In his excellent book, A First Course in Atmospheric Radiation, Grant Petty introduces a number of spectral measurements of atmospheric radiation which are very illuminating.
In this article I am going to reproduce them, along with a lot of Petty’s comments and explanations – hard to improve on what he has to say. (See the book recommendation).
For people confused about how the atmosphere absorbs and emits radiation these might be helpful. For people spreading confusion about how the atmosphere absorbs and emits these will be hard to explain.
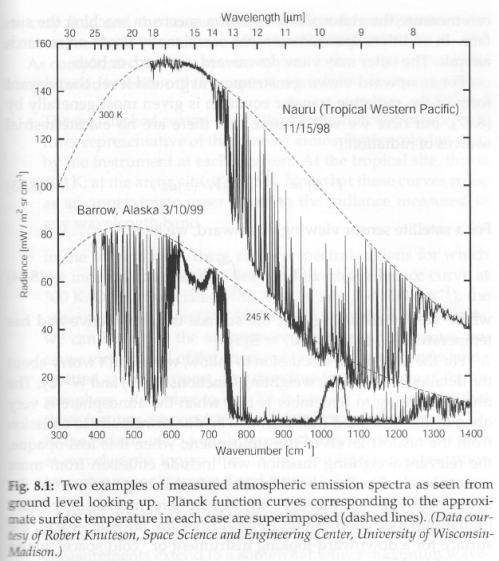
Figure 1 – The atmosphere above Nauru and Alaska under cloud-free conditions
A few basics first of all – if the atmosphere didn’t emit radiation then the upward looking spectrometer would measure a flat line at zero, as the (extremely low) emission from space at 3K would not even register on the spectrometer. (Note 1).
So where we see very low radiance measurements this is because the atmosphere is transparent at these wavelengths/wavenumbers. (Note 2).
Nauru is in the tropical western Pacific where atmospheric temperatures are warm and humidity is high. Barrow is in the Arctic, and so temperatures in winter are very cold and the atmosphere contains only small amounts of water vapor.
1. The two dashed curves are the Planck function (note 3) at the warmest atmospheric emission seen by the spectrometer at each location.
2. In the tropical example there are two spectral regions where the measured radiance is very close to the 300K reference curve: >14 μm (<730 cm-1 ) and <8 μm (>1270 cm-1). Therefore, the atmosphere must be quite opaque in these bands because the radiation is being emitted from the warmest – and, therefore, lowest – levels of the atmosphere. The >14 μm region is the region of strong absorption of CO2 and, above 15 μm, of water vapor, and the <8 μm region is the water vapor region.
3. In the arctic example we can also see these two water vapor regions, but it’s clear that these are somewhat weaker, with variable radiance. This is because the water vapor concentration is much lower in the colder arctic.
4. In the tropical example, in the region from 8 – 13 μm, the radiances are well below the 300K reference curve and in some cases even a little below the 245K reference curve – this is because the atmosphere is quite transparent in this region.
5. In the arctic example this is much clearer. The 8 – 13 μm region (with the exception of 9.6 μm) is almost at zero radiance because, with much lower water vapor concentration, the spectrometer is almost measuring the radiance of space.
6. The 9.6 μm region in both examples is due to ozone emission. It’s not as obvious in the tropical example because water vapor emission extends across this band.
7. The 15 μm band in the arctic example has an interesting feature. At the center of the band the radiance is a little lower than at the edges of the band. Why is this? The center of the band is the most opaque so it should be measuring the temperature of the lowest levels of the atmosphere, almost at the surface. And the edges of the band – a little less opaque – should be measuring the temperature a little higher up. The reason is that in the arctic in wintertime it is very common to see a temperature inversion, where the surface is colder than the atmosphere a few hundred meters above.
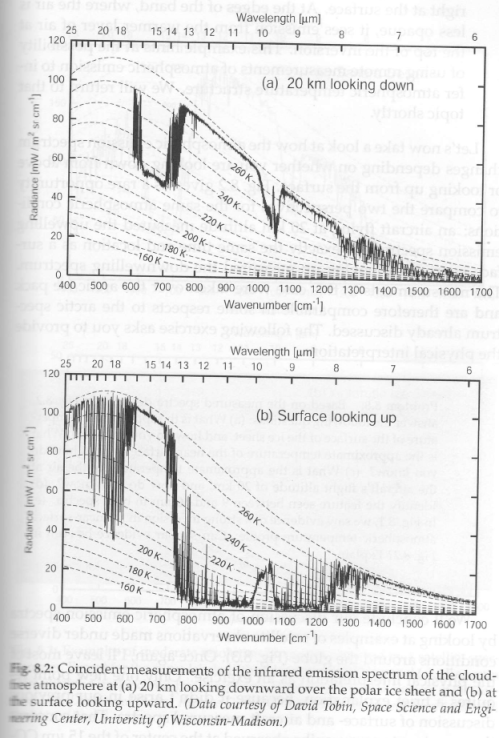
Now let’s review a very interesting pair of measurements. One from 20km looking down – with the simultaneous surface measurement looking up:
Figure 2 – Upwards and downwards measurements at the polar ice sheet
Petty now asks a few questions – in the manner of all good textbook writers. I attempt to answer these questions and if I embarrass myself by getting them wrong, please speak up. I know someone will..
a) what is the approximate temperature of the surface of the ice sheet and how do you know?
b) what is the approximate temperature of the near-surface air, and how do you know?
c) what is the approximate temperature of the air at the aircraft’s flight altitude of 20km, and how do you know?
d) identify the feature seen between 9 – 10 μm in both spectra
e) in fig 1 (fig 8.1) we saw evidence of a strong inversion in the near-surface atmospheric temperature profile. Can similar evidence be seen here?
If you want to check your understanding – try and answer the above questions before reading on. Anyway, you can’t rely on my answers..
My answers, for review:
a) the surface temperature is approx. 268K. The atmosphere is most transparent at 900 cm-1 & 1150 cm-1, so, looking downward at these wavelengths we should see the surface emission. And ice, like water, emits at very close to a blackbody at these wavenumbers.
b) the near surface temperature is approx. 268K. Looking upwards where the atmosphere is most opaque (15 μm) we should see the temperature of the atmosphere closest to the surface. At 15 μm we see a temperature of 268K.
c) the approx. temperature of the air at 20km is 225K. Looking downward from the aircraft where the atmosphere is most opaque (15 μm) we should see the temperature of the atmosphere closest to the measurement device. At 15 μm we see a temperature of 225K (corrected Jan 28th, 2014 thanks to Mike B). Note that we don’t want to rely on the brightness temperature seen at the strongest water vapor absorption (<8 μm) because the water vapor concentration is low when the atmosphere is very cold.
d) the feature seen between 9 – 10 μm in both spectra is the ozone absorption. In the downward looking spectrum from the aircraft we see a colder brightness temperature than the rest of the 8 – 13 μm band – because the rest of the band is viewing the surface, while the ozone absorption centered at 9.6 μm sees the atmosphere much closer to the aircraft. Looking upwards from the surface, the rest of that band sees (very cold) space, while the ozone band reflects the temperature of the lower stratosphere, around 235K.
e) No.
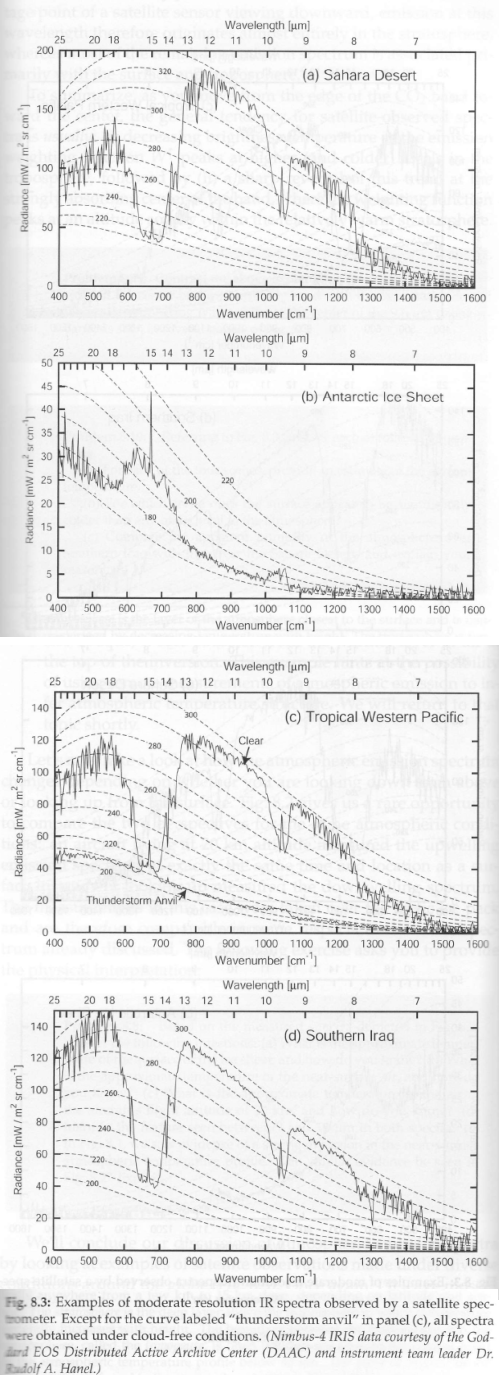
Lastly, four satellite spectra (upwards radiance) from different locations:
Figure 3 – Four satellite measurements from different locations
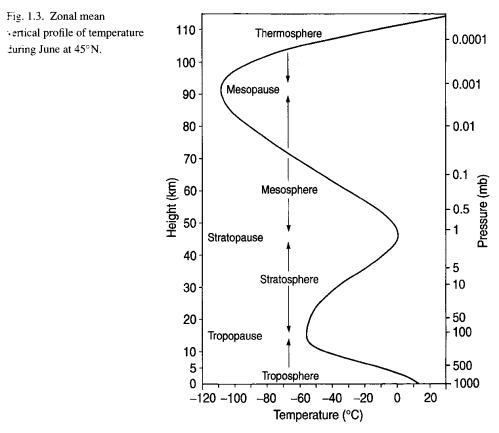
Notice the 15 μm radiance compared with the surrounding band. Remember that the stratosphere warms from the tropopause to the stratopause:
Figure 4 – Temperature profile of the atmosphere
The reason the brightness temperature (radiance) for the first graph – the Sahara – is at the low temperature of 215K between 14-16 μm is because the satellite is measuring the temperature of the region around the tropopause. But at the very opaque 15 μm the satellite is measuring even closer to itself – higher up in the stratosphere, which is why the brightness temperature is around 230K.
Contrast that with the Antarctic (2nd graph in Fig 3). There we see that the ice sheet is colder than the stratosphere. This is why the 15 μm radiance is higher than the radiance at all the other wavelengths.
If you compare c) Tropical Western Pacific with d) Southern Iraq you can see the effect of water vapor. In the desert of Southern Iraq where the water vapor is low the radiance is measured from close to the surface – and therefore, is high. In the Western Pacific, where the water vapor concentration is much higher, the radiance is measured from closer to the satellite, i.e., higher up in the atmosphere, where the temperature is lower, and so is the radiance.
Comparing c) and d) for the 15 μm radiance you see that they are almost the same. Water vapor is overwhelmed by CO2 in this region and so water vapor concentration has no effect here.
Conclusion
Even though water vapor and CO2 are present in very low concentrations, they have a very strong radiative effect.
This is not something which is a subject of debate in spectroscopy or in atmospheric physics. The fact that many people find it difficult to understand how a gas present in 360ppm concentrations can have such a strong effect is of no scientific interest. This is because it isn’t a scientific argument.
The changing transparency/emissivity of the atmosphere at various wavelengths provides us with very valuable information about:
a) the concentration of water vapor
b) the temperatures at different heights in the atmosphere
Other articles:
Part One – a bit of a re-introduction to the subject.
Part Two – introducing a simple model, with molecules pH2O and pCO2 to demonstrate some basic effects in the atmosphere. This part – absorption only.
Part Three – the simple model extended to emission and absorption, showing what a difference an emitting atmosphere makes. Also very easy to see that the “IPCC logarithmic graph” is not at odds with the Beer-Lambert law.
Part Four – the effect of changing lapse rates (atmospheric temperature profile) and of overlapping the pH2O and pCO2 bands. Why surface radiation is not a mirror image of top of atmosphere radiation.
Part Five – a bit of a wrap up so far as well as an explanation of how the stratospheric temperature profile can affect “saturation”
Part Six – The Equations – the equations of radiative transfer including the plane parallel assumption and it’s nothing to do with blackbodies
Part Seven – changing the shape of the pCO2 band to see how it affects “saturation” – the wings of the band pick up the slack, in a manner of speaking
Part Eight – interesting actual absorption values of CO2 in the atmosphere from Grant Petty’s book
Part Nine – calculations of CO2 transmittance vs wavelength in the atmosphere using the 300,000 absorption lines from the HITRAN database
Part Ten – spectral measurements of radiation from the surface looking up, and from 20km up looking down, in a variety of locations, along with explanations of the characteristics
Part Eleven – Heating Rates – the heating and cooling effect of different “greenhouse” gases at different heights in the atmosphere
Part Twelve – The Curve of Growth – how absorptance increases as path length (or mass of molecules in the path) increases, and how much effect is from the “far wings” of the individual CO2 lines compared with the weaker CO2 lines
And Also –
Theory and Experiment – Atmospheric Radiation – real values of total flux and spectra compared with the theory.
Notes
Note 1: If it was daytime and the sun was directly overhead (not possible in Barrow, Alaska in March) then the sun’s radiance would add about 1 mW/m2.sr.cm-1 at 1000 cm-1, 1.7 mW/m2.sr.cm-1 at 1400 cm-1 (right hand edge of the graph), and 0.1 mW/m2.sr.cm-1 at 300 cm-1 (left edge of the graph).
Note 2: According to Kirchhoff’s law, if the atmosphere absorbs at any wavelength it also emits at that wavelength – and in equal strength. Absorptivity = Emissivity (but very very important, at the same wavelength, or range of wavelengths). See Planck, Stefan-Boltzmann, Kirchhoff and LTE.
This means that if the atmosphere is transparent at any given wavelength it doesn’t emit at that wavelength. And if it is opaque at any given wavelength it is a strong emitter.
If the absorptivity = 1 at any wavelength (or range of wavelengths) then the emissivity = 1 at that wavelength, meaning it emits like a blackbody at that wavelength.
Note 3: The Planck function is the formula for emission of thermal radiation from a blackbody – a perfect emitter and perfect absorber. There is plenty of unscientific confusion about blackbodies on the web. A blackbody is simply the maximum radiator for any given temperature. Nothing can radiate with a greater intensity at any wavelength than a blackbody and while no real surface is a perfect blackbody many surfaces come close. For example, the ocean has an emissivity of about 0.96. The atmosphere – at some wavelengths – emits very close to a blackbody, while at other wavelengths it is almost transparent and, therefore, the emissivity is close to zero.


SOD,
great graphics and citations as always.
Adding CO2 has the effect of raising the height of emissions to space in the CO2 band.
Would you agree that in the cases of Antarctica (figure B) and Tropical Thunderstorm Anvil (figure C), the emissions in the CO2 band are actually warmer than the lower emission surface ( ice sheet, very high cloud top ), and therefore -increase- total emission to space above what they would be with a lower level of CO2?
In these cases (and probably Greenland in winter also), additional CO2 acts to cool which is probably not included in the CO2 forcing estimates.
SoD,
Saying that nothing can emit more than a black body isn’t strictly true. You have to qualify that by saying thermal emission of radiation cannot be higher than a black body. There are lots of non-thermal sources of radiation like LED’s and fluorescent lights. In the atmosphere there is lightning and the aurora.
I’ve only read a few paragraphs so may be way premature, but it sounds (so far) like “opaque” and “transparent” are turned around.
OOPS! Scratch earlier comment. I missed the looking up and looking down stuff. Sorry
Well done.
Part of the problem is that most people do not have a good feel for how strong IR absorption is. The number which scales it for me is that the 1/e distance for the CO2 bending mode is about 10 m at sea level.
There are some neat puzzlers in the appearance of the CO2 Q branch btw
Thanks for the excellent summary. Cleared up many points in a very concise way for me that seemed a bit confused in textbooks and on other sites.
SoD wrote.. >>>Even though water vapor and CO2 are present in very low concentrations, they have a very strong radiative effect.<<<
SoD… This seems to make the argument that most of the heavy lifting in regards to the GHG effect from both CO2 and H2O are done at very low concentrations. Is that the case?
In regards to low concentrations, I know that the CO2 level is the range of 360 ppmv. If you know, what was the water vapor level in ppmv?
Also, how does the fact that H20 changes from vapor to liquid at the dew point which I believe occurs at a relatively low elevation, impact the measurements?
Thanks.. great site. I am learning a lot as I wade through all of this. Although I am an engineer,the science of climate science is new to me. Thinking about this helps us geriatrics stay alert.
sod,
I’d like to get your thoughts on the issue of CO2 *increasing* the emissions at the poles, as raised by ClimateWatcher in the first response.
I believe there is a net transport of energy from the equator to the poles, with CO2 then being one extra way to get rid of the energy by radiation into space. As seen in the graphs, there is not much water in the athmosphere, but can we see contributions from other greenhouse gases (methane, …) also? Do your calculations give an estimate for the magnitude of such an effect?
PeterF,
True. See this graph:
I should probably invert the difference graph to make it emission – absorption rather than absorption – emission.
The increase is because without CO2, the temperature at the poles would be a lot lower.
For sub-Arctic winter with a surface temperature of 257.2 K or -16 C, surface emission (100-1500 cm-1) is 234.2 W/m2. At 375 ppmv CO2 emission to space at 100 km is 196.6 W/m2. Remove CO2 and emission is 211.9 W/m2. So the total forcing is 37.6 W/m2 with CO2 contributing 16.3 or 43%. That percentage drops to about 25% in the summer when it’s warmer and the absolute humidity goes up.
Increasing CO2 cools the stratosphere because it increases emission with almost no increase in absorption. CO2 warms the surface everywhere.
Eli,
10m must be for broad band absorption/emission. At the 667 cm-1 peak, it’s about 25 cm.
Payne,
thanks. Are your arguments taken from a publication to which you could provide a link? Would be appricaiated.
The data on heat transfer is from Grant Petty, A First Course in Atmospheric Radiation ( http://www.sundogpublishing.com/AtmosRad/index.html ).
The other data was calculated using MODTRAN accessible on the web at: http://geoflop.uchicago.edu/forecast/docs/Projects/modtran.orig.html thanks to David Archer.
thanks. had ordered the book already and will take a look and dig further.
A black body is an idealized physical body that absorbs all incident electromagnetic radiation.
The above from Wiki. If you agree than CO2 is not a black body as it does not absorb all incident radiation.
What is the emissivity of CO2?
mkelly:
Everyone agrees that CO2 is not a blackbody.
The emissivity of CO2 depends on:
a) the pathlength
b) the pressure & temperature
And consequently, the direction of the path – so a 10km path horizontally at the earth’s surface is different from a 10km path vertically from the surface up to the top of the troposphere.
The temperature is important for two reasons:
1. it affects the line width (as does the pressure)
2. we need to know the emission spectrum that the emissivity is relevant for
Point 2 is very important as the emissivity of CO2 emitting at a temperature of 1000K is very different from CO2 emitting at 200K. This is because the Planck curve moves to shorter wavelengths in the 1000K case.
And after you tell me the conditions I will have to calculate it.
If it includes a significant path in the stratosphere then I will need to complete the work I am investigating at the moment – to include the Voigt profile to take account of the Doppler broadening at lower pressures. (See Part Nine for more on this point).
Or maybe someone has provided this data for the particular path you want to consider. That would save working it out from scratch.
mkelly:
And if you are interested in a very rough number under typical atmospheric conditions you can make an estimate yourself from the data provided in this article.
Take figure 1 for the Barrow, Alaska case and estimate the area under the curve between the 600 – 750 cm-1 – call this value A. Then estimate the total area under the 245K Planck curve and call this B.
A/B = emissivity estimate for these colder temperatures.
It won’t be 100% correct because the atmosphere absorbs and emits all the way down.
However, the emissivity of CO2 in this wavenumber region (600 – 750 cm-1) is very close to 1 -as you can see from the measurements.
And so the contribution from higher up is of no importance (emissivity can never exceed 1).
It is difficult using this method to see the effect of CO2 around the edges of the band. What is CO2 and what is water vapor? And this is where absorption and re-emission make the subject more complex.
However, assuming you have arrived at this question because of a certain blog article, the value you calculate from experimental evidence will demonstrate that very low emissivities for CO2 are not correct.
However, assuming you have arrived at this question because of a certain blog article, the value you calculate from experimental evidence will demonstrate that very low emissivities for CO2 are not correct.
I ask because my heat transfer book (Holman) shows emissivity for CO2 to be very low. i.e. the Hottel charts.
It’s only low for a short path length. I’ve had this discussion before. If the path length is 1 m and the partial pressure is 0.00038 atmospheres then the Hottel emissivity curve to use is the one for 0.00038 atm m. If the path length is 10 km, then the mass path length is 3.8 atm m, which is well above the range of the Hottel charts I’ve seen, but puts the emissivity value for CO2 at ~0.2.
mkelly,
Here is an example calculation. The linked graph is the emission from a cell with length 7998 m at 1 atm total pressure with a CO2 partial pressure of 0.00034 atm and a temperature of 308 K. The path length come from the length of a vertical column of the atmosphere if it were all at a pressure of 1 atm:
The radiance is 34 W/m2/sr. Multiply by π to get the total flux = 106.8 W/m2. Divide by the blackbody flux for 308 K ( 510.3 W/m2 ) to get the integrated emissivity of 0.209. Or something like that. I may have a factor of two and/or π error here but I’m not going to pay SpectralCalc another $25 to find out.
mkelly:
What exactly does your book state about emissivity of gases?
And does it come with some curves for emissivity of CO2?
I have seen Leckner’s curves (1972). Leckner also covered the flame and combustion field.
Leckner has a set of curves on the graph. Each curve has different emissivity for different “bar.cm”. This is the partial pressure of CO2 x path length.
I’ll see if I can track it down.
Can you scan your graph with surrounding text and email it to me? (scienceofdoom, you know what goes here, gmail.com)
mkelly:
Here is the estimate of atmospheric CO2 emissivity from the real data (measurements) shown in the article:
It is an under-estimate as I will explain.
First, I extended the graph from 0 cm-1 to 2000 cm-1 and reviewed a calculated 245K Planck curve to extend the lines by hand.
Second, I took the area under the curve for the unambiguous CO2 portion, 600-740 cm-1. You can see that it is probably an under-estimate. I converted it from mW to W and just did an area calculation. Area (CO2) = 8.7 W/m2.sr.
Third, I calculated the area under the complete 245 K Planck curve by using the Stefan Boltzman equation, and divided by pi to get the same comparison as with the CO2 result in W/m2.sr. (rather than W/m2)
Total area = 65 W/m2.sr.
But if you don’t understand this calculation – the reason for dividing by pi – it might be easier for you to simply estimate the area graphically. It is easy to see this number 65 is approximately correct. Average height of the graph, something like 30/1000. Width of the graph = 2000. Multiply for area = 60.
Area (CO2)/ Total area = 8.7/65 = 0.13
Remember this is an under-estimate – CO2 emission/absorption extends lower than 600 cm-1 and higher than 740 cm-1. Emissivity in the center of the band is under-estimated here because the temperature close to the ground is probably lower than the temperature 245K.
The point of the exercise is to get an approximate value.
If this is much higher than some other calculated value then either another gas has very high emissivity around 600 – 740 cm-1 or the other calculation is wrong.
I hope this is the right place to ask this question. Can someone (SoD or others) point me at good measurement data showing the earth emitting a black body radiation curve? I see a lot of theoretical estimates, but what would be nice would be a real spectrum obtained by pointing at the ground (perhaps some nice dark podzols in the Russian Steppe on a sunny day)? Sorry if its on the site and I’ve missed it! Thanks in advance.
Kevin Parker,
No one would produce a paper with the radiance vs wavelength of some particular earth surface at some particular temperature because it wouldn’t be of interest. Well, it would be of interest if someone found that the thermal emission of radiation didn’t follow established theory. Back in the late 1800s lots of papers were published on the spectral intensity of radiation of various surfaces at various temperatures. It was groundbreaking stuff then.
There are lots of papers from the last few decades where researchers do unexciting work on measuring emissivity of various materials vs wavelength.
For example, in Emissivity of the Ocean you can see some results of measured emissivity of the ocean vs wavelength.
Emissivity is simply the comparison of the actual measured radiance vs the expected “blackbody” radiance.
HI SoD, thanks for this. The reason I was asking was that there is often a cry of ‘show me real data, not models, that show global warming’. To me the satellite data showing the CO2 absorption peak are very telling, as is the spectrum of the back radiation. What I was looking for was a nice smooth spectrum of radiation coming up from the earth so one could say ‘look this is what the earth emits, and this is what happens to that spectrum as it goes up through the atmosphere’.
If we are talking to a lay/young audience (which I often do), then saying ‘this is the earth’s emission pattern according to the Stefan Boltzman equation’ sounds like jargon. I know its important and necessary jargon, its just that illustrating it by pictures has much greater impact. I’ll have a look around the references on the emissivity page. Thanks, KP.
I see your point.
If I had a lazy million sitting around then it would be fun to get an FT-IR on a flight taking downward looking spectra every 1km all the way up to the stratosphere (or as high as the plane could go), then point it up to the sky and take the upwards measurements every km on the way down.
Might as well get two spectrometers and make simultaneous measurements. Dollars to doughnuts says something like that has been done already. It’s just a question of finding the data. I say ‘just’ as if that would be trivial, but it’s probably not. No one would do it now because the results would simply confirm what we already know. If you have an emissivity close to one, it’s very unlikely that the emission spectrum could be very different from a Planck curve. I believe it would violate the Second Law for the emission at any wavelength to exceed the Planck value for a black body.
Hi guys back again. In my searches I found a diagram on this website
http://www.geol.umd.edu/sgc/lectures/climatepart2.html
At the top is a graphic ‘Radiation transmitted by the atmosphere’ On the right is a blue spectrum labelled ‘upgoing Thermal radiation 15-30% transmitted’ Just to clarify, is that a spectrum taken near ground level or as I understand, something taken from higher up, perhaps a satellite. It does look different to some of the other satellite spectra though.
Thanks again for your help – K
The upgoing spectrum has to have been calculated. That’s surface radiation transmitted directly to space. A real spectrum would have CO2 and water vapor emission from altitudes well above the surface as well as the transmitted surface radiation. Here’s a calculated TOA spectrum. A real spectrum won’t go to as low a frequency because nobody has a spectrometer that can be put in a plane or satellite that has any sensitivity at the low frequency (long wavelength) end of the spectrum yet. Compare that to a measured spectrum. Note that the ground looking up spectrum cuts off at 400 cm-1 (25 μm) while the spectrum from the plane cuts off at 600 cm-1 (16.7 μm). It is possible to get down to 30 cm-1 for a bench top analytical IR absorption spectrophotometer, but those use high energy sources. It’s not clear that the detector would have sufficient sensitivity to measure atmospheric emission frequencies that low.
Hi Scienceofdoom!
In the first part of this subject you introduce a graph with: “The typical radiation from the earth’s surface at 15°C measured just above it ..” as coming from a black body. At this temperature it has a peak of radiative intensity at 10 microns, which should compare to 1000 wavelengths/cm.
In this part you show spectral measurements from Petty’s book. But what has happened with the Planck curves? If I compare with the Shara Desert above, I find that the Planck curve which represents 288K, now has a peak at about 18 microns. I thought these curves should black body comparisions.
What’s the explaination?
Both curves are correct and represent the same distribution of energy. The difference is that one curve gives the distribution in terms of wavenumber and the other in terms of wavelength. As the wavenumber is the inverse of wavelength the peak is seen at a different point.
The effect is seen also on the uneven wavelength scale in the graphs of this post.
Thanks’ for your reply Pekka, but I still don’t understand. I can see that those two graph examples have have different directions on the horizontal scale (according to the microns (wavelength) and the wavenumber), but the “Sahara graph” is represented with both scales (up and down in different directions). I thought that the peak of radiative intensity still should be at the same value (choosing one scale) relentless of the direction on the horizontal axis. If I choose the micron scale, the peak is represented with two different values for a temperature of 288K (15C). 10 microns (in the first post of this subject) AND 18 microns (in this tenth post). They can’t both be correct, can they? Or what? What is it I don’t see? Could you try once more please?
Trond,
Look at the first graph of this thread. There you notice that the wavelengths 17 and 18 um are closer to each other than wavelengths 14 and 15 um. The amount of energy radiated between two wavelengths is proportional to the product of the value of the curve and the distance between those wavelengths. Because the distance is larger at smaller wavelengths the peak of the wavelength distribution is shifted towards smaller wavelengths from the the top position of the curve.
Similar things happen with all distributions when the variable is changed to another which is not just a multiple of the other one, but has a non-linear relationship.
These two curves have both been calculated in Excel from the Planck law. The upper gives the distribution in wavelength, the lower in wavenumber (the unit for the function refers to frequency, but that’s just a multiplicative factor that does not change the shape of the curve). The difference is only a change of variables.
Trond A,
You are correct, there appears to be something wrong with these graphs. I will look into it and then make further comment.
Pekka,
I think the curves have the right shape related to each other. And the scales up and down given in wavelenght and wavenumer seem to fit each other. But the scales does not seem to fit the curves. A black body curve for a temperature of 288 Kelvin has it’s peak very near to 10 microns (or 1000 wavelengths/cm). That does not match what you see here taken from Grant Pettys book.
Trond,
That the curves do actually fit together is the point I have tried to explain. I haven’t used any equations as I that might not be useful. If you could gain understanding from the equations you would probably already understand the point.
I see now that it is all summed up in your first sentence: “Both curves are correct and represent the same distribution of energy.”.
Trond,
Here are the two graphs of spectral intensity, one plotted vs wavelength and one plotted vs wavenumber:
These were created from the fundamental equations.
Pekka’s explanation is correct.
The way to think about it is, what is spectral intensity?
It is how much energy is available between each 1 μm (wavelength);
At least that is the case for the first graph.
But in the second graph it is how much energy is available between each 1 cm-1 (wavenumber).
Between 4-5 μm there are 500 cm-1 (10000/4 – 10000/5)
Between 29-30 μm there are 11.5 cm-1 (10000/29 – 10000/30)
If there is a strongly changing amount of wavenumbers for each wavelength then the peak of one function will be different from the peak of another function.
Does this make sense?
Yes! I think I got it now. Very instructive. Thanks very much to both you and Pekka. The second coordinate is indeed not just a “one dimensional value”. I’ve learned a lot.
In your answer (c) I think you have made a slip. After saying that the temperature is 225K you subsequently say “ At 15 μm we see a temperature of 268K”. I think that should be 225K
That does appear to be a mistake. But because there is a peak at the center of the band from stratospheric emission, the temperature at 15 μm appears to be 230K.
MikeB,
Thanks, I fixed the article.