What is the surface upward radiation minus the total solar radiation?
Well, doesn’t matter if we can’t figure out what these terms might mean. Instead we will just do some maths, using the fact that the surface energy must balance and the atmospheric energy must balance.
First let’s write down the atmospheric energy balance:
AA + K + F = EU + ED [10] – I’m jumping the numbering to my equation 10 to avoid referencing confusion
This just says that Surface radiation absorbed in the atmosphere + convection from the surface to the atmosphere + absorbed solar radiation in the atmosphere = energy radiated by the atmosphere from the top and bottom.
Given the (incorrect) premise that AA = ED, we can rewrite equation 10:
K + F = EU [10a]
We can see that this matches M5a, which is correct, as already stated.
So first, let’s write down the surface energy balance:
F0 – F + ED = SU + K [11]
This just says that Solar radiation absorbed at the surface + downward atmospheric radiation = surface upward radiation + convection from the surface to the atmosphere.
Please review Figure 1 to confirm this equation.
Now let’s rewrite equation 11:
SU – F0 = ED – F – K [11a]
and inserting eq 10a, we get:
SU – F0 = ED -EU [11b]
Which agrees with M6a.
And as an aside only for people who have spent too long staring at these equations – re-arrange the terms in 11b:
Su – Ed = F0 – Eu; The left side is surface radiation – absorbed surface radiation in the atmosphere (accepting the flawed premise) = transmitted radiation. The right side is total absorbed solar radiation – upward emitted atmospheric radiation. As solar radiation is balanced by OLR, the right side is OLR – upward emitted atmospheric radiation = transmitted radiation.
Now, let’s see the mystery step :
This equation M7 makes no sense. Note that again I have removed the tiny P0 term.
Let’s take [11b], already demonstrated (by accepting the premise) and add (ED -EU) to both sides:
SU – F0 + (ED – EU) = ED – EU+ (ED -EU) = 2(ED -EU) [12]
So now the left side of eq 12 matches the left side of M7.
The M7 equation can only be correct if the right side of eq 12 matches the right side of M7:
2(ED -EU) = F0 [13] – to be confirmed or denied
In concept, this claim is that downward radiation from the atmosphere minus upward radiation from the atmosphere = half the total planetary absorbed solar radiation.
I can’t see where this has been demonstrated.
It is not apparent from energy balance considerations – we wrote down those two equations in [10] and [11].
We can say that energy into the climate system = energy out, therefore:
F0 = OLR = EU + ST [14] (atmospheric upward radiation plus transmitted radiation through the atmosphere)
Which doesn’t move us any closer to the demonstration we are looking for.
Perhaps someone from the large fan club can prove equation 7. So many people have embraced Miskolczi’s conclusion that there must be a lot of people who understand this step.
Conclusion
I’m confused about equation 7 of Miskolczi.
Running with the odds, I expect that no one will be able to prove it and instead I will be encouraged to take it on faith. However, I’m prepared to accept that someone might be able to prove that it is true (with the caveat about accepting the premise already discussed).
The more important point is equating the kinetic energy of the atmosphere with the upward atmospheric radiation.
It’s a revolutionary claim.
But as it comes with no evidence or derivation and would overturn lots of thermodynamics the obvious conclusion is that it is not true.
To demonstrate it is true takes more than a claim. Currently, it just looks like confusion on the part of the author.
Perhaps the author should write a whole paper devoted to explaining how the upwards atmospheric flux can be equated with the kinetic energy – along with dealing with the inevitable consequences for current thermodynamics.
Update 31st May: The author confirmed in the ensuing discussion that equation 7 was not developed from theoretical considerations.
Other Articles in the Series:
The Mystery of Tau – Miskolczi – introduction to some of the issues around the calculation of optical thickness of the atmosphere, by Miskolczi, from his 2010 paper in E&E
Part Two – Kirchhoff – why Kirchhoff’s law is wrongly invoked, as the author himself later acknowledged, from his 2007 paper
Part Four – a minor digression into another error that seems to have crept into the Aa=Ed relationship
Part Five – Equation Soufflé – explaining why the “theory” in the 2007 paper is a complete dog’s breakfast
Part Six – Minor GHG’s – a less important aspect, but demonstrating the change in optical thickness due to the neglected gases N2O, CH4, CFC11 and CFC12.
Further Reading:
New Theory Proves AGW Wrong! – a guide to the steady stream of new “disproofs” of the “greenhouse” effect or of AGW. And why you can usually only be a fan of – at most – one of these theories.
References
Greenhouse Effect in Semi-Transparent Planetary Atmospheres, Miskolczi, Quarterly Journal of the Hungarian Meteorological Service (2007)
Notes
Note 1 – A good paper on the virial theorem is on arXiv: The Virial Theorem and Planetary Atmospheres, Victor Toth (2010)
Read Full Post »