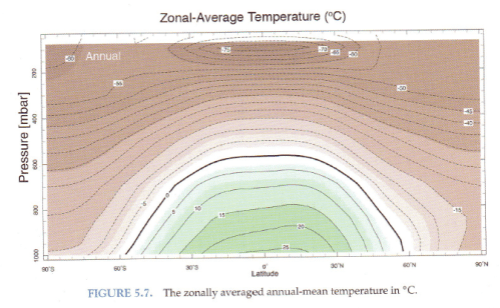
Here is the annual mean temperature as a function of pressure (=height) and latitude:
Figure 1 – Click for a larger image
We see that the equator is warmer than the poles and the surface is warmer than the upper troposphere (“troposphere” = lower atmosphere). No surprises.
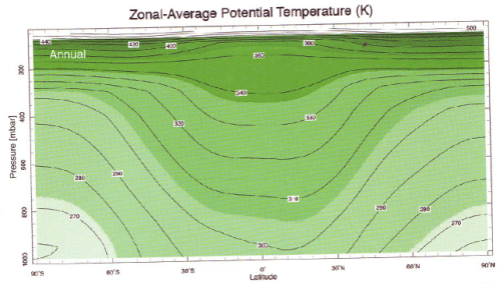
Here is “potential temperature”, whatever that is..
Figure 2 – Click for a larger image
We see that – whatever “potential temperature” is – the equator is warmer than the poles, but this version of temperature increases with height.
Why does temperature decrease with height? What is potential temperature? And why does it increase with height?
The Lapse Rate
Atmospheric pressure decreases with height. This is because as you go higher up there is less air above you, and therefore less downward force due to the weight of this air.
Because pressure decreases – and because air is a compressible fluid – air that rises expands (and air that sinks contracts).
Air that expands does “work” against its surroundings and because of the first law of thermodynamics (conservation of energy) this work needs to be paid for. So internal energy is consumed in expanding the parcel of air outwards against the atmosphere. And a reduction in internal energy means a reduction in temperature.
- Air that rises expands
- Expanding air cools
A little bit more technically.. adiabatic expansion is what we are talking about. An adiabatic process is one where no heat is exchanged with the surroundings. This is a reasonable approximation for typical rising air. It is reasonable because conduction is an extremely slow process (= negligible) in the atmosphere and radiative heat transfer is quite slow.
So if heat can’t be exchanged between a “parcel of air” and its surroundings it is relatively simple to calculate how the temperature changes. An example which contains way too much detail (because it is debunking a “debunking”) at Paradigm Shifts in Convection and Water Vapor?
The essence of the calculation is to equate internal energy changes with work done on the environment.
Textbooks usually start off with the simplest version, the dry adiabatic lapse rate, or DALR. (The “lapse rate” is the change in temperature with height of a parcel of air).
The DALR is for air without any water vapor. Now water vapor is very influential in our climate. The reason for neglecting it and starting off with this simplification is:
- the calculation is easy and everyone (almost) can understand it
- it represents one extreme of the atmosphere (polar climates and upper troposphere)
The result from this simplification:
Change in temperature with height = -g/cp ≈ -10 °C/km, where g = acceleration due to gravity = 9.8 m/s² and cp = heat capacity of air at constant pressure ≈ 1 J/kg.K
So for every km we displace air upwards it cools by about 10°C – so long as we displace it reasonably quickly. Well, this is true if it is dry.
A note on conventions – dry parcels of air moved upwards cool by 10°C per km, but the lapse rate is usually written as a positive number. So a cooling of 10 °C/km = -10 °C/km, but by convention, equals a “lapse rate” of +10 °C/km. This makes it very confusing when people say things like “the environmental lapse rate must be less than the adiabatic lapse rate“. Are we talking about the number with the minus sign in front? Or not?
It’s not easy to think about negative numbers being less than other negative numbers when the “less than” test is applied after they have been made into positive numbers. Not for me anyway. I have to write it down each time.
The Saturated Lapse Rate
If a parcel of air contains water vapor and it cools sufficiently then the water vapor condenses. This releases latent heat.
As a result, moist rising air cools slower than dry rising air
So the saturated adiabatic lapse rate is “less than” the dry adiabatic lapse rate.
E.g. the change in temperature with height of a dry parcel of air ≈ -10 °C/km, while the change in temperature with height of a moist parcel of air in the tropics near the surface ≈ -4 °C/km.
Conventionally we say that the saturated adiabatic lapse rate is less than the dry adiabatic lapse rate. Because we write them as positive numbers.
Now note the caveats around the value for the moist parcel of air rising. I said “..in the tropics near the surface..”, but for the DALR there are no caveats. That’s because once we consider moisture we have to consider how much water vapor and the amount varies hugely depending on temperature (and also on other factors – see Clouds and Water Vapor – Part Three).
The maths is somewhat harder for the saturated adiabatic lapse rate but it’s not conceptually more difficult, there is just an addition of energy (from condensing water vapor) to offset the work done.
Potential Temperature
Potential temperature is usually written with the Greek letter θ.
θ = T.(p0/p)k
where T = (real) temperature, p = pressure, p0 = reference pressure (usually at 1000 mbar) and k = R/cp = 2/7 for our atmosphere (more on this in a later article)
With a bit of tedious maths we can prove that θ stays constant under adiabatic conditions (for dry air).
Let’s look at what that means.
Suppose the surface (1000 mbar) temperature = 288 K (15°C) so also θ = 288K.
Now the air is moved (adiabatically) to 800 mbar, so T = 270 K. That’s what you expect – temperature falls with height. And no change to potential temperature, so θ = 288 K.
Now we move the air to 600 mbar, and T = 249 K. More reduction of temperature. And still θ = 288 K.
So is this a useful parameter – move the air (adiabatically) and the potential temperature stays the same?
The parameter is mathematically sound, but whether it is useful remains to be seen. As an artificial construct no doubt many people will be shaking their heads..
Stability and Potential Temperature Profile
In Density, Stability and Motion in Fluids we saw that for a fluid to be stable, lighter fluid must be above heavier fluid. No surprise to anyone.
And we saw that in mechanical terms equilibrium is different from stability.
An unstable equilibrium can exist, but a slight displacement will turn the instability into motion. Whereas with a stable equilibrium a slight displacement (or a large displacement) will result in a restoring force back to its original position. For the simplest case – an incompressible fluid – this means that the temperature must increase with height.
If you watched the accompanying video of a tank of water being heated from below you would have seen that the instability caused turbulent motion until finally the tank was well-mixed.
We left the more complex case of compressible fluids (like air) until today. What we will find is that with a compressible fluid potential temperature is effectively the same as “real” temperature for an incompressible fluid.
So if potential temperature increases with height the fluid is stable, but if potential temperature decreases with height the fluid is unstable.
Let’s look at two examples:
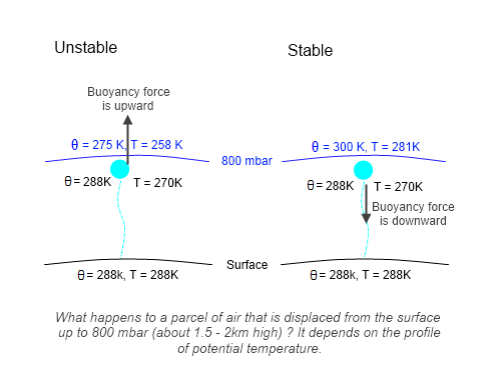
Figure 3
On the left hand side we see an example where potential temperature decreases with height. At the surface, θ = 288 K but at 800 mbar, θ = 275 K. A parcel of air displaced adiabatically from the surface to 800 mbar will keep its potential temperature of 288 K. Now we convert that to real temperatures. The environmental temperature at 800 mbar is 258 K, but the parcel of air cools to only 270 K. This means the displaced parcel is warmer than the surroundings, so it is less dense – and therefore it keeps rising.
This case is unstable – clearly any air that starts rising or falling (perhaps due to atmospheric winds, pressure differentials, etc) will keep rising or falling.
On the right hand side we see potential temperature decreasing with height. The parcel of air displaced from the surface to 800 mbar reaches the same temperature as on the left – 270 K. But here the environmental temperature is 281 K. So the parcel of air is cooler than the surrounding air, so it is more dense – and so it falls.
This case is stable – any air that starts rising or falling experiences a restoring force.
So the potential temperature profile with height tells us whether the atmosphere is stable, neutral or unstable. If potential temperature increases with height the atmosphere is stable, and if potential temperature decreases with height the atmosphere is unstable.
This is exactly the same as comparing the actual temperature change with the lapse rate.
Both answer the same question about atmospheric stability.
Moist Potential Temperature
The previous section slightly over-simplified things because potential temperature is with reference to dry air and yet moisture changes the way in which temperature decreases with height.
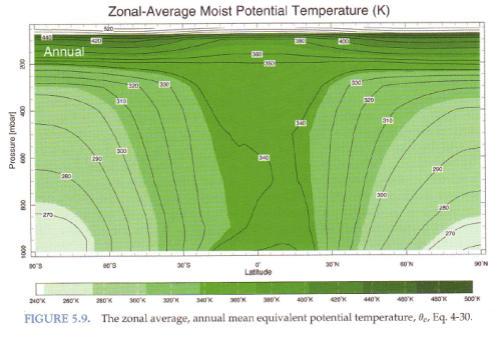
So here is the real deal – moist potential temperature. This is also known as equivalent potential temperature:
Figure 4 – Click for a larger image
Here we see the “real potential temperature” and notice that especially in the tropics moist potential temperature is almost constant with height – up to the tropopause at 200 mbar. This is due to convection creating a well-mixed atmosphere. In the polar regions we see that the atmosphere is still quite stratified, which is due to the lack of convective mixing.
Conclusion
Potential temperature is very useful. It is a method of comparing the temperature of air at two different heights.
And if potential temperature is constant or increasing with height then the atmosphere is stable.
The atmosphere is mostly stable for dry air. If you refer back to figure 2 you see that (dry) potential temperature is quite stratified which means any displaced air experiences a restoring force. So it is moisture in the air that is the enabler for most of the convection that takes place. Figure 4 shows us that the atmosphere is “finely” balanced as far as moist convection is concerned.
(Remember of course that these graphs are annual mean values. It doesn’t mean that dry convection does not occur).
Potential temperature is also a useful metric because the change of potential temperature with height can be used to calculate the strength of the restoring force on displaced air. The result is the buoyancy frequency and the period of internal gravity waves.


[…] « “Blah blah blah” vs Equations Potential Temperature […]
For me the biggest surprise is that the coldest area is at top of the equator at high altitude (figure 1). At the surface the equator is warmer then the poles, but at altitude, the equator is colder than the poles.
Why is that?
Andrejs Vanags,
I haven’t read an explanation that (to me) seems complete. Perhaps it’s so basic that it’s “obvious” and no need to go into detail..
Please give me a guess. Fig. 2 and 3 show that the atmosphere is more stratified in the regions of poles. So it’s less mixed there than over the equator and the heating by ozone is more efficient.
Right?
I wtnaed to make another comment but it may raise the ire of the local climate modelers who hold their model outputs in such high esteem. For years we’ve been saying the models are crap when, in all fairness, the computers calculate what they’re told to calculate. The models themselves are amazing. Works of art. Lines and lines of code. Years and years of tweeking. Amazing they calculate anything at all. However, to quote the conclusion of the paper: The main implication is that temperature increase predictions based on CO2 driving models are not reliable. The broader implication: The United States of America should NOT have under any circumstances used the results from the models for policy decisions. It doesn’t matter if 10 runs out of 10 runs or 1000 runs out of a 1000 runs suggest the earth will warm 2C over 100 years. Garbage programmed, garbage out. The sad truth is, it’s pretty much over. The EPA has run amok. We can only hope that common sense prevails and the House of Representatives sneaks in back room deal legislation to defund stupidity.
Rani,
I think you are new to commenting on this blog. Please read Etiquette.
This is a science blog. And this article is not about models (there are articles on this blog about models). I’m not sure what paper you are talking about. Generally I ask people to stick to the topic else there is always the chance that comments will be snipped or deleted.
If you have a comment here related to potential temperature or atmospheric stability I’m sure it will be of interest. If you want to write an essay about unrelated topics then there are much better blogs with much wider audiences.
The cold air of the equator which is below -60 degK has a higher moist potential temperature than the air immediately below it, so it normally would not reach this altitude by convection. Energy is conserved in a parcel of air undergoing adiabatic convection and moist potential energy accounts for thermal energy (kT), PV work, and latent heat; so convection shouldn’t change a parcel’s moist potential energy. However, moist potential energy doesn’t include the kinetic energy contained in the bulk motion of a parcel. In thunderstorms, the vertical motion of air can reach 100 km/hr.
Therefore, a sensible hypothesis is that the surprisingly cold air above the equator was driven there by the kinetic energy of bulk motion.
In that case, where does the kinetic energy of bulk motion originate? Convection alone shouldn’t be able to drive air from the surface about about 200 mb or θ = 345 degK.
You forget there’s convergence near the equator as well as simple convection. The surface flows in the Hadley cells above and below the InterTropical Convergence Zone are towards each other. That reinforces the upward flow driven by moist convection. There’s plenty of energy to drive circulation up to the tropopause. The Wikipedia article linked is a good place to start.
Throw a bombshell in at the end. Now I don’t understand what the “parcel of air” is in the tropics if the whole column is well mixed by convection.
Also this might be a bit dumbass but how big is the “parcel of air”?
HR,
The concept is a standard one in fluid mechanics. A simple explanation in Wikipedia.
“Less than the adiabatic lapse rate”: I like to say that the actual temperature gradient must be less STEEP than the adiabatic lapse rate.
SoD for Head of the IPCC!!!
Andrejs Vanags
“For me the biggest surprise is that the coldest area is at top of the equator at high altitude (figure 1). At the surface the equator is warmer then the poles, but at altitude, the equator is colder than the poles.
Why is that?”
I think part of the reason is that the earth spins on its axis. That centrifugal force makes the atmosphere at the equator much thicker than at the poles.
That 2 or 3 times thicker atmosphere at the equator allows higher altitudes of atmosphere to MEASURE a lapse rate..
On VENUS, with a “day” of 250 or so earth days, that centrifugal force is negligible, the atmosphere thickness between equator and poles should be similar, there should be negligible difference in Venus’ temperatures at high altitudes between equator and poles.
Alan D McIntire,
Have you ever tried to prove that?
An alternative explanation is that the thickness of the atmosphere is calculated via the ideal gas laws and the hydrostatic equilibrium.
Height of a given pressure, z(p) = R/g.∫T.dp/p, where the integral is from p to ps (the surface pressure), R = the ideal gas constant, g = acceleration due to gravity.
Or in simple terms, warm air expands and so the height of a given pressure surface at the equator is significantly higher than the same pressure surface at the poles.
Another way to view the geopotential height is the 500mbar surface which is around 5.5km high. Here is the contour plot of Jan 2003 mean value:
Click for a larger image
Alan D McIntire:
..That centrifugal force makes the atmosphere at the equator much thicker than at the poles..
Let’s take an element of mass in the atmosphere above the equator.
It experiences a gravitational acceleration, g = 9.81 m/s2
and a centrifugal acceleration, ac = ω2r, where ω = earth’s rotation rate = 7.27 x 10-5/s, r = distance from axis of rotation = 6.36 x 106m (at the equator)
At the equator, ac = 0.034 m/s2
Compare that with g = 9.81 m/s2 and you see it is tiny.
At the poles this value reduces to nothing and of course as the latitude increases to 90′ the direction of this acceleration with respect to the direction of gravity increases from 0′ to 90′.
The earth bulges slightly at the equator as a result of this.
Centrifugal acceleration doesn’t explain how 500mbar can be 5km at the poles and 5.9km at the equator.
I smell a very big rat with Scientific Research Publishing . It is run by Chinese. It has the hgsheit page rank in Iran. Anything can be published in their journals. The above article is mostly just junk, unfortunately.
SoD,
All interesting stuff; lots of work gone into this.
However, you say:
“[The DALR is for air without any water vapor.]”
This is not quite right. The DALR is for unsaturated air (less than 100% relative humidity), so it can contain water vapour. A parcel of air rising at DALR will cool at appx 3 deg C/thousand ft until (and if, depending on the amount of water vapour) it reaches its dew point. At that point cloud will form. Thereafter, the parcel of air will cool at the SALR (appx 1.5 deg C per thousand ft.
Arfur Bryant,
You are correct.
Another way to look at the potential temperature is as a stand in for entropy. The adiabatic lapse rate is then the isentropic lapse rate.
Higher average temperature is indeed the reason the atmosphere is higher at constant pressure in the tropics than at the poles. Note that the pressure difference increases with altitude. The resulting pressure gradient force is one of the main drivers of atmospheric circulation. But to describe that flow you also have to deal with more complex concepts like how the atmospheric density changes as a function of pressure and temperature, i.e. whether it’s baroclinic or barotropic. The Coriolis force also plays a big role in directing air flow.
Holy crap! DeWitt Payne has it right.
It should not be called the adiabatic lapse rate. That is an incomplete description. It is, in fact, the isentropic lapse rate.
kdk33,
The adiabatic lapse rate is the complete description because it is the process where a parcel of air rises with no exchange of energy with the atmosphere.
What is incomplete about this description?
The adiabatic lapse rate requires the expansion to be adiabatic AND REVERSIBLE. This can be easily proven. See isentropic expansion in Wiki and then look at lapse rate in wiki. I can reproduce the math if you’d like…
There is no general requirement that adiabatic processes be reversible. And you cannot derive the DALR with only the adiabatic requirement.
I think I know the origin of this oversight in metorology – a convecting parcel of air always expands against a differential pressure gradient; so, the pressure part is always reversible. Once the temperture gradient equals the DALR (for dry gas) there is no heat transfer, so the process is adiabatic (and reversible).
Outside of meteorology, in the more general world, the first part isn’t true.
In engineering we talk of adiabatic processes, and we talk of isentropic processes, and they are not the same.
And nobody ever gets this right.
SoD,
great post!
What about the influence of greenhouse gases? Is it right that the partition of atmosphere in troposphere and stratosphere is due to GHGs?
Yes, as the pressure drops, the path length in LW increases. There are less absorbing molecules in the path of out going radiation.
So the inversion between the tropopause and upper stratosphere is caused by the absorption of UV by O2/ O3(UV disassociates O2, freeing O to form O3) heating it from the top, CO2, and H2O act to cool it, by collisional exchange, and radiating away the energy. So with increased CO2, or H2O in a given area(as the pressure increases/or the ppm concentration increases), it increases cooling via radiation, It still absorbs LW coming up from the troposphere, but it radiates approximately twice what it absorbs…. the net flow of energy between the stratosphere and troposphere is actually from the stratosphere to the troposphere… kinda counter intuitive.
SoD has a good post on this somewhere here, and in comments he links a paper (Ramanthan and someone else, memory not what it used to be) that nicely quantifies the exchanges between troposphere and stratosphere.
err ummm… a correction to my above comment on reading it… the path length shortens, not lengthens… obviously, less molecules in path, less opaque, more transparent.
I think you were right the first time. Fewer molecules implies longer path length – a photon goes further on average before it hits a molecule.
However, strictly, isn’t it density, not pressure, which is important?
Good post SoD,
If anyone goes through a standard undergraduate atmospheric science curriculum, in a required thermodynamics class you are bound to learn at least a dozen or so different “temperatures” and it will make your head spin (temperature, potential temp, equivalent potential temp, virtual temperature, dew point temperature, wet-bulb temperature, etc, etc). All of them have varying degrees of practical use, but potential temperature is used frequently as a vertical coordinate (e.g., instead of height or pressure) in meteorology.
Physically, the potential temperature is the temperature a parcel of air* would have if it were brought dry adiabatically to the reference pressure of ~1000 mb. Thus, it can compare air parcels at different elevations to determine which one is warmer or cooler when brought to the same elevation. The conservation of potential temperature is also significant , since it means a potential temperature surface is an actual material surface along which air parcels must move. In contrast, air parcels are not constrained to move along a surface of constant pressure or constant height. Conversely, it is also informative to know that if an air parcel moves across surfaces of constant potential temperature, there must be non-adiabatic (diabatic) processes relevant to the situation, and any non-zero value of dθ/dt implies diabatic heating. Therefore there is often a better relationship between air moving along a θ surface than a horizontal or topographic surface. The equivalent potential temperature θe, is nearly perfectly conserved for dry and moist processes, and can act as a tracel to identify, and can be used for example to understand the inner structure of convective weather systems such as thunderstorms. where parcels originated from. As someone also pointed out, θ is a representation of the dry entropy of the air.
Under idealized situations, the pressure advection on a surface of constant potential temperature can also give an indication of the sign of vertical motion (whether air is rising or sinking in a region is probably the most important diagnostic in forecasting, in order to determine whether clouds will have a tendency to form). It can also be shown that is related to the sign of the temperature advection. There a number of other useful applications, in addition to characterizing the vertical stability field.
* I put a note by the term “air parcel” since someone asked about it. It is somewhat an informal term, but we typically think of it as a “blob” of air that is small enough to be considered homogenous in its properties (of temperature, water mixing ratio, etc) but also large enough to be a good statistical representation of molecules that one could apply thermodynamic principles to. The concept is a good starting point for understand thermodynamic relations between parcels (its temperature, density, pressure, etc and how they are related) as well as its movement through space and time in dynamical considerations.
1)What is meant by stable? Does stable mean that there is no convective heat transfer, and thus a radiative atmospheric layer? and does an unstable atmosphere means that heat is transferred convectively?.
At the stratosphere -dTdz is negative or << Gama so it is stable. I assume anything in between -dTdz of zero and about 5 C/Km is also stable.
2)Does that imply that the mesosphere lapse of 2.8 degC/Km rate is purely radiative? (if so, its surprising that it is so linear)
The other part that's confusing to me is that suppose (for dry air) the lapse rate at any instant is greater (slope more shallow) than the adiabatic lapse rate, heat will rise and the actual lapse rate will readjust until it equals the adiabatic lapse rate. So if we are talking at the steady state conditions the lapse rate will always equal the adiabatic lapse rate, so what new information (at steady state) does the stability criteria provide?
3)Would it not be better to compare the adiabatic lapse rate to the corresponding radiative lapse rate? then if the radiative lapse is greater than the adiabatic you know you will get convection and the actual lapse rate will follow the adiabatic lapse rate. If the radiative lapse rate is less than the adiabatic lapse rate, there will be no convection and the actual atmosphere will settle into the radiative lapse rate. Or am I off on that?
Andrejs Vanags says
3)Would it not be better to compare the adiabatic lapse rate to the corresponding radiative lapse rate? then if the radiative lapse is greater than the adiabatic you know you will get convection and the actual lapse rate will follow the adiabatic lapse rate. If the radiative lapse rate is less than the adiabatic lapse rate, there will be no convection and the actual atmosphere will settle into the radiative lapse rate. Or am I off on that?
Interesting question and I hope SoD answers with an explanation of:…….
What exactly is the radiative lapse rate?
It isn’t a constant like the dry adiabatic lapse rate. It’s the temperature profile the atmosphere would have if it had infinite viscosity. The initial slope at the surface for the atmosphere as we know it is considerably higher than the dry adiabatic rate. That’s why the convective component of the outgoing energy balance at the surface is higher than the radiative component.
DeWitt
Frank wrote: “In that case, where does the kinetic energy of bulk motion originate? Convection alone shouldn’t be able to drive air from the surface [above] about 200 mb or θ = 345 degK.”
Dewitt replied: “You forget there’s convergence near the equator as well as simple convection. The surface flows in the Hadley cells above and below the InterTropical Convergence Zone are towards each other. That reinforces the upward flow driven by moist convection. There’s plenty of energy to drive circulation up to the tropopause.”
Frank continues: Before commenting, I spent a long time reading about what determines the height of the tropopause at the top of the ITCZ, but I didn’t find a satisfying answer. If one assumes that the Hadley circulation is driven only by convection in the ITCZ, such convection shouldn’t extend above θ = 345 degK. There could/should be other sources of energy driving the Hadley circulation that enable convection to reach above θ = 345 degK. The trade winds are part of the Hadley circulation and the direction of the trade winds is modified by the Coriolis force. Does the Coriolis force do work (force times distance?) on the Hadley circulation and increase the energy in it bulk motion? What about the other psuedoforce, centrifugal force, which is greater where the flow is upward (near the equator) than downward (at higher latitudes).
The Coriolis Force does no work. In that respect it’s no different than the effect of a static magnetic field on a moving charged particle.
The Yoda-speak is not the fault of the autohr but of the journal. The AGW papers are by and large very professional looking and well written. At first you may think they know something.This paper seems to me to be one of those baby got thrown out with the bath-water papers.This is a bit worrisome [square brackets added by me]: The first [point to be considered] is that if a causal relation exists, then removing external trends, increase of residuals in CO2 must imply an increase of residuals in T I think AGWers would point out that the trend (in rising temperatures) is due to a rise in CO2 levels. So removing external trends (or looking at residuals, or flux changes) would seem to be the wrong thing to do. Estimating trends has been what this whole mess is all about. I don’t think anyone believes correlation in the short term variations in CO2 and temperature mean anything with respect to climate change.Won’t a cross-plot of the data from Fig.1 show a strong correlation between CO2 and T, unless I am missing something. Help?
DeWitt, your analogy between the Coriolis Force and a magnetic field makes sense. Still, we have jet streams in the atmosphere moving at almost 100 km/h against some sort of viscous friction, which should require some input of power. The limited reading I have done is very vague on this subject, but some sources suggest that they are driven by the earth’s rotation.
Deep convection in the tropics seems to rise higher than expected from potential temperature, suggesting it is driven by more than latent heat and buoyancy. Is there another energy source that powers the Hadley circulation? and the jet stream?
Frank
The jet streams are easily explained by conservation of angular momentum.
See an earlier comment.
Although my own conceptual issue is that the thermal wind equation also explains the jet streams, but I think I have almost mentally resolved that one.
And hope to write about it shortly.
Frank,
What makes you say that (“higher than expected from potential temperature”)?
Latent heat and buoyancy appear adequate as an explanation.
SOD: The plot of Zonal Average Moist Potential Temperature vs Latitude appears to show paths where large scale (therefore adiabatic) motion COULD occur: a) θ = 340-345 degK appears to correspond to upward convection in the ITCZ. b) θ = 320-330 degK could be downward potion of the Hadley circulation. c) θ = 280 degK (which is slanted, not vertical) could be the upward portion of the Polar cell. d) And so on. Every contour line is a potential pathway for adiabatic convection, but that doesn’t mean that a particular pathway is actually used at a significant rate, or in which direction. For example, is the flat contour at θ = 350 degK and 200 mb the result of convection?
Exc. post and I will end up sending a lot of folks to it. Theta and Theta e are not easy to explain, and when I was a student in metr. it took me awhile to get my head around it! Potential temp. charts are becoming more and more popular with forecasters I might add.
Off topic: I saw an illustration of the Webb Telescope in the news today.
http://graphics.latimes.com/towergraphic-james-webb-telescope/
I am curious about how well their cooling device will work given that it’s to be in the vacuum of space. Perhaps the sunward side of each layer is highly reflective while the opposite is black?
Anything facing the sun will be protected with a layer of sputter coated aluminum on plastic. Aluminum is very reflective over a wide range of wavelengths, see space blankets for example. I’m pretty sure the gold color you see on spacecraft like the lunar lander is because a polyimide plastic, Kapton® is the substrate and it’s yellow colored, not because the vapor deposited metal is actually gold rather than aluminum. I think.
As for cooling, radiating to deep space is actually quite effective at normal temperatures. At 300K, it’s 459 W/m².
What puzzles me is the five layers. The design seems based on heat sinks we see in electronic devices; but those rely on heated air moving out from between the layers and being replaced by cooler ambient air. To me the middle of the five layers will only radiate to space through a very narrow window. Put another way the heat from the sunward layer radiatively transfers heat energy layer by layer almost as if the layers were touching. The NASA web page has more illustrations. http://www.jwst.nasa.gov/sunshield.html
as well as info about tests. http://www.jwst.nasa.gov/images_sunshield11.html
It seems to me that rather than having the five layers close to each other they should be farther apart to open up the “window” available to radiate out of. Shouldn’t the heat radiated to space increase as the distance between the layers increases?
Science of Doom,
It appears to me that the day/night, latitude, and storage (ground and ocean) variation have not been taken fully into account. For example, day heating of a relatively cool ground or SST for the initially cooler atmosphere would cause strong force buoyant rising early in the day, the movement of the heated areas as day progresses cause wind mixing, and so on. Weather is fairly local in most cases, and just looking at average levels misses much of the dynamics.
SoD, when, in your comment to the EPT figure, you say that
“Here we see the “real potential temperature” and notice that especially in the tropics moist potential temperature is almost constant with height – up to the tropopause at 200 mbar. This is due to convection creating a well-mixed atmosphere.”,
it seems you suggest that the “well mixed atmosphere” has the same water load aloft as it has at the surface.
Of course it is not!
And, probably, you didn’t mean that.
Just for clarity, water is squeezed out inside the convective towers and what is left aloft and outside is just the usual dry (very dry) air.
Water (mass of) is always in the lower troposphere (and a bit also in the convective towers).
What that image suggests is that most of the latent heat is released in the low and medium troposphere and then transported upwards by the ascending air.
Now we can understand why the presence of that so cold air at the tropopause above the Equator.
First of all, the tropopause exists because of the convective mixing which, at the Equator, is very strong. That’s why the tropopause is higher there than at a greater latitude.
Then, since most of the water is rained out from the lower troposphere, the updraft has very less heat left to be released during its final route. So the dry adiabatic is approached up to the tropopause and air cools a lot more.
Paolo M,
What makes you think that I suggest the well-mixed atmosphere has the same water load high up as at the surface?
The fact that the atmosphere is “well mixed”, or the fact that moist potential temperature is almost constant against height? Or something else?
I said: “it seems you suggest…” and added “…you didn’t mean that”.
In event someone could misinterpret your words, I supplied an unrequested help.
You introduced the concept of a “well mixed atmosphere”, right when dealing with moist potential temperature.
I wanted just to restate that water is never well mixed, whereas a well mixed troposphere is, meteorologicaly speaking, referred to the air over a desert in summertime, with water having no role and the vertical profile of temperature right that of the dry adiabatic.
Then I went further to the why the tropopause is so cold over the Equator.
Didn’t you like my comment? I’m sorry.
“and cp = heat capacity of air at constant pressure ≈ 1 J/kg.K”
That should be 1 kJ/kg·K – i.e., 1000 times larger.
Ed Davies says
“I think you were right the first time. Fewer molecules implies longer path length – a photon goes further on average before it hits a molecule.
Oh dear, this is proof that video games rot yah brain like crack, yes you are right… (a pox on skyrim and bf3)
And yes, it is density that is more relevant… But inline with this, would someone be able to enlighten me with the blah blah, on the temperature effect on transparency.. Is a warmer parcel of air, of the same density(ignoring quietly ideal gas law), and make up of a colder parcel of air, more opaque? (im just wondering, if the increased excitation, stretches molecular bonds, causing larger molecules with T… kinda irrelevant, just got me thinking along those lines)
To a first approximation, no. To a second approximation, the individual line width is dependent on pressure broadening near the surface and Doppler broadening at high altitude. Pressure broadening is not, to first order, affected by temperature, but Doppler broadening is. For a short path cell where the rotational branches can be resolved, the population of the rotational energy levels will change.
Spectra of CO2 in a 1 m length cell at 1 atm and a volumetric mixing ratio of 380 ppm at 296 and 200 K are visibly different, but I would have to pay $25 to subscribe to Spectralcalc to tell you if the integrated absorbances were actually different.
200K http://i165.photobucket.com/albums/u43/gplracerx/CO2200K1m0000381013mbar.png
296K http://i165.photobucket.com/albums/u43/gplracerx/CO2296K1m0000381013mbar.png
And I didn’t correct for density change by reducing the path length, but should have. The path length for 200K should actually be 0.676m. It would be important if I were actually calculating the integrated absorbance. Of course, if I subscribed, Spectralcalc would do it for me.
Thanks for the response, probably not really what i was pondering, but gave me some interesting stuff to read up on, appreciated.
Frank wrote: Deep convection in the tropics seems to rise higher than expected from potential temperature, suggesting it is driven by more than latent heat and buoyancy. Is there another energy source that powers the Hadley circulation? and the jet stream?..
SOD asked: “What makes you say that (“higher than expected from potential temperature”)? Latent heat and buoyancy appear adequate as an explanation.”
Frank now responds: The coldest region (<-70 degC) above the equator at 50 mb in Figure 1 does not have the same moist potential temperature in Figure 4 as the air below it from 1000 mb to 200 mb. However, we know that this location is the top of the deep convection associated with the ITCZ. Above I speculated that there is an additional source of energy – probably the kinetic energy associated with bulk motion – that allows convection extend higher than it should based solely on the basis of moist potential temperature. Unfortunately, my suggestion implies that the bulk kinetic energy in the Hadley circulation arises from more than just buoyancy considerations in the air rising above the ITCZ. The discussion, so far, hasn't identified any viable candidate for this putative energy source.
There are jet streams circling the planet that contain a large amount of bulk kinetic energy with no obvious source for the power that the circulation must be consumed overcoming friction.
The obvious source is the average temperature difference between the equator and latitudes 30N and 30S caused by the difference in insolation (the cosine effect), not to mention the tipped axis of the planet. So now you have a heat engine driven not only by a vertical temperature gradient but a horizontal one too. In fact, this temperature difference would likely drive large scale circulation in a planet with a perfectly transparent, non-condensable atmosphere.
DeWitt: This seems like a good place to look. Unfortunately, bulk kinetic motion of the atmosphere (wind) is usually driven by pressure gradients rather than by temperature gradients. So your temperature gradient probably needs to produce a pressure gradient between 30 degN/S and the equator (at the surface or in the opposite direction at the tropopause) if it drives the convection higher than expected from moist potential temperature.
In agreement with this idea, the Hadley circulation is shown by Wikipedia with high pressure at 30 degN/S and low pressure at the equator.
Of course it does. Think about it. If it’s warmer at the equator than at 30N, the pressure at a given altitude at the equator will be higher than for the same altitude at 30N and the difference will increase with altitude. That pressure gradient causes air flow. Coriolis force deflects the flow and eventually you get the jet streams.
SOD,
I have enjoyed the post and the thread discussion over the past weeks. I don’t have anything to add really except thanks.
Thanks Jeff, appreciate the note.
I think I read your own lament on your blog a little while ago that your time is very limited for blogging.
Currently I am suffering the same sad situation.
Frank,
Sorry to be late in picking up on this thread but I just got around to reading it.
You said in your first post:
Your scientific instincts are correct.
During strong convection, driven by latent heat release, a large portion of that latent heat release is converted into PV work expansion rather than thermal energy. This PV work energy/thermal energy ratio is a direct function of the water vapor concentration (mixing ratio) at the surface. The higher the surface specific humidity, the higher the PV work/thermal energy ratio will be. And a higher ratio will result in a stronger upward convection (PV work) and a higher maximum convective height. (More latent heat is converted into gravitational potential energy (height) rather than thermal energy (temperature)). This is an example of non-equilibrium thermodynamics at its best.
The basis for this is spelled out in my E&E paper, which is indirectly linked by SOD above, and can be found here:
Click to access Gilbert-Thermodyn%20surf%20temp%20&%20water%20vapour.pdf
This can best be understood by referring to equation #3 in my paper (the first law):
dU = CvdT + gdh+ Ldq – pdV (3)
where CvdT is the change in thermal energy, gdh is the change in gravitational potential energy, Ldq is latent heat release and pdV is expansion work energy.
The first two terms on the RHS effectively represent “potential temperature” (Θ) and the first three terms on the RHS effectively represent “equivalent potential temperature” (Θe). The excess pdV term (beyond equilibrium) is the missing energy source you are looking for. It is not fully accounted for in either Θ or Θe. They are both calculated assuming thermodynamic equilibrium conditions (CpT for thermal energy).
When I plot Θe versus altitude for the three scenarios in my paper, Θe declines with altitude until condensation has ceased. Ldq is being converted to pdV, which is not fully reflected in Θe; only the CpdT term is. In fact, at maximum condensation, up to 80% of the latent heat release is converted to PV expansion work (see my Figure 6.) At that point, Θe then begins to increase until maximum convective height is achieved. This is due to the continued buoyancy which is causing a steady increase in gdh. At maximum convective height, Θe at altitude equals Θe at the surface. The pdV component is exhausted.
The rate of decrease in Θe from the surface increases as the surface specific humidity increases. Maximum convective height also increases as surface humidity increases.
You can see this effect from another perspective in my figures 3 – 5. The “thermal adiabat” represents the temperature profile if all latent heat released was converted to thermal energy. The difference between this theoretical profile and the actual temperature profile represents the excess pdV expansion work that is occurring. Figure 6 quantifies this PV work with respect to altitude and surface humidity in terms of % of latent heat released.
It is worth noting that this phenomenon may explain the missing hot spot that many climate scientists are frantically searching for. My guess is that the GCM’s do not handle the non-equilibrium thermodynamics correctly. They are probably assuming a constant Θe with altitude.
I hope I have explained this clearly enough. I have crammed a lot into a few paragraphs.
If you want another perspective, please take a look at this paper by Noor van Andel:
Click to access CO2_and_climate_v7.pdf
It is quite an impressive take on empirical atmospheric thermodynamics.
Bill Gilbert
Hi William; I haven’t looked in for a while; good to see that SoD is still going great guns,educating the masses! You say:
“a large portion of that latent heat release is converted into PV work expansion rather than thermal energy.”
If I may pose the question to you and SoD, is that why the Tropical Hot Spot has proved so elusive?
Hi Cohenite; Good to hear from you. I haven’t heard from you in awhile, but I have been slack in keeping up with the various climate blogs.
Yes, I am pretty sure the “hot spot” is an aberration of the climate models in that they do not handle the non-equilibrium thermodynamics correctly. I think the models do not properly take into account the fact that the latent heat of condensation is dispersed both as heat and PV work. Thus, where the “hot spot” is supposed to be, there is still much convection (PV work and mass transfer) going on which doesn’t show up as temperature. The actual thermodynamic behavior within the troposphere is not just a heat transfer mechanism, but a combined heat and mass transfer phenomena. The models’ parameterization of the Navier-Stokes equations may not do this properly (if the models use Navier-Stokes at all?)
I had corresponded with Noor van Andel before his death and he had done considerable work with KNMI in the Netherlands. He believed that the models assumed a constant equivalent potential temperature with altitude but was not able to confirm it. I have asked the question to several people, including Will Happer at Princeton, but I have not gotten an answer. There seems to be a dearth of actual model knowledge out there – which is scary since the entire academic field of climate science is based on these things.
Anyway, the figures in my E&E paper demonstrate the empirical facts pretty clearly. I could show you the corresponding plots of Θe with altitude but I don’t think I can copy figures on this site. They are very illuminating.
Bill Gilbert
williamcg,
An undiscovered tool of many people who strangely call themselves skeptics is called Google Scholar.
Google Scholar provides papers which reveals a laughable wealth of actual model knowledge. An embarrassment of riches. A veritable treasure trove of learned information.
Try Recent behavior of the Hadley cell and tropical thermodynamics in climate models and reanalyses by Mitas & Clement, GRL (2006), which is free – I found by using St. Google with the search phrase:
“potential temperature” in climate models
Found it quicker than it would take people to type out an amazing theory even once..
To get a better understanding of the detail of climate models, have a read of Description of the NCAR Community Atmosphere Model (CAM 5.0).
A good and more readable summary of an earlier version of CAM is in The Formulation and Atmospheric Simulation of the Community Atmosphere Model Version 3 (CAM3) by Collins et al, Journal of Climate (2006) but seems to be behind a paywall, happy to email a copy to anyone, email me at scienceofdoom you know what goes here gmail.com.
Assuming constant equivalent potential temperature would be pretty strange for climate models given what they actually calculate. An hour or two spent reading papers, even a few minutes probably, would dispel that illusion for anyone.
SOD,
Great!
Now that you have studied the models, how do they handle the allocation of latent heat of condensation between heat and PV work in a dynamic atmosphere? That is the question I have been asking. I look forward to your answer.
Thanks,
Bill Gilbert
Well guys; I did look at SoD’s model link [the NCAR community model] and I’m going to have to spend a little more time digesting the info.
But, at the end of the day, or lapse rate, the models emphatically predicted a THS, which is yet to appear. I just thought it may be a matter of interest consistent with this thread’s topic.
williamcg,
You want me to do your research?
I have other interests and little time.
Here is a diagram of the Earths Energy Budget; taken from NASA.
Globally, and yearly averaged, 484 W/m2 are asymmetrically generated by the atmosphere; 324 W/m2 downward and 165 upward.
To what atmospheric band are these 484 W/m2 emissions in your figure 1?
I only ask as now one has ever placed an Energy Budget and atmospheric temperature profile side-by-side, with connecting arrows or photon fluxes on the stratified atmospheric profile.
I would rather like to see and ‘onion’ model of Figure 1, showing the absorbance and emittance flux at each of your lines, both vertically and horizontally.
DocMartyn
All of the atmosphere radiates. If you take one thin layer of the atmosphere it radiates equally in all directions, which means the up and down emissions are the same.
The reason why the emission upwards from the top of the atmosphere is much lower than the emission downwards from the atmosphere to the surface is the absorption and reemission that takes place through the atmosphere.
The results are different in different locations, dates and times. The emission and absorption depend on the concentration of various gases, especially water vapor and the temperature profile through the atmosphere.
It’s easy to calculate a simplified “gray model” where no convection takes place. But for real life you need the results of a GCM.
A more interesting result is the graph of heating and cooling rates vs height for different gases and in total.
You can see this at Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Eleven – Heating Rates
“The reason why the emission upwards from the top of the atmosphere is much lower than the emission downwards from the atmosphere to the surface is the absorption and reemission that takes place through the atmosphere.”
Which one would, a prior, believe would have the flux gradient in the opposite direction. Given the much lower concentration of absorbers going up, the path to freedom is much shorter than the path to the surface.
DocMartyn,
I can’t comment on what any individual might, a priori, believe.
The number of absorbers from the surface to TOA is the same no matter which direction you travel in. The atmosphere near the surface is hotter than the atmosphere at the tropopause. Therefore the emitted flux is higher near the surface than at TOA.
If there was no absorption – for example if there was only a thin layer of atmosphere at one temperature, then the flux into space would be the same as the flux to the surface.
But the atmosphere close to the surface – the hottest level – emits directly to the surface with no absorption. The upward flux from this lowest level of the atmosphere gets mostly absorbed before it makes it to TOA.
Have a read of Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Ten to understand these points better.
O.K., so as the atmosphere near the surface is warmer than the atmosphere at high altitudes, then we have an asymmetry in the upward/downward fluxes.
Hope all is well
Eli
This is interesting and relevant …
An internal study by the U.S. EPA completed by Dr. Alan Carlin and John Davidson concluded the IPCC was wrong about global warming. One statement in the executive summary stated that a 2009 paper found that the crucial assumption in the Greenhouse Climate Models (GCM) used by the IPCC concerning a strong positive feedback from water vapor is not supported by empirical evidence and that the feedback is actually negative. Water vapor in the atmosphere causes a cooling effect, not a warming one. Carbon dioxide also causes a slight cooling effect but it so small it could never be measured by man’s instrumentation.
EPA tried to bury the report. An email from Al McGartland, Office Director of EPA’s National Center for Environmental Economics (NCEE), to Dr. Alan Carlin, Senior Operations Research Analyst at NCEE, forbade him from speaking to anyone outside NCEE on endangerment issues. In a March 17 email from McGartland to Carlin, stated that he will not forward Carlin’s study. “The time for such discussion of fundamental issues has passed for this round. The administrator (Lisa Jackson) and the administration have decided to move forward on endangerment, and your comments do not help the legal or policy case for this decision. …. I can only see one impact of your comments given where we are in the process, and that would be a very negative impact on our office.” A second email from McGartland stated “I don’t want you to spend any additional EPA time on climate change.”
McGartland’s emails demonstrate that he was rejecting Dr. Carlin’s study because its conclusions ran counter to the EPA’s current position. Yet this study had its basis in three prior reports by Carlin (two in 2007 and one in 2008) that were accepted. Another government cover-up, just what the United States does not need.
Eliminate this regulation immediately. This is a scientific tragedy.
Graham, what are you smoking? I can’t tell what you’re trying to say. Are you trying to say that water vapor and CO2 do not act as GHGs? If they do not, then what do you propose is creating this nice warm troposphere that allows Important People to go out in shorts and golf 2-3 times a week? If that’s not what you’re trying to say, then perhaps you should say it in a different way.
Yes, I know this is a drive by. I just can’t resist answering the phone when the 800 number comes through.
If you look at Solar Radiation absorption graphics you will see that water vapor absorbs quite a lot of incoming solar radiation and then re-emits the energy straight back to space, thus preventing the surface getting as warm as it would otherwise have done during the day. Even carbon dioxide absorbs some incident solar radiation around 2 microns.
The only effect that radiation from a cooler atmosphere can have is to slow the radiative component of surface cooling. There cannot be any actual transfer of heat to the surface, though. Water vapour is indeed about 40% as effective as a true blackbody in slowing the radiative cooling, but carbon dioxide has far fewer frequencies in its spectrum and so its effect on radiative cooling is absolutely minuscule.
Radiative cooling of the surface (excluding that which goes direct to space) is less than a third of total surface cooling. Evaporation and sensible heat transfer account for most of the rest. These non-radiative cooling processes cannot be affected by radiation from the cooler atmosphere because there can be no heat transfer from the cooler atmosphere to the warmer surface as such would violate the Second Law of Thermodynamics.
So, as I said, all that such radiation can do is slow the rate of cooling. It does so by supplying electromagnetic energy, not thermal energy. Hence the surface does not have to convert some of its own thermal energy to electromagnetic energy when emitting its quota of radiation. The important thing to remember is that the energy in the radiation is not converted to thermal energy in the surface. So other rates of non-radiative cooling cannot be affected.
Now, if carbon dioxide does have even just this minuscule effect on the rate of radiative surface cooling, then a wider temperature gap would open up and consequently non-radiative processes would speed up to compensate.
So the overall net effect of carbon dioxide on the cooling rate of the surface would be nothing at all. Meanwhile, it and water vapor will have a slight cooling effect as they absorb incident solar radiation, as explained above.
Graham Thompson,
Please supply your equations for radiative transfer along with your method of calculation, assumptions, and final results.
(Of course, I realize you won’t do that).
Any handwaving arguments about the effects of more or less of this or that constituent are handwaving arguments and not physics.
It’s good to start with handwaving arguments, it gets you “out of the starting blocks” but then you need to use the correct physics equations and calculate the results.
Otherwise you are engaging in poetry and not science.
I might theorize that getting the space shuttle to the moon would be more fuel efficient if they just started a little bit to the left, used less rocket fuel and it would all work out better. I might write an article to that effect without using a single equation of Newton’s laws of motion and continue to insist that my words are true because they sound nice – but the humor would most likely be wasted on the people it’s aimed at.
Work to do unfortunately, so no time for fun like that.
Don’t forget to supply your equations.
Or alternatively, critique the equations in Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Six – The Equations.
That’s incredible! AGW has been destroyed!
Hey wait. How does a slowing of the cooling rate not create a warming trend? It sounds like you’re a semantics troll. Oh, and how does the water vapor, with a residence time of about nine days, keep from pretty much condensing out of the atmosphere in your model–esp during ice ages? Oh, and how does your model match up to the findings of Puckrin et al. 2004? Based on actual measurements, Puckrin seems to have found that CO2 can be quite a significant factor in radiative flux, particularly when it shares the atmosphere with H20. I’m not sure your model also accounts for interglacial warming.
At any rate, you should probably have a look around this site. I am sure you’ll find a number of points with which to disagree, causing no end to the joy of intellectual stimulation.
If I were to refer you to the publications which contain the equations and the graphics supporting them, you would merely launch into criticism of the people involved, rather than discuss the physics.
Your own equations are incorrect because, as in the Backradiation series, you assume that if radiation is not reflected or transmitted then it must be absorbed. And when you say “absorbed” you in fact assume there is a coinciding conversion of its electromagnetic energy to thermal energy. In other words, you assume there is a heat transfer.
But there can be no heat transfer due to one-way spontaneous radiation from a cooler atmospheric source to a warmer surface because total entropy would decrease in such a process. Any subsequent returning radiation is irrelevant, and that component absorbed by the atmosphere may be less anyway when other sensible heat transfers also operate and some radiation goes to space anyway.
Try applying your equations to blocks of ice receiving intense radiation in your microwave oven. Work out how quickly they should warm and melt. Then observe what really happens.
Absorptivity is not only a function of the wavelength of the radiation – it is also a function of the temperature of the receiving body. Unfortunately not enough empirical work has been done on this, but no one has ever proved that backradiation actually causes a measurable heat transfer to a warmer surface. Yes it can slow the component of cooling which is due to that component of radiation which is not going directly to space. But water vapor is hundreds of times more effective (in total) than carbon dioxide, based on its wider bands of frequencies and greater abundance in the atmosphere. That fact alone puts carbon dioxide in its place.
And nowhere do you even discuss my key point that water vapor (and even carbon dioxide) send some of the incident solar radiation back to space before it even gets to the surface. They also help to radiate away the thermal energy which oxygen and nitrogen first acquired from the surface by sensible heat transfer. Where are your own equations regarding these cooling effects?
When I first started studying atmospheric physics, I too was originally bluffed by the GH hoax. Then I became skeptical. Now I am no longer just skeptical, I am absolutely certain that carbon dioxide and water vapor have a very slight net cooling effect.
Natural long and short term cycles can and do explain absolutely all climate change. But that’s another matter I really can’t be bothered explaining to those reading this site. I will not be returning here, as no one here has an open mind to the facts.
Graham Thompson
If you had read a textbook on atmospheric radiation you would realize that all of your amazing discoveries were questions already answered or confusion about basics on your own part.
A fabulous start to your treatise. I will let my record of articles on this blog speak for itself.
I’ll pick up a couple of points for other readers interested in the subject.
In a two body problem the second law of thermodynamics tells us that both bodies move towards a common temperature. That is, the hotter body cools down and the cooler body warms up.
In the three body problem of the sun, the earth and the atmosphere (count them, 3 “bodies”) – the temperature of the atmosphere affects the temperature of the earth.
In The Three Body Problem I proved that a warmer atmosphere increases the temperature of the earth compared with the case of a colder atmosphere. And the entropy calculations are there for replication – in both cases entropy increases which means the second law of thermodynamics is not violated.
I realize that the army of advocates of the imaginary second law of thermodynamics don’t know how to do an entropy calculation otherwise one of the following two responses would have been obtained:
a) here is the error in your calculation..
b) you are right, the second law of thermodynamics is not violated!
Of course there is a third possibility, embarrassed silence. I consider that highly unlikely of course, not even worth considering and chide myself for mentioning it.
And just to absolutely clarify yet again, if the sun turned off then after a very very long time the sun, the earth and the atmosphere would all end up at the same temperature (in a 3 body problem, i.e., if these were the only bodies in a closed system).
The analyses of the changes in CO2 and other trace gases do of course include the absorption and scattering of solar radiation by these gases. You find it’s not a “key point” once you have done the mathematical analysis, it is simply an adjustment to the longwave forcing.
This is from Radiative forcing by well-mixed greenhouse gases: Estimates from climate models in the IPCC AR4, Collins et al, JGR (2006):
Click for a larger image
My equations are in the post I pointed you to earlier.
The equations are not solvable analytically (means no solution in the form of an equation) – they must be solved numerically for different cases.
As before – I have provided equations and their derivation from first principles. These equations have been well established for more than 60 years. I have also shown solutions in various posts on this blog. You have provided no equations but made handwaving claims. This is a science blog. Provide your equations.
1. You produced no computations matching with reality for ice cubes in a microwave oven.
2. Your three body “proof” is garbage because you continue to assume radiation from a cooler body is absorbed by a warmer one and thus that there is a heat transfer associated with this one way passage of radiation. Wrong again, my friend. The energy in radiation from a cooler source is never converted to thermal energy in a warmer recipient.
3. I carefully explained a one way radiation scenario which had no dependent subsequent reverse radiation that was greater in magnitude. Yet with hand-waving you twist it into a standard two body scenario with simultaneous radiation and no other sensible heat transfer – theoretically. You assume that the “net” result is sufficient to satisfy the 2LoT but it is not. Every one way process must satisfy the 2LoT, except where there is dependency.
4. You say the atmosphere warms the surface. Wrong again. At the most it might slow the radiative cooling process, but that does not stop the other cooling processes accelerating. It is you who needs to prove otherwise. All the IPCC and KT energy diagrams clearly imply that there is heat transfer to the surface. These are wrong.
5. You continue to ignore the fact that water vapor has a far greater influence on such radiative cooling than does carbon dioxide, because the effect depends on emissivity and carbon dioxide cannot possibly have anything but very low emissivity because it radiates in very limited frequencies which are not at their maximum at most levels in the troposphere and stratosphere – only in the upper mesosphere around -70 to -90 C. All the carbon dioxide has less than 1% of the effect of all the water vapor, so where does that leave AGW?
That’s enough. I’m out of here because you have demonstrated that you are not prepared to attempt to answer questions about the low frequency radiation in a microwave oven, which does not cause heat transfer by the normal atomic absorption processes which would occur in the same objects (plastic etc) when they are placed in the radiation from the Sun. You are stumped on this one because you refuse to admit that radiation with frequencies lower than those being emitted by the target does not get absorbed in the sense that its energy is converted to thermal energy. Any such conversion in such a one-way radiation process would indeed violate the 2LoT and so does not happen.
I’m sure you know that this is what Prof Claes Johnson explained.
I don’t have to produce any “equations” because I use the same ones you do for calculating radiative heat transfer between two blackbodies. But the heat transfer is only associated with and caused by the radiation from hot to cold. Not any of the heat transfer depends on the radiation from cold to hot. The result is no different whether that radiation is more or less than the radiation from hot to cold, as can be the case in the real world. All that matters is the relative temperatures and the emissivity of the hot body and the absorptivity of the cold body. Absorptivity becomes zero when the source of the spontaneous radiation is cooler than the target.
I await your computations of what you think the radiation in a microwave oven ought to do to ice cubes in, say, 90 seconds.
Your equations were very compelling, but I might be more convinced I you would add some more poetic descriptions of what your equations signify…
If only.
First, you never raised the question but only complained that it wasn’t answered, at least in this thread. Second, the intensity of microwave radiation from incoming solar radiation is infinitesimal compared to a microwave oven. Third, microwave radiation is not absorbed by atoms but by molecules. Finally, the absorptivity of 2.4GHz radiation is different for different molecules. Plastics, not so much. Water, very high.
You misunderstand me, De Witt. I was not meaning just microwave radiation from the Sun, which is of course infinitesimal. I meant SW radiation. The surface acts in the way which Prof Claes Johnson describes in the quote in my comment below.
I am only discussing atomic level absorption which also leads to conversion of electromagnetic energy to thermal energy (KE if you wish.) When whole water molecules in a MW oven are rotated once with each wave of radiation (and thus generate frictional heat) that is a totally different process, which is unique to water molecules and a few fat and sugar molecules at MW oven frequencies.
It depends how you define absorptivity. Frictional heat generated at the molecular level is a totally different process which happens only within narrow bands which resonate with the natural physical frequencies of those whole molecules. There is absolutely no atomic-level absorption (with conversion to thermal energy) in any matter whatsoever receiving radiation in a microwave oven, when such matter obviously has a temperature above 1K.
There is an exact analogy with low frequency radiation from the atmosphere. Its energy is not converted to thermal energy in a warmer surface. Instead it supplies EM energy for the surface to do some of its own radiating. This EM energy never gets converted to KE because it is re-emitted immediately – just as quickly as reflected radiation, though the processes are different. So it is not “absorbed” in the sense I am talking about. None of its energy gets converted to thermal energy.
The IPCC thinks it does and thinks it thus raises the surface temperature so the surface radiates more. But the extra radiation does not come from surface thermal energy – instead its energy comes from EM radiation from the atmosphere which never got converted to thermal energy.
Yes, there is a lot of radiation thus coming from the Earth’s surface. But it is mostly reflected and “scattered” radiation which is just re-emitting the energy from downwelling LW radiation without any heat transfer to the surface.
Only a small portion of the radiation from the surface is actually transferring energy that was converted to EM energy from thermal energy that was in the surface. This portion does some of the surface cooling. More than half the thermal energy transferring from the surface to the atmosphere does so by non-radiative processes. If this were not the case then there is no way that the ground-level air below 2m (which is what we measure for climate) would stay as warm as it does. Radiation could not have this effect. But molecular collisions at the surface do have this effect.
Radiation from the atmosphere cannot have any effect on the rate of these non-radiative cooling processes. If radiatve cooling were slowed by a very small amount due to radiation from carbon dioxide, then a wider temperature gap would open up between the surface and ground level air. Thus, according to well known physics, conduction and evaporation rates would increase. This compensating increase has not been considered in IPCC models.
Nor have they considered the “backradiation” to space of energy absorbed by CO2 and water vapor from incident SW solar radiation. SoD thinks this is accounted for in an adjustment to LW radiation. But that is inappropriate. There is no reason why the downwelling radiation from the atmosphere should be shown as being more than the upwelling radiation to space, especially when you consider the incident solar radiation captured in the upper atmosphere right up to and including the thermosphere..
Graham,
BTW, you’re barking up the wrong tree with claims of a 2nd law violation. If anything, it’s the 1st law that is broken by the so-called ‘consensus’ climate science (i.e. the 3C or more rise hypothesis).
Graham Thompson
Despite my best efforts, some continue to confuse themselves regarding HEAT transfer in a two way radiative exchange.
This is wrong, but understandable, if someone is in a muddle about what exactly the difference is between heat, energy, work and radiation.
There is no heat flow from a colder to hotter object!
However;
1. There is a two way radiative exchange
2. There is a two way energy exchange
3. But there is only a one way heat transfer.
Carnot and Clausius were practical men who thought about the most efficient way to extract work from a heat engine.
They found that work (such as a moving piston output) can only be obtained with a high temperature source and a low temperature sink.
Use this as the test to see if an energy transfer qualifies as being a HEAT tranfer.
1. For a complete cycle extract thermal energy from a higher temperature source do work then dump unused energy to lower temperature sink.
This is a HEAT TRANFER and happens all the time!
2. From colder to hotter object, spontaneously extract thermal energy do work and dump unused energy to higher temperature sink .
This never happens.
So this energy transfer CANNOT be called HEAT.
So if HEAT can only BE transferred spontaneously in one direction why do some still insist in calling it NET?
Graham you see that you can test as to whether an energy transfer can be described as a heat transfer.
The transfer must be capable of doing thermodynamic work in the given situation
Its clear then that heat is a Macro Quantity.
The question as to whether or not a molecule absorbs a photon is a Micro phenomena.
This is where we differ
1. There is a two way radiative exchange which you say is impossible.
This is despite such an exchange being standard physics for over one hundred years.
2. There is a two way energy exchange if the hotter surface absorbs ANY radiation from the colder object.
Take for example 3 objects in local thermodynamic equilibrium with a vacuum separating them and all three can ‘see’ each other ;
A one at 270K
B one at 300K
C one at 330K
All three will include 10um radiation within their Planck spectrum
We are both in agreement that B can accept a 10um photon from C.
However you think that B will reject an identical 10um photon from A
This makes no logical sense!
“Take for example 3 objects in local thermodynamic equilibrium with a vacuum separating them and all three can ‘see’ each other ;
A one at 270K
B one at 300K
C one at 330K
All three will include 10um radiation within their Planck spectrum
We are both in agreement that B can accept a 10um photon from C.
However you think that B will reject an identical 10um photon from A
This makes no logical sense!”
Yes, to me and virtually everyone else, but good luck trying to convince Graham of this.
Prof Claes Johnson has already explained what happens …I quote him
“A blackbody acts like a transformer of radiation which absorbs high-frequency radiation and emits low-frequency radiation. The temperature of the blackbody determines a cut-off frequency for the emission, which increases linearly with the temperature: The warmer the blackbody is, the higher frequencies it can and will emit. Thus only frequencies below cut-off are emitted, while all frequencies are being absorbed.
A blackbody thus can be seen as a system of resonators with different eigen-frequencies which are excited by incoming radiation and then emit radiation. An ideal blackbody absorbs all incoming radiation and re-emits all absorbed radiation below cut-off. Conservation of energy requires absorbed frequencies above cut-off to be stored in some form, more precisely as heat energy thus increasing the temperature of the blackbody.
As a transformer of radiation a blackbody thus acts in a very simple way: it absorbs all radiation, emits absorbed frequencies below cut-off, and uses absorbed frequencies above cut-off to increase its temperature.
A blackbody thus acts as a semi-conductor transmitting only frequencies below cut-off, and grinding coherent frequencies above cut-off into heat in the form of incoherent high-frequency noise. We here distinguish between coherent organized electromagnetic waves of different frequencies in the form of radiation or light, and incoherent high-frequency vibrations or noise, perceived as heat.
A blackbody thus absorbs and emits frequencies below cut-off without getting warmer, while absorbed frequencies above cut-off are not emitted but are instead stored as heat energy increasing the temperature. A blackbody is thus like a high-pass filter, which re-emits frequencies below a cut-off frequency while capturing frequencies above cut-off as heat.”
Graham Thompson,
Claes “no photons” Johnson has explained what he believes happens..
And if his theory is correct, he has overturned the last 100 years of physics – as far as quantum mechanics and statistical thermodynamics is concerned. Which is quite a lot.
It is marvelous to see that so many people (including yourself) have realized modern physics needs to be overturned in order to demolish the inappropriately named “greenhouse” theory.
However, this blog is based on standard science and the Etiquette of this blog states:
So, if you want to discuss whether quantum mechanics & statistical thermodynamics are wrong, please find a blog interested in that field.
Future comments promoting the demolition of modern physics, entertainingly bad though they may be, will probably be deleted.
Firstly, in answer to Bryan’s puzzle, B will “scatter” all the 10 micron radiation from cooler sources because all can resonate. This happens because the Planck curve for a cooler body is always beneath and fully contained by the Planck curve for a warmer body. However, there will be an excess of 10 micron radiation from the warmer body and that component of such which corresponds to the area between the Planck curves will not resonate (because the cooler body can’t emit that excess) and so its energy becomes thermal energy. Hence the heat transfer is caused only by that radiation represented by the area between the Planck curves. This is not new physics – engineers have been using this calculation for decades when they subtract the two S-B calculations to get the difference.
There;s nothing “new” in the physics I use. Clausius stated the 2LoT in 1850 and it has never been disproved.
It should be perfectly clear that I agree radiation can travel “both” ways – in fact it travels in all directions.
And there’s nothing particularly new about microwave ovens which demonstrate the validity of what I am saying that the energy transported by low frequency radiation is not converted to thermal energy in a warmer target but is immediately re-emitted in a “pseudo” scattering process. After all, what could happen to that radiation which is neither reflected nor transmitted by ice cubes in a MW oven?.
It is you, SoD who is making up an imaginary 2LoT which you say permits a heat transfer from cold to hot just so long as there is also a greater heat flow (by radiation) from hot to cold.
It is the IPCC who fail to explain why the air just above the ground is kept warm. It can’t be by radiation, now can it? After all, if it were then it would be far colder when relative humidity is low on a hot summer day.
So maybe you can tell me just how and why carbon dioxide is supposed to cause the surface to warm and somehow have a significant effect not in any way dominated by big brother, water vapor? And have that effect in the first two meters of the air just above the ground.
And if you do try to tell me there’s any heat flow from the cold atmosphere to the warmer surface then I rest my case. But if you agree that only the radiative component of surface cooling can be affected, and that effect is far less than the effect of water vapor, and even when the radiative cooling is reduced, 99.9% or so of the extra warmth is above the 2m mark, then I also rest my case.
Graham Thompson says
“Firstly, in answer to Bryan’s puzzle, B will “scatter” all the 10 micron radiation from cooler sources”
The object B is at an intermediate temperature.
You say It absorbs 10um radiation from C while simultaneously rejecting 10um radiation from A.
Apparently encoded information about the Planck curve is carried by the photon.
Have you any evidence for this?
Some engineers use a subtractive short cut to quickly solve problems.
If you ask any who had studied thermodynamics 101 they would agree that real objects that emit 10um radiation will also absorb it regardless of its temperature source.
Yes, Bryan, Body B can handle all the 10 micron radiation from the cooler A, but there is more 10um radiation from the warmer C than it can handle. It resonates (thus scatters) the surplus 10 micron radiation up to the intensity for such on its own Planck curve. But that is all it can re-radiate because the Planck curve represents the maximum at each frequency, being, after all, that for a true BB. So the excess radiation (that is represented by the extra intensity which is between the Planck curves) is what does the warming. Nothing else ever warms anything.
By the way, this hypothesis, to the best of my knowledge, was first published by our group of about 40 scientists among whom are climatologists, professors (including Claes) atmospheric physicists, an astrophysicist, industrial chemist and many others with PhD’s in various disciplines. All are very much aware that AGW is a hoax.
KL only applies in certain circumstances. Note, for example, that the Earth’s surface absorbs 2 micron radiation in the IR band from the Sun (though carbon dioxide stops some of it getting through to warm the surface) but does the surface re-emit just as much 2 micron radiation as it absorbs? No. And, for much shorter wavelengths it re-emits virtually nothing at those wavelengths compared with what it absorbs. Why, because that is the radiation which, (as explained by Claes in the quoted paragraphs above) is having its energy converted to thermal energy in the surface.
It will surprise you to learn that Solar radiation also supplies direct EM energy for nearly all the radiation coming from the surface when it is actually shining on the surface. This small component of the Sun’s incident EM energy does not get converted to thermal energy. So the surface is not actually using its own thermal energy for what it is radiating during sunlit hours. Rather there is pseudo scattering of a small part of the lower frequencies of the incident solar radiation. You see, the Sun does also radiate everything represented by the Planck curve for the Earth’s surface. It’s just that this portion is such a small percentage of its total radiation that we tend to ignore it. However, it is there and it does resonate with the surface. As far as energy is concerned, it is just as if all this small percentage of the total solar radiation is reflected back into the atmosphere, along with the downwelling IR radiation from the atmosphere itself. It slows the rate of radiative cooling of the surface, but it does not actually add to the thermal energy in the surface. So most of the radiation from the surface which is lit by the Sun is not actually cooling the surface, just like when light is reflected by a mirror it is neither warming nor cooling the mirror.
Seeing that evaporative cooling is compensated for by precipitation, by far the majority of the heat transfer from the surface to the first 2 meters of the atmosphere is by physical conduction processes during molecular collisions at the interface. This is why and how the air temperature is maintained close to that of the surface, though usually just 1 to 4 degrees cooler in calm conditions.
You are right, though Bryan, in pointing out that engineers take short cuts in assuming two-way heat transfer or, as you put it, that radiation is heat. This of course is what the IPCC and KT imply in their Energy Budget diagrams.
Maybe some readers are starting to understand why the simplistic, flat Earth IPCC models are as far from reality as was flat Earth science.
This fact explains why
Graham Thompson,
Have a read of Amazing Things we Find in Textbooks – The Real Second Law of Thermodynamics where extracts from six heat transfer textbooks demonstrate exactly that point.
Basic atmospheric physics textbooks will give you the insights you need into why all this happens.
You appear to enjoy inventing ideas and putting them into the mouths or pens of others. But the random mix of ideas you present to debunk has no relationship to anything claimed in atmospheric physics textbooks.
Take a look at Find Stuff Out and Book Reviews – pick one that you think best suits and go visit your nearest university library, or stretch to $50 or less on Amazon or Book Depository. The trip or the cash will be well worth it. The only downside is you will realize what a total ass you have been making of yourself.
Alternatively, please cite a reference which claims what you are trying to debunk in the extract above. Textbook and section, IPCC report & page number, or peer reviewed paper.. My prediction – more blather in response.
Graham Thompson
Unfortunately SoD’s “Find Stuff Out and Book Reviews” contains several quotes from ‘engineers’ who take a shortcut and imply radiation is always heat.
For a practical problem at a certain level this ‘pass with a shove’ definition will not cause too many problems.
For a discussion that involves the second law of thermodynamics and the radiative interaction between the atmosphere and the Earth surface needless confusion will be created.
For this reason this post cannot be recommended.
Yes, Bryan, the information about the Planck curves is contained in the distribution of frequencies in the radiation. The resonance which occurs corresponds to the radiation which is represented by the area under the Planck curve for the cooler body. That is exactly how and why radiation from a cooler body has an effect on the radiative rate of cooling of the warmer body. The 2LoT is a macro law, so we have lots of molecules and lots of radiation. Thus we don’t need to discuss individual photons because it’s all a statistical process.
Have you any evidence for this? Yes I do have evidence of pseudo scattering. Do you have any evidence to the contrary?
Here’s the evidence which you can check for yourself, because, like all physics experiments, it should be repeatable.
Obtain two identical microwave bowls – ones which are not transparent to sunlight. Fill one about two thirds with water and tape the other one over the top so that no radiation can strike the water directly. Operate the MW oven for about 2 minutes. Now, obviously the bowls do not reflect all the radiation, and we know that they are not warmed significantly by the radiation either. So will the water get hot? If you think not, then place a glass of water in with it.
According to Claes Johnson the atoms in the bowls will absorb and immediately re-emit all the radiation, without any conversion of its energy to thermal energy. He says this happens because the frequency of the radiation is below the cut-off frequency for the bowl.
But the re-emission (unlike reflection) will be in random directions and so, for a thin bowl, it will follow random paths and so about half will get through the material in the bowl and warm the water. Note that of course the lower bowl will then get warm due to conduction from the warmed water, but the top bowl will remain cool.
How else can you explain this transmission which occurs without warming the bowl, when even sunlight cannot be transmitted but does warm the bowl?
Graham Thompson says
“Yes, Bryan, the information about the Planck curves is contained in the distribution of frequencies in the radiation.”
I will repeat my question which you failed to answer
………………………………………………………
The object B is at an intermediate temperature.
You say It absorbs 10um radiation from C while simultaneously rejecting 10um radiation from A.
Apparently encoded information about the Planck curve is carried by the photon.
Have you any evidence for this?
………………………………………………………..
If any radiation from the colder object is absorbed by B then an energy transfer has occurred.
As far as physics is concerned the 10um photon from A is identical to the 10um photon from C and object B would treat them in exactly the same way.
You will be well aware that photons are bosons with the usual properties that that implies.
But just in case you need an update
http://physicsworld.com/cws/article/news/2010/jul/01/ultra-precise-test-confirms-photons-are-bosons
You seem to rely totally on the theories of Claes Johnston!
Apart from yourself I know of no qualified physicist who accepts Claes ideas about photons.
If Claes abandons his little theory, does that mean you will have to accept the CO2 driven greenhouse gas theory?
Graham: A microwave bowl is transparent to the microwave radiation passing through it, but it absorbs visible and/or infrared light. Glass is transparent to visible light and absorbs infrared. Nitrogen gas doesn’t absorb infrared emitted by the earth’s surface, water vapor does. Materials only absorb and emit photons when they can use the energy to make an allowed transition between different electronic, vibrational, or rotational (for microwaves) energy states. There is nothing happening here that demands new physics.
As best I can tell, a microwave oven provides evidence a microwave energy can be absorbed by molecules that are hotter than source that emitted them.
The early physicists a century ago were not into it enough to realise exactly what happens when radiation strikes an object, so they assumed there was two-way heat transfer between two metal plates, that being the reason, they thought, why the cooling rate of the warmer one is reduced when a cooler plate (still warmer than the surrounds) is placed near it. Claes Johnson has a better explanation which still gives the same mathematical result. His views are now widely believed and understood amongst at least 30 or 40 scientists in the know with whom I have frequent contact. Don’t expect all your textbooks to contain this information. But you can confirm it with your microwave oven as explained in my other comment today.
Q.1: So what is your explanation as to how the air near the surface stays as warm as it does if conduction is really as low as IPCC energy diagrams would have us believe?
Q.2: And what is your explanation as to exactly how all the photons in a microwave pass through plastic without warming it?
Your last post (on this blog) on Claes “no photons” Johnson.
No doubt the physics journals have a whole raft of upcoming papers from these “scientists in the know” explaining how the last 100 years of physicists have been mistaken and deriving the new theory.
I will watch out for them.
No, SoD, it is you and those inventing “post physics” (with its political agenda) who are trying to overturn physics that dates back to Clausius in 1850.
“No process is possible whose sole result is the transfer of heat from a body of lower temperature to a body of higher temperature.”
Your “must be absorbed” backradiation is my proof. Read what your friend Bryan says here about your textbook thread.
I won’t write here again, but I will occasionally check in to see if you think you can explain how and why all those photons just pass right through the microwave plastic bowls and heat the water on the other side just as quickly as if it were in an open glass bowl. It’s got you stumped, hasn’t it?
Graham: Clausius may have said in 1850: “No process is possible whose sole result is the transfer of heat from a body of lower temperature to a body of higher temperature”, but physicists afterwards learned much more about meaning of the key words in this law, TEMPERATURE, HEAT and BODY. They now realized that every body having a temperature is composed of molecules (unproven in 1850) with a distribution of kinetic energies (unknown in 1850) and that energy is continuously being transferred both ways between slower-moving and faster-moving molecules by random collisions and by radiation composed of discrete quanta (understood to be continuous in 1850). Clausius did recognize radiation as a form of heat. Due to the Maxwell-Boltzmann distribution of molecular velocities, the kinetic energy of any one molecule in a body tells one nothing about an object’s temperature. Collisions change the kinetic energy of an individual molecule millions of times per second, but the mean kinetic energy of a large group of molecules in a body is unchanged by their internal collisions. So temperature was defined as being proportional to the mean kinetic energy of a group of molecules that collide frequently enough (to maintain a Maxwell-Boltzmann distribution). Unlike kinetic energy, temperature therefore is a bulk, statistical property that is meaningless for individual molecules. Physicists now apply Clausius’s law only to NET energy transfer between large groups of frequently-colliding molecules. They don’t apply the 2LoT to the one-way transfer of radiation between one molecule in the atmosphere and one molecule on the surface, because neither has a well-defined temperature.
What would happen if the 2LoT actually prevented a transfer of a photon from a molecule of higher kinetic energy to a molecule of lower kinetic energy? DLR would still be possible! Due to the Maxwell-Boltzmann distribution of molecular velocities, at least 40% of the faster-moving molecules in the cooler atmosphere have more kinetic energy than the slower-moving half of the molecules on the warmer surface. DLR can flow from faster-moving molecules in the atmosphere to the slower-moving molecules on the warmer surface without violating this version of the 2LoT. Upward radiation from the surface to the atmosphere would do the opposite.
For the 2LoT to prevent DLR photons from traveling from a cooler atmosphere to a warmer surface, the following would need to happen: a) The emitting molecule would need to know the LOCAL temperature by somehow determining the mean kinetic energy of its neighbors. b) It would need to emit a photon containing this information. This information can’t be the frequency of the photon, because objects with a given temperature emit photons with a range of frequencies. c) A molecule on the surface would need to know its local temperature by somehow determining the mean kinetic energy of its neighbors and decide whether to absorb or scatter an incoming photon based on the temperature information that is contained in the photon. Alternatively, one could abandon the kinetic theory of gases and the Maxwell-Boltzmann distribution and postulate that all molecules in the atmosphere have a kinetic energy that is lower than those on the surface.
From my perspective, applying the 2LoT to molecules and photons quickly leads to apparent absurdities that appear unnecessary when one understands what is NOW meant by the words “higher temperatures” and “lower temperatures” in the above version of the 2LoT.
Graham Thompson previously said on May 10, 2012 at 1:40 pm:
I responded on May 10, 2012 at 7:56 pm:
Graham replied on May 11, 2012 at 12:20 am:
No.
You made the claim. So please either:
a) show where this idea originates seeing as you claimed it came from the IPCC
b) admit you made it up
I don’t have any interest in explaining climate basics to people who invent ideas and claim they came from others – then don’t have the integrity to back up their claims or admit their mistakes.
1. The KT energy diagrams show a very small percentage of energy from the surface is “thermals.”
2. It is a common observation that the air near the surface is at a similar, slightly cooler temperature.
3. Standard physics formulas for conduction and convection can easily be used to show that such a low rate of conduction is not only incorrect but coould not maintain the temperature.
4. The first 2 meters is obviously a very small percentage of the atmosphere, so obviously does not get much of a share of the radiative warming.
5. Engineers find that radiation only starts to exceed conduction at temperatures above about 100 C, yet the energy diagrams show far more radiation.
6. David (in comments above) points put the error in assuming that radiation always causes a heat transfer.
You have no idea of my knowledge in both climatology and atmospheric physics. But your own knowledge of radiative heat transfer mechanisms is primitive, whereas I have written peer-reviewed science on the topic.
Sorry in para 6 above I should have said “Bryan” – see this comment.
Bryan, you can read more in about 250 comment in this thread.
“Graham Thompson” = anonymous
Sorry Bryan, but it seems that links to a certain tallbloke’s talkshop go into moderation, so look for a topic there on the Second Law/
Bryan, I have answered your A, B, C problem in two posts above – please be sure you read both.
You and others seem to think that radiation carries no information about the temperature of the source, or that at least the photons don’t.
I gave you an example with two bowls in a MW oven which you all try to ignore because you have no answer, do you?
The high frequency photons from the Sun warm a microwave bowl. but the low frequency photons in the microwave oven, despite their much greater intensity, pass right through the bowl without warming it.
I rest my case. You are not just arguing against myself – there’s a rapidly growing team of over 40 of us who are all very convinced of the errors in the AGW conjecture. You will see in due course.
Goodbye
Bryan, the second post (actually the third) that I don’t think you read was this one.
Graham Thompson says
“I rest my case. You are not just arguing against myself – there’s a rapidly growing team of over 40 of us”
Don’t dodge the question!
…………………………………………………
A surface that will accept a 10um photon from a hotter object will have to treat a 10um photon from a colder object identically.
This means that there is a two way energy transfer.
………………………………………………..
You will have to stick to a point if you ever hope to reach a conclusion.
Would your 40 include Doug Cotton?
He is regarded as a hopeless case (or perhaps a practical joker) by all sides in the climate debate.
Its remarkable that microwave ovens have waited for 70years before someone(Claes Johnson) explained how they worked.!
A surface that will accept a 10um photon from a hotter object will have to treat a 10um photon from a colder object identically.
Yes indeed it will – up to a certain intensity limited by the Planck curve. Now go and re-read the comment I linked above, which you still don’t comprehend.
This means that there is a two way energy transfer.
Who said there was no two way flow of electromagnetic energy flow in your ABC example? Not me. After all, you defined the scenario.
Its remarkable that microwave ovens have waited for 70years before someone(Claes Johnson) explained how they worked.!
We all know how microwave ovens work heating water molecules by rotation. But that is not what I was asking – and not what Claes’ scattering explained.
He is regarded as a hopeless case
If you can’t knock the physics, knock the person, eh? A typical arrogant approach that I’ve noticed is common among AGW hoaxers. You can always argue over there on the tallbloke thread where a certain peer-reviewed paper is being discussed.
You show less and less understanding of what I have written and asked with every post you write.
Well, I see no one has explained yet why that MV radiation passes right through the plastic without heating it. It seems you are all still stumped – as I knew you would be.
Goodbye
The only thing I’m stumped at is why I responded. The wise squirrel says, “Some nuts are best left buried.”
Graham Thompson,
I assume you mean MW for microwave. Your answer to me above clearly indicates that you have no understanding whatsoever of molecular and atomic absorption and emission of electromagnetic radiation even before you mention Claes Johnson. You seem to think that absorption of microwave radiation by a water molecule makes it rotate when it would otherwise not be rotating. No. It makes it rotate faster, i.e. change its rotational energy level, which isn’t zero to start with. But since statistical mechanics requires that large numbers of molecules divide their total energy equally between all degrees of freedom of motion by collisional energy transfer, including translational motion (kinetic energy), any increase in rotational energy by a molecule will be rapidly equilibrated to other molecules and thermalized. Friction has nothing to do with it.
Asking why 2.4 GHz microwave energy heats water and doesn’t heat plastic is the same as asking why sunlight doesn’t heat glass (much) but does heat a black surface. One is transparent and the other isn’t. And in order to get atomic absorption, one needs free atoms, not solids or liquids. The usual method of doing this is by using an air/acetylene or nitrous oxide/acetylene flame or a heated graphite cylinder.
No DeWitt.
You just make an assertive claim that the plastic is transparent to the MW oven radiation. This is not so, because the radiation does not pass through in the same way that it passes through glass, retaining its direction perhaps with some refraction. Instead it is totally scattered and some goes back out of the boundary through which it entered, just as happens when backradiation is scattered by the Earth’s surface without transferring heat. But some also gets through the plastic, following a random path between molecules which scatter it. This is totally different from normal transparency.
Apart from anything else, normal transparency happens with far shorter wavelengths. If glass is not transparent for LW radiation, and the same plastic is not transparent for sunlight (which does warm it) then why on Earth would it start to become transparent to such long wave radiation? Why doesn’t that radiation warm it, especially when its intensity is greater than the Sun’s radiation which reaches us?
But, if you accept that it does not warm it, then why do people assume LW radiation from the cooler atmosphere warms the surface? All it can do is slow down the cooling which is due to radiation. It cannot affect the majority of cooling of Earth’s surface which is by non-radiative processes. Has the IPCC or anyone ever proved that these other processes do not accelerate if radiative cooling slows? No. They never even thought about the possibility, and neither have you or SoD, now have you?
But that’s what happens, and that is why carbon dioxide has no net effect.
“You just make an assertive claim…”
🙂
To all:
The scattering to which I referred in my reply to DeWitt above, is being called “pseudo scattering” by some scientists now because it is actually a process of absorption followed by immediate re-emission at the same frequency and intensity. Technically it is new radiation, but it looks like scattered radiation. The main point is that none of its energy is converted to thermal energy if the target is hotter than the source. So there is no heat transfer involved and no energy left behind.
However, the new radiation is part of what the target would otherwise have emitted using its own thermal energy. Instead, it is using the EM energy in the incident radiation. But this does mean that it cools more slowly which is, of course, a well known fact. It is also well known that the quantification of this depends on the emissivity of the source which, in the case of CO2 at atmospheric temperatures, must be very low because of its limited range of emission frequencies. Each water vapor molecule has a greater effect, so the total water vapor in the atmosphere has a far greater effect than the total carbon dioxide. Water vapor also forms clouds which reflect solar radiation and thus have a cooling effect, by day at least. And all such molecules help to radiate energy from the atmosphere to space, and also send back some of that incident solar radiation which is in the IR band.
.
” The main point is that none of its energy is converted to thermal energy if the target is hotter than the source.” So if the source was 0.00001 degrees hotter than the target, the target accepts the incoming photons and they are converted to thermal energy. If the source is 0.00001 degrees cooler than the target, it “pseudo scatters” or rejects them? Thats a pretty smart target, and quick on its feet! I would appreciate more details, what happens if the source and target are the exact same temperature?
Graham,
“However, the new radiation is part of what the target would otherwise have emitted using its own thermal energy. Instead, it is using the EM energy in the incident radiation. But this does mean that it cools more slowly which is, of course, a well known fact.”
This doesn’t make any sense because even if it were true, the net effect is the same as if it was converted to thermal energy. If it’s just offsetting energy that would otherwise have to radiated and lost, there’s no difference and the net effect is to warm or reduce the rate of cooling.
I see that you’re stumped about what happens when the temperatures are the same. I surmised that would be the case…
I see that Mr. Cotton is still stumped by my question. I hereby declare complete and total victory, I knew he could never answer this question…
Strictly speaking Doug Cotton no longer gets to reply on this blog. This is because he does not respect the blog etiquette.
Those who want to engage with the amazing mind of Doug “occupy blog street” Cotton should engage him elsewhere. I realize that most (all?) are simply responding to the [deleted due to blog etiquette].
Well he had his chance before he outed himself so I still declare complete and total victory 🙂
Still not much point attempting to have a discussion with someone who believes they have “debunked” the last 100 years of quantum mechanics and statistical mechanics.
What would be the starting point? What is the equation for the emission of thermal radiation from a solid surface or a gas? What is the equation for convective heat transfer? What is the equation for latent heat transfer?
No common ground. Not much point having a discussion.
Especially someone who states so definitively that they understand how others explain particular processes, when that someone clearly hasn’t even read a standard textbook on atmospheric physics.
Graham Thompson,
Please cite experimental evidence that a plastic container scatters microwaves and a glass container doesn’t. Most plastics are not polar enough to interact with microwaves significantly. When I put a plastic cover over a plate of food in my microwave oven, the food still heats up. In fact, it heats faster because evaporative cooling is reduced. But the cover itself only heats up by sensible and latent heat transfer from the food, not by absorption of microwave radiation.
The wavelength of 2.45GHz microwave radiation is 12.5 centimeters. That’s a bit large for it to be “following a random path between molecules which scatter it.” If that were possible, then glass doors could not be used on microwave ovens. The glass door is covered on the inside with a metal screen. The holes in the screen are large enough for visible light to pass through, but too small for microwave radiation to pass so it’s reflected.
As far as pseudo scattering of microwave radiation, if the radiation isn’t absorbed or reflected, it can’t be scattered. If there were absorption, there would be heating. If there were reflection, it would be like putting a metal container in the microwave, lots of sparks and such.
SoD,
Pardon me for continuing this, but it’s actually becoming enjoyable. Thompson is even more clueless than Nasif Nahle.
No, GW, the net effect is not the same as if the energy had been converted to thermal energy, because thermal energy could then escape the Earth’s surface by sensible heat transfer. Not even the IPCC energy diagrams show an increase in sensible heat transfer resulting from backradiation. They show all the backradiation coming back out of the surface again, which is actually correct, but it does not transfer any energy to the surface.
Yes, SoD, there is nothing incorrect about the existing physics regarding S-B equations for emission or the calculations of heat transfer based on such. No new equations are needed – just an understanding of what is actually happening, namely that radiation in one direction is an independent process not in any way dependent on radiation the other way. Only the rate of heat transfer is affected by opposing radiation such as backradiation, not the total amount of radiation emitted by the surface.
Because each one-way process is independent, then it must do what Clausius said in 1850 and obey the Second Law of Thermodynamics, for which I suggest there is plenty of empirical “evidence.”
“No process is possible whose sole result is the transfer of heat from a body of lower temperature to a body of higher temperature.”
You cannot excuse a violation of this law by combining two independent processes (as SoD does in the “Backradiation” posts) and mathematically deducing a net effect. The mathematics is not explaining the physics, nor proving your false assumption to be correct.
So, yes, DeWitt, we both totally agree that the microwave radiation passes right through the plastic, as it does through a cube of ice also, without imparting any energy whatsoever to either. So how do all those photon particles that you believe in somehow dodge each and every molecule, yet in water, where the molecules are much further apart, they obviously have an effect?
If this long wave radiation does not warm these solids, what makes you think that long wave radiation from a cold atmosphere will warm the solid surface of the Earth? If it is transmitted (rather than scattered) then I suppose it would eventually come out on the other side of the globe. Either way, I don’t really care about where or at what angle it comes out.
The point is that all the backradiation does come back out of the surface somewhere and somehow without being absorbed and without transferring any energy to the warmer surface.
Hence …
(1) Radiation is not transferring as great a proportion of the surface energy as the energy diagrams imply because some is just backradiation coming back out, like the radiation that passes through the plastic in a MW oven and heats the water just as quickly.
(2) Thus non-radiative components are transferring a larger proportion – probably over 70% of the total energy departing from the surface after it was warmed by the Sun.
(3) The backradiation can have no effect on that component of the rate of surface cooling which is by non-radiative processes.
(4) If carbon dioxide has any measurable effect on the rate of radiative cooling of the surface, it must be far less than, and completely dwarfed by the effect of water vapor because there are always many more WV molecules and each WV molecule radiates more than each CO2 molecule because its emissivity is greater.
(5) Even if the effect of CO2 were measurable, it would lead to a larger temperature gap at the surface/atmosphere interface, and so the non-radiative components of cooling would accelerate to compensate. So there would be no net effect.
Well, for one we can measure the absorptivity of the solid surface of the Earth in the 5-50μm band (note that’s micrometers, not centimeters) and it’s pretty close to one. We can point an IR thermometer at the ground and the observed temperature is pretty close to what you would measure with a contact thermometer.
Also, there are lots of solid materials that absorb microwaves like clays, kaolin, talc, silicates, alumina, aluminosilicates and mixtures thereof. Gee, that sounds a lot like what the solid surface of the Earth is made of. Water molecules in a liquid are free to move, specifically rotate as the radiation passes through. They can’t in ice, so the interaction with microwaves is much reduced.
Actually, it’s about 60% by convection (102/168 W/m²) and 40% by radiation.
Smaller than, but not dwarfed. This stuff can be calculated, you know, or apparently don’t. And the emissivity of water vapor in the 15μm band where CO2 absorbs and emits is much lower than CO2. Plus, CO2 is a well-mixed gas and water vapor isn’t. So the effective emission height of CO2 at 15 μm is much higher, and therefore colder, than for water vapor. You can easily see the effect of CO2 on the emission spectrum of Earth as measured at high altitude, if you bothered to look. It’s right here.
Please show your calculations rather than just waving your hands. If there is no temperature increase, there can be no increase in convective cooling. It is true that convective cooling is a negative feedback, but a feedback, by definition, can’t eliminate the signal. If there is no temperature increase to drive it, how could increased convection continue? If it could do that, it would. In fact, that’s probably one of the causes of variability in the measured temperature. But that’s variability around a baseline. Increasing CO2 and other ghg’s will raise the baseline.
Firstly, there is no convection between the solid surface and the air. Go back to school and learn what convection is. I don’t care if you call it conduction or diffusion, but it sure isn’t convection. Convection takes place after air molecules have been warmed by the surface during collisions therewith.
Of course we can measure temperature with an IR thermometer: did I claim that the radiation from the surface is not in accord with S-B calculations? No.
The radiation is not reduced by backradiation, only the rate of radiative heat transfer. Go back and study more carefully what I have in fact said. I don’t care if you don’t believe it, but at least demonstrate next time that you understand what I have said – because several times you demonstrate that you have not done so, and that is a matter of poor comprehension, not science.
Yes, I won’t argue about whether it’s 60% or 70% that is transferred by such a process. But go back to your KT energy diagram and work out what percentage is shown there – far, far less I suggest. Something wrong perhaps?
I would be extremely interested to see your links to peer-reviewed empirical measurements of backradiation being absorbed by a warmer surface. Until you produce such I will treat it as hand waving.
I never said the surface would be warmed by the backradiation and I don’t believe you will ever produce evidence of high absorptivity of 50μm radiation by the surface. What I did say is that the rate of radiative cooling would be slowed by a minuscule amount by CO2 and that this would widen the gap. If I need to spell it out, the air would keep cooling at the same rate but the surface would cool slower. It’s not hand-waving to say that well known physics then says that the rate of energy transfer by conduction (diffusion) would increase because the temperature difference increased. But the increase is not due to the surface getting warmer.
CO2 will only emit at the peak of its Planck curve at temperatures around -80C as would be found only in the mesosphere. How much of that radiation gets back to the surface anyway? Water vapor emits at many frequencies and absorbs far more of the incident radiation from the sun also, even though CO2 does absorb some around 2 microns. So both have this cooling effect during the day. At typical atmospheric temperatures the 15μm band of CO2 is strongly attenuated because it is out to the side of the Planck curve. Nothing can radiate more than a BB at any frequency. It should be obvious that WV radiates more at such temperatures, just as it is clearly seen empirically that it absorbs more incident solar radiation. It moderates temperatures, cooling by day and warming by night, the net effect being cooling.
This bit of “news” is yet another example of possible evidence that WV has a net negative feedback. So that pretty much wipes out AGW doesn’t it if that is the case?
Moderator – please delete first version of my comment – sorry I had wrong info in my clip board regarding the link in the last paragraph. This is corrected in the second comment.
Firstly, there is no convection between the solid surface and the air. Go back to school and learn what convection is. I don’t care if you call it conduction or diffusion, but it sure isn’t convection. Convection takes place after air molecules have been warmed by the surface during collisions therewith.
Of course we can measure temperature with an IR thermometer: did I claim that the radiation from the surface is not in accord with S-B calculations? No.
The radiation is not reduced by backradiation, only the rate of radiative heat transfer. Go back and study more carefully what I have in fact said. I don’t care if you don’t believe it, but at least demonstrate next time that you understand what I have said – because several times you demonstrate that you have not done so, and that is a matter of poor comprehension, not science.
Yes, I won’t argue about whether it’s 60% or 70% that is transferred by such a process. But go back to your KT energy diagram and work out what percentage is shown there – far, far less I suggest. Something wrong perhaps?
I would be extremely interested to see your links to peer-reviewed empirical measurements of backradiation being absorbed by a warmer surface. Until you produce such I will treat it as hand waving.
I never said the surface would be warmed by the backradiation and I don’t believe you will ever produce evidence of high absorptivity of 50μm radiation by the surface. What I did say is that the rate of radiative cooling would be slowed by a minuscule amount by CO2 and that this would widen the gap. If I need to spell it out, the air would keep cooling at the same rate but the surface would cool slower. It’s not hand-waving to say that well known physics then says that the rate of energy transfer by conduction (diffusion) would increase because the temperature difference increased. But the increase is not due to the surface getting warmer.
CO2 will only emit at the peak of its Planck curve at temperatures around -80C as would be found only in the mesosphere. How much of that radiation gets back to the surface anyway? Water vapor emits at many frequencies and absorbs far more of the incident radiation from the sun also, even though CO2 does absorb some around 2 microns. So both have this cooling effect during the day. At typical atmospheric temperatures the 15μm band of CO2 is strongly attenuated because it is out to the side of the Planck curve. Nothing can radiate more than a BB at any frequency. It should be obvious that WV radiates more at such temperatures, just as it is clearly seen empirically that it absorbs more incident solar radiation. It moderates temperatures, cooling by day and warming by night, the net effect being cooling.
There is some news out about an experiment showing negative feedback for WV, but when I try to post the link it is not accepted. That’s what I was referring to in the above comment.
Graham Thompson,
Read Amazing Things we Find in Textbooks – The Real Second Law of Thermodynamics – extracts from six heat transfer textbooks showing exactly that.
Of course these people also embrace the last 100 years of physics and believe photons exist..
Graham Thompson,
I wrote a long post rebutting your gobbledegook (i.e. sounds scientific but isn’t), but it got lost. It’s not worth reconstructing. Suffice it to say that except for where you nitpick about terminology you’re absolutely and completely wrong.
No DeWitt and SoD that doesn’t suffice. (Next time DeWitt write your posts in a word processor first and paste them in so you have a copy, as I obviously did of mine.) I don’t know why SoD left both copies of the post above – feel free SoD to delete the second and the comment addressed to the moderator.
There is nothing incorrect in what Rudolf Clausius wrote back in 1850 …
“No process is possible whose sole result is the transfer of heat from a body of lower temperature to a body of higher temperature.
DeWitt talks about LW radiation up to 50 microns being absorbed by various materials. Sure. That is artificially generated radiation which obviously involves external energy input, as in a microwave oven or a CO2 laser. Such processes do not violate the 2LoT. They are in fact involving molecular absorption rather than atomic absorption, and this is such a different process that we really need a different word for it. It should not be counted in absorptivity measurements because the graph of such would have crazy spikes where resonance occurs, as in a MW oven.
But, you will never find spontaneously emitted radiation from a cooler source transferring energy into a warmer target. If you think you will, then it is you and your text book authors who are imagining things.
Radiation from cooler regions of the atmosphere undergoes what scientists are starting to call “pseudo scattering” where pseudo scattering by their definition is a process involving temporary absorption at the atomic level followed by immediate re-emission of identical radiation without any energy being transferred to the target. The re-emitted radiation does, however, count as part of the radiation which the target would otherwise have used its own energy to emit. The re-emitted radiation cannot lead to the target emitting more than its Planck curve permits at any frequency. Hence the target’s rate of cooling by radiation is in fact slowed down. But because no energy is transferred to the target (and so no heat transfer as per Clausius) then the other non-radiative cooling processes are not affected.
Hence it is totally wrong of the IPCC and KT energy diagrams to imply that wherever radiation goes, heat also goes.
I still await just one reference to any published empirical evidence which would disprove what I have said and show a heat transfer from cold to hot in a spontaneous one-way independent process such as radiation passing from a small region in the atmosphere to a small region of the surface. None of you can produce a single link to such, now can you?
If you want a more detailed explanation relating to how the energy in the radiation represented by the area between the Planck curves is that which transfers the heat one way (and none the other way) then this peer-reviewed paper (widely discussed among 40 or so members of PSI) may help your understanding.
Goodbye.
You must put a lot of faith in Wikipedia, whig made up 6/10 references in that paper…
Which not “Whig”. Another question: How come kids can’t use Wikipedia in science class, but Doug Cotton can in a “peer reviewed” paper?
Graham Thompson commented:
So I produced six textbook extracts from Professors of Heat Transfer and our new entertaining commenter replies:
My work is done.
What is it that you misunderstand about the word “empirical.” If your professors don’t quote actual experiments then why should I believe them, rather than professors I am in contact with who do carry out experiments which prove otherwise?
Graham Thompson,
If only…
Please cite any example of a peer reviewed paper or any other source for that matter where a qualified scientist explained the greenhouse effect as a net transfer of energy from colder to warmer.
Does a blanket transfer heat from colder to warmer? Does a greenhouse transfer energy from colder to warmer? No, they don’t. Nor does radiation from the colder atmosphere transfer net energy to the surface. The net energy flow is from the hotter sun to the colder Earth which then transfers energy to even colder deep space. If you actually understood thermodynamics, you wouldn’t quote Clausius in your defense. But you don’t. You string together terms you don’t understand to defend an untenable position.
CO2 for example, radiates according to the number of molecules in the bending mode excited state. The fraction of molecules in the excited state is dependent only on the temperature and the energy of the excited state provided the number density of molecules is sufficient to maintain Maxwell-Boltzmann statistics. The emission intensity is then the fraction excited times the number density times the Einstein A21 coefficient, provided the sample is optically thin so self absorption can be neglected. One can also calculate the optical density and convert it to absorptivity and multiply by the Planck equation for the temperature and wavelength. In either case, the emission intensity always increases with temperature over the range of interest. In other words, the emission of CO2 will be a lot higher at 300K than it is at 80K.
DeWitt: There is nothing you have said that I am not aware of or disagree with. Where did I ever say anything to the contrary? Once again, you display a lack of comprehension of what I am writing. Where did I refer to 80K for example? Where do you give values for emissivity of CO2 at various atmospheric temperatures compared with water vapor? Pretty low I suggest. All you do is compare it with itself at different temperatures. What does that tell anyone? It does not radiate a full spectrum under its Planck curve at any temperature and so it cannot radiate anywhere near what a true BB would.
And where do you in any way even discuss, let alone disprove the main thrust of my argument that non-radiative components of surface cooling (which you agree to be at least 60%) are not affected by backradiation and would have a tendency to compensate for slowing of radiative cooling?
Have I yet made it clear that you are all missing this fundamental issue?
I misread -80C as 80K, big deal. CO2 in the atmosphere emits more total flux at 300K than it does at 193 K. Are you happy now?
At high optical depth, the emissivity of CO2 is very close to 1 so the emission is nearly identical to the intensity of blackbody radiation over the wavelength range of the peak of the CO2 band. From the surface looking up at 299.7K and a tropical atmosphere where there’s a lot of water vapor, that amounts to about 7.2 W/m² out of 348.2 W/m² for 390ppmv CO2. While it’s 30 W/m² out of 163 W/m² for sub-Arctic winter conditions where there isn’t much water vapor. In the tropics, however, the effect on outgoing radiation at the tropopause is much greater. At a surface temperature of 299.7 K, 390 ppmv CO2 reduces emission at 18 km looking down by 35 W/m² from 323 W/m² to 288 W/m². For sub-Arctic winter, the corresponding numbers are 16 W/m² from 213 W/m² to 197 W/m².
For low cloud cover, you don’t see an effect of CO2 at the surface looking up at all because the cloud cover acts as a blackbody source at nearly the surface temperature. But there is still a reduction in emission from the cloud tops by CO2.
PS And DeWitt falls for the “argument” about “net” heat flows being sufficient to justify the 2LoT. Garbage my friend! I could just as easily say that if water flowed up one side of a hill (spontaneously) and then further down the other side of the hill that the “net” flow was downhill and so entropy increased and the Second Law of Physics was thus not violated. Your argument is just as fallacious.
There is absolutely no transfer of heat in the one way process of radiation from a cold source to a warmer target.
What happens in the other direction is irrelevant – simply because it is an independent process. As Clausius said, the law applies to any (and every) process.
Apparently the SLoT doesn’t “apply to any (and every) process”. There is a ‘loophole’ in the SLoT involving nanoparticles for short periods of time, up to a couple of seconds. vide: “Experimental Demonstration of Violations of the Second Law of Thermodynamics for Small Systems and Short Time Scales” – G. Wang et al (2002)
The analogy of water ‘spontaneously’ flowing up one side of a hill and another is not a good one as it violates Newton’s Universal Law of Gravitation. In terms of the SLoT, the work done to non-spontaneously get the water up over the hill would increase the entropy of the surroundings.
Wrt to the ‘one way process of radiation’. The oscillating electric and magnetic field of the EMR from a cold source that is incident upon an opaque warmer source would interact with its molecules’ electric dipoles, its free electrons or its ions resulting in their vibration and the production of heat. This would be less so for a transparent warmer source.
To comply with the SOD etiquette of posting something mathematical: (P + p)(V – v) = NkT
George Montgomery,
You realize you are talking to someone who doesn’t accept statistical thermodynamics, quantum mechanics, or the existence of the photon.
In my case I could spend a lot of time digging out very old papers to show that surface % absorption of incident radiation is ONLY a function of:
i) the absorptivity of the surface material at that wavelength, and
ii) the wavelength of the photon
– but well, there are no photons, they don’t exist so…
Bit of a waste of time. Like being asked to produce a paper showing the earth goes around the sun. Hard to find, then when produced the use of the telescope in the experimental procedure gets questioned..
Let’s just agree that NO textbook on heat transfer produced in the last 50 years agrees with the claim of Graham Thompson.
One day when the Illuminati produce their complete theory and explain the last 100 years of experimental evidence in the light of that theory I will take a look.
It will need to be quite extensive and I am not holding my breath for that one.
When I get some time free from work I plan to dive into subjects which accept as their foundation the physics basics as taught in textbooks. People who think all textbooks are wrong on physics basics – please post on other blogs. When you have a complete theory I will be happy to post a link to it.
“There is absolutely no transfer of heat in the one way process of radiation from a cold source to a warmer target.”
I’ll see Doug Cotton’s Wikipedia references and raise him one USA today article. So why are cloudy nights warmer than clear nights?
http://www.usatoday.com/weather/wcldwarm.htm
George Montgomery,
The Second Law is statistical so obviously if there are a small number of particles, then a ‘violation’ of the Second Law is possible. However, for the Earth’s atmosphere, the probability is vanishingly small.
Whoever said there was? In this case Bryan’s favorite definition of heat applies which means heat does not transfer from cold to hot with the usual caveat that the number of particles is sufficiently large.
So if you have two parallel infinite planes with non-zero absorptivity in vacuum at temperatures T1 and T2 in degrees K, you don’t need to know both temperatures to calculate the flux? I think not. Assuming equal absorptivity/emissivity, the formula is εσ(T1^4-T2^4) where ε is the emissivity and σ is the Stefan-Boltzmann constant. Gee, that looks exactly like flux in minus flux out. Your interpretation of Clausius’ use of the term process is incorrect.
Of course you need to know both temperatures because the heat transfer relates to the radiation which does not resonate each way. This radiation is represented by the area between the Planck curves. This, and only this radiation transfers heat. You obviously have not got past page 1 of the paper I referred you to. Read some of the 250 or so comments thereon here. No, SoD, you won’t find it in text books yet. These things take time.
If there were any evidence of any effect of CO2 on climate it might be a different story, but, as you can read in the Appendix to the above peer-reviewed paper, the rate of increase in SST has been declining at least since the year 1900, suggesting an approaching natural maximum much like the MWP and others at about 1,000 year intervals prior to that.
DeWitt: Further to my comment at 12:54pm (awaiting moderation) let me make it quite clear that we both agree regarding the quantitative effect on heat transfer between two parallel plates. But I say the heat transfer relates only to that radiation represented by the area between the Planck curves. This is of course the same as deducting the small area from the large one, so mathematically the result is the same. It’s all in the paper I referred you to a few days ago and again in the 12:54pm comment today.
Furthermore, DeWitt, the Earth’s surface-atmosphere interface is a far cry from two infinite plates in a vacuum. There are other heat transfer processes which do not involve radiation. Do you seriously think that one-way radiation from the atmosphere to a warmer surface can actually transfer heat? If it did, then that transferred energy could exit by evaporative cooling or sensible heat transfer – or at least 60% of it would. So only 40% comes back as radiation. How does that fit with your formula?
What happens each morning when the Sun is warming the surface? There is net energy into the surface. Is backradiation going to warm it more? Does backradiation penetrate beneath the surface of the water and warm a sub-surface layer? After all, LW radiation gets through that plastic in the MW oven. If the subsurface layer is warmed where is the returning radiation? Maybe the water evaporated.
What I say does explain what actually happens in all circumstances you care to dream up. Nothing else does. You obviously have not taken the time to really try to comprehend what I am explaining. Go read my paper and some of the comments on it on the Tallbloke thread re “Radiated Energy and the Second Law of Thermodunamics.”
OK, the cat’s out of the bag, so there’s no point in my writing more here.
Please produce a quote where I or SoD said or implied that. Heat, defined as net energy, can only be transferred from hot to cold. That’s not the point. The point, which you seem to miss completely is that for a constant rate of heat transfer, the temperature of the colder object affects the temperature of the warmer object. The rate of heat transfer for the Earth’s surface is determined by the absorption of incident solar radiation. Absent a change in albedo, that’s constant. So when the atmosphere warms, the surface must warm also. But the energy for the warming comes from the incident solar radiation, not the atmosphere.
Subtracting spectra or Planck curves is mathematically identical to subtracting fluxes, which are the areas under the spectra. Why you think that somehow invalidates the greenhouse effect is beyond me.
We have been dealing with one of the many faces of Doug “no ethics” Cotton.
Enough said.
I believe the term of art is “sock puppet”. Now I’m wondering if Arfur Bryant at The Blackboard isn’t another one.
Speaking of “sock puppets” and Janus-like commenters, add Graham Young at the Air Vent blog as another possible.
Having wasted some hours checking out posts on other websites, “carpet bombing” is another term of the art that may, given their sheer volume, apply in this circumstance. The aptness of this phrase is given extra meaning by “The Cotton War” article on “the Air Vent”. It allegedly reveals another side to the prolific poster of comments.
KE (rotational) = 1/2 I w2
Adieu
[…] CO2 the emission of radiation to space from the climate system is from some altitude. And because temperature reduces with height due to other physics the surface must be warmer than the effective radiating point of the atmosphere. This means the […]
[…] lapse rate has already been covered in Potential Temperature and for those new to the subject Density, Stability and Motion in Fluids is also worth a […]
Hello
Thank you for useful information really.
M Ahmadi
Oh i get the confusion. Graham Thompson was talking about “Radiated Energy and the Second Law of THERMODUNAMICS.” I always get those mixed up.
[…] this air. This hot moist air rises. As this air rises it cools, due to adiabatic expansion (see Potential Temperature), and water vapor condenses out, releasing the latent heat stored. The strongest examples are known […]
[…] equations (already explained). Convection moves heat if the atmosphere above is too cold (see Potential Temperature and Density, Stability and Motion in Fluids). Energy retained increases the temperature of the […]
Hi,
Just wanted to double-check what I think is correct. Say we’re looking at the ARGO float data. And say that we want to compute the heat added to a cell of water dV = (dA)(dz), where dA is the area of the float’s cell, and dz is a small vertical slice defined by the resolution of the float’s measurement process.
As long as the pressure remains constant for dV, am I correct in assuming that we don’t need to worry about potential temperature to compute heat changes? Reason: 1st Law: dU = c_v dT – PdV, and the change in volume is 0 because isobaric condition guarantees isochoric condition in this situation.
Thanks,
Jeff Cagle
I don’t see how isobaric guarantees isochoric. Quite the opposite in fact. The sea level does increase with warming as one would expect for isobaric conditions.
Great information. Lucky me I recently found your website
by chance (stumbleupon). I’ve bookmarked it for later!
This is really interesting, You are an overly professional
blogger. I’ve joined your feed and stay up for in quest of extra of
your magnificent post. Additionally, I’ve shared your web site in my social networks
…an well-explained article on the vertical profile of potential temperature…thanks a lot.
Very Nice Explanations.. Keep It Up 🙂
Cool explanation.. Thanks!
[…] same at all latitudes. (On Earth, the average temperature in the upper troposphere at a pressure of 190 mbar is only weakly dependent on latitude, so this assumption of constant temperature isn’t […]
[…] at all latitudes. (On Earth, the average temperature in the upper troposphere at a pressure of 190 mbar is only weakly dependent on latitude, so this assumption of constant temperature isn’t […]
[…] same at all latitudes. (On Earth, the average temperature in the upper troposphere at a pressure of 190 mbar is only weakly dependent on latitude, so this assumption of constant temperature isn’t […]