Here is an article from Leonard Weinstein. (It has also been posted in slightly different form at The Air Vent).
Readers who have been around for a while will remember the interesting discussion Convection, Venus, Thought Experiments and Tall Rooms Full of Gas – A Discussion in which myself, Arthur Smith and Leonard all put forward a point of view on a challenging topic.
With this article, first I post Leonard’s article (plus some graphics I added for illustration), then my comments and finally Leonard’s response to my comments.
Why Back-Radiation is not a Source of Surface Heating
Leonard Weinstein, July 18, 2012
The argument is frequently made that back radiation from optically absorbing gases heats a surface more than it would be heated without back radiation, and this is the basis of the so-called Greenhouse Effect on Earth.
The first thing that has to be made clear is that a suitably radiation absorbing and radiating atmosphere does radiate energy out based on its temperature, and some of this radiation does go downward, where it is absorbed by the surface (i.e., there is back radiation, and it does transfer energy to the surface). However, heat (which is the net transfer of energy, not the individual transfers) is only transferred down if the ground is cooler than the atmosphere, and this applies to all forms of heat transfer.
While it is true that the atmosphere containing suitably optically absorbing gases is warmer than the local surface in some special cases, on average the surface is warmer than the integrated atmosphere effect contributing to back radiation, and so average heat transfer is from the surface up. The misunderstanding of the distinction between energy transfer, and heat transfer (net energy transfer) seems to be the cause of much of the confusion about back radiation effects.
Simplest Model
Before going on with the back radiation argument, first examine a few ideal heat transfer examples, which emphasize what is trying to be shown. These include an internally uniformly heated ball with either a thermally insulated surface or a radiation-shielded surface. The ball is placed in space, with distant temperatures near absolute zero, and zero gravity. Assume all emissivity and absorption coefficients for the following examples are 1 for simplicity.
The bare ball surface temperature at equilibrium is found from the balance of input energy into the ball and radiated energy to the external wall:
To = (P/σ)0.25 ….(1)
Where To (K) is absolute temperature, P (Wm-2) is input power per area of the ball, and σ = 5.67×10-8 (Wm-2T-4) is the Stefan-Boltzmann constant.
Ball with Insulation Layer
Now consider the same case with a relatively thin layer (compared to the size of the ball) of thermally insulating material coated directly onto the surface of the ball. Assume the insulator material is opaque to radiation, so that the only heat transfer is by conduction. The energy generated by input power heats the surface of the ball, and this energy is conducted to the external surface of the insulator, where the energy is radiated away from the surface. The assumption of a thin insulation layer implies the total surface area is about the same as the initial ball area. 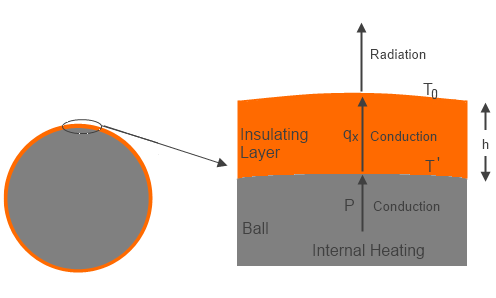
Figure 1 – Ball with Insulation
The temperature of the external surface then has to be the same (=To ) as the bare ball was, to balance power in and radiated energy out. However, in order to transmit the energy from the surface of the ball to the external surface of the insulator there had to be a temperature gradient through the insulation layer based on the conductivity of the insulator and thickness of the insulation layer.
For the simplified case described, Fourier’s conduction law gives:
qx=-k(dT/dx) ….(2)
where qx (Wm-2) is the local heat transfer, k (Wm-1T-1) is the conductivity, and x is distance outward of the insulator from the surface of the ball. The equilibrium case is a linear temperature variation, so we can substitute ΔT/h for dT/dx, where h is the insulator thickness, and ΔT is the temperature difference between outer surface of insulator and surface of ball (temperature decreasing outward).
Now qx has to be the same as P, so from (2):
ΔT = (To-T’) = -Ph/k ….(3)
Where T’ is the ball surface temperature under the insulation, and thus we get:
T’ = (Ph/k)+To ….(4)
The new ball surface temperature is now found by combining (1) + (4):
T’ = (Ph/k)+(P/σ)0.25 ….(5)
The point to all of the above is that the surface of the ball was made hotter for the same input energy to the ball by adding the insulation layer. The increased temperature did not come from the insulation heating the surface, it came from the reduced rate of surface energy removal at the initial temperature (thermal resistance), and thus the internal surface temperature had to increase to transmit the required power.
There was no added heat and no back heat transfer!
Ball with Shell & Conducting Gas
An alternate version of the insulated surface can be found by adding a thin conducting enclosing shell spaced a small distance from the wall of the ball, and filling the gap with a highly optically absorbing dense gas. Assume the gas is completely opaque to the thermal wavelengths at very short distances, so that he heat transfer would be totally dominated by diffusion (no convection, since zero gravity).
The result would be exactly the same as the solid insulation case with the correct thermal conductivity, k, used (derived from the diffusion equations).
It should be noted that the gas molecules have a range of speeds, even at a specific temperature (Maxwell distribution). The heat is transferred only by molecular collisions with the wall for this case. Now the variation in speed of the molecules, even at a single temperature, assures that some of the molecules hitting the ball wall will have higher energy going in that leaving the wall. Likewise, some of the molecules hitting the outer shell will have lower speeds than when they leave inward. That is, some energy is transmitted from the colder outer wall to the gas, and some energy is transmitted from the gas to the hotter ball wall. However, when all collisions are included, the net effect is that the ball transfers heat (=P) to the outer shell, which then radiates P to space.
Again, the gas layer did not result in the ball surface heating any more than for the solid insulation case. It resulted in heating due to the resistance to heat transfer at the lower temperature, and thus resulted in the temperature of the ball increasing. The fact that energy transferred both ways is not a cause of the heating.
Ball with Shell & Vacuum
Next we look at the bare ball, but with an enclosure of a very small thickness conductor placed a small distance above the entire surface of the ball (so the surface area of the enclosure is still essentially the same as for the bare ball), but with a high vacuum between the surface of the ball and the enclosed layer.
Now only radiation heat transfer can occur in the system. The ball is heated with the same power as before, and radiates, but the enclosure layer absorbs all of the emitted radiation from the ball. The absorbed energy heats the enclosure wall up until it radiated outward the full input power P.
The final temperature of the enclosure wall now is To, the same as the value in equation (1). 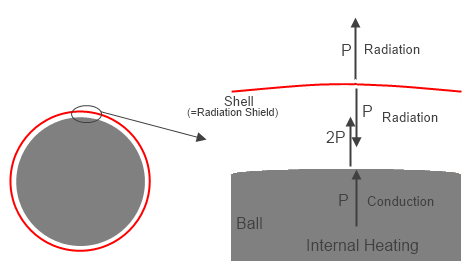
Figure 2 – Ball with Radiation Shield separated by vacuum
However, it is also radiating inward at the same power P. Since the only energy absorbed by the enclosure is that radiated by the ball, the ball has to radiate 2P to get the net transmitted power out to equal P. Since the only input power is P, the other P was absorbed energy from the enclosure. Does this mean the enclosure is heating the ball with back radiation? NO. Heat transfer is NET energy transfer, and the ball is radiating 2P, but absorbing P, so is radiating a NET radiation heat transfer of P. This type of effect is shown in radiation equations by:
Pnet = σ(Thot4-Tcold4) ….(6)
That is, the net radiation heat transfer is determined by both the emitting and absorbing surfaces. There is radiation energy both ways, but the radiation heat transfer is one way.
This is not heating by back radiation, but is commonly also considered a radiation resistance effect.
There is initially a decrease in net radiation heat transfer forcing the temperature to adjust to a new level for a given power transfer level. This is directly analogous to the thermal insulation effect on the ball, where radiation is not even a factor between the ball and insulator, or the opaque gas in the enclosed layer, where there is no radiation transfer, but some energy is transmitted both ways, and net energy (heat transfer) is only outward. The hotter surface of the ball is due to a resistance to direct radiation to space in all of these cases.
Ball with Multiple Shells
If a large number of concentric radiation enclosures were used (still assuming the total exit area is close to the same for simplicity), the ball temperature would get even hotter. In fact, each layer inward would have to radiate a net P outward to transfer the power from the ball to the external final radiator. For N layers, this means that the ball surface would have to radiate:
P’ = (N+1)Po ….(7)
Now from (1), this means the relative ball surface temperature would increase by:
T’/To = (N+1)0.25 ….(8)
Some example are shown to give an idea how the number of layers changes relative absolute temperature:
N T’/To
——————-
1 1.19
10 1.82
100 3.16
Change in N clearly has a large effect, but the relationship is a semi-log like effect.
Lapse Rate Effect
Planetary atmospheres are much more complex than either a simple conduction insulating layer or radiation insulation layer or multiple layers. This is due to the presence of several mechanisms to transport energy that was absorbed from the Sun, either at the surface or directly in the atmosphere, up through the atmosphere, and also due to the effect called the lapse rate.
The lapse rate results from the convective mixing of the atmosphere combined with the adiabatic cooling due to expansion at decreasing pressure with increasing altitude. The lapse rate depends on the specific heat of the atmospheric gases, gravity, and by any latent heat release, and may be affected by local temperature variations due to radiation from the surface directly to space. The simple theoretical value of that variation in a dry adiabatic atmosphere is about -9.8 C per km altitude on Earth. The effect of water evaporation and partial condensation at altitude, drops the size of this average to about -6.5 C per km, which is the called the environmental lapse rate.
The absorbed solar energy is carried up in the atmosphere by a combination of evapotransporation followed by condensation, thermal convection and radiation (including direct radiation to space, and absorbed and emitted atmospheric radiation). Eventually the conducted, convected, and radiated energy reaches high enough in the atmosphere where it radiates directly to space. This does require absorbing and radiating gases and/or clouds. The sum of all the energy radiated to space from the different altitudes has to equal the absorbed solar energy for the equilibrium case.
The key point is that the outgoing radiation average location is raised significantly above the surface. A single average altitude for outgoing radiation generally is used to replace the outgoing radiation altitude range. The temperature of the atmosphere at this average altitude then is calculated by matching the outgoing radiation to the absorbed solar radiation. The environmental lapse rate, combined with the temperature at the average altitude required to balance incoming and outgoing energy, allows the surface temperature to be then calculated.
The equation for the effect is:
T’ = To -ΓH ….(9)
Where To is the average surface temperature for the non-absorbing atmospheric gases case, with all radiation to space directly from the surface, Γ is the lapse rate (negative as shown), and H is the effective average altitude of outgoing radiation to space. The combined methods that transport energy up so that it radiated to space, are variations of energy transport resistance compared to direct radiation from the surface. In the end, the only factors that raise ground temperature to be higher than the case with no greenhouse gas is the increase in average altitude of outgoing radiation and the lapse rate. That is all there is to the so-called greenhouse effect. If the lapse rate or albedo is changed by addition of specific gases, this is a separate effect, and is not included here.
The case of Venus is a clear example of this effect. The average altitude where radiation to space occurs is about 50 km. The average lapse rate on Venus is about 9 C per km. The surface temperature increase over the case with the same albedo and absorbed insolation but no absorbing or cloud blocking gases, would be about 450 C, so the lapse rate fully explains the increase in temperature.
It is not directly due to the pressure or density alone of the atmosphere, but the resulting increase in altitude of outgoing radiation to space. Changing CO2 concentration (or other absorbing gases) might change the outgoing altitude, but that altitude change would be the only cause of a change in surface temperature, with the lapse rate times the new altitude as the increase in temperature over the case with no absorbing gases.
One point to note is that the net energy transfer (from combined radiation and other transport means) from the surface or from a location in the atmosphere where solar energy was absorbed is always exactly the same whatever the local temperature. For example, the hot surface of Venus radiated up (a very short distance) over 16 kWm-2. However, the total energy transfer up is just the order of absorbed solar energy, or about 17 Wm-2, and some of the energy carried up is by conduction and convection. Thus the net radiation heat transfer is <17 Wm-2, and thus back radiation has to be almost exactly the same as radiation up. The back radiation is not heating the surface; the thermal heat transfer resistance from all causes, including that resulting from back radiation reducing net radiation, results in the excess heating.
In the end, it does not matter what the cause of resistance to heat transfer is. The total energy balance and thermal heat transfer resistance defines the process. For planets with enough atmosphere, the lapse rate defines the lower atmosphere temperature gradient, and if the lapse rate is not changed, the distance the location of outgoing radiation is moved up by addition of absorbing gases determines the increase in temperature effect. It should be clear the back radiation did not do the heating; it is a result of the effect, not the cause.
—— End of section 1 ——
My Response
I agree with Leonard. Now for his rebuttal..
Ok, a few words of clarification. I agree with Leonard about the greenhouse mechanism, the physics and the maths but see a semantic issue about back radiation. It’s always possible it’s a point of substance disguised as a semantic issue but I think that is unlikely.
A large number of people are unhappy about climate science basics but are unencumbered by any knowledge of radiative heat transfer theory as taught in heat transfer textbooks. This group of people claim that back radiation has no effect on the surface temperature. I’ll call them Group Zero. Because of this entertaining and passionate group of people I have spent much time explaining back radiation and physics basics. Perhaps this has led others to the idea that I have a different idea about the mechanism of the inappropriately-named “greenhouse” effect.
Group Zero are saying something completely different from Leonard. Here’s my graphic of Leonard’s explanation from one of his simplified scenarios:
Figure 2 – again
From the maths it is clear that the downward radiation from the shell (shield) is absorbed by the surface and re-emitted. Here the usual graphic presented by the Group Zero position, replete with all necessary equations:
Figure 3 – how can you argue with this?
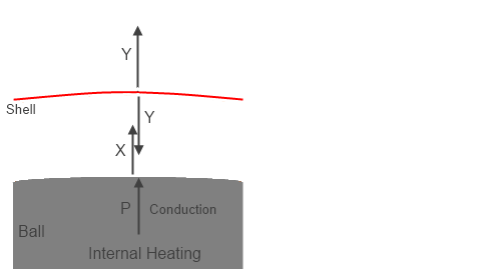
And here’s an interpretation of a Group Zero concept, pieced together by me from many happy hours of fruitless discussion:
Figure 4 – Group Z?
In this case P, the internal heating, is still a known value. But Y and X are unknown, which is why I have changed them from the solution values shown in figure 2.
Now we have to figure out what they are. Let’s make the assumption that the shell radiates equally inwards and outwards, which is true if it is thin (and so upper and lower surfaces will be at the same temperature) and has the same emissivity both sides. That is why we see the upward flux and the downward flux from the shell both = Y.
Because, according to Group Zero, the downward radiation from a colder atmosphere cannot “have any effect on” the surface, I’m going to assume their same approach to the radiation shield (the “shell”). So the surface only has the energy source P. Group Zero never really explain what happens to Y when it “reaches the ground” but that’s another story. (Although it would be quite interesting to find out along with an equation).
So at the surface, energy in = energy out.
P=X ….(10)
And at the shell, energy in = energy out.
X = 2Y ….(11)
In figure 2, by using real physics we see that the surface emission of radiation by the ball = 2P. This means the surface temperature, T’ = (2P/σ)0.25.
In figure 4, by using invented physics we see that the surface emission of radiation by the ball = P. This means the surface temperature, T'(invented) = (P/σ)0.25.
So the real surface temperature, T’ is 1.19 times larger than T'(invented). Because 20.25 = 1.19.
And back to the important point about the “greenhouse” effect. Because the atmosphere is quite opaque to radiation due to radiatively-active gases like water vapor and CO2 the emission of radiation to space from the climate system is from some altitude. And because temperature reduces with height due to other physics the surface must be warmer than the effective radiating point of the atmosphere. This means the surface temperature of the earth is higher than it would be if there were no radiatively-active gases. (The actual maths of the complete explanation takes up a lot more room than this paragraph). This means I completely agree with Leonard about the “greenhouse” effect.
If back radiation were not absorbed by the surface lots of climate effects would be different because the laws of physics would be different. I’m pretty sure that Leonard completely agrees with me on this.
—— End of section 2 ——
Leonard’s Final Comment
I think we are getting very close to agreement on most of the discussion, but I still sense a bit of disagreement to my basic point. However, this seems to be mainly based on difference in semantics, not the logic of the physics. The frequent use of the statement of heat being transferred from the cold to hot surface (like in back radiation), is the main source of the misuse of a term. Energy can be transferred both ways, but heat transfer has a specific meaning. An example of a version of the second law of thermodynamics, which defines limitations in heat transfer, is from the German scientist Rudolf Clausius, who laid the foundation for the second law of thermodynamics in 1850 by examining the relation between heat transfer and work. His formulation of the second law, which was published in German in 1854, may be stated as: “No process is possible whose sole result is the transfer of heat from a body of lower temperature to a body of higher temperature.”
The specific fact of back radiation and resulting energy transfer does result in the lower surface of the cases with radiation resistance going to a higher temperature. However, this is not due to heat being transferred by back radiation, but by the internal supplied power driving the wall to a higher temperature to transfer the same power. The examples of the solid insulation and opaque gas do exactly the same thing, and back heat transfer or even back energy transport is not the cause of the wall going to the higher temperature for those cases. There is no need to invoke a different effect that heat transfer resistance for the radiation case.
An example can give some insight on how small radiation heat transfer can be even in the presence of huge forward and back radiation effects. For this example we use an example with surface temperature like that found on Venus.
Choose a ball with a small gap with a vacuum, followed by an insulation layer large enough to cause a large temperature variation. The internal surface power to be radiated then conducted out is 17 Wm-2 (similar to absorbed solar surface heating on Venus). The insulation layer is selected thick enough and low enough thermal conductivity so that the bottom of the insulation the wall is 723K (similar to the surface temperature on Venus). The outside insulation surface would only be at 131.6K for this case.
The question is: what is the surface temperature of the ball under the gap?
From my equation (6), the surface of the ball would be 723.2K. The radiation gap caused an increase in surface temperature of 0.2K, which is only 0.033% of the temperature increase. The radiation from the surface of the ball had increased from 17 Wm-2 (for no insulation) to 15,510 Wm-2 due to the combined radiation gap and insulation, and back radiation to the ball is 15,503 Wm-2. This resulted in the net 17 Wm-2 heat transfer. However, the only source of the net energy causing the final wall temperature was the resistance to heat transfer causing the supplied 17 Wm-2 to continually raise the wall temperature until the net out was 17 Wm-2. Nowhere did the back radiation add net energy to the ball wall, even though the back radiation absorbed was huge.



I dislike the use of the term back-radiation as well (at least it in the context it seems to be used often). It seems to imply it is somehow the surface radiation (or heat which would be somewhat more accurate) that is “reflected” back to the earth by the atmosphere, which is not a correct representation of it in my opinion.
What is called back-radiation is actually (again, from my understanding of things) mostly atmospheric long-wave radiation measured at the surface of the earth. I would guess it is actually a rather localized effect that it’s value is very dependant on the temperature of the air relatively close to the surface. As a thought experiment – if we packed all the absorbing gases to the first 1 km above the surface – the back-radiation value would initially increase a bit, but this would lead to significant cooling of the surface temperature of the planet as Weinstein wrote).
The terminology would be better, if the atmospheric heat transfer was similar to the shell model you presented, but very large of heat transfer through the atmosphere should be actually due to convection (and some of the heating is actually caused by absorbed solar radiation in the atmosphere I think). In the example of Venus the radiative heat transfer on the surface is probably not even particulaly worth mentioning.
Why I don’t like it being called that particular thing, is that it seems to create some unnecessary confusion how things work. For example a statement, that the surface of Venus is so hot because of all the back-radiation there is. The causality of this statement is upside down for this particular term – the back-radiation is so high, because the temperature of the gases above the surface is so high. The resistance terminology used by Weinstein seems somewhat more descriptive for the process of transfering heat. I would imagine separating all the radiation sources is better for modelling calculations though, but even then why not call it atmospheric downwards radiation and separated reflected radiation (I think clouds reflect some longwave) alltogether. Would avoid misconceptions a bit better in my opinion.
Very good and greatly informative as usual. Please stop semantic discussions and drop the misleading “back radiation” idea 🙂
Two questions:
1. Is the “average height where radiation occurs” anything more than a mathematical layer? After all, the adiabat goes up to the tropopause, below which convection is king
2. Would a doubling of atmospheric mass really have no consequences?
omnologos,
I’m not sure I understand this question, the meaning of “anything more than a mathematical layer”.
We discussed this at Air Vent, and agreed I think that our differences were semantic. The thing is, I can’t see any substance here.
As I’m sometimes solemnly informed at WUWT, the Sun is our energy source and essential to our climate. No-one disputes that. So the air is not a nett energy source.
But accounting for what happens to energy from the sun, as in Trenberth’s diagram, is useful. The individual fluxes follow various laws that relate them to temperature. It is informative to calculate them on that basis, and account for energy balances on the basis that each is heating or cooling according to the energy that it carries.
If you insist that back radiation – an observable, measurable flux – is not heating because there is a larger upward flux, then why stop there? Insolation is balanced by various upward fluxes, so it’s not heating either. In fact, energy flow is a steady state; all the fluxes add to nothing, so nothing is heating anything.
It’s a sterile argument.
Nick Stokes says;
“It’s a sterile argument.”
Leonard is quite correct to identify thermodynamic work as an essential part in the definition of thermodynamic heat.
Heat is not exactly the same as energy.
Anyone taking physics 101 will have thought through the Carnot Cycle to get a grasp of the second law and the meaning of heat transfer.
The definition of heat then, is perfectly well understood.
If someone wants to distort its meaning what harm is done?
Probably in most cases, none!
However if the discussion involves the second law then the distortion causes needless confusion.
If a child calls a whale a fish what harm is done?
What if a biology textbook says the same thing?
A great deal of confusion results from the misuse of definitions.
Nick,
You still miss the point. All of the energy transfer, storage, and back radiation do not heat the ground. It is the input energy combined with the lapse rate and the fact that the elevation of outgoing radiation to space is increased, that is the cause. Since back radiation and the raising of the elevation are tied together (you can’t have one without the other), is may be difficult to see that the driver is not back radiation. Here is an example that may make this more clear. Look at the pure solid insulation case on the ball. Clearly the ball surface got hotter. Thus the wall of the ball radiated at the higher level, however, the insulator absorbed or reflected this radiation in a very short distance (atoms thick), and back radiated it to the ball surface where it was absorbed. There is huge back radiation here, but net transfer of radiation energy of zero (the energy is conducted by molecular vibration). Are you claiming this back radiation contributed to the high temperature? How is this different from the planet cases. The only radiation effect that matters is NET energy transfer.
Leonard,
The lapse rate can’t heat anything. It’s not a flux – it’s just the temperature pattern arising from a balance of fluxes. And the only way “input energy” can heat anything is via a flux.
As you say below, the distinction that we’re talking about can be characterized as eq vs non-equilibrium. And you say you’re talking about the inability of back-radiation to heat the surface at equilibrium, because there is no heat transfer. But my complaint is that that statement is empty – you can never have heat transfer at equilibrium. Then the sun does not heat, a furnace does not heat.
Of course, the sun does clearly warm the surface in the morning. But back-radiation can warm the surface too. If you have a clear dry night and then clouds come over, the surface will warm, from back-radiation.
Nick,
In reply to the comment “Insolation is balanced by various upward fluxes, so it’s not heating either. In fact, energy flow is a steady state; all the fluxes add to nothing, so nothing is heating anything”. I do agree that in the steady state all the fluxes add to nothing, and nothing is heating anything. However, the level of temperatures are determined by the fluxes. Heating implies changing temperature, and I am discussing the equilibrium cases.
Here’s a question of great importance (at least I think so)!
Can a hot non GH gas transfer heat to a GhG?
One answer is of course Yes but this is molecular motion. A wizzy hot O2 hits a cold less wizzy CO2 and the CO2 gets warmer (more wizzy) and the O2 gets cooler (less wizzy). However this is not a photon absorption.
Can the wizzy CO2 eject a photon of LWIR and cool itself if it has not been excited by a photon?
I.e.an a wizzy O2 hit CO2 and excite the CO2 enabling a photon release?
If it cannot emit a photon of IR then it cannot emit heat/energy to space just because it has been made wizzy through conduction with Non GHGs
If this is the case then for hot non-GHGs to have anything to do with cooling the planet they must conduct to a solid,
which can then emit photons
which then might hit a GHG molecule
which will then emit a photon in a random direction (one of which will be space)
I believe a conductive transfer will not enable a photon release.
Confused – I am
Nick,
I think I found the cause of many of our disagreements. You are looking at problems for changing conditions (change one parameter, and how does the system change dynamically). These are non-equilibrium cases, and can be quite different depending on details of internal processes. For these cases, fluxes are not yet balanced, and storage and internal flux transfers drive the system toward equilibrium. For the non-equilibrium system, back radiation may be a factor in how fast the system goes toward equilibrium. For these systems, detail does matter. However, once equilibrium is established, the only factors that define temperatures are net fluxes in and out, and any source of average temperature gradients within the system.
thefordprefect,
The greenhouse gas will emit photons if the surrounding temperature is in the correct range. In fact, the absorption of thermal photons generally results in the energy to be passed to the surrounding gas by collisions. The surrounding gas also can cause the greenhouse gas to emit a photon. This is possible because the gas has a range of molecular speeds at a given temperature (Boltzmann distribution), and some collisions are more energetic and raise the vibration energy state of the greenhouse gas, which then emits to drop back to unexcited state. Thus the absorption and emission are effectively independent events.
thefordprefect,
The concept of local thermal equilibrium requires that the vast majority of energy transfer be collisional rather than radiative. So only a tiny fraction of ghg molecules that absorb a photon emit again before they can lose energy by collision. If that were not true, then the gas would not exhibit a Boltzmann energy distribution. At very high altitude where the gas density is very low, the kinetic energy distribution is no longer Boltzmann.
Leonard Weinstein,
You can’t dismiss back radiation so casually. No one who knows anything about thermodynamics says, when speaking or writing carefully, that back radiation heats the surface. What is said is that for an atmosphere that is not transparent in the thermal IR, the surface must be warmer than the atmosphere if there is a net flow of energy from the surface to the atmosphere and to space. That’s where energy balance diagrams come into play. Or, for that matter, energy flow between parallel planes at different temperatures. That’s described by σ(T1^4-T2^4) where T1 and T2 are the temperatures of the two planes. Note that I have left out emissivity. That’s because as long as the emissivity of the planes isn’t identically zero, which is impossible in the real world, the radiation flux (photon gas) between the planes is not dependent on the emissivity. Note also that you are subtracting the radiation emitted by the lower temperature plane from the radiation emitted by the higher temperature plane to calculate the net flux. This is back radiation in a nutshell. It is a necessary concept.
One might as well ask as to the cause of debt.
Is it due to borrowing or lending?
To hunt for the essential component in any system with multiple interrelated, interdependent aspects tied in a web of cause and effect is not always fruitful.
To highlight one aspect as more essential than another is not always illuminating.
Even when after consideration one finds an aspect that affects the system but is not an effect of the system, and from that assert that it is a cause; one may not have understood much about the system. For instance asserting that sunlight is essential to climate is very true but not all that insightful.
One can ask as to the nature of each aspect, what it causes and what it is caused by. Whether it is one of many aspects that is of moment to, or diagnostic of the state of the system. One may be able to formulated a description of the system that takes one apsect as a starting point, or another apsect as a starting point. Eventually one has to include all significant aspects and all their interdependencies. In that process one may introduce abstract properies that can be neither located in the system nor measured from it.
I think there is more than semantics at stake whenever one aspect is asserted to be more essential than another, when one model is asserted to be superior to another when in truth both lead to the same conclusions albeit via different insights. That we choose one aspect to take precedence over another may tell us much about ourselves but little else.
Alex
SoD,
You keep saying this. To quote Inigo Montoya in The Princess Bride: “You keep using that word. I do not think it means what you think it means.”
Do you actually believe that Wood was correct in 1909 and that the optical properties of horticultural greenhouse glazing are unimportant? That would certainly be a surprise to BC Greenhouse Builders Limited
[emphasis added]
Horticultural greenhouses and the planetary greenhouse effect are the same. Planets are just better insulated than horticultural greenhouses.
DeWitt Payne
Here is a paper that you once advised me to read.
The way I read the paper is it gives massive support for the conclusions of the famous Woods experiment.
Basically the project was to find if it made any sense to add Infra Red absorbers to polyethylene plastic for use in agricultural plastic greenhouses.
Polyethylene is IR transparent like the Rocksalt used in Woods Experiment.
The addition of IR absorbers to the plastic made it equivalent to “glass”
The results of the study show that( Page2 )
…”IR blocking films may occasionally raise night temperatures” (by less than 1.5C) “the trend does not seem to be consistent over time”
Click to access penn_state_plastic_study.pdf
Bryan,
I’ve told you many times that paper is not the be-all and end-all with respect to the validity of the Wood experiment, yet you keep bringing it up. A single layer tunnel greenhouse is poorly insulated. The effect of IR blockers in the film on temperature will be small and difficult to measure. Dew or frost formation on the film on the outside or condensation on the inside at night is an additional confounding factor as that will act as an IR blocker on non-blocker containing film. The point, though, is that even if the effect isn’t consistent, it is there. And that in itself contradicts Wood. Whether using IR blocking polyethylene film is economic in tunnel greenhouses or not is irrelevant.
DeWitt Payne
“The point, though, is that even if the effect isn’t consistent, it is there.”
No!
I would think that an anything grand enough to be called an ‘effect’ would always be there.
Does gravity in you vicinity behave in a capricious fashion?
Grand enough to be called an ‘effect’???? What is that supposed to mean? You’re grasping at very weak straws. Your analogy to gravitation is ludicrous. In the real world, lots of effects that exist are difficult to measure because the magnitude of the effect is small relative to the measurement error, both random and systematic. Einstein’s General Relativity comes to mind. You need very precise atomic clocks or very precise astronomic orbital measurements to detect it.
DeWitt,
You have not stated anything that disagrees with what I stated. However, it is a matter of fact that many people do exactly say the back radiation heats the surface. Since the net average energy flow is up, the back radiation is not heating it, but is deminishing the NET flow so that the solar energy has to drive the temperature higher to overcome it. This is exactly like a thermal insulating layer, and you don’t call that heating the surface with back conduction do you? I think you are caught in the semantics of the issue.
SoD – is there such a thing like an average layer from where radiation is emitted to space or is it like the proverbial mathematical point, a useful concept that doesn’t exist in reality?
PS no takers for question #2?
Omnologos,
On your #1, there is no single layer where radiation is emitted to space for real atmospheres. The layer was defined as the integrated average location where the temperature of a black body causing outgoing radiation would match the total absorbed solar radiation. It appears that even with large variation in actual outgoing radiation, this approach gives a good approximation of the net process. It is a useful concept.
On #2, the doubling of the mass of the atmosphere would have an effect on the surface temperature. However, the details are important as to what the doubled atmosphere is like. Did the % composition of all gases, including CO2 and water vapor stay the same, or just O2 and N2 increase? If all gases held the same % increase, the surface temperature would increase significantly. In the end, it is only the increase in average altitude of outgoing radiation that would be the main cause of the increase, since the lapse rate (which is a gradient, not a level) would be about the same. If the absorbing gases did not increase along with the O2 and N2, the increase in temperature would be less, but probably still slightly larger that present.
Hey, Leonard Weinstein gets almost everything right here, congratulations!
But I feel I must quibble with two incorrect statements I noticed, despite the generally good description of the state of affairs in the main body of the text. Neither has anything to do with the argument, but I thought I ought to address the misstatements on their own there.:
* zero gravity does not imply no convection. Convection is driven by pressure differences that can be created through temperature gradients even within a system at constant pressure and under no gravity. Horizontal convection on Earth happens and has no relationship to gravitational forces. T
* a 1/4 power is very different from a “semi-log like” relationship. If the relationship was logarithmic, the increment from 1 to 10 would be the same as the increment from 10 to 100, but in this case the increment from 10 to 100 is quite a bit larger. Logarithmic relationships are extremely slow-growing, slower than any power.
Other than that, as SoD put it, “I agree with Leonard”. I wonder who he thinks might disagree with him?
Arthur Smit,
You couldn’t possibly be more wrong. If the system isn’t gravitationally bound, there would be no pressure difference and no convection. Take a sealed container in orbit. Heat one end. Does that cause convection. Not hardly. The container will have no pressure gradient because the pressure equilibrates on a time constant proportional to the dimension of the container and the speed of sound. There will be a temperature and density gradient, but without gravity, there will be no pressure difference and no convection. If you turn the air circulation fans off and light a candle in the space station, it barely glows because the combustion is supported by diffusion, not convection.
https://flightline.highline.edu/iglozman/sciphy/sphinx/candle_zerog.htm
“buoyant convection” is absent in zero-g, but that is not the same as convection in general. Convection is simply the motion of parcels of a fluid relative to one another – Navier Stokes and all that. Gravity is not required though it certainly has a strong effect when it’s present. This was only a quibble anyway.
Your definition of convection is a tad broad. Most people think of advection when they hear convection. Microscale convective or eddy diffusion, which is all you would get in micro-gravity, is orders of magnitude slower than advection. What would happen in true zero g isn’t clear either. Even the Navier-Stokes equations distinguish between diffusion and advection.
Arthur,
The ball models I used were uniformly heated (and I said so), so no lateral convection. Planets are clearly different, and I never said otherwise. Also, I did not mean to be making a mathematical comment on the relationship, just commenting that the increase in temperature grows much slower than the value of N. You took it more literally than I meant, but I am glad you paid close attention to the details.
Arthur,
Many people on both sides of the AGW issue disagree with what I said. Heating by back radiation has remained a talking point for a lot of uninformed comments. That is why I said what I did here as clearly as I could.
Can you find a specific example of somebody saying something that clearly disagrees with your presentation (other than the “dragon slayers” and friends)?
Leonard Weinstein,
You’re the one tied up in semantics, not me. You’re as bad as Gerlich and Tscheuschner at nitpicking (See chapter 3 of their paper). At least you don’t use loose terminology as a disproof of the concept. Even in thermodynamics the word “heat” has several meanings. You and Bryan insist that the term means one and only one thing. You’re wrong. There are at least five sensible and widely-used but mutually-inconsistent technical meanings (not to mention innumerable nontechnical and metaphorical meanings) of the term “heat” in thermodynamics.
Bryan will blow a gasket when I source this (somewhat modified) quote ( John Denker, Modern Thermodynamics, Chapter 16 ), but he has yet to do more than quote some textbook(s) and rant about Denker’s qualifications. Neither of which is proof that Denker is, in fact, wrong about this.
Leonard Weinstein,
Your case is that back radiation cannot “heat the surface because “heat” can mean one, and only one, thing. Accusing me of arguing semantics requires more than a little audacity when your whole case is semantic not substantive.
DeWitt Payne
There are some people like Dr Roderich Graeff, Professor Claes Johnston and John Denker who are uncomfortable with various aspects of mainstream physics.
John Denker has only one supporter that I know of.
You are correct to point out that it is possible that John is correct.
However if you study the history of science as outlined by Thomas Kuhn you will realise that the existing paradigm will only give way if it fails to account for some critical problem.
Denkers wish to get the rest of mainstream physics to depart from orthodox thermodynamic definitions will not catch on in my opinion.
‘if its not broke don’t fix it’ is a wise saying.
Besides SoD wishes these discussions to stay within the framework of the broad mainstream science.
Denker is hardly Claes “photons don’t exist” Johnson or Roderich “perpetual motion” Graeff. He isn’t throwing out a century or more of modern physics. He is pointing out logical inconsistencies in how thermodynamics is taught, not in how thermodynamics works.
But it is broken, or aren’t you aware of the agony suffered by countless (hyperbole, they are obviously countable) undergraduate Chemistry majors during the rite of passage called Physical Chemistry?
DeWitt Payne
Denker wants more than just a change in terminology.
In the last manifestation of his book he saw no need for the Third Law of Thermodynamics.
His new version (you linked) shows he is now not so sure.
The index shows this topic on page 53.
However the page is now blank!
If someone particularly wanted to say that heat transfers spontaneously from a colder to a warmer surface then they would have to make clear that they were not using the term in the orthodox thermodynamics way.
Work also has a specific meaning in thermodynamics.
“aren’t you aware of the agony suffered by countless undergraduate Chemistry majors during the rite of passage called Physical Chemistry?”
Its a little hard to explain to them that no matter how hard they tried they did not do any thermodynamic work.
It may be that thermodynamic terminology could be improved but this would require international agreements.
So stick with the orthodox terms or else set out right at the beginning your own set of definitions.
Does anyone disagree with the values shown in figure 2?
Energy emitted by the surface = 2P, along with the consequences for the surface temperature; T’ = (2P/σ)0.25
Any dissenters?
The energy emitted by the surface is 2P but the heat (net energy) being transferred is P. For a solid insulation, with the same internal surface temperature, the energy emitted from the surface is also 2P (a very short distance, order of atomic, into the insulation), and P of this energy is emitted backwards, with the net heat being transferred also P (in this case by solid conduction). For the opaque gas case, with the same surface temperature, the energy emitted from the surface is 2P, for a very short distance, but the heat being transferred is P (back radiation equals 1/2 radiation out, or P, but the molecular interaction transports the net energy). Why do you consider the radiation case basically different?
Excuse the error, but the insulation and opaque gas case have back radiation 2P, not P. The conduction by phonon and molecular interaction are P. Thus we actually have 3P going out, 2P of which is radiation, and 2P which is back radiation to cancel 2P of the 3P, with the remaining 1 P (the net input) carried by conduction or diffusion.
Leonard Weinstein,
I said nothing about conduction. My question is a simple one.
It is aimed at identifying:
a) who understands physics yet differs on semantics and
b) who doesn’t understand physics and uses semantics to hide that fact
If you are really interested in how the conduction and radiation shield case are different.. well, the two principal equations are different.
Case A – the ball with no insulating layer nor radiation shield, T’ = (P/σ)0.25
Case B – the ball with a radiation shield, T’ = (2P/σ)0.25
Case C – the ball with an insulating layer, T’ = (Ph/k)+(P/σ)0.25 (your equation 5) where h = thickness of the insulating layer and k is the thermal conductivity.
Of course, you agree because you wrote these equations.
Interesting to see what someone like Bryan might say.
In case B compared with case A I might say that the radiation shield has “increased the temperature” of the surface.
Bryan will say that people should study the Carnot cycle a little more and won’t commit to one equation or the other and definitely not supply an English sentence to describe the two cases.
Just my prediction. I’d like to be wrong. Here’s hoping I am wrong.
And so..
if we say b = T’ with radiation shield / T’ of “bare ball” (i.e., the ratio of temperature increase due to a radiation shield)
and c = T’ with insulating layer / T’ of “bare ball” (i.e., the ratio of temperature increase due to an insulating layer)
b = 20.25 = 1.189
c = P0.75σ0.25h/k + 1
– so the two cases are only equal when P0.75h/k = 12.3, which only happens under special circumstances.
Are they “basically different”? I think it depends on the definition of “basically different”. Both increase the surface temperature due to their role in effectively reducing energy loss from the surface. But they do it via a different physical process and according to a different mathematical equation.
One could ask as to how:
a pursuer can dog when no cannines are involved,
the ancients could truck before lorries came to be,
the sun may shine without a reflective surface,
some could fail to notice that verbs are not well constrained by homonymous nouns.
Alex
scienceofdoom,
I agree with what you stated.
scienceofdoom,
The point I was making is that in all three cases there is back radiation, and it is absorbed by the wall, but in the cases of a solid insulator and opaque gas, no one refers to the back radiation as causing the higher temperature. However, the pure radiation transfer does have people describing the back radiation as heating the surface. All three process transfer the net heat differently. I agree different equations need to be used. The point I make is that it does not matter which process occurs, only net energy fluxed determine the resultant temperature, and back radiation (which occurs in all cases) is a result of, not cause of the increase in temperature.
scienceofdoom
“Bryan will say that people should study the Carnot cycle a little more and won’t commit to one equation or the other”
I think it is almost impossible to understand the second law without understanding the Carnot cycle.
I would recommend a post from you on this very topic and I say this in the most constructive and helpful way.
It is after all the almost universal introduction to the second law.
Leonard has gone out of his way to show backradiation,backconduction and back convection do not transfer heat to a warmer surface from a colder atmosphere.
The equations that Leonard provided will of course vary with the nature of the insulating layer.
What heat transfer processes are appropriate,conductive,convective or radiative will depend on the specifics.
As an example air has an almost constant value of heat capacity (Cp) in the atmospheric range of temperature (250K to 350K)
In contrast the heat capacity(Cp) of CO2 changes by 13% in this range This is because of the increased radiative activity of CO2 at higher temperatures.
It seems clear that this would have to be taken into account in a particular calculation.
I was right.
scienceofdoom
There is no evidence that you have ever studied the Carnot cycle and that’s not something to be proud of!
Bryan,
Do you disagree with the values shown in figure 2?
Energy emitted by the surface = 2P, along with the consequences for the surface temperature; T’ = (2P/σ)0.25
scienceofdoom
I dont have any problems with the equation.
Here are some additional calculations for you to think about.
The light hearted way equations are used in climate science is certainly liberating.
I thought perhaps I might do likewise and show the difference between the first and second law of thermodynamics.
Using KT97 numbers.
Total solar insolation at Earth orbit = 1368W/m2
This radiation is very nearly black body
KT maintain that this is thermodynamically equivalent to 4 lots of 342W/m2.
How can we test if this is true?……- a possible experiment!
There is no problem with the first law for indeed 4 X 342 = 1368W/m2
Take a cube of capacity one cubic metre filled with (say) water at 20degrees Celsius.
The container is a black body absorber
Test one
Arrange for a radiation source of exactly 1368 W/m2 to be incident on one face of the container.
The other five faces covered with a ‘perfect’ insulator
With perfect radiative coupling the maximum temperature that could be reached can be worked out from
T = (P/σ)0.25 ….(1)
Where To (K) is absolute temperature, P (Wm-2) is input power per area of the cube, and σ = 5.67×10-8 (Wm-2T-4) is the Stefan-Boltzmann constant.
gives T = 396K or 123C
Discretion can be used to stop this part of the experiment once its outcome is clear .
The heat flow is into the cube as you would have expected.
Test two
Arrange for four radiation sources of exactly 342 W/m2 to be incident on four faces of the container.
The other two faces covered with a ‘perfect’ insulator
With perfect radiative coupling the maximum temperature that could be reached from the incident radiation can be worked out from equation above
Giving T = 279K or 6C
So even though the cube is absorbing 1368 Joules every second its temperature is dropping.
The heat flow is now out of the cube.
Despite the fact that KT maintain that 1368W/m2 insolation is thermodynamically equivalent to 4 lots of 342W/m2 we can see that it is not.
Have you never been exposed to statistical thermodynamics? The Carnot cycle is completely unnecessary for understanding of the second law when viewed from the perspective of statistical thermodynamics. The Carnot cycle is classical thermodynamics. The field has since moved on. Apparently you haven’t.
DeWitt Payne
To approach the second law initially via SM would seem a very awkward way to go about it.
Via the Carnot cycle it can be done in physics 101 but via SM the required maths hurdle would push that back to physics 301.
Can he name some physics departments that ignore the Carnot route?
Eli must agree with DeWitt. The problem with the Carnot cycle is that it ignores molecules. It is basically moves a fluid through a set of equilibria looking only at the net balance at each position. On the molecular level, that is not what is happening.
The Carnot cycle is elegant, and historically important, but it is training wheels for thermo. Time to grow up Brian and Leonard
So DeWitt Payne and Eli think its time to drop the Carnot cycle.
Apparently statistical thermodynamics contradicts classical thermodynamics!
We should ‘move on’.
The way I understand it is there is no contradiction whatever only alternative ways at looking at the same situation.
The Carnot cycle is simpler and still very relevant when looking at atmospheric heat transfer.
My next post will explain this further.
In fact the Carnot cycle(CC) gives the best basic structure to build atmospheric heat transfer theory around.
Combine CC with parcel theory as a used to derive the DALR
Earth surface heated by Sun.
Air parcel in contact with Earth surface will leave with the characteristic temperature of the surface.
1. Isothermal expansion stage
The concept of an air parcel is almost universally adopted to describe the temperature profile of the troposphere.
The parcel expands isothermally absorbing heat from the Earth surface
Air parcel considered to be in hydrostatic equilibrium is used to analyse thermodynamic behaviour of troposphere.
Air parcel in hydrostatic equilibrium means it is either stationary or moving with constant velocity i.e. no unbalanced force acts on body.
Given a small nudge the air parcel will rise under balanced forces as Newtons First Law states.
The dry adiabatic lapse rate(DALR) can be derived for dry air by combining the laws of thermodynamics with the hydrostatic condition.
DALR = – g/Cp = – 9.8 K/km
Derived from stationary condition showing DALR does not depend on convection being present.
2. Adiabatic expansion stage.
For ascending parcel
The air parcel does work Pdv in expanding the air parcel.
This work is supplied by the internal energy of the air parcel causing the temperature inside the parcel to drop.
This loss in internal energy is stored in the atmosphere (surroundings).
3. Isothermal Contraction stage
On ascending the slight loss of energy by radiation becomes much more significant at higher altitudes as heat is radiated to space.
The thinner colder air becomes more ‘transparent’ to long wavelength infra red
4. Adiabatic contraction stage
For descending parcel (or back convection if you like)
The air parcel is slightly denser than surrounding atmosphere and so descends.
The surrounding atmosphere does work Pdv compressing the air parcel.
This results in an increase in the temperature of the parcel.
The internal energy of the air parcel rises causing the temperature inside the parcel is increasing.
This gain in internal energy is matched by the loss of atmosphere(surroundings) energy.
The parcel arrives back at the start of the cycle .
The cycle can now be repeated
With water vapour present in air lapse rate decreases and is called the environmental lapse rate and an average figure of around -6.5 K/km is obtained
The average effective radiation altitude(AERA) is taken to be about 5 Kilometres where the temperature is 255K.
By working back from AERA to the surface the air temperature increases to give the average surface temperature of 278K or 15C.
Other points to notice is that during adiabatic stages no significant amount of heat enters or leaves the air parcel.
Heat absorbed in the isothermal expansion at Earth surface
Heat emitted during isothermal contraction at TOA.
This means that near reversible conditions are observed for the whole cycle.
No cycle can be as perfect as the Carnot cycle but the atmospheric transfer is reasonably modelled by it.
Here are two modern papers where the Carnot cycle is very much part of the analysis.
Click to access rspa.2009.0581.full.pdf
ftp://texmex.mit.edu/pub/emanuel/PAPERS/Physics_Today_2006.pdf
No Lamb Chop, the Carnot cycle is to radiative transfer like Newtonian Mechanics is to General Relativity, there is less in the former than the latter, and when you hit something that involves the latter you are screwed and deluded if you only consider the former.
scienceofdoom,
One reason I brought the subject of the effect of back radiation effect on heating (or not) the surface, is the previous discussions we had on Venus. It so happens that the near total absorption length for thermal radiation from the hot
surface on Venus occurs in a very short distance. In fact, as I stated in my final response in the writeup, the hot surface is radiation is 15,510 Wm-2, and back radiation is between 15,510 and 15,503 Wm-2 to result in the net heat transfer of 17 Wm-2, part of which is conduction and/or convection. The maximum temperature due to net radiation alone is only 0.2 K for this case, and likely is much smaller. For a lapse rate on Venus of 9 K per km, this means the effective distance the radiation penetrates is <22 m (and this is out of 50,000 m to radiation out level). This is very close to the totally opaque gas case. In the limit, it is the same process. For that ideal case, as for the pure conduction in a solid case, no one talks about back radiation being the cause of a hot surface, even though is still exists (and exactly balances outward radiation). Yet, several very bright people still keep saying it is the back radiation that is the cause of the hot Venus surface.
I just noticed a mistake I made several places. 15,503 should be 15, 493 Wm-2. I am surprised no one caught it. This slightly changes the delta T and absorption length, but both are still very small.
Leonard Weinstein
thefordprefect,
The greenhouse gas will emit photons if the surrounding temperature is in the correct range. In fact, the absorption of thermal photons generally results in the energy to be passed to the surrounding gas by collisions. The surrounding gas also can cause the greenhouse gas to emit a photon. This is possible because the gas has a range of molecular speeds at a given temperature (Boltzmann distribution), and some collisions are more energetic and raise the vibration energy state of the greenhouse gas, which then emits to drop back to unexcited state. Thus the absorption and emission are effectively independent events.
DeWitt Payne
thefordprefect,
The concept of local thermal equilibrium requires that the vast majority of energy transfer be collisional rather than radiative.
is an excited co2 molecule warm or just excited? the molecules in the enclosure will be thermally at the same temperature (same average velocity)
So only a tiny fraction of ghg molecules that absorb a photon emit again before they can lose energy by collision. If that were not true, then the gas would not exhibit a Boltzmann energy distribution.
but gasses as I understand them do not exhitit a Boltzmann distribution they emit discrete frequencies the measured outward radiation from earth has a modified Boltzmann distribution because the earth is a BB radiator and the unabsorbed waveklengths gets through to be part of the upward spectrum
At very high altitude where the gas density is very low, the kinetic energy distribution is no longer Boltzmann.
————————————————-
Sorry people still not sure on this one.
Obviously 1 photon in to a co2 molecule will almost immediately give 1 photon out. If a collision occurs with co2 that has just “absobed ” one photon surely that collision can result in between no transfer to full transfer of atomic vibration of co2 to kinetic energy of o2 (say) depending on the collision type. But if less than 1 photon’s worth of energy is transfered to thermal motion then that would leave the impossibility of a co2 molecule being partially excited, never being able to absorb another photon nor able release what is left.
I do not understand how internal atomic vibrations can transfer to thermal kinetic energy.
Here’s a thought.
lets put some CO2 + O2 into space and hold it there with magic gravity i.e. it’s not going to expand and cool and the gravity souce has no thermal properties.
Now heat the mixture using a thermally conductive hot probe
Does this gas cool once the probe is removed. I think it may not! (think thermos flask without container to radiate).
Now start with the same gas and hit it with radiation of co2 absorption wavelengths. obviously the co2 will absorb then retransit the same wavelength as the initial stimulation. But then will:
1) the mixture warm thermally by transfer of energy from co2 to o2 then slowly cool by transfer of energy from o2 to co2 to radiation
2) or will the emissions of radiation stop as soon as the stimulus stops
I favour 2)
What is the energy of collision required to excite a co2 molecule to a state where it can emit radiation?
But individual molecules have different energies. That energy distribution is, for large numbers of molecules in a small enough volume, the Boltzmann distribution. Assuming that the energy of excitation is higher than the average energy, the majority of the molecules will not be in an excited state. The concept of temperature is a thermodynamic bulk property and has no meaning for an individual molecule.
You don’t understand correctly. For a gas having a Boltzmann energy distribution does not require a complete black body emission spectrum at all wavelengths. The maximum flux at any given wavelength cannot exceed the black body flux for that temperature, but there is no requirement for emission at wavelengths where the structure of the molecule does not allow emission.
Obviously. But they do. In fact, the Virial Theorem requires it. The proof is that the heat capacity of a molecular gas isn’t constant with temperature. It increases as the energy content increases to levels high enough to populate rotational and vibrational energy states. These are called additional degrees of freedom. In the limit, the amount of energy in each degree of freedom that is significantly populated is equal to the energy in every other degree of freedom. CO2, for example, has three translational, two rotational and four vibrational degrees of freedom. The two bending vibrational modes are only slightly populated at normal surface temperature. At Venusian surface temperature, all 9 modes are populated.
That would depend entirely on whether the temperature was high enough for emission to occur at allowed wavelengths.
No. O2 has a microwave emission spectrum (60GHz,used for remote temperature sensing by satellite) so even at quite low temperature, the gas would cool by radiation.
Leonard Weinstein,
Radiation resistance is an empty concept. There is no such animal. An object at a given temperature and with a given emissivity will emit the same amount of radiation whether it is in deep space or surrounded by a shell at a higher temperature than deep space. Whether the object’s temperature is stable is determined by the energy balance, not some completely unnecessary and potentially confusing concept of ‘resistance to radiation.’ As Alexander Harvey pointed out so eloquently above, you attempt to disentangle inextricably entangled concepts in your futile denigration of back radiation. Like Humpty-Dumpty, you seem to think that words mean only what you want them to mean, nothing more and nothing less.
DeWitt,
A better choice of words would be that the surface temperature increases in the presence of a source of back radiation (or other heat transfer resistance). This is due to the fact that there would be a net reduction in radiation (or conducted heat transfer) at the initial temperature. Thus the continual supplied power (the actual only source of the increase in temperature) raises the surface temperature (by not being removed as fast as it is supplied) to bring the net emitted energy to the supplied energy for equilibrium.
If you say the back radiation heats the surface, you have to say back radiation heats an insulated surface, or an opaque conducting gas over a surface also, because both also have back radiation. Heating, as I define it, and which is technically correct, implies heat transfer, which can only be hot to cold. Use of the term heating otherwise may be used and understood (and commonly is), but gives the wrong emphasis of the actual physics. I just want the words to correctly convey what is actually doing the heating. If you do not like the word radiation resistance, just use reduced net radiation, or radiation heat transfer.
DeWitt,
Since both you and Alexander Harvey think my words mean only what I want them to mean, nothing more and nothing less, then why does there seem to be such a big issue on the subject. It continually keeps coming up. I think most of the problems derive exactly from common uses of terms that mean different things to different people.
DeWitt,
Do you think back radiation is the cause of the high surface temperature on Venus?
Leonard Weinstein:
You claim the “location of outgoing radiation is moved up”
1. Where is the observational evidence of this?
2. Why do theoretical calculations on MODTRAN show the temperature profile of the atmosphere in the troposphere and stratosphere does not change at all between CO2 concentration inputs of 350 and 3500:
http://forecast.uchicago.edu/Projects/modtran.html
The profile temperature gradient (lapse rate) would not change. This gradient is due to Cp and g, with condensation effects included. The absolute level would change if there were no feedbacks, but skeptics (including me) think the feedbacks are likely negative, reducing the small effect from CO2 alone.
Hockey Schtick
I’d love to hear your theory of radiative transfer.
Here’s the first set of observational evidence that I could find in a few minutes (from Grant Petty, A First Course in Atmospheric Radiation, p.9).
Panel b & c have light shades corresponding to cold temperatures and dark shades corresponding to warm temperatures.
Panel b is in the IR window where the atmosphere is not very opaque, while panel c is at a wavelength where water vapor is very opaque.
[And panel a, in the visible spectrum is what you would see with the naked eye, except in black & white, and so shows reflectivity of the various climate features to sunlight].
Why is the 6.7 μm band so cold compared with the 10.7 μm band? Because the atmosphere is more opaque at this wavelength and so the emission of thermal radiation is coming from a higher (and colder) part of the atmosphere.
Now your specific question would be finally demonstrated by finding two shots of the same location with different amounts of water vapor.
However, for you to believe that this is not the case would be quite extraordinary. How could it possibly not be the case if the emission of radiation from a more opaque wavelength is a lower temperature than the emission of radiation from a less opaque wavelength?
Perhaps if you can articulate some sensible reason why – then it might be worth searching for relevant data…
Probably because you don’t know what this tool actually does. What do you think it is calculating?
Is it calculating the temperature of the atmosphere from the change in concentration of “greenhouse” gases, or is it calculating the spectral intensity for prescribed atmospheric temperature profiles?
Please provide a link to a published paper that proves from observations that increased GHGs raise the “location” and thereby lower the temperature of outgoing radiation.
for e.g. a paper in which
“Now your specific question would be finally demonstrated by finding two shots of the same location with different amounts of water vapor.”
Hockey Schtick,
Finding papers about physics basics is difficult because these papers got published when the physics basics were being worked out or controversial. Papers from more than 40 years ago are hard to find online.
Did you understand the graphic I reproduced and why it was relevant?
Why do you think the emission of radiation at 6.7 μm is lower than at 10.7 μm?
Do you doubt that the temperature of the atmosphere reduces at altitude?
Do you doubt that the transmissivity of the atmosphere reduces with more radiatively-active gases?
If you can explain what part of the physics doesn’t make sense to you it will make finding relevant data easier.
Here is the average OLR for January 2009 measured by CERES:
You can see that the tropics with high humidity have much lower OLR than the subtropics with very low humidity. Why do you think that is?
“Why do you think that is?”
I’ll bite. It’s clouds. The ITCZ is full of thunderstorm clouds that go up to at least the top of the troposphere. Radiation from those cloud tops will be quite low compared to clear sky emission.
I don’t think that necessarily provides a reply to Hockey Schtick.
SoD,
What I think is irrelevant to the simple question of whether or not a peer-reviewed paper exists that confirms theory with observations that the “characteristic emission height” shifts to a higher altitude due to increased greenhouse gases. Are you claiming there are papers “from more than 40 years ago” with this observational evidence? Are you suggesting that nobody has written a peer-reviewed paper with satellite data showing e.g. “two shots of the same location with different amounts of water vapor” to confirm or quantify the theory?
Secondly, how does the theory explain these observations?
Hockey Schtick: For a discussion of how GHG’s raise the height of the “characteristic emission level” (the altitude from which most photons escaping to space are emitted), you could see Lindzen’s E&E article “Taking Greenhouse Warming Seriously”. Google the article title or try this link.
http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.160.448&rep=rep1&type=pdf
Lindzen is in agreement with Leonard.
If the earth had no atmosphere or an atmosphere that lacked GHG’s, all of the photons emitted to space would be emitted by the surface. When you add GHG’s, some of the photons are emitted from higher than the surface. In this trivial case, adding GHGs clearly raises the average emission level.
You can also approach this problem from the point of view of the mean free path of a photon emitted by CO2. When there is little CO2 around (low pressure or low mixing ratio), a photon can travel a long distance without being absorbed and possible escape to space. This perspective is useful near the surface of Venus (90 atm of CO2, 2 million times the earth), where the mean free path is so short that there is little temperature difference between absorbing and emitting molecules. So there is little vertical radiative heat transfer (upward radiative energy flux minus downward) = o(T2^4-T1^4) despite the high temps.
Hockey Schtick,
Can you find a peer-reviewed paper proving that gravity is responsible for Titan orbiting Saturn?
The thing is, a very long time ago researchers found that if you add more molecules of a substance that absorbs in a certain band the transmissivity of the gas is reduced. The Beer-Lambert law expresses this mathematically. This law dates from the 1700s.
And a very long time ago researchers had found that bodies at terrestrial surface temperatures emit radiation mainly in the 4-50 μm range – in the late 1800s.
A very long time ago researchers found that water vapor and CO2 absorb wavelengths between 4 – 50 μm. I don’t know how long ago.
And the knowledge of the atmosphere reducing in temperature with altitude was definitely ancient knowledge, although the temperature profile of the troposphere was probably not known in much detail until the 1800s.
Now if all of these points are correct then it follows inescapably that the average emission of thermal radiation moves to a higher colder point in the atmosphere with more radiatively-active gases. Who would try to publish a paper on this? Who would want to publish a paper proving gravity is responsible for Titan orbiting Saturn?
There are thousands of papers on measurements of top of atmosphere radiation since satellites were launched in the late 1970s. Probably some of those incidentally demonstrate the piece of information you are so concerned about.
Just looking at the OLR values already provided to you should prove to you that the humid tropics have a lower OLR than the dry sub-tropics. Why is this?
You can go to the CERES website yourself and produce values of TOA radiation and humidity.
Spend some time and prove it wrong. Or explain which bit you think is wrong.
Otherwise why should I sift through 100 papers to find you a paper when you can’t be bothered to even explain your area of doubt let alone do anything yourself?
Nature has a way of confounding the most beautiful of theories. Apparently, you are unable to cite a single paper based on satellite observations to confirm or just as importantly quantify from observations the shift in “characteristic emission height” / “OLR trapping” due to increased GHGs.
Please elaborate on how the theory explains the following observations:
1. CO2 and outgoing OLR have no correlation

2. OLR, if anything, has decreased since 1974

3. There is even less of a correlation between CO2 and outgoing LW radiation in the tropics

4. ARID stations warm faster and cool faster than WET stations
5. Negative water vapor feedback found from satellite observations (Lindzen & Choi, Spencer & Braswell) and radiosonde observations (Paltridge)
Hockey Schtick,
It’s hard explaining to people who haven’t grasped a basic theory why their “evidence” only demonstrates their confusion over what said theory would imply.
If you can explain why the OLR of the tropics is so much lower than the OLR of the subtropics I will have a go at addressing your points (= explain observational evidence provided with “your theory”).
Actually if you manage to explain this you might understand the relevance or otherwise of your “points”.
It won’t do any good, but here’s my explanation of the altitude dependence of emission, with satellite images from many different wavelengths:
http://blog.chron.com/climateabyss/2010/12/the-tyndall-gas-effect-part-2-seeing-is-believing/
Whoops #2 should have been
2. OLR, if anything, has increased over the satellite era:

On your point 4, I suggest you look up enthalpy of moist air.
To be frank, I think virtually everyone here has missed some of the most fundamental aspects of Earth’s greenhouse effect. I also think that some of the flaws of Leonard’s physical understanding have went unexplored because of the rambling over semantics.
Basically, it is the difference between the longwave radiative fluxes at the ground surface and the top of the atmosphere that defines the greenhouse effect. That is, G = (σTs^4)-OLR, where Ts is the surface temperature, and OLR is the outgoing longwave radiation at the top of the atmosphere. Here, G is expressed in flux units. This is, in principle, directly observable and well-defined.
Simple layer models employing Stefan-Boltzmann type simplifications are not a useful approximation for a real planetary atmosphere, for instance in a simple one-layer model using Ts = Te[(N+1)^0.25]… one cannot in the real world determine the surface temperature with knowledge of the emission temperature of the planet alone, and full knowledge of the surface temperature must account for the temperature and absorber distributions, along with the spectral properties of the absorbing substances. Moreover, the key mechanism behind the greenhouse effect is not just that you can have a high temperature emitter sending radiation to the surface, but that you can efficiently reduce the outgoing radiation to space by means of a cold emitter aloft.
This is why low clouds are horrible contributors to the greenhouse effect, as they emit at a temperature very near that of the surface (it is noteworthy that the absorptivity of a substance is not very dependent on temperature). In contrast, “high temperature” radiation from the surface is absorbed by high clouds, which then emit at a very cold temperature, and thus correspondingly weaker radiation. Viewed from space, this is manifest as “blips” in the emission spectrum seen by the observer, as shown in this figure from my site:
One of the best ways to heat the Earth would be to enrich the upper troposphere with very cold, thick clouds, but ones that aren’t too reflective.
The area within these blips in the image above represents energy that is no longer being emitted to space at the same temperature. The only way to compensate for the decreased emission is to increase emission via temperature change in other transparent spectral regions. If the atmosphere were isothermal, there would be no ditches in the spectrum, no reduction in outgoing radiation, and no need for the planet to become warmer than its emission temperature, even in the presence of opacity. If the temperature structure were inverted, as in the stratosphere or Antarctic winter, the ditches would be “inverted” and increasing greenhouse gases could create a “negative greenhouse effect” in which decrease surface temperature would need to come at the expense of increased emission aloft.
The greenhouse effect also determines how the longwave emitted from the surface is transformed from its initial emission all the way to its final destination out to space. The greenhouse effect concept could be perfectly well-defined even on a planet like Jupiter that had no “surface.”
The argument that a “lapse rate” can heat a planet is one of the most misinformed lines of thought on the internet. Nick’s response is correct. The temperature structure of the atmosphere is the result of the radiative heating and cooling rates by SW and LW radiation, in addition to the dynamics that gives freedom for convection to set the temperature profile that must arise due to the instability caused by radiative-only balance. In order to export all of the energy radiated by the ground surface out to space in an optically thick atmosphere (with only radiation), the resulting temperature gradient would need to be very steep, resulting in a greenhouse effect at least twice its current value of ~33 C. This radiatively driven profile becomes unstable to convective activity, which then redistributes heat in the vertical, in order to assure stability to convection. If there was no source of atmospheric opacity, the atmospheric temperature structure would necessarily lead to a rather isothermal structure.
In contrast, if the optical thickness of the atmosphere is very high (as on Venus), then there is a very high tropopause, and one needs to extrapolate down the adiabat a further distance to reach the surface. You can also get a higher tropopause with a low lapse rate (e.g., by releasing latent heat of condensate), as with the present-day tropics, but if one were to inject enough CO2 in the atmosphere then one can go all the way up to the top of the atmosphere without a transition to a temperature inversion (even with ozone). It is also possible to whipe out the troposphere altogether, or create a situation in which convection cannot occur, as in nuclear winter where a sufficiently good incoming solar absorbing layer is placed high in the atmosphere.
I believe some of these “lapse rate” people starting with idiots like Steve Goddard think that you can arbitrarily set the starting point of the adiabat anywhere you want high up in the atmosphere, and then “explain” the high surface temperature if the distance between the top and bottom is large enough. But convection only establishes an adiabat insofar as the conditions are conducive for convection to occur. There is a large literature discussing the radiative and dynamical factors governing a planet’s tropopause height, and it’s not a trivial issue.
The surface back-radiation is largely a red herring. It must necessarily contribute to the surface temperature, since it represents a real physical flux of energy, but it is just one term in the surface energy budget (along with upward IR, and evaporative/sensible fluxes), and one that also changes with the atmospheric temperature. Consider for example, a regime in which thick clouds covered the lower atmosphere, such that the bottom boundary radiated like a blackbody at its temperature. In this case, one can increase the CO2 all they want and not directly impact the downwelling IR (this is not to suggest this is really relevant for most locations on Earth). However, you still reduce the outgoing radiation, and must warm the whole atmospheric profile in order to reach equilibrium again. This includes the lower atmosphere, which then can radiate more downwelling IR to the surface. However, one cannot think about this problem without considering the energy budget of the planet as a whole, or the efficient heat trapping that is determined by the temperature profile.
Chris,
It is not necessary to call people idiots just because you disagree with them. As to the statement the greenhouse effect is caused by the difference in radiation between the ground and upper atmosphere, this is nonsense. Whatever would cause the ground to be hotter than the upper atmosphere would cause the exact same difference, so it is not a cause but effect. If you look at the radiation from one layer that is warmer to a layer that is cooled, you can represent the heat transfer by a single parameter that has Thot to the 4th minus Tcold to the 4th in the equation. The only heat flux is the net. This is not two separate equations, but one. Thus to consider back radiation as a distinct effect rather than a part of a single effect is where you go wrong. The effect of an insulating solid layer, and a totally opaque gas and a back radiating gas all do exactly the same thing to a surface receiving a fixed amount of power. They result in the surface temperature increasing to overcome the reduction in heat transfer that would occur to the bare surface case if the temperature did not increase. However, these are all distinct processes. One is solid thermal heat transfer, the second is gas diffusion heat transfer, the third is radiation heat transfer. The boundary conditions for all three can be different, or the same, depending on the exact process. However, the equations are all different. The first two depend only on:
1) Supplied power
2) Thickness of insulating layer or opaque gas layer
3) Effective heat transfer conductivity
The radiation case depends on:
1) Supplied power
2) Effective net heat transfer (independent of spacing)
However, for an atmosphere on a planet, there is a mix of convection and radiation heat transfer, and other factors such as clouds and water evapotransporation and condensation, and even aerosols. However, atmospheres are special cases in that they have an imposed temperature gradient if they are well enough mixed (due to Cp, g, and wet effects), and this lapse rate is essentially independent of which process transfer the energy. There has to be absorbing and emitting gases to radiate at higher altitude, or all radiation would be from the ground to space once the atmosphere was warmed. This imposed gradient, and the effective average location of outgoing energy exactly determine the temperature increase.
Oh, please. There is no other whatever and you should know it. No matter how hard you try, it’s not possible to separate cause and effect in this case. Pretty soon we’ll be back to the idea that the temperature of the surface is determined only by the gas pressure at the surface.
Hi Leonard,
If you refer to this sentence:
“Basically, it is the difference between the longwave radiative fluxes at the ground surface and the top of the atmosphere that defines the greenhouse effect.”
when inferring:
“As to the statement the greenhouse effect is caused by the difference in radiation between the ground and upper atmosphere, this is nonsense.”
There is a difference in meaning between defines and caused by.
Chris continues:
“That is, G = (σTs^4)-OLR, where Ts is the surface temperature, and OLR is the outgoing longwave radiation at the top of the atmosphere. Here, G is expressed in flux units. This is, in principle, directly observable and well-defined.”
I think he states that the GHE is not just a temperature effect but also a flux effect; that seems to be worthwhile point.
You end with:
“However, atmospheres are special cases in that they have an imposed temperature gradient if they are well enough mixed (due to Cp, g, and wet effects), and this lapse rate is essentially independent of which process transfer the energy. There has to be absorbing and emitting gases to radiate at higher altitude, or all radiation would be from the ground to space once the atmosphere was warmed. This imposed gradient, and the effective average location of outgoing energy exactly determine the temperature increase.”
I concern myself with your term “imposed.gradient”
I think this may mean different things to different people insofar as whether gradient should imply just the abstract slope or the extended decline of temperatures with height conforming to that slope.
I believe that the abstract slope is given as the solution to an equation based on adiabatic considerations. The extent of the slope is a different question. The abstract slope in itself does not inform us as to the physical extent of the gradient. The abstract notion does not impose upon the realised physical extent but it does constrain the gradient to the slope throughout its extent.
I believe that the extent of the lapse rate gradient, the height of the tropopause, is determined by dynamical and radiative factors. Further that the radiative regime up to the tropopause is characterised by a local radiative deficit which is resolved dynamically.
I am not saying that you think otherwise, but some seem to, and the term gradient is ambiguous as to this point due to bearing both abstract and physical interpretations. Your use of “this lapse rate” seems to me to refer to your prior “imposed temperature gradient” and is open to the interpretation that an extended physical gradient is imposed by the abstract slope or lapse rate. I think any that make that interpretation would be mistaken for my given reasons. That said; provided that they continue to follow your argument they may not be misled but there is no guarantee that they will indeed follow..
Alex
Hi Chris,
“Moreover, the key mechanism behind the greenhouse effect is not just that you can have a high temperature emitter sending radiation to the surface, but that you can efficiently reduce the outgoing radiation to space by means of a cold emitter aloft.”
That is a point well put on at least two levels. It is insightful as to the physics but also as a guide to an approach to our thinking. At first it seems to pose something antithetical which needs to be resolved at a higher level to obtain a fuller understanding. A mechanism that causes temperatures higher than that of an IR transparent atmosphere, or the no atmospere case, at one location, does, in combination with other aspects of the system, cause temperatures lower than that case at another. IR opacity doesn’t, and in reasonably earthlike systems perhaps cannot, produce a general increase in temperatures.
I believe I would be correct in saying that previously it was the presence, the necessary formation, of cold IR active regions aloft that was highlighted as the instructive insight and not the downward path thermal radiation (back raditaion). I think this is certainly true of Manabe’s instruction (c. 1970s).
That is not to say that the view looking down from space is inherently more insightful than the view looking up from the surface. They don’t exist in isolation.
I think that the formation of atmospheric regions that are colder than the transparent atmosphere case gives an insight that there much be some mechanism that has the properties of IR activity as it would be difficult to explain the persistence of such temperature regions otherwise. I think we could have inferred atmospheric radiative activity based purely on temperature observations and the laws of thermodynamcs even without the wit to measure it directly. I have such idiosyncrasies.
There is an opportunity to understand the system by focussing on its apparent contradictions, or conflicting aspects, and resolving them at some higher level.
The lapse rate is an important aspect, it is real, measurable, and in most part a persistent quality. In turn is has not just these real but abstract aspects. We can deduce an abstract part based on an assumption of adiabatic behaviour yet doubt that its formation is a result of adiabatic behaviour alone. We can observe convection and we infer a transport of sensible heat and have to resolve this with the abstract adiabatic case which seems to be posed on the absence of such transport.
Simple models of an IR active atmosphere prescribed to have both a lapse rate and tropopause characteristically have local radiative deficits. Each volume aloft up to the tropopause emits more IR than it absorbs. This is antithetical to any notion of heat trapping and so needs resolution. One part can be resolved by recourse to convection, perhaps exploiting some deviations in the lapse rate sufficient to maintain an energy equilibrium. Another part by recourse to the asymmetry of the system. Above there is the cold depths of space, below a surface and atmosphere of middling temperatures which give rise to an asymmetry in regional atmospheric IR absorption that must be weighed against a near isotropy in that regions contribution to the radative IR field. That is to say that radiative absorption in a asymmetric IR field when coupled to near isotropic emission is an aspect of the system and gives rise to and arises from both the radiative energy deficit, the fluxes that balance it and the persistence of a lapse rate.
Hopefully we can come to see heat trapping as one aspect of a system that has a heat releasing aspect (local IR energy deficit) and perhaps that the two are mutually inevitable when viewed at some higher level. Consideration of one aspect in the absence of the other may not only fail to explain the whole but also its own existence and hence explain nothing.
It is my view that the tendency to establish an essential aspect of the system and assert it as the explanatory factor has had its day. It has lead to failures to instruct and impaired reasoning. From backradiation, to lapse rates that will their own formation and persistence, it engenders debates irresolvable in their inherently simplified view.
I expect I have read too much of mine own thinking into what you wrote yet I note that you have considered the circumstances under which the effect of atmospheric IR activity could not only expand or diminish a surface warming effect but also reverse the sign to produce surface cooling. I think such extensions are worthwhile when their apparent difference can be resolved into an entirety.
Alex
Chris: You can DEFINE the greenhouse effect to be the difference between surface and TOA upward radiative flux. This definition ensures that the greenhouse effect exists, but it doesn’t immediately explain why an atmosphere with GHGs will have a higher surface temperature than an atmosphere without GHG’s. In a FAQ explaining “what is the greenhouse effect?” in AR4, the IPCC talks about DLR.
I don’t recall having much of an issue with the AR4 explanation (which has been a few years since I looked at it) but I did think it could have been better. That said, the AR4 is not a textbook, and I don’t really consider that to be the best of sources on “textbook matters” such as this.
Frank:
– I agree with Chris on what is meant by the GHE.
– The reason there is a temperature difference between the upper atmosphere and ground level is because air cools when it rises (or is forced to rise), as long as there is an atmosphere. This is true with or without GHGs.
– However, unless there are GHGs, this temperature difference does not affect the rate at which radiation is released by the earth & atmosphere. The GHGs link these two issues.
– When the GHGs are added, the TOA point is defined; and this is the point at which the outgoing flux must equal the incoming flux, to achieve a steady state. If this is not initially the case, the system is not in steady state; and the resulting imbalance of incoming and outgoing radiant energy will increase all temperatures, until there is a balance. At that point, the temperature difference (which is roughly set by the DLR) remains the same, but the TOA temperature should now be the same as the “pre-GHG” ground-level temperature.
Hi Frank,
From AR4 WGI Chapter 1 FAQ 1.3:
“The Sun powers Earth’s climate, radiating energy at very short wavelengths, predominately in the visible or near-visible (e.g., ultraviolet) part of the spectrum. Roughly one-third of the solar energy that reaches the top of Earth’s atmosphere is reflected directly back to space. The remaining two-thirds is absorbed by the surface and, to a lesser extent, by the atmosphere. To balance the absorbed incoming energy, the Earth must, on average, radiate the same amount of energy back to space. Because the Earth is much colder than the Sun, it radiates at much longer wavelengths, primarily in the infrared part of the spectrum (see Figure 1). Much of this thermal radiation emitted by the land and ocean is absorbed by the atmosphere, including clouds, and reradiated back to Earth. This is called the greenhouse effect. The glass walls in a greenhouse reduce airflow and increase the temperature of the air inside. Analogously, but through a different physical process, the Earth’s greenhouse effect warms the surface of the planet. Without the natural greenhouse effect, the average temperature at Earth’s surface would be below the freezing point of water. Thus, Earth’s natural greenhouse effect makes life as we know it possible. However, human activities, primarily the burning of fossil fuels and clearing of forests, have greatly intensified the natural greenhouse effect, causing global warming.”
The topic here seems captured by two of those sentences:
“Much of this thermal radiation emitted by the land and ocean is absorbed by the atmosphere, including clouds, and reradiated back to Earth. This is called the greenhouse effect.”
insofar as they state a definition of the GHE, plus the following two sentences:
“Analogously, but through a different physical process, the Earth’s greenhouse effect warms the surface of the planet.”
which states a consequence of the GHE, and finally:
“Without the natural greenhouse effect, the average temperature at Earth’s surface would be below the freezing point of water.”
which states the implicative link.
I don’t think there is anything amiss here. The implication seems sound. If the GFE as defined ceased to be, the described elevation in temperature should cease to be.
I think there is a difference between implication and cause and effect. The FAQ does not I think demonstrate cause and effect, I doubt that it was intended to argue at that depth.
In earthlike worlds, which is all the FAQ covers, downward path thermal radiance (their reradiation, your DLR) is I think a sound indicator of the existence of an elevated surface temperature. Further I believe its is both necessary and sufficient evidence of the existence of an elevated surface temperature for our world. Yet I will hold that evidence is no more the same as proof, than implication is the same as cause and effect. There remains a case to be argued, and people do.
The radiance the originates in the atmosphere and is absorbed at the surface is the answer to an important question:
“When the average of the radiated, sensible and latent energy fluxes from the surface exceeds the solar and mechanical fluxes into the surface, by what additional process is an average flux balance maintained?”
I would argue that the path thermal radiance in its totality, all the radiative energy that originates in the atmosphere, is necessary for the elevation of the surface temperature and sufficient for a production of the latent and sensible fluxes. That the path thermal flux acts in response to the latent and sensible fluxes and that they in turn act in response to the totality of the path thermal fluxes. Not in isolation but to a sufficient degree whereby I cannot claim for any to be the essential cause of another. The cause that is not also an effect.
It is my current personal opinion that too much emphasis is given by some, particularly those with a science background, to identifying some unique and essential cause. This may work well for them but the populace may be used to more subtle thinking. People familiar with systems with multiple parts most of them necessary, many acting in response to a multiplicity of actions by the the others.
Alex
Alexander:
So your conclusion is that scientists are too stupid to think about complicated things. So that is why they only develop simplistic and unsophisticated ideas, like:
– Quantum mechanics
– General relativity
– Superconductivity
– Lasers
– GPS location systems
It’s a pity we didn’t set the English majors or computer scientists to working on all this stuff: I’m sure they would have done a more complete and sophisticated job at it.
“Much of this thermal radiation emitted by the land and ocean is absorbed by the atmosphere, including clouds, and reradiated back to Earth. This is called the greenhouse effect.”
The main problem with this (the AR4) definition of the greenhouse effect is that it implies that the strength of the greenhouse effect ought to be proportional to the amount of thermal radiation absorbed by the atmosphere and reradiated back to earth.
The counterexample is high and low clouds, discussed earlier. Take clouds with arbitrary (but equal) albedo and unit IR emissivity. Low clouds have little effect on surface temperatures (because they emit to space at almost the same temperature as the Earth) while high clouds have a large effect (because they emit at much lower temperatures/intensities). Yet low clouds produce strong back-radiation and high clouds produce weak back-radiation.
Focusing on back-radiation as the key to the greenhouse effect will get you into trouble very quickly.
Bryan on July 24, 2012 at 10:39 pm:
.
No they don’t. You have invented a claim. You don’t have the slightest idea what their paper is about.
KT show the globally annually averaged values across the surface. You could just as well multiply those numbers by the surface area of the earth to get the total energy per second. Or multiply those numbers by the surface area and the number of seconds in a year to get the total annual energy values.
The values simply identify the surface energy balance and the top of atmosphere energy balance, partitioned between different processes.
You can measure these values.
See What’s the Palaver? – Kiehl and Trenberth 1997: “KT97 is a paper which attempts to quantify the global average numbers for energy fluxes at the surface and the top of atmosphere. And to quantify the uncertainty in these values.”
For those who are confused on climate basics like this I recommend reading a good textbook like Hartmann, Global Physical Climatology.
Reading a good textbook beats inventing ideas about a paper that attempts to isolate and quantify one idea in climate science.
For Bryan, please go ahead and quantify the correct values for globally annually averaged surface and top of atmosphere fluxes.
KT97 represents a dead world.
If the globally averaged temperatures were as stated there would be no photosynthesis possible.
No biosphere, very little photochemistry.
Which I suppose is why no reference to them can be found in the energy distribution.
KT 97 should be grouped with bare rock Earth and snowball Earth as unfit for life as we know it.
Once again.. For Bryan, please go ahead and quantify the correct values for globally annually averaged surface and top of atmosphere fluxes.
I realize you prefer to get on your soap box and proclaim instead of providing any science, but if you want dialogue then you should provide some values yourself.
Otherwise readers will assume you don’t provide values because you don’t know the right answer. And if you don’t know the right answer how do you know KT97 is the wrong answer? Etc.
In response to my statement:
Bryan said on July 24, 2012 at 10:39 pm, proving me happily wrong, and lo there was much rejoicing:
Just to finish up this happy encounter, please supply an English sentence to describe what happens to the surface temperature when the radiation shield is added to the ball in space.
“The radiation shield ________ the surface of the ball”.
Chris Colose
You do write a load of tosh.
“it is noteworthy that the absorptivity of a substance is not very dependent on temperature.”
Can you explain why the heat capacity of Carbon Dioxide (Cp) varies by 13% from 250K to 350K?
“a greenhouse effect at least twice its current value of ~33 C.”
The 33K figure is a meaningless number, wrongly calculated, as you should well know.
Using KT97 figures
I know of no-one who is now prepared to defend this figure by coherent logical argument.
1. In the absence of strongly active IR gases the cloud formation chemistry would be different hence no 308W/m2 reflection guaranteed
2. No IR absorbers in atmosphere a further 268W/m2 reaches the ground
3. The ground albedo or 0.17 would be reduced to nearer 0.12 because of much larger IR component of incident radiation.
This brings the figure down from 33K to around 16K
SoD says;
“Just to finish up this happy encounter, please supply an English sentence to describe what happens to the surface temperature when the radiation shield is added to the ball in space.”
“The radiation shield ________ the surface of the ball”.
The radiation shield insulates the surface of the ball.
I will assume that there are ‘surroundings’ outside the shield
This means that if the shield and the ball are at the same temperature – no change.
If the surroundings are at a higher temperature than the ball the shield will reduce the heat flow from the surroundings to the ball.
Balls surface temperature rises more slowly
If the surroundings are at a lower temperature than the ball the shield will reduce the heat flow from the ball to the surroundings.
Balls surface temperature drops more slowly or if constantly internally heated will rise to new equilibrium temperature.
Bryan,
Do you not understand the equations you claim to have accepted?
The surface temperature of the ball, T’, is higher in the case where the radiation shield is added.
That is, 20.25=1.19 > 1.
What English sentence would you use about the surface temperature of the ball after adding the radiation shield?
“The radiation shield insulates the surface of the ball.” is not a statement about how the surface temperature changes.
A reasonable person would think that you don’t want anyone to know your views on the absolute basics of physics of this simple scenario. Why is it so hard to state your position?
Please supply a description in English about the surface temperature of this object that indicates that you understand the word “increase”, or that clarifies what you really think about the ratio 20.25 : 1.
Or maybe you want to rethink your acceptance of the equations.
SoD
Perhaps you did not read this part of my previous post
“If the surroundings are at a lower temperature than the ball the shield will reduce the heat flow from the ball to the surroundings.
Balls surface temperature drops more slowly or if constantly internally heated will rise to new equilibrium temperature.”
I don’t think its possible to be clearer than that.
This argument really strikes me as the same conversation about ocean “acidification” where as reducing the pH is making it more “acid” even tho it is actually pulling the value closer to neutral.
The earth heats the atmosphere but adding insulation creates a temporary imbalance requiring the amount of total flux to be reduced for a time period until the new equilibrium is reached. The new insulated temperature will be higher if measured at the same point after equilibrium is reached. I think we all see and agree with that.
Reducing outflow is not heating just as reducing pH is not acidifying the ocean in real terms but the General public would and do accept both as adequate descriptions. In fact most would say a blanket warms them up when they get in bed.
Exactly. It’s all semantics. It depends on what you mean when you say acidify or heat because those words do not have one and only one meaning. However, some people who should know better, Gerlich and Tscheuschner come to mind, use semantics as part of their ‘proof’ that the greenhouse effect doesn’t exist.
DeWitt Payne says
“Exactly. It’s all semantics.”
However as Leonard Weinstein says above he is using language which would be recognised as technically correct.
Why is it that yourself , Nick Stokes and Chris Colose seem uncomfortable about using the correct terminology?
When all three of you get a chance to fully explain what you mean a lot of the disagreement vanishes.
But why cause the confusion in the first place?
I would think that after the climategate scandal the supporters of IPCC science should frame their point of view with unquestionable rigour if they want to be taken seriously.
Bryan,
Sure. The reason is that the bending modes of CO2 become more active over that temperature range. More degrees of freedom, higher heat capacity.
Specifically, the fraction of molecules in the first bending mode excited state at 250K is 2.16%. At 350K, the fraction is 6.46%. The calculation of the actual change in heat capacity based on increasing fraction in the excited state is not trivial. Fully populated, the heat capacity at constant volume would increase from 5/2R to 7/2R or 40% and the ratio of Cp/Cv would decrease from 1 + 2/5 to 1 + 2/7. While this is related to emission of radiation at 15μ because the bending mode changes the dipole moment of CO2, the heat capacity will also increase when the symmetrical stretch mode becomes active. That mode does not produce radiation. So your idea that the heat capacity change is related to emission/absorption of radiation by CO2 is not correct.
DeWitt Payne
“While this is related to emission of radiation at 15μ because the bending mode changes the dipole moment of CO2, the heat capacity will also increase when the symmetrical stretch mode becomes active”
When CO2 absorbs a 15um photon what do you think happens?
I would like to hear your explanation
I think the bending mode is activated.
When CO2 emits a 15um photon what do you think happens?
I would like to hear your explanation
I think the bending mode is relaxed.
The bending stretching modes can also be affected by collisions.
I did a calculation along the lines of Maxwell – Boltzmann for the degree of freedom dependence on temperature and got very close to the measured 13%.
While the energy is initially deposited in the bending mode, it doesn’t stay there. Back in the early 1970’s people thought they could direct chemical reactions by activating particular molecular modes using lasers. It didn’t work. The redistribution of energy to the other modes was too fast. The time scale for redistribution is femtoseconds. And the more energetic molecule doesn’t stay in that state for long because it redistributes its energy to other molecules by collision. That time scale is on the order of a microsecond or less.
It loses 1.324 E-20J. The point is that a molecule with a total energy level of greater than 1.324E-20 J is considered to be in an active state for purposes of emitting radiation at 15 μm. That doesn’t mean it’s sitting there barely moving but vibrating. The states are mixed. You clearly don’t understand the implications of quantum mechanics. Molecules are not, in fact, balls on springs. A molecule with a total energy of 1E-20J does not need to have and additional 1.324E-20J added to emit a 15μm photon. It only needs 0.324E-20J.
Again you fail to understand the basic principles of quantum mechanics.
DeWitt Payne says
“Molecules are not, in fact, balls on springs. A molecule with a total energy of 1E-20J does not need to have and additional 1.324E-20J added to emit a 15μm photon. It only needs 0.324E-20J.”
With increasing temperature the degrees of freedom become activated.
At low temperature, only the translational mode is available and fits in well to the classical kinetic theory of gases.
At higher temperatures certain molecules like CO2 have the possibility of additional modes.
Quantum mechanics is required for a complete picture but there are several little bendy-stretchy animations of balls showing almost classical motion and it seems to work most of the time.
For an isolated molecule with values above after emitting a 15um photon it would have no translational energy left.
Where would it get the additional 0.324E-20J.? to emit the photon.
I would suggest by a collision but you seemed to have ruled that one out
Correct. But if it had a total energy of 1.324E-20J and it was all in the bending mode, it couldn’t have any translational energy too or the total would be higher. You can’t explain this sort of thing in plain language without running into apparent paradoxes. The correct explanation is mathematical and involves solving the Schrödinger wave equation. The states of the molecule at an energy level high enough for a photon to be emitted are superposed until the wave function collapses and you now have two wave equations, one for a 15 μm photon with its associated energy and momentum and a another for the molecule with energy and momentum reduced by the energy and momentum of the emitted photon.
I see now why you are so hung up about the Carnot cycle. You’ve never actually progressed beyond classical mechanics.
Why do you say that? Of course it’s a collisional energy transfer. 99.99+% of all energy transfer in a molecular gas at local thermodynamic equilibrium is by collision. It has to be or it wouldn’t be at LTE.
Strictly speaking, you can’t even say a particular molecule has a specific energy at a given time because energy and time are linked by the uncertainty principle as are momentum and position.
DeWitt Payne
I said
I would suggest by a collision but you seemed to have ruled that one out
you said
“Why do you say that? Of course it’s a collisional energy transfer.”
Well perhaps I said it because above…….
The bending stretching modes can also be affected by collisions.
But you said
“Again you fail to understand the basic principles of quantum mechanics.”
DeWitt Payne
I find it hard to understand that you do not believe that the emission /absorption spectrum of CO2 is related to the stretching/rotational degrees of freedom.
This in turn directly links to the specific heat capacity of CO2.
I think you will find this link helpful
http://farside.ph.utexas.edu/teaching/sm1/lectures/node70.html
Bryan,
Your reading comprehension sucks. Energy levels do not contribute to the heat capacity unless they are populated. Did you notice they didn’t talk about CO2 in your link? Of course not.
The symmetrical (7.2 μm) and asymmetric (4.26μm)stretch modes aren’t significantly populated until you get to a lot higher temperature. The population of the first excited symmetric stretch state at 250K is 0.03% and 0.3% at 350K. The population of the first asymmetric stretch level will be orders of magnitude less.
Two, the symmetric stretch mode doesn’t change the dipole moment of CO2, so it doesn’t play a direct part in emission/absorption. It does play a part in CO2 lasers, though. The 10.4 μm laser wavelength results from a transition from the first asymmetric stretch energy level at 2349 cm-1 to the first symmetric stretch energy level at 1388 cm-1. But there is no emission of 1388cm-1 (7.2 μm) radiation. No emission, no absorption. If you had a significant population of the first symmetric stretch level, you might get significant absorption of 10.4μm radiation, but you don’t.
The only modes in play are the in-plane and out-of-plane vibrations. So yes for the Earth’s atmosphere and temperature, the <b<stretching/rotational transitions have little to do with emission/absorption of CO2 in the thermal wavelength band or the heat capacity. There is a small absorption of solar radiation amounting to about 2% of the total incident radiation.
Any increase or in molecular energy affects all the modes of a molecule. That was the point of my comment of your lack of understanding.
DeWitt Payne
Its healthy that you admit you can make mistakes (see Neal)
I can make make mistakes and hope I can be realistic and honest enough to learn from an interaction.
There is no point in having ego baggage.
In this case however I remain to be convinced that you are correct.
The v2 mode of CO2 can be activated by two modes.
By collision (classical kinetic theory) or by absorption of a 15um photon (non classical).
As temperatures rise the classical prediction for Cp is increasingly out of line with measured values pointing to the non classical reality.
In the case of our atmosphere
CO2 will thermalise 15um in the mixed N2, O2 atmosphere causing local heating.
H2O vapour (the major player) provides several longer wavelength exits for the thermalised energy.
So back to my first post here; for a mainly CO2 type atmosphere a constant lapse rate cannot be used.
Simpson’s rule or similar must track the changing Cp for an actual heat transfer calculation as temperatures change.
For our air atmosphere the variable CO2 component is too small to change the overall Cp which can be taken as a constant.
However the main point I raised a few months ago here is that the bulk thermodynamic quantity Cp also includes the radiative effects.
The DALR of g/Cp = – 9.8K/km gives the temperature change differential.
Since the radiative properties are still ongoing they must be subsumed into Cp.
For LTE to hold the radiative effect of adjacent gas volumes must cancel.
Only at the top of atmosphere will this no longer hold and a net heat loss to space results.
If an actual heat tranfer calculation for DALR conditions extracts the radiative transfer from the total heat tranfer is undertaken.
For the other non radiative transfer comonents must a modified value of Cp be used?
The question is not whether a mode can be activated by collision, which is true, but whether at a given temperature what fraction of the molecules will be activated. I have calculated the dimensionless heat capacity from the full statistical mechanical formula. At 250 K, the bending mode contributes 0.673 degrees of freedom of a maximum of 2 to the dimensionless heat capacity. The symmetric stretch is 0.0298 of 1 and the asymmetric stretch is 0.00026 of 1. At 350K, it’s 1.117, 0.133 and 0.006. The asymmetric stretch contribution is insignificant, the symmetric stretch contributes some. The majority, ~80%, of the increase comes from the activation of the bending modes. Again, photon emission from the symmetric stretch mode is forbidden because the dipole moment doesn’t change.
So the DALR for the Venusian atmosphere isn’t constant. That was known back in 1969 and probably earlier: http://journals.ametsoc.org/doi/pdf/10.1175/1520-0469%281970%29027%3C0219%3ATALRIT%3E2.0.CO%3B2
Again, whether an energy level absorbs or emits radiation has no effect whatsoever on its contribution to the heat capacity. The symmetric stretch mode does not absorb or emit, yet it contributes to the heat capacity when the temperature gets high enough. The heat capacity of molecular nitrogen will increase when the temperature gets high enough for the stretching mode to become populated, but molecular nitrogen will still not absorb or emit radiation. The bending mode of CO2 contributes no more and no less to the heat capacity because it can absorb or emit radiation. If there is an excess of radiation absorbed or emitted, and there is no other source of energy like convection, the gas will warm or cool, but the rate of heating and cooling is the same whether the mode of energy change is radiation or convection.
Oh, wait. Your link did mention CO2 in the fine print. Which wasn’t much other than CO2 had a non-classical heat capacity, which I explained in my first post on the subject. There was nothing there to suggest that the stretch modes of CO2 contributed to its heat capacity at normal temperature. So as usual, your link wasn’t really what you thought it was and I’m right and you’re wrong.
DeWitt Payne says
“Oh, wait. Your link did mention CO2 in the fine print. Which wasn’t much other than CO2 had a non-classical heat capacity, which I explained in my first post on the subject.”
What exactly did you think non classical meant?
It means that the bending stretching modes are increasingly being populated by photons and that a quantum mechanical interpretation is now required.
When you look at the rates of increase in the heat capacity of CO2 they begin to tail off as the temperatures move beyond the thermal range.
Your contention that absorption/emission of radiation is unrelated to the bending stretching modes is clearly wrong.
Further your lack of understanding (if believed) undermines the case that absorption of IR radiation can increase thermal energy.
What do you think heat capacity means?
You say
” Energy levels do not contribute to the heat capacity unless they are populated. ”
My point was as the temperature rises they are increasingly BEING populated and this contributed directly to the increased values of Cp that are confirmed by measurement
So, I’m right and you’re wrong.
Bryan,
Pay attention.
Populated by photons??? As I said, your understanding of quantum mechanics is severely lacking. No photon exists until it is emitted. You have molecules with sufficient energy to be in a particular excited state or not. At atmospheric pressure, being in an excited state rarely leads to emission and a molecule that absorbs a photon distributes that energy by collision to the surrounding molecules far more often than it emits a photon. The bending modes start to contribute to the heat capacity of CO2 in the temperature range from 250 to 350 K. The stretching modes don’t because they’re not significantly populated in that temperature range.
Again, your reading comprehension sucks. There are two bending modes and two stretching modes. The bending modes are degenerate but the stretching modes are not. Conflating the modes into “bending stretching modes” is wrong. I never said or implied that absorption/emission is unrelated to the bending modes of CO2. Those are the modes responsible for the absorption and emission of 15 μm radiation.
I did say that the symmetric stretch mode doesn’t contribute to absorption/emission of radiation. That is correct. I also said that the asymmetric stretch mode contributes little to absorption and hardly at all to emission at Earth surface temperature. That is correct too.
Only the bending modes are being significantly populated in the range 250-350K as I demonstrated above.
You’re still wrong and apparently willfully misinterpreting my comments too.
omnologos:
From the perspective of radiative transfer theory, the altitude of the effective radiative temperature is defined roughly as the point at which an IR photon has a better than half chance of escaping out to the emptiness of space. Because the atmosphere does not have a sharp edge, this notion is a bit fuzzy, but could be defined as the point which has opacity value 1 (for the photons in question – fitting into the CO2 emission windows) as measured from outer space down into the atmosphere. Another way of putting it: The “top of the atmosphere” is one mean free path away from outer space, for the IR photons.
If you double the amount of atmosphere, you should also be increasing (in absolute numbers) the number of CO2 molecules. So that moves the altitude of the top of the atmosphere to higher (and colder) levels. In order for the effective radiating temperature to be the same as it was 150 years ago, the ground level has to warm up enough to satisfy equation (9): the ground-level temperature moves up, equation (9) pushes the effective temperature up, until it matches the pre-atmospheric temperature of the Earth.
Neal,
It only matters a little if you double the total atmosphere and thus double the mass path of CO2 or just add sufficient N2 and O2 to double the mass of the atmosphere. The altitude where the optical density reaches 1 from space to ground will still go up a lot, meaning so will the surface temperature. I actually ran some calculations on the Venusian atmosphere. Removing half the CO2 and replacing it with N2 only reduced the surface temperature a few degrees, less than 20, IIRC. For significant cooling, you have to reduce the total mass.
DeWitt,
I made the argument you just stated (but with no calculations to back it) on a previous writeup that scienceofdoom put up for me about Venus. At that time I argued that both composition and total atmosphere mass (and thus height) affected outgoing radiation altitude, but once there was a reasonable amount of absorbing gas, adding a larger amount has much less additional effect. I stated that for Venus, a fairly large change in CO2 (but replacement with a non absorbing gas, so that total mass remained the same) would only result in a relatively small surface temperature change. I do not remember if you agreed with me or not at the time, but many, including Rabbit and Chris disagreed with me. Those two especially do not seem to get it that the outgoing altitude time lapse rate gives the temperature increase. It is, in the end, solar heating, and the altitude increase in conjunction with the lapse rate, not back radiation, that results in the temperature increase. The back radiation is a consequence of the cause of the increased temperature, which is resistance to direct radiation to space at the ground.
DeWitt:
Yes, the opacity is proportional to the integrated CO2 mass. But the question then is, How is the distribution of CO2 molecules changed by different ways of adding mass?
– If all atmospheric constituents are doubled, it is clear that everything scales up (though not by a factor of 2) and the CO2 top moves up.
– If you double the non-CO2, the CO2 remains well-mixed throughout the atmosphere, and moves out with the N2 & O2? Using an adiabatic model for the gas, I find that the scale height is:
[(gamma)/(gamma-1)]*(k*T(0))/(mg)
where m is the average mass of a molecule.
So if you double everything, m doesn’t change, so the spatial distribution of molecules scales linearly with the total number: Hence the opacity-1 length should halve.
– But if you double the non-CO2 but leave the CO2 unchanged, you also barely change the value of m (because CO2 is such a minority). So the spatial distribution is doubled for non-CO2 and unchanged for CO2, so the opacity-1 length should be unchanged.
– Likewise, removing half the CO2 and replacing it with N2 should barely change m (and the scale height), but double the opacity-1 length.
So I don’t understand your claim. How does your model describe the number density of molecules as you vary the constituents?
Neal,
I was exposed to the same argument, and you are correct for a small CO2 minority composition, and simple adiabatic assumptions. For Venus, this is not true, since CO2 is in fact the main component. Even for cases where CO2 is a small minority, there will be some effect for large non-CO2 additions (say doubling O2 and N2). Pressure broadening, and non-linear lapse effects near the top of the Troposphere will result in some change in outgoing effective altitude.
The above is written in a confused fashion, as I was thinking my way through it when I wrote it. Let’s try again:
– For an adiabatic atmosphere, with Pressure (P), density (ρ), average molecular mass (m), Temperature (T), gravitational strength (g) and Boltzman’s constant k:
P = (kρ/m)T
P ∝ ρ^γ
dP/dz = -ρg
In this simple model, the atmosphere does come to an end, at scale height:
Zt = (γ/(γ-1))(kT(0)/(mg))
– If m does not change, the spatial structure of the distribution of molecules does not change.
– Because CO2 is such a small fraction of the atmosphere (4 x 10^(-4)), nothing that is done to the CO2 molecules will affect m significantly, or the scale height.
– So the opacity for a given distance will be proportional to the total number of CO2 molecules: If you halve the number, the mean free path for IR photons will be twice as large. If you increase the number of N2 and O2 (but maintain the ratio), but maintain the same number of CO2 molecules, the mfp doesn’t change at all.
– So I fail to understand your statement about needing to change the total mass. If I were to remove all the CO2 in the atmosphere, it would hardly affect the mass; but it would remove a good part of the greenhouse effect.
Look at it in terms of an absorption spectrophotometer cell. If I double the mass of the other components, and halve the mixing ratio of CO2, I need a cell twice as long for the same absorption. Pressure broadening of the lines makes things more complicated, but I’m pretty sure that means that the altitude where the optical density equals 1 goes up. You halve the number locally, but not globally.
I could be wrong about this when CO2 is a minor component. The math isn’t entirely making sense right now. I’m sure it works for Venus, though.
Obviously. If you removed all the CO2 in the Venusian atmosphere it would be a lot cooler too. But you would have to remove way more than half of it before it made much difference.
“If I double the mass of the other components, and halve the mixing ratio of CO2, I need a cell twice as long for the same absorption.”
No, the length of the cell is determined (to 1st order) by the amount of CO2, not by the ratio of CO2 to N2 or O2.
And as long as the average mass, m, is unchanged, the spatial dimensions of the density distributions will be unaffected.
Neal,
The amount of CO2 is the product of the volumetric mixing ratio and the total volume. If I double the volume and halve the mixing ratio, the amount of CO2 remains constant.
At constant pressure, the volumetric mixing ratio and the partial pressure are the same.
The point is that the absorption/length by CO2 depends almost entirely on the amount and density of CO2.
Playing games with N2 and O2 and then undoing them with ratios doesn’t change anything.
Neal,
To a first order, you’re right and I’m wrong. (See Bryan, unlike you I can admit I made a mistake and learn from it.) Venus works because adding lots of N2 increases the scale height because it reduces the average molecular weight.
But to a second order, pressure broadening will make some difference. I believe it will increase the effective altitude of emission and increase DLR at the surface (where emission isn’t already saturated) at the same temperature. Broader lines absorb/emit more total radiation and the pressure will double at every altitude where emission/absorption is significant. It’s not quite interesting enough, though, to make me cough up a month’s subscription to Spectralcalc to calculate the difference.
Leonard et al.
Even if we were only talking about differences in semantics (and I don’t think we are), resolving disagreements and confusion arising from semantics is extremely important. I’d rather understand why G&T and friends say the 2LoT is incompatible with the existence of a greenhouse effect (DLR from the atmosphere “heats” the surface) rather than treat their confusing statement with disdain.
In physics, there frequently are a number of different approaches that can be used to analyze problems. For example, some simple problems are best approached by analyzing the forces and others are more tractable using conservation of energy. I personally find it easier to understand the high surface temperature of Venus by working my way down via the adiabatic lapse rate from the altitude that is in radiative equilibrium with incoming solar radiation, while others prefer to follow the energy flux up from the surface. The more tools one has in his problem-solving tool box, the better.
I greatly appreciate Leonard’s post and the comments.
having read numerous papers (some not understandable by an electronics engineer like myself!) I think that transfer of kinetic energy to atomic vibration is plausible.
Using some of the calculators on line it seems that the energy of collision is sufficient 1 in 5*10^4 times to excite a co2 molecule sufficiently to enable a 4um photon emission. This I assumes is true only if 100% of the kinetic energy is transfered to the co2 molecule. What is the occurrence of this?
http://hyperphysics.phy-astr.gsu.edu/hbase/hframe.html
Warm non GHGs do not significantly radiate to space. A mixture of GHG and non-GHG will radiate but ony at GHG wavelengths this will continue until absolute zero when there will be no colisional speed distribution that is capable of exciting the GHG.
Is this correct?
I have found nothing that states that an excited ghg molecule will transfer vibrational energy to other substances – any references?
A ghg molecule cloud with a temperature of 0K could absorb 4um radiation and retransmit it without warming?
Thanks
thefordprefect,
Have a read of Planck, Stefan-Boltzmann, Kirchhoff and LTE, especially the section Local Thermodynamic Equilibrium. It has some textbook extracts.
If you read a textbook on the subject it would help you a lot. Making sense of papers without understanding the foundations is very difficult.
thefordperfect:
When considering whether a vibrationally-excited molecule will emit a photon, you may be intuitively considering a two-step calculation: 1) The fraction of molecules that the Boltzmann distributions predicts will have enough energy to be in a vibrationally-excited state. 2) The rate at which these vibrationally excited molecules emit a photon. It turn out that the Planck function B(v,T) has incorporated the net result of both processes into a single equation.
http://en.wikipedia.org/wiki/Vibrational_energy_relaxation
Quantum mechanics demands that molecules in stable states have discrete, not continuous, amounts of energy. This is true of rotational energy, vibrational energy and electronic energy (associated with where an electron is likely to be found with respect to a nucleus), but not the kinetic energy of translation. Every change in energy state (that doesn’t involve a collision) is associated with absorption or emission of a photon with precisely the appropriate wavelength (E = hv) to conserve energy. Changes in electronic state usually involve visible or UV photons; vibrational state, infrared photons; and rotational state, microwave photons. Symmetrical diatomic molecules without a dipole such as N2 and O2 (the main components of earth’s atmosphere) are forbidden to change vibrational energy state by emitting or absorbing an infrared photon, so they don’t absorb or emit the pervasive blackbody infrared radiation emitted by the surface of the earth or the infrared radiation emitted by GHGs. These gases do absorb and emit UV and microwaves. These non-GHGs can gain or lose energy by collisions with another molecule and if that molecule is a GHG, it could absorb or emit an infrared photon. At atmospheric pressure and temperature, molecular collisions occur every few picoseconds, but vibrational excited states typically emit photons every few microseconds. When a GHG absorbs an infrared photon and enters a vibrationally excited state, that GHG molecule is much more likely to lose its extra energy via molecular collisions than by emitting a photon and we say that local thermodynamic equilibrium exists.
Frank,
Translational energy is also quantized, think particle in a box. But since the difference between energy levels depends on the size of the box, the difference can be arbitrarily small. Some speculate that even time is quantized with the minimum time step ranging upward from Planck time of 5.39E-44 seconds.
2nd para totally wrong!
1 in 6*10^2 times at 288C
at different temperatures:
1 in 3*10^5 at 15C
1 in 3*10^6 at -30C
Nope.
Try looking up ‘local thermodynamic equilibrium’ and Kirchhoff’s Law. Here’s one link:
Click to access TheorAstro6_handouts.pdf
Note the comment on page 2:
As I stated above, vibrational energy in a molecule does not stay isolated in the vibrational mode. That’s classical thinking. The redistribution time constant is on the order of femtoseconds. That’s even faster than the average time between collisions (most of which are elastic) of about 1 ns at surface pressure.
The article from Leonard Weinstein looks almost perfect to me. The back radiation argument could also be applied to the sun. Thermonuclear reactions in the core radiate to outer layers and via convection to the surface which the radiates to space. Presumably then back radiation is also supposed to “warm” the core. Isn’t this all just reflected by the effective heat capacity of the sun ?
Bryan,
What correct plain language terminology (and you left out Alex Harvey)? The only correct terminology for thermodynamics, like quantum mechanics, is mathematical. But try using that on a lay audience, or even a relatively scientifically literate audience. Frankly, much of the classic works of Ramanathan goes over my head without spending too much time and effort to acquaint myself with the mathematical conventions he is using.
On a side note, Richard Feynman admitted in his ‘lost lecture’ that he didn’t completely understand Newton’s geometric derivation of the inverse square of the distance relationship for gravitational attraction from Kepler’s Laws because it used a lot of the mathematics of conic sections which has largely gone out of favor. So he invented his own.
It would probably fill a page to write out the full equation describing the temperature of the surface of the planet without using some sort of shorthand. And as soon as you do that, someone like you will nitpick the shorthand. But that equation will include atmospheric flux absorbed by the surface, DLR for short. But let’s try anyway.
Let:
Fs equals the average incoming solar flux/unit area at the top of the atmosphere calculated by integration over a sphere with the radius and orbit of the Earth around the sun over a full year and a suitable wavelength range. From 0-4 μm will get most of it.
Asw equals the average absorption of incoming solar radiation by the atmosphere integrated…
DLR equals the average atmospheric flux as observed from a unit area of the surface, integrated as above but over a different wavelength range that includes at least 4-50μm.
C equals the average convective flux/unit area from the surface to the atmosphere, integrated…
A equals the integrated… average planetary reflectivity for incoming solar radiation (planetary albedo) including cloud and surface reflectivity.
Ts equals the average surface temperature integrated…
B(λ,Ts) is the Planck equation for temperature Ts and wavelength λ
εσTs^4 is the Stefan-Boltzmann equation where
σ is the Stefan-Boltzmann constant
ε is the average surface emissivity for radiation in the equation ε(λ,θ,φ, lat,lon)B(λ,Ts) integrated over λ,θ,φ, lat,lon such that the integral is equal to the Stefan-Boltzmann equation using the same ε and Ts
Oh, forget that and set ε = 1, it won’t be too far off.
At energy balance (which also shouldn’t be too far off)
Ts =((DLR + (1-A-Asw)* Fs -C)/(σ))^0.25
Plugging in the TFK09 numbers but not including their net imbalance:
Fs = 341.3 W/m²
A = 0.2986
Asw = 0.2285
DLR = 333 W/m²
C = 97 W/m²
σ=5.67E-08
Ts = 289.3K =16.1 C instead of the 15C in TFK09. As I said, not too far off.
If we make the atmosphere transparent and infinitely viscous so we can ignore vertical, meridional and latitudinal heat transfer:
Asw=0, DLR=0, C=0 (no convection = no convective loss)
Ts = 254.9 K
Now if you can write an equation for the surface temperature that doesn’t include DLR, I would have to admit that DLR does not “heat” the surface using “heat” in the plain language sense that a lay audience would understand, meaning make warmer than it would be otherwise. Anyone who claims this violates the Second Law or is somehow misleading is either engaging in nitpicking on a massive scale or being willfully misleading (see, for example G&T).
Also, don’t nitpick the TFK09 numbers unless you can prove they are wrong by coming up with better numbers of your own that are consistent with observations. Good luck with that.
The ‘lost lecture’ link above didn’t seem to work so here it is:
http://en.wikipedia.org/wiki/Feynman%27s_Lost_Lecture:_The_Motion_of_Planets_Around_the_Sun
It’s available on CD for anyone who wants to hear Feynman lecturing.
DeWitt Payne
Dont you think its a bit odd that the all three of you seem uncomfortable with the correct thermodynamic terms yet the ‘sceptics’ are quite happy to use the appropriate terms.
It almost seems that you need to ‘redefine’ the meaning of the accepted language to prove your point.
Phil Jones also was keen to ‘redefine’ the meaning of peer review.
Its not a good idea either way.
Gerlich & Tscheuschner pointed out that many of the popular expositions of the greenhouse gas theory had heat moving spontaneously from the colder atmosphere to the warmer Earth surface.
This is of course contradicting the second law as they pointed out.
You consider them a bit mean to do this indicating that G&T should have ‘mind read’ their true intention and overlook this mistake.
But what if it was no mistake and was fully intended?
Chris Colose and Joshua Halpern repeated this mistake in the international physics journal.
Another co – author Joel Shore now admits this was an error.
You might consider it a cheap point but why leave yourselves open to it?
It must surely be possible to present a coherent presentation of IPCC science using the correct thermodynamic terms.
Semantics are again involved with the degrees of freedom of Carbon Dioxide.
You agree that the v2 motion contributes to Cp above a certain temperature.
However the related v2 photon energy of 15um wavelength is unrelated to this condition you imply.
The v2 motion can be induced by collision or absorption of 15um photon.
I cant see much difference in looking at it either way.
The test for equipartition holding is
kT >> photon energy
For 250K at the low end of atmospheric temperatures
kT = 3.45 x 10^-21J
hf = 13.2 x10^-21J
So CO2 even at this low temperature has a non classical specific heat capacity.
Some argue that for every 15um photon absorbed another is almost instantly released.
This would imply no overall heating at all.
I think that thermalisation occurs mainly by collision with N2and O2 and cooling by the availability of more easily attainable longer wavelength paths provided by water vapour.
Bryan,
I’m not uncomfortable at all. I just don’t believe that what you call ‘correct thermodynamic terms’ are, in fact, universally accepted. And by universally accepted, I mean understandable by a high school graduate who has probably never heard of the laws of thermodynamics.
Gerlich and Tscheuschner are arguing semantics, not facts or logic. They also willfully misinterpret anything that might contradict their preconceived conclusion like energy balance diagrams, which they call “scientific misconduct.” (Chapter 3.7.2). Quite frankly, so is Leonard. Arguing semantics has been called the “Last bastion of the small-minded.” ( http://boards.straightdope.com/sdmb/showthread.php?t=80984 ) Although a comment at the end stated that the last bastion of the small-minded is the same as the first bastion, unwavering certainty. Sound familiar?
In a word: Duh! That’s a requirement for Local Thermodynamic Equilibrium, as has been pointed out to you many times.
Indeed. That’s called “steady state”, which, of course, is only approximately true on average.
I will try to summarize the points I have been trying to make as briefly as possible and that responders still seem to miss the reason why I have been making them. The three ball examples were ideal cases with a common theme. The surface temperature increased with fixed supplied energy over the bare ball case due to a resistance of outgoing energy from some sort of external structure.
In the first case, a solid insulator totally blocked the outgoing radiation, so that all net energy was transmitted by (phonon) conduction. Note that the radiation out for the final surface temperature was still there, but back radiation occurred immediately just above the surface, and exactly balanced outgoing, so the radiation term could be left out, and we can still solve the relevant thermodynamics. However, each component was still there. We could just simplify by leaving out a net zero term.
In the second case, an opaque gas totally blocked the outgoing radiation, so that all net energy was transmitted by gas conduction (diffusion). Note that the radiation out for the final surface temperature was still there, but back radiation occurred immediately just above the surface, and exactly balanced outgoing, so the radiation term could be left out, and we can still solve the relevant thermodynamics. However, each component was still there. We could just simplify by leaving out a net zero term.
The third case was somewhat different in that a solid shell with vacuum only transmitted the energy by radiation. The radiation out was there, but back radiation was smaller, in order to transmit the net energy flux. For this case, we have to keep the radiation terms to solve the problem.
The first point here is that the third case has LESS back radiation than the other two, yet we do not say the first two are higher temperature due to back radiation, we say they are higher temperature due to something that cause some resistance to direct radiation out, and this resistance allowed energy to accumulate in the ball until it heated up enough to rebalance energy flows. That exact same comment can be used to describe the third.
The second point relates to the case of a planet with absorbing gas. There is a combination of energy transmitting methods, including some internal net radiation energy transfer, through an atmosphere that resists direct outgoing radiation. However, there is a kicker for planets. Instead of an energy flow resistance like an insulator, where something forces a temperature gradient who’s magnitude is dependent on level of temperature difference and thickness, the atmosphere forces a (essentially) constant temperature gradient independent of the source OR MAGNITUDE of the resistance. Under this imposed FIXED gradient, only the altitude of out going radiation needs to be determined to find the temperature increase, and this is true for any initial ground temperature or any level of input power. Thus this is true for any level of forward and back radiation, as long as the lapse rate is constant, and as long as the height of outgoing radiation is the same. This would also be true for an opaque gas on a planet like case two (and essentially like Venus), with no net (significant) radiation energy transfer. Where is the back radiation heating here?
DeWhitt,
In response to you question, the effect of back radiation is only needed when you need to break out the individual components of the resistance to direct radiation (for whatever reason). If you can separately determine the altitude of average outgoing radiation, it can have an infinite number of solutions of back radiation, with the same surface temperature. More opaque means shorter distance to absorption and thus smaller temperature difference for back radiation in a constant lapse rate. Adding a small amount of absorbing gas increases altitude of outgoing radiation, and thus heats the ground. Calculating back-radiation is not useful, since there will also be changes in convection, and the only important effect occurs at the level of outgoing radiation.
Back radiation does not now and never has ‘resisted’ direct radiation. That would be absorption. And calling absorption ‘resistance’ is quite a stretch if you’re trying to use ‘correct’ terminology. You can conduct an energy balance at the surface without knowing anything about the lapse rate or the effective altitude of emission to space. You simply measure the fluxes. Actually that’s not so simple for convection. Vertical convection isn’t too difficult, but horizontal convection is a real problem. The same applies at the top of the atmosphere.
And there aren’t an infinite number of solutions with different (constant) lapse rates at constant surface temperature. For a given atmospheric composition, the lapse rate has a large effect on the relative amounts of upward and downward IR emission, so there’s only one tropospheric lapse rate that will give you energy balance at the TOA and the surface.
Prevosts theory of exchanges deals with the two way simultaneous radiative exchange between a colder and a hotter radiator.
Note the word simultaneous because these are two parts of one process not two separate processes
Prevost’s theory of exchanges was first proposed (1792)
Only with climate science is it seriously suggested that the flow from the colder object is a heat source.
Anyone who has had the benefit of studying the Carnot cycle will interpret this a suggestion that it is possible to intercept this flow with a heat engine to extract work.
In less than a microsecond they will reject this interpretation as erroneous.
Oh, please. That’s a strictly semantic argument. You can only make that claim by torturing the language. No rational intelligent person with an open mind who has spent any time reading the literature, scientific or otherwise, believes that statement.
I thought Leonard’s carefully organized presentation was superb, but I’m not sure if it is sensible to apply it to the real earth. The problem is the assumption that a fixed lapse rate will faithfully transmit to the surface the temperature changes associated with radiative equilibrium near the tropopause (where radiative forcing is calculated). Unless there is a reliable linkage between the upper troposphere and the surface, GHG’s can change the characteristic emission level without changing surface temperature as shown in equation 9.
The real earth doesn’t have a uniform lapse rate linking surface temperature to temperature of the upper troposphere where radiative equilibrium with the sun dominates. Radiosondes show that the local environment lapse rates are highly irregular and differ with location and time in a seemingly chaotic manner. There appears to be little linkage between the upper troposphere and lower troposphere/surface by radiation: The atmosphere is too opaque at wavelengths the atmosphere emits strongly and surface emission in the “atmospheric window” escapes to space. Although we can calculate a lapse rate which is too steep to be stable to buoyancy-driven convection, the observed global lapse rate is not derived from such calculations. The maximum stable lapse rate depends on humidity, but humidity (and relative humidity) are highly variable. The environmental lapse rate changes at altitudes where clouds form and release latent heat and clouds are highly variable.
Most of the surface of the planet seems to be convectively stable – not always on the edge of instability (the assumption made by models). There seem to be with a few relatively small locations (the ITCZ, thunderstorms, hurricanes, some fronts) where massive amounts of energy are convected to the upper troposphere. Winds such as the Hadley circulation collect latent heat from convectively stable regions and bring it to the ITCZ. There seems to be no practical limit to how much energy can be convected through these unstable regions (hurricanes for example) as long as the heat delivered to the top of the troposphere moves away or radiates away from the top of the unstable region fast enough so that the region remains unstable. Since evaporation depends on wind speed (as well as surface temperature), the AMOUNT of energy convected upwards is not a simple function of the temperature or temperature gradient. The OPPORTUNITY to convect is controlled by the temperature gradient, and the opportunity to convect may be ended by the total amount of energy convected upwards, but the relationship doesn’t appear to be simple.
Finally, at the bottom of the troposphere (where most DLR originates), we find complex phenomena associated with the boundary layer. Dramatic changes in the lapse rate in the boundary layer are associated with the diurnal cycle.
Perhaps all of these phenomena are “trees” and I’m having trouble seeing the “forest” through the trees – the “forest” being a compelling reason for an unchanging mean global environmental lapse rate determined by some fixed combination of the moist and dry adiabatic lapse rates (which are known from first principles of physics).
Frank,
It seems to me that the lapse rate gives the maximum temperature gradient, and thus gives an upper limit to the temperature difference between TOA and ground level; and thus to the GHE.
Neal: A maximum possible temperature gradient doesn’t imply an unchanging gradient. A 1% shallower gradient can enhance can compensate for a 1% higher characteristic emission level.
Frank,
You have it backwards. A shallower temperature gradient at the same surface temperature means the atmosphere is warmer at any given level and radiates more energy upward and downward. Since specific humidity is a factor in determining the value of the lapse rate, it is extremely unlikely that a lower surface temperature would be associated with a shallower lapse rate. There are other constraints on the value of the lapse rate and convective energy transfer from the surface to the atmosphere. See Alex Harvey’s reply to you below.
Frank,
You are correct. All of my comment are for idealized models, which show the basic physics, but the real situation is much messier. However, the average lapse rate seems to fit the calculated wet lapse rate for measured evapotransporation fairly well, and the lapse rate for Venus seems very well defined. However, the point I have been making is that whatever the resistance to direct radiation to space, it is the solar heating that raises the surface temperature to balance outgoing with incoming, not back radiation. Back radiation can be one of the sources of the resistance, but is not a source of heat transfer. Also, the integrated lapse rate and the (by definition) location of average outgoing radiation exactly define the temperature increase, even if they are poorly connected locally.
Hi Frank,
I started this reply some hours ago since when Leonard has added:
“Also, the integrated lapse rate and the (by definition) location of average outgoing radiation exactly define the temperature increase, even if they are poorly connected locally.”
This renders some or most of what follows this paragraph unnecessary. I am puzzled over the benefit of a sentence that defines some relationship by definition. My prior thoughts follow.
After presenting equation 9, Leonard gives the following interpretation:
“In the end, the only factors that raise ground temperature to be higher than the case with no greenhouse gas is the increase in average altitude of outgoing radiation and the lapse rate. That is all there is to the so-called greenhouse effect.”
with the caveat:
“If the lapse rate or albedo is changed by addition of specific gases, this is a separate effect, and is not included here.”
bearing in mind his prior inclusion of “effective” in a key phrase:
“effective average altitude of outgoing radiation to space”.
Given that this leaving the questions of how that altitude is determined and could be located, and how the lapse rate is determines and also why it is applicable to the troposhere unresolved, in what sense has there been a representation of “… all there is …”.
You point out that the achieved lapse rate arises from a complex system, I will add that the altitude is an abstract notion, in the sense that examination of the location at that altitude would reveal nothing special about it. It is above the surface and below the tropopause, both of which have observational definitions, but beyond that it is difficult to position except by recourse to some relationship such as equation 9, e.g. it is defined by such an equation, that it takes the value that makes a equation balance. When that remains the case such equations will be true by definition.
Equation 9 could be made to balance by determining the altitude by the finding locations that are on average the same temperature as the surface would be absent of a GHE. Given knowledge of that altitude and the lapse rate the surface temperature can be determined by the method stated but that seems tautologous,
I expect that you know more about the convective regimes than I. However I will venture the following.
What is the impetus to which convection responses or acts to resolve?
I believe that a candidate would be the characteristic tendency for regions in the troposhere to express a local radiative deficit. To contribute more radiance to the radiative field than is absorbed from it. That the local temperature at which radiative balance would occur is below that at which convective equilibrium would occur and that these two processes are inconflict which is resolved in a stand off close to but plausibly not quite the same as temperature indicated by purely adiabatic process.
The radiance eminating from such reasons is I believed refered to as path thermal radiation and can be resolved into two components, upward and downward, the latter expressing itself as DLR at the surface. The downward component is I believe of greater magnitude than the upward component.
If the DLR is seen as an expression of a process that acts to cause local radiative deficits; it is at least an expression of a need for a convective regime to resolve that deficit from a fund of energy by way of its convective transport. To that degree it plays a key role in a system that in turn expresses an elevated surface temperature and a lowered troposheric temperature compared to the notional non-GHE surface temperatures.I am saying that convection by enabling a transport of heat into the troposhere acts to resolve a radiative deficit of which DLR is one terminal effect.
I stop at this point having been overtaken by events.
Alex
Leonard and SoD,
I fail to see the point of this exercise. If it’s designed to produce a plain language exposition of the planetary greenhouse effect that can’t be misinterpreted, it’s doomed to failure from the start. People who want to will always be able to find a way.
In a previous post shown in Judith curry’s site, several explanations for the planetary greenhouse effect were shown. One in particular is shown next:
In what was arguably the most lauded comment on the two threads, Nullius in Verba provides this eloquent explanation:
The greenhouse effect requires the understanding of two effects: first, the temperature of a heated object in a vacuum, and second, the adiabatic lapse rate in a convective atmosphere.
For the first, you need to know that the hotter the surface of an object is, the faster it radiates heat. This acts as a sort of feedback control, so that if the temperature falls below the equilibrium level it radiates less heat than it absorbs and hence heats up, and if the temperature rises above the equilibrium it radiates more heat than it is absorbing and hence cools down. The average radiative temperature for the Earth is easily calculated to be about -20 C, which is close enough although a proper calculation taking non-uniformities into account would be more complicated.
However, the critical point of the above is the question of what “surface” we are talking about. The surface that radiates heat to space is not the solid surface of the Earth. If you could see in infra-red, the atmosphere would be a fuzzy opaque mist, and the surface you could see would actually be high up in the atmosphere. It is this surface that approaches the equilibrium temperature by radiation to space. Emission occurs from all altitudes from the ground up to about 10 km, but the average is at about 5 km.
The second thing you need to know doesn’t involve radiation or greenhouse gases at all. It is a simply physical property of gases, that if you compress them they get hot, and if you allow them to expand they cool down. As air rises in the atmosphere due to convection the pressure drops and hence so does its temperature. As it descends again it is compressed and its temperature rises. The temperature changes are not due to the flow of heat in to or out of the air; they are due to the conversion of potential energy as air rises and falls in a gravitational field.
This sets up a constant temperature gradient in the atmosphere. The surface is at about 15 C on average, and as you climb the temperature drops at a constant rate until you reach the top of the troposphere where it has dropped to a chilly -54 C. Anyone who flies planes will know this as the standard atmosphere.
Basic properties of gases would mean that dry air would change temperature by about 10 C/km change in altitude. This is modified somewhat by the latent heat of water vapour, which reduces it to about 6 C/km.
And if you multiply 6 C/km by 5 km between the layer at equilibrium temperature and the surface, you get the 30 C greenhouse effect.
It really is that simple, and this really is what the peer-reviewed technical literature actually uses for calculation. (See for example Soden and Held 2000, the discussion just below figure 1.) It’s just that when it comes to explaining what’s going on, this other version with back radiation getting “trapped” gets dragged out again and set up in its place.
If an increase in back radiation tried to exceed this temperature gradient near the surface, convection would simply increase until the constant gradient was achieved again. Back radiation exists, and is very large compared to other heat flows, but it does not control the surface temperature.
Increasing CO2 in the atmosphere makes the fuzzy layer thicker, increases the altitude of the emitting layer, and hence its distance from the ground. The surface temperature is controlled by this height and the gradient, and the gradient (called the adiabatic lapse rate) is affected only by humidity.
I should mention for completeness that there are a couple of complications. One is that if convection stops, as happens on windless nights, and during the polar winters, you can get a temperature inversion and the back radiation can once again become important. The other is that the above calculation uses averages as being representative, and that’s not valid when the physics is non-linear. The heat input varies by latitude and time of day. The water vapour content varies widely. There are clouds. There are great convection cycles in air and ocean that carry heat horizontally. I don’t claim this to be the entire story. But it’s a better place to start from.
DeWitt,
Do you have any comments on Nullius in Verba’s version? Is he wasting his time also? BTW, my version was not based on Nullius’s. In fact it was presented about the same time as his, but in a less complete form, without the examples.
Leonard: We can calculate the greenhouse effect by considering radiative forcing at the tropopause and the temperature change needs to occur to restore radiative equilibrium high in the troposphere. Above, I questioned whether that temperature change will be transmitted to the surface via an unchanging lapse rate.
Judith Curry briefly discussed radiative forcing at the surface and the temperature change needed to restore energy balance at the surface. Since one can’t calculate changes in latent heat flux/convection from first principles, one can’t calculate a temperature change. However, with 2X CO2, the molecules that emit DLR reaching the surface will be lower and warmer and additional energy will be absorbed. One could call this the greenhouse effect.
Surface energy balance will be restored by a combination of increased upward convection and radiation. The latter requires an increase in surface temperature, but the former may not. Evaporation is limited by diffusion/convection of water vapor from surfaces and increases dramatically with wind speed. Assuming that all of the energy from increased DLR is removed by increased radiation allows one to calculate a maximum temperature rise (which will be modified by water vapor and especially cloud feedback). The “future of the planet” may depend on how much convection can reduce this maximum temperature rise, but our climate models aren’t very good describing convection in the boundary layer, the source of most DLR. One could define a “surface climate sensitivity” as the ratio of the surface forcing to the surface temperature change. (Our concerns can’t end when the energy from extra DLR leaves the boundary layer: It must rise to the upper troposphere, where it can escape to space as radiation.)
Frank,
As I have pointed out with the solid insulation and totally opaque gas examples, the outward and back radiation are still there, and back radiation has been maximized to exactly match outgoing radiation for the final temperature. Do you consider the hotter wall as being due to back radiation heating for those cases? What is the difference? Why is back radiation not included in those cases just because it is exactly a balance, since it is still there? I have agreed that the effect of back radiation is needed for a greenhouse effect, and it is a resistance to net radiation transfer, but the heating of the surface is totally due to the slower removal of solar energy from all causes, allowing a build up of temperature. The rest is semantics.
Frank,
Since the absorption path decreases with concentration increase of CO2, and the lapse rate is essentially constant, the temperature difference between the back radiating layer and surface decrease inversely to concentration. Double CO2, half distance, half temperature difference. Meanwhile, the increase in surface temperature is about 1.2 C per doubling (all other things being equal). Due to the non-linear (4th power) radiation law with temperature, this implies that radiation net energy flux decreases at the same time that total energy flux is constant (solar energy in has not changed here), and surface temperature is increasing. The greater the CO2 concentration, the less the radiation heat transfer, and the more the convection and other energy transport. The end point is an opaque gas out, so no radiation heat transport, but maximum back radiation (and it is exactly like the insulation case). Yet these are exactly the condition for increased heating due to raising the outgoing radiation to space from increase greenhouse gas.
Leonard:
“Double CO2, half distance, half temperature difference.”
No, if you double the CO2, it’s true that the photon’s mean free path will halve, but that means that the distance between the physical TOA and the photonic TOA will decrease. And that means that the temperature difference between ground level and photonic TOA INCREASES, not decreases; because the distance between the ground level and the TOA increases (though not by half).
Leonard:
Personally, I rather like Nullius in Verba’s version. I would nitpick on three points:
– He doesn’t take into account the fact that CO2 has two significant IR windows for GHE application. Because they also have different absorption strengths, that means these two windows will have different photonic “top of atmosphere”s (TOAs). So the real effective radiating temperature will be somewhere between these two TOA temperatures; such that the total emission through these two windows is what it would have been without the CO2.
– Likewise, if you want to take into account other GHGs, each of their spectral windows also has a different TOA. So to figure it out, you calculate the total emission through all windows and add it up; then adjust the ground-level temperature until it all adds up to the total pre-GHG insolation in all these windows.
– To cover the case where two GHGs have overlapping windows (as in the case of CO2 and H20), you have to realize that it’s the windows that are primary, not the flavor of molecule. So you calculate the TOA for a frequency based on both populations of molecule; but since CO2 gets 100 km high, and water vapor only about 10 km, it’s very likely that the TOA will be determined by the CO2 and not by the H20 for overlapping frequencies. As long as you know the TOA, it doesn’t matter if there is more absorption below or not.
Nullius in Verba has an overly simplistic view of the lapse rate. The lapse rate is determined not just by the force of gravity, the heat capacity of air and the latent heat of evaporation of water. Radiation plays a part as well. They don’t call one dimensional models of the atmosphere “radiative/convective” for nothing. A one-dimensional R/C model can be initialized with an isothermal atmosphere far from steady state and will evolve to essentially the same steady state whether the initial temperature is higher or lower than the steady state temperature. The steady state lapse rate can’t be greater than the adiabatic rate, but it can be and is different from the moist adiabatic rate.
The 1976 US standard atmosphere, for example, has a constant lapse rate of 6.5 K/km up to the tropopause at 11km. But the relative humidity never goes above 52%. So you can’t invoke the moist adiabatic lapse rate as the sole controlling factor. For the same water vapor content, the temperature drops less rapidly with altitude than it would if the RH was 100% at all altitudes, i.e. the moist adiabatic rate.
For the tropical atmosphere, the lapse rate in the upper troposphere is still 6.6K/km when the relative humidity is in single digits. The moist adiabatic rate would be approaching the dry adiabatic rate of 10 K/km at that level of specific humidity.
Saying that the magnitude of the greenhouse effect depends only on the lapse rate and average altitude of emission implies that you know both of these quantities. But while you can measure the lapse rate, the average emission altitude is a mathematical construct with no direct physical meaning because the atmosphere isn’t gray in the thermal IR. It amounts to a cartoon explanation of the greenhouse effect. It’s useful as a pedagogical tool, like the single slab gray atmosphere model, but it’s not reality.
The difference between TOA emission upward to space and the sum of atmospheric and solar radiation absorbed by the surface as advocated above by Chris Colose and as detailed in, for example, TFK09, makes a lot more sense to me. These are measurable quantities and do not require knowledge of the structure of the atmosphere. That puts an upper limit on the surface temperature which is then reduced to the actual value by convective energy transfer from the surface to the atmosphere. That can also be measured, at least in principle. One can also make the reasonable assumption that the proportion of convective and radiative energy transfer from the surface to the atmosphere and space won’t change for a small change in forcing.
Nullius specifically stated that his statement was a simplified one, and he gave examples of radiation changing the lapse rate. You are nit picking.
I’ve only just found this discussion. It’s been very interesting. I don’t know if anyone is still watching it, but I’ll comment anyway, for posterity. 🙂
I agree that the model was simplistic. The aim was to present the minimum complexity needed to get across the essential idea of the physics.
The lapse rate is not always adiabatic, true. But there’s a reason they assume it for the standard atmosphere. I disagree about the average emission altitude being just a mathematical construct, any more than a centre of gravity is in mechanics. The fact the atmosphere is not grey does not pose a problem, the radiation is averaged over all wavelengths too. It’s physical meaning is where you would ‘see’ the top of the planet/atmosphere if you could see in infrared, and is as meaningful a concept as the height of the cloud tops on Venus as seen in the visible spectrum.
Regarding the GHE and backradiation, my current favourite example is the pond of water with a black bottom. Sunlight shines through the water warming the bottom, but water is opaque to IR so the thermal radiation from the bottom is absorbed and radiated back. We have lots of backradiation from the water, much more than under an open sky, but (virtually) no greenhouse warming, due to the convection.
In considering this conflict between explanations – backradiation or adiabatic lapse rate – the best test would be to find a situation where the predictions conflicted. For example, if the adiabatic lapse rate was zero, but backradiation existed and was powerful, you could test which was the essential element. Zero adiabatic lapse rate would result in no warming. Powerful backradiation results in warming. Physics decides.
There is an element of semantics to it, but it is also about explanatory power. The pot of boiling water is at a temperature of exactly 100 C because of a detailed balance of energy flows – conduction, convection, evaporation, and radiation from the sides of the pan. Put the pot lid on, and the energy flows change, it bubbles more vigorously, but in such a way that the temperature stays the same. What is the best explanation for the temperature? Do we draw a complicated Kiehl-Trenberth diagram for the pot and its surroundings, and calculate the temperature needed to radiate that much heat from the pan sides, or do we point out that the water is boiling? And if I insist that the right explanation is the boiling point of water, does that deny the role of the radiation and other energy flows in the physics?
Heat (a noun) in thermodynamics is given specific meaning. It is energy in transit mediated by a thermal interaction that cannot of itself, in isolation from other processes, pass from a body at a lower temperature to a body at a higher temperature.
E.G.
From Wikipedia on Heat
“Heat is energy transferred from one system to another by thermal interaction.”
in addition to:
“No process is possible whose sole result is the transfer of heat from a body of lower temperature to a body of higher temperature.” (Clausius via Wikipedia)
Heat must have such an interpretation if verbal forms of the Laws of Thermodynamics are to hold.
This characterisation of heat allows its distinction from other energetic interactions. It engenders the notion thermodynamic work in contrast to itself. Work is said to have many catagories including mechanical, chemical, and electromagnetic.
Processes involving work can result in a transfer during which a body of a lower temperature loses internal energy to a body of higher temperature. I believe processes involving friction are a case in point. When rubbing two sticks together I may raise the temperature of parts of those sticks well above my own temperature. Another example might be the refrigeration cycle. There is something unsatisfactory in these examples which I associate with the processes that are left out of view. This may resolve itself at some other level of view.
In the case in question we should consider the nature of radiation.
What is radiation? Should it be considered as heat, or work, both, or neither?
In the case of “Thermal radiation” Wikipedia has the following to offer:
“In engineering, thermal radiation is considered one of the fundamental methods of heat transfer, although a physicist would likely consider energy transfer through thermal radiation a case of one system performing work on another via electromagnetic radiation, and say that heat is a transfer of energy that does no work. The difference is strictly one of nomenclature.”
it also adds that:
“The radiative heat transfer from one surface to another is equal to the radiation entering the first surface from the other, minus the radiation leaving the first surface.”
Accordingly I might consider radiation to be not heat but work with the caution that the notion of radiative heat derives from difference in energy flows due to interactions mediated by such work.
This allows us to think that internal energy from the Sun is transfered to the earth by radiation not because the Earth is the colder but because it got in the way, and that internal energy from the Earth is transfered to the Sun by radiation not because the Sun is the colder, for it is not, but because it got in the way. Work is not constrained in the same way as heat is.
In this view, the process by which the temperature of surface of the Earth is elevated from the non-GHE equilibrium to its with-GHE equilibrium could be viewed in terms of an excess of work being done by the Sun and Earth’s atmosphere in combination above the work done by the Earth’s in its entirety, on all other bodies, during the period of passage between the two equilbrium states.
Sucn an action in combination through the common process of work shields us from a distinction as to whether some partition of the work done by the Earth’s surface is due to energy most recently transfered from the Sun or the almosphere.
In this view the elevation of the surface temperature is due to neither the action of the Sun nor the atmosphere alone but in combination.
A flow of energy from a colder atmosphere to a warmer surface has occurred, which in isolation cannot be consider to be heat for to do so would violate the Laws of Thermodynamics, but it does not act in isolation. Heat can be defined as a difference between two opposed radiative flows and at that level the Laws of Thermodynamics hold. This conformity at the level of differences does not imply that the underlying processes are restricted to flows from hot to cold or that there is some partition in which only energy flows from a hotter bodies can aggregate into a resultant flow of energy to a colder body.
I cannot see any sense in which it can be said that a colder body acting in combination may not act to raise the temperature of a hotter body, nor can I see that the energy flows once combined can be meaningfully partitioned as to source in a subsequent transfer.
Some seem to argue from polar positions be it the Sum that did the deed or the backradiation, rather from the notion of concerted action.
Alex
A correction:
“In this view, the process by which the temperature of surface of the Earth is elevated from the non-GHE equilibrium to its with-GHE equilibrium could be viewed in terms of an excess of work being done by the Sun and Earth’s atmosphere in combination above the work done by the Earth’s in its entirety, on all other bodies, during the period of passage between the two equilbrium states.”
should have been:
“In this view, the process by which the temperature of surface of the Earth is elevated from the non-GHE equilibrium to its with-GHE equilibrium could be viewed in terms of an excess of work being done by the Sun and Earth’s atmosphere in combination. above the sum of the work done by the Earth’s in its entirety, on all other bodies, and any heat similarly transfered, during the period of passage between the two equilbrium states.”
Alex
Alex:
I think your attempt to use the term “work” in this way is deeply misguided. I cannot see any potential advantage to this terminological shot in the foot.
Alex,
I agree with Neal. I think you could rephrase your argument in terms of energy and entropy instead of ‘work’. That would avoid using the term ‘heat’ as well.
Leonard,
Let me engage in reductio ad absurdum here. The way I read this article is that you want to rule out the use of energy balance diagrams to explain the greenhouse effect. Correct or not? If not, then why write the article? The concept of effective emission altitude and lapse rate has been around for a long time and you have, in my opinion, added nothing to it.
Back-radiation from the perspective of radiative transfer theory:
I have mostly skipped over all the discussions above on back-radiation, because I find it irrelevant.
Back-radiation is the radiation emitted/scattered downward from the atmosphere. If I think of a beam of radiation starting deep in outer space and terminating in a spot on the Earth’s ground, it begins with radiation appropriate to the 3-degree K cosmic background, and begins to pick up photons emitted downward by the atmosphere as it crosses in. As I described above regarding the upward beam, its temperature changes as it proceeds through the atmosphere; but since it is headed downward, this temperature is on the increase; until finally it hits the ground. Its radiative temperature will be that of the atmosphere, at optical depth = 1 ABOVE ground level. So it will not be too different from ground-level temperature.
What does it tell us about the magnitude of the GHE? Not much.
Does it warm the surface of the Earth? Yes, but it doesn’t tell you by how much.
Basically, what happens is that, since the upward IR beams are not carrying out enough radiant energy to balance the incoming solar insolation, the ground will gain heat, and increase temperature until the adiabatic lapse rate has forced the TOA to increase enough to radiate outwards a balancing power of IR. When balance is achieved, you get a steady state.
Neal,
Yes as far as you went. See my comment to DeWitt about the convective response compensating the change in back radiation.
As stated above, I find that discussion on the back-radiation issue is not particularly helpful: It entails more complexity and doesn’t advance any result.
(And I really object to designating it as “resistance”: This scratches my blackboard in lots of ways.)
Neal,
You cannot consider upward IR emission from the surface in isolation. It’s the net emission, up-down, that counts. If down went away, there would be too much up and the surface would cool. The converse is also true.
DeWitt:
No, I don’t agree. The radiative transfer equation I’m relying on relates the intensity of the beam to a convolution of the emissions from the preceding parts of the beam, and includes both emission and scattering/absorption. To find out how much is going out, I look at the intensity at the TOA, defined as optical depth = 1 from vacuum.
Downward IR can be determined by using the same formalism, but with the beam starting in space and headed to the ground. The BOA can be set to optical depth = 1 from the ground. The difference from the upward IR is: a) the direction of the beam; and thus, b) the order in which the convolution is done. The basic input, temperature and CO2 density as a function of altitude, is the same.
The point is not that the downward radiation doesn’t play a necessary role, but that in this formalism, it can be sidestepped from the calculation. The basic criterion for steady state is that the outgoing IR power is the same as the portion of the incoming insolation power that would have been emitted in the same spectral window, w/o CO2.
(Complexities concerning multiple windows and multiple molecules were alluded to earlier.)
And this relates to a measurement with, say, a pyrgeometer or an FT-IR spectrophotometer integrated over the wavelength range in orbit precisely how? The TOA is not where the optical depth equals 1. That’s the effective emission height for a saturated line. The TOA is where emission at that altitude is insignificant compared to the emission from altitudes below that or where looking up, you can no longer detect atmospheric emission, i.e. ~100 km for the Earth. What about the wings? It’s possible for the OD there to never reach 1.
The BOA is not defined by an optical density. It’s defined by a physical surface.
DeWitt,
I’ve been consistently using the terminology as follows:
– TOA: effective top of atmosphere with regards to photonic emission
– physical TOA: top of atmosphere with regard to atoms (in the adiabatic-gas model, this is fortuitously a sharp edge, where the number density drops to zero)
Analogously, I use the terms:
– BOA: effective bottom of atmosphere with regards to photonic emission
– physical BOA: bottom of atmosphere with regards to atoms => ground level. [I actually haven’t had occasion to use this yet, but this is how I would]. The point being that the radiation temperature for downward radiation is not the ground-level temperature, but the temperature optical-depth 1 upward.
I do not believe there is any need to consider downward traveling IR radiation when calculating the TOA.
DeWitt,
I do not want to rule out energy balance for some information and use. All possible details are useful for something. I just want to make clear that it is not the back radiation heating the ground, it is the reduction in energy removal at the initial temperature due to a combination of causes (resistances), of which back radiation is one, and thus solar energy is accumulated, causing the ground to increase temperature until balance is restored, which also results in raising the average altitude of outgoing radiation to space up from the ground level. Since total heat transfer resistance is constant at equilibrium (and output equals input solar power), when back radiation increases, radiation resistance increases (less heat transfer by radiation), and convective process adjust to maintain the balance. Since there is NO change in heat flux at equilibrium, the increased back radiation is NOT heating the ground. Thus only lapse rate and altitude of outgoing radiation are critical to determine temperature increase. This to me is an issue of cause and effect, which seems to repeatably come up. Since this is not a cause of confusion to you or some others, I am specifically addressing those to whom it is.
The no back radiation argument is so 19th century, before we knew about atoms (weel a few suspected), electrons, and much more. It arises from a static picture of thermodynamic equilibrium. Chemical equilibria provide an excellent example of why LWs argument is useless.
Consider a simple chemical equilibrium at some temperature T, something like an isomerization
A = B
where there is a barrier to rearrangement. In such a case, molecules of A (the surface) are continually converting to B (the atmosphere), but molecules of B are also converting to A (backreaction or if you will radiation).
The equivalence with an energy balance model can be pushed a lot further but the point is that heat transfer is a two way street, it;s just that the number of lanes in one direction exceed those in the other.
BTW, welcome back SOD. E missed you
Eli,
I agree, it is both ways. However, the pure solid resistor and totally opaque gas examples I gave also have radiation both ways (immediately above the surface), but they exactly equal. Thus other processes than radiation are needed to explain the net heat transfer. How is this different? Why is the radiation not invoked there? In the extreme case, like Venus, the back radiation term is almost exactly equal to the outward (and both are huge), and convection plays a part. Why is this not essentially like the totally opaque gas, where radiation only plays a significant part at the outer portion of the atmosphere?
Because you don’t have the equivalent of gravity imposing a thermal gradient on the system. You are also missing the point that the part of the atmosphere that provides the back radiation to the surface which limits cooling rates is within a few meters of the surface and that it is important to consider radiation in such cases. While this is mostly done for very hot situations given the T^4 scaling of the SB law, because of the scale of the earth, you have to consider it here also.
Basically you are applying classical thermo to a statistical mechanics problem. As such you capture the net, but ignore the mechanisms which affect the net.
I think I understand some of the disagreement. It comes from discussing the non-equilibrium in-between state as the surface warms from a step or ramp increase in the greenhouse gas. However, I am discussing final equilibrium cases. There are still dynamic process (energy does go both ways) but they balance out to zero net.
nealjking,
One dictionary definition of Thermal Resistance is “a measure of difficulty of heat transfer through a substance”. This is the way I am using the term, although I don’t always use both words together. What is your problem with this?
That definition probably refers to the inverse of thermal conductivity, and is, I would guess,
R_thermal = Temperature_difference/heat_flux
You are using it for the back-radiation flux.
They are not even close.
Neal, No I am not using it for the back-radiation flux, but for the decrease in net outward flux. Where did you get the idea I was using it for back flux? Read my write ups.
There is an old saying that classical thermo does not tell you why something is happening, or whether it will happen, but whether it cannot happen. You are missing the trees for the forest. It is clear that there are flows of radiation between the earth and the atmosphere and visa versa, and that both fields are thermal, in the sense that you can assign a temperature to them. That makes them heat.
What does Leonard’s description of the GHE imply about water vapor feedback?
If convection automatically compensates for inadequate radiative cooling below the characteristic emission level, does the amount of water vapor below this level make any difference?
SOD once wrote a post about the influence of altitude on water vapor feedback, which I think were based on 1D radiative-convective models. I don’t know if these models assume, like Leonard, that convection compensates when radiative cooling fails to produce a stable lapse rate.
Frank,
I specifically stated that the discussion did not consider feedback such as water vapor effects. In fact, feedback is the big area of disagreement between those on extreme ends of the debate. In fact, I do believe CO2 caused a small increase in temperature, but only very small (with mostly negative feedback), and that it’s main effect is to delay return to glacial conditions.
Frank wrote above: Quantum mechanics demands that molecules in stable states have discrete, not continuous, amounts of energy. This is true of rotational energy, vibrational energy and electronic energy (associated with where an electron is likely to be found with respect to a nucleus), but not the kinetic energy of translation.
DeWitt replied: Translational energy is also quantized, think particle in a box. But since the difference between energy levels depends on the size of the box, the difference can be arbitrarily small. Some speculate that even time is quantized with the minimum time step ranging upward from Planck time of 5.39E-44 seconds.
Frank now adds: I’ve always thought of the particle in a box to be an electron being constrained within an atom of molecule (by external walls rather than central forces). Chemists would normally call these quantum effects different electronic states, not quantized translational kinetic energy. When walls of the box have a finite energy (potential energy is not infinity outside the box), the Schrodinger eqn yields both real quantized solutions and continuous complex wave-like solutions. I’m under the impression these continuous solutions are equivalent to translation.
Does the possibility of quantization have any practical impact on translation in the kinetic theory of gases?
Yes, the collision cross-section is the box into which all the qm is piled.
Eli,
Radiation resistance can be simply expressed as the ratio of energy flux that would occur if there were no back radiation divided by the actual net energy flux for radiation. Back radiation is the sole source of resistances to net energy transfer for pure radiation cases such as the thin radiation barrier in vacuum, and with space considered essentially to be 0 K. For cases where more than one mode of heat transfer exists, such as conduction, diffusion, and convection, as well as radiation, there are several parallel resistances. For a pure heated and insulation covered surface, back radiation at the wall exactly equals outgoing radiation, so radiation resistance is infinite within the insulator. However, the (phonon) conductivity of the solid insulator is in parallel, so there is a net resistance that causes a temperature increase to occur to obtain the final outward required energy flux.
An atmosphere has several sources of parallel resistance, and radiation resistance is one of them. However, the atmosphere has (in an approximation for small perturbations, and on average) a fixed temperature gradient, independent of absolute temperature level, and energy flux level. Only the total resistance is important, and since increasing CO2 increases the radiation resistance a lot (doubling it doubles the radiation resistance), and since the total resistance only increases a little (1.2 K increase out of 288 K for doubled CO2 in an ideal case), the adjustment in net resistance is only weakly related to back radiation level. Unless you can determine all resistance components to a very high precision, you can’t realistically determine their net effect to a useful level. However, the process that occurs at the upper atmosphere level (movement of the level of outgoing radiation) has a direct one-to-one effect on the increase in temperature.
In the extreme case of Venus, the net radiation energy flux through the atmosphere is less than 0.001 of the surface radiation out. In that case measuring the radiation and back radiation is not any help to determine surface temperature. If the atmosphere were totally opaque, the radiation resistance would be infinite, but convection would still carry the energy up. However, the lapse rate is reasonably well behaved and thus just determining altitude of outgoing radiation to space is all that is needed to define surface temperature. Increasing or decreasing back radiation with a modest change in CO2 would have only a little effect on surface temperature, since it would not change the net energy flux resistance significantly. The most important effect would occur at the upper portion of the atmosphere, where radiation is leaving to space.
What energy? If the atmosphere were totally opaque, there would be no input of energy from solar radiation either. I don’t think you can have a gas that’s totally opaque. Even in the depths of the solar atmosphere, there is still radiative energy transfer. For very large optical densities one uses the Rosseland diffusion approximation to calculate radiative transfer.
This comment is for the end point of a trend. Venus is an example with a small solar heating of the surface, but huge radiation and back radiation, that approaches that level. That and the model of an internally heated object with a theoretical opaque gas is what is being discussed.
1. Conduction in the atmosphere can be neglected.
2. In the absence of greenhouse gases the temperature of the surface would be much lower.
3. The fixed temperature gradient (e.g. the lapse rate) is ~independent of temperature, 9.8 K/km
4. However the temperature at any altitude depends on the surface temperature T(z) ~ T(s) – 9.8 K/km x height (in km)
5. Since the radiation rate at any altitude depends on the temperature, the radiation rate depends on the amount of greenhouse gas in the atmosphere.
You have it backwards.
Leonard,
I still maintain your description is not a mechanism, it’s a result. As such, it isn’t all that helpful except as a cartoon. Worse, it’s not even clear that it’s correct.
Let’s instantaneously double the CO2 concentration. Now what? The temperature of the atmosphere hasn’t changed, the effective height of emission to space has changed, resulting in less emission to space, but what does the surface know about that? Nothing. The lapse rate and the atmospheric temperature profile hasn’t changed at all. Yet.
The surface and the atmosphere immediately above it will warm because the atmosphere is now emitting more radiation toward the surface than it had been before the CO2 concentration was doubled and there is an energy imbalance, but not because effective height of emission has increased and the atmosphere is emitting less radiation to space. That will affect the temperature of the atmosphere, not the surface. More radiation is being absorbed by the surface than it is emitting because the atmosphere near the surface is emitting more radiation than it had been. Energy then accumulates at the surface. This must result in an increase in temperature since the heat capacity of the surface is finite. But the temperature profile of the atmosphere hasn’t changed. Yet.
If it were possible to instantaneously double the CO2 concentration, which it isn’t, what would actually happen is that the lapse rate would decrease because the top of the atmosphere would warm because it’s emitting less radiation than it’s absorbing while the atmosphere near the surface would cool because it’s emitting more radiation than it’s absorbing. The radiative imbalance at the TOA and the surface would be lower, but would still exist. As the surface warms, the lapse rate would increase until it reached a steady state value again. There is no guarantee, though, that the new steady state lapse rate would be identical to the previous steady state lapse rate if, as expected, specific humidity increases.
Talking about effective height of emission and a constant lapse rate is not a mechanism and may not even be correct.
“I still maintain your description is not a mechanism, it’s a result. ”
I’ve always thought that, too. What is the mechanism by which the effective emitting height rises. Using the running sink analogy, what is the mechanism by which the water height rises when the drain is partially obstructed?
Actually, should be “what’s the mechanism by which the temperature rises.” The mechanism you propose makes sense to me, but reading Leonard’s description, a physical mechanism is not evident.
DeWitt:
You wrote above:
“Let’s instantaneously double the CO2 concentration. Now what? The temperature of the atmosphere hasn’t changed, the effective height of emission to space has changed, resulting in less emission to space, but what does the surface know about that? Nothing. The lapse rate and the atmospheric temperature profile hasn’t changed at all. Yet.
The surface and the atmosphere immediately above it will warm because the atmosphere is now emitting more radiation toward the surface than it had been before the CO2 concentration was doubled and there is an energy imbalance, but not because effective height of emission has increased and the atmosphere is emitting less radiation to space. That will affect the temperature of the atmosphere, not the surface. More radiation is being absorbed by the surface than it is emitting because the atmosphere near the surface is emitting more radiation than it had been. Energy then accumulates at the surface. This must result in an increase in temperature since the heat capacity of the surface is finite. But the temperature profile of the atmosphere hasn’t changed. Yet.”
The way I understand this: If you instantaneously double the CO2 concentration, you halve the photonic mean free path everywhere, so the “top of the atmosphere” (= effective radiating height) halves its distance to the physical top of the atmosphere (= where the gas runs out). Also, the “bottom of the atmosphere” (= effective radiating height for downward radiation) halves its distance to the ground.
Immediately, the radiation escaping the atmosphere outward will decrease, because the effective radiating temperature will be cooler, due to the higher altitude. However, this local reduction of IR emission does NOT communicate itself downward. The downward radiation is proceeding in line with the downward traversal of the atmospheric temperature profile, so the temperature of the radiation is still about optical-depth 1 behind (= above) the local gas temperature. So the beam is more tightly coupled to the gas (reflects the temperature half the distance away than before), but it doesn’t really seem to be an effect of radiation “bouncing” all the way from the TOA to the ground.
However, as previously stated, the BOA is closer to the ground than before (half the distance), so it is at higher temperature, in accordance with the adiabatic lapse rate. So that means that the downward radiation is characteristic of a significantly higher temperature than before, when the BOA was higher up, even though the temperature profile HAS NOT CHANGED: the downward radiation flux is just tied in more closely to the ground-level temperature, so it’s more intense. And where is it getting the power from? From the air near the ground, in other words from the ground-level heating of air.
And as the power of the downward radiation increases, we expect the local environment to increase in temperature, heating up more air, and gradually lifting the entire temperature profile. At some point, the TOA temperature will be high enough to effectively radiate the same power level in the CO2 spectral window that it had before, and we attain steady state.
So it all makes sense:
a) The steady state is indeed determinable by consideration only of the TOA and upward traveling IR (the downward traveling IR is part of the physical picture but doesn’t need to be calculated);
b) the pre-steady-state heating of the atmosphere does indeed come from the downward traveling IR (= back radiation);
c) the source of the extra heating of the downward traveling IR is the air heated by the ground, which has absorbed the visible radiation from the Sun.
From this perspective, I can also now understand what everyone was saying about Venus: Since the Venusian atmosphere is about 95% CO2, and fairly dense, the distance between the TOA and the physical TOA is not very large. If you instantaneously take out half the CO2, you double the distance; but “twice nothing is still nothing”, so the TOA has barely shifted. But that means that the effective radiating temperature has barely shifted. So the “reduced greenhouse effect” (RGHE) is rather small for high concentrations (and high absolute densities) of CO2.
Neal,
See my reply about emission height change to Leonard below:
If you double the concentration of CO2, the emission height for a saturated line goes up by a factor of the scale height, about 6 km, not by a factor of two. But that’s just for CO2, not water vapor, N2O, ozone and all the other ghg’s.
DeWitt,
– I can’t locate your comment to Leonard regarding emission height; nor does the link do anything.
– “If you double the concentration of CO2, the emission height for a saturated line goes up by a factor of the scale height, about 6 km, not by a factor of two.” I did not say the emission height went UP by a factor of two, I said the DISTANCE BETWEEN the emission height and the physical end of the atmosphere went DOWN by a factor of two.
Sorry, it’s in moderation.
The long and short is that it doesn’t go down by a factor of two, not for the whole atmosphere anyway. And for most of the CO2 band, that’s so close to the surface that the temperature difference between the locations is miniscule so the flux doesn’t change, which means the effective temperature doesn’t change either. We’re talking about the difference between, say 1 meter and 0.5 meters or less. That might matter in the polar winter when there’s a massive temperature inversion, but not so much everywhere else.
DeWitt:
– At different altitudes, the photonic mean free path is different. So a factor of 2 in the mfp at the physical TOA gives a much bigger difference in distance than the same factor of 2 applied to the mfp at the physical BOA.
– The shift in the TOA altitude determines the temperature change of the EGHE.
– The shift in the BOA altitude determines how quickly the EGHE acts to warm the atmosphere. The fact that it’s small explains why it takes decades for the full warming impact of an increase in CO2 to take effect.
=> DeWitt, cont’d:
The other point is that the EGHE effect is known to be sizable for small values of CO2 concentration: the effect is generally described as being logarithmic in that quantity. Since the photonic mfp’s are greater for low concentration, this fits into the picture quite well: at higher and higher concentrations, the induced changes in altitude for each factor of 2 in mfp become less and less.
Neal,
Only if the lapse rate doesn’t change. A constant lapse rate would be only a first order approximation. As SoD points out below, that’s a decent conceptual viewpoint, but it’s not definitive. The surface energy balance, which can, at least in principle, be measured, is definitive. The change in the effective altitude of emission, being a mathematical construct, can only be calculated if you already have enough information that you don’t actually need it. That’s especially true if you’re only changing one component of the system, like doubling CO2.
Here’s a plot of the MODTRAN calculated spectrum for the US 1976 Standard Atmosphere, clear sky, surface looking up 375 ppmv CO2, all other settings default and a plot of the difference between the radiance spectrum at 750 ppmv and 375 ppmv CO2. There appears to be a slight increase in emission in the center of the CO2 band, but there’s a lot of rounding error. Most of the increase in emission comes from the wings of the CO2 band. There is no single emission altitude in the wings. It varies with frequency and the OD may never even reach a value of 1. At 758 cm-1, close to the peak of the difference between 750 and 375 ppmv CO2, the transmittance is 0.494 at 375 ppmv for an optical density (exponential, not log10) of 0.705 and 0.351 at 750 ppmv or 1.046 OD. So what was the effective altitude of emission at 375 ppmv CO2 again? Saying that the emission altitude when looking up from the surface decreases doesn’t tell you much other than emission will increase, which you already know. You have to calculate the spectrum with at least a moderate resolution band model like MODTRAN.
DeWitt,
The first goal of an explanation is to provide insight. To that end, it’s often necessary to start with a simplified case and understand how it works. As I’ve mentioned before (July 28, 2012 at 2:59 pm ), we should also be thinking about multiple IR windows, interaction with other GHG windows, etc.
It’s always possible to retrofit complexification. The fact is that there are a lot of dimensions by which the workings of the GHE can be made more conceptually accurate; to the extent that the explanation of the GHG becomes more conceptually confusing. The aim of a good explanation is to capture the essence of what might be a surprising or unexpected result, and fit it into a framework in which it seems more natural.
Even regarding numerical simulations, as Hamming said, “The purpose of computing is insight, not numbers.” So much more the case for conceptual explanations.
Neal,
The simplest case doesn’t involve lapse rates and effective heights of emission, ghg concentrations, interactions or any complications. It’s simply two parallel planes of infinite extent. We put a constant flow of energy into one plane and we control the temperature of the other plane. If we raise the temperature of the temperature controlled plane above the cosmic microwave background temperature, the temperature of the other plane must also go up. It’s that simple. The greenhouse effect can then be demonstrated by pointing a simple hand held IR thermometer at the night sky. Well, that’s true for most of the year and providing that the IR thermometer goes down to -60C, some don’t. If the sky has a measurable temperature, then there must be a greenhouse effect at the surface. That’s “how the greenhouse effect works.” Everything beyond that is details, like how much the effective temperature of the sky goes up if you double CO2.
What are you measuring with your IR thermometer? Back-radiation. It’s certainly not the effective height of emission or the lapse rate. That’s how you might calculate the magnitude of the sky temperature, not prove its existence, which seems to be the stumbling block for many. If you start with lapse rate and height of emission, you’ve already lost a lot of people.
DeWitt:
I find that a model w/o the ALR misses the whole force behind the phenomenon. To me, that’s no better than the greenhouse pane of glass, as an explanation.
That’s also why I have no taste for the vacuum with conducting shells: Yes, that’s how a sphere surrounded by conducting shells would act, but the Earth isn’t a sphere surrounded by conducting shells.
Neal,
A model of the atmosphere without a lapse rate doesn’t have a greenhouse effect. Even in the single slab gray atmosphere toy model the atmosphere slab has a lower temperature than the surface, assuming the absorptivity of solar radiation is less than the absorptivity of thermal IR. If the opposite is true you get the stratosphere with the temperature increasing with altitude, a reverse greenhouse effect. The limit of that toy model is an opaque shell, btw.
The ALR and the pane of glass fulfill exactly the same function, to create a temperature difference that causes a difference between upwelling and downwelling radiation (that nasty back radiation again). Now some will tell you that Wood proved in 1909 that a pane of glass and a pane of rock salt were no different. He was wrong then, as was pointed out to him at the time in no uncertain terms by the leading authority on the intensity of solar radiation at the time, Charles Greeley Abbot (a fact that seems to have been missed by the modern champions of Wood), and they are wrong now. By putting a second pane of glass above the glass covered box, he decreased the total solar radiation entering that box. By putting a pane of glass above the box covered by a pane of rock salt, he increased the downwelling thermal IR seen by that box as well as reducing the solar energy. Since the inside of glass covered box now sees less total energy than the rock salt covered box, the fact that their temperatures are nearly the same proves that the thermal IR transmission characteristics of the glazing does make a difference. Abbot was right, Wood was wrong.
The difference between a planet and a horticultural greenhouse is that a planet is better insulated for energy loss by means other than radiation. The better insulated your horticultural greenhouse the more difference the radiative properties of the glazing makes. If that weren’t the case, they wouldn’t be selling low emissivity double and triple pane windows for greenhouses in northern climes.
Leonard,
What is your definition of ‘back radiation’?
BTW, Leonard – I generally agree with your description as does SoD.
I think the biggest problem here is semantic and that there is too much focus on the complexity of all the physics on a more micro or local level as opposed to the bigger picture.
The net energy flux entering the surface to sustain the surface temperature is just the sum of the radiative flux and non-radiative flux from the atmosphere to the surface. Much of the direct radiative flux from the atmosphere to the surface is replenishing non-radiative flux leaving the surface but not returned. Moreover, much of the energy entering and leaving the surface is that which is circulating within the system (i.e. surface -> atmosphere -> surface). This is why Trenberth’s depiction confuses everyone and is really incomplete.
So one should create two versions of the Trenberth diagram: the conventional one with absolute energy fluxes and a second with net energy flux = heat transfer. The net LW radiative flux from surface to the atmosphere is 396 (OLR) – 333 (DLR) – 40 (surface to space) = 23 W/m2. An interesting question is whether the heat transfer by each mechanism needs to be from hot to cold, or whether only the net heat transfer by all mechanisms need to be from hot to cold. The total heat transfers are:
161 W/m2 from sun to surface
78 W/m2 from sun to atmosphere
120 W/m2 from surface to atmosphere
40 W/m2 from surface to space
199 W/m2 from atmosphere to space
(1 W/m2 radiative imbalance warms the earth)
When viewed this way, the relative opacity of the atmosphere to LWR is impressive: Only 23 W/m2 of the outgoing total of 239 W/m2 is net LWR. The atmosphere gets three times as much HEAT from the sun as it does from the surface of the earth.
My point is Trenberth’s depiction doesn’t give the full picture of what’s going on and confuses many. The fundamental mechanism of the GHE is that of downward re-emitted radiative effects in the atmosphere. This is often incorrectly thought to be caused by the amount of direct downward LW from the atmophere to the surface (erroneously all designated as ‘back radiation’). The reason the atmosphere directly radiates more down to the surface than out the TOA is due to the lapse rate (i.e. because the lower boundary is significantly warmer than the top boundary) and not because each layer of the atmosphere anisotropically radiates more downward than upward.
The key thing to understand is every photon in the atmosphere has a 50/50 probability of being emitted up or down, and this is the case whether the emission occurs 1 inch from the surface or 1 inch from the top of the atmosphere. It is also the case if the absorption of a photon by a GHG molecule causes the immediate emission of another, as I would imagine is quite often the case. This is further why the 50/50 split is an emerging property when Conservation of Energy is applied to the surface and TOA boundaries. The complexity of all the physics (convection, latent heat, radiation, etc.) only involves how the atmosphere manifests the net energy flow of about 390 W/m^2 into the surface to sustain 288K.
Frank
The KT97 and similar diagrams each show one possible reconciliation of energy flows that satisfy the first law of thermodynamics.
But since there appears no attempt to accommodate the second law of thermodynamics its questionable if these diagram has any useful purpose.
See my earlier post on why one 1368W/m2 is not the same as four lots of 342W/m2
I see no violation of the 1st law with the K&T ’97 diagram. Only an incomplete picture that can be easily misinterpreted as violating the 1st law.
RW
I agree that there is no violation of the first law of thermodynamics.
The numbers all balance out.
However an infinite number of balanced out versions such as KT97 can be created.
If they do not balance out in terms of the second law what use are such diagrams
Not for the Earth. KT97 and TFK09 are based on observations. There are not an infinite number of versions that are still consistent with observation, unless you consider minor variations inside the error ranges of the measurements. That would be yet another straw man.
To put it another way. If there are so many different versions, show me one that is significantly different yet still conforms to all known measurements.
And that manages to violate the Second Law.
There is no real problem with the KT diagram, there is a problem with those who try and use it as a strawman which they can try to trample. It is a simple description of net heat energy flows between the four major heat reservoirs, earth, air, fire and water (bunnies sense a classical theme emerging with the sun being fire) and does not show explicitly the flows interior to any of the three, thus the lack of layers in the atmosphere which capture much else of what is being talked about here.
Bryan,
Your “1368 is not four lots of 342” is a straw man as was pointed out to you above. Look at equation (80) in G&T, which is something they actually get right, if you want to know why the solar constant is divided by 4 to get an average surface flux. It has to do with integrating cos(θ) and cos(φ) where θ is latitude and φ is longitude.
You have also consistently failed to show anything in KT97 or TFK09 that violates the second law. Net energy flows are always from hot to cold. And please don’t tell me that Clausius never said anything about net energy. His often quoted statement about the flow of heat and temperature dates from 1854. He didn’t invent the concept of entropy until 1865, so using that statement to define the second law is both dated and limited to heat transfer. The more correct statement is that for an isolated system, the entropy can never decrease. The statistical mechanical expression of the second law is even further removed. The absolute prohibition goes away. Instead, the probability of a decrease in entropy of an isolated system goes down as a function of the number of possible system states and is never identically zero.
Bryan,
How does KT97 violate the 2nd law? I don’t see that.
RW and DeWitt Payne
1368W/m2 at Earth orbit falling on a circle is equated to 342W/m2 falling on a sphere that’s where the four times figure comes from.
1368W/m2 produces a temperature of 123C at equator.
The higher value of radiative power/m2 initiates processes that cannot be represented by the averaged down lower power/m2
Even though this seems alright by first law it is unrealistic and does not represent the reality that the second law insists on.
A 342W/m2 is equivalent to 6C.
Would this 6C produce 24W/m2 of thermals?
Would this 6C produce 78W/m2 of latent heat?
Photosynthesis and most photochemistry is impossible.
No biosphere
How realistic is this picture and how useful is such a diagram?
I appreciate the explanation, but I don’t understand it.
RW,
I’ll interpret for you. Bryan knows that the values in KT are correct but doesn’t like admitting that. So instead, Bryan is taking issue with the usefulness of the concept of “a global annual average”. It’s a fascinating example of cognitive dissonance.
If the numbers were wrong Bryan would produce the right numbers. If the second law of thermodynamics were violated then Bryan would produce an entropy calculation to prove it.
To tie this back to the present discussion KT97 would be represented by an Earth surrounded by a constantly radiating sphere concentric with the Earth.
The sphere would produce a uniform radiation of 342W/m2 directed normal to the Earth surface.
Wien’s law tells us that this would represent a spectrum with a maximum wavelength in the region of 10um.
This is well into the Infra red so the 107W/m2 reflection by clouds would not happen.
Several other problems occur if you take these numbers seriously.
They add up WRT the first law of thermodynamics in the sense that the numbers balance.
Perhaps expecting it to be compliant with the second law is a bit unfair since it perhaps was never intended to.
But then how useful is such a diagram?
Bryan,
There is no violation of the 2nd law, because the GHE is not about heat flowing from cold to warm through a conduction process. Radiative effects are different, and the GHE effect at its core is radiatively induced. The other processes such as latent heat and convection are 2nd order effects that influence the 1st order radiative effects ultimately driving the process.
1368 W/m² produces a temperature of 121 C when the sun is directly overhead if and only if the heat capacity of the surface is zero or at least very low. But if that were the case, then when the sun went down the temperature would also be very low. For a heat capacity of zero the night time temperature would be -270 C.
342 W/m² would produce a temperature of 5.5 C for a blackbody if there were no greenhouse effect. But an average ( a concept which seems to be beyond you) temperature of 5.5 C would be higher than the average temperature for a blackbody with a heat capacity of zero.
You can’t calculate peak emission wavelength by assuming that the incoming radiation is from a blackbody close by. If that were the case, then the solar constant of 1368 W/m² would be from a blackbody at a temperature of 121 C. No photosynthesis from that radiation either. But you know that, you’re just floundering. In fact, the brightness temperature of the solar radiation will still be 5800K whether you use the solar constant or the radiative flux averaged over time and space.
You usually do better than this.
The Plank Radiation curve describing emission from a body at 290K extends to roughly 4 microns on the blue side and 50 on the red. There is plenty of IR to be reflected. Bunnies lose patience rapidly with dissemblers.
DeWitt Payne
“1368 W/m² produces a temperature of 121 C when the sun is directly overhead if and only if the heat capacity of the surface is zero or at least very low. But if that were the case, then when the sun went down the temperature would also be very low. For a heat capacity of zero the night time temperature would be -270 C.”
Yes in the real world the atmosphere moderates the extremes.
Highest recorded temperature 57.8C in Libya.
Lowest temperature however was not the same night in Libya but in Antarctica at -89C.
Very little attention is paid to the cooling effect of the atmosphere in the real world.
RW
The second law is not so much violated in KT97 it is just not applicable.
Any attempt to use realistic calculations falls apart as I have indicated now in several posts.
Bryan,
This is a golden opportunity for you to prove I’m wrong about you, that you can admit you’re wrong about something.
I’m throwing a concept you raised back to you. The quality of solar radiant energy is not changed by distance from the sun or averaging over time and space. The peak wavelength is still ~0.5 μm. whether the sun is directly overhead or at the horizon (in the absence of an atmosphere).
True or not true?
If true, then the KT97 and TFK09 assumption on average flux is not the equivalent of putting an opaque shell at a temperature of 5.5C around the Earth. It’s putting a nearly transparent shell at a temperature of ~5800 K around the Earth. The emissivity of the shell would be 342/(σ*(5800)^4) = 5.33E-06. In which case you are wrong about at least this aspect of KT97 and TFK09.
Here’s a question for you: Suppose we take a rotating sphere with the diameter of the Earth and put it in orbit around the sun in the Earth’s orbit. Now suppose we put equally spaced thermometers on the surface so every square meter of surface has exactly one thermometer on it. Let’s set several boundary conditions:
1. The surface of the sphere is superconducting.
2. The surface of the sphere has zero conductivity and zero heat capacity.
3. The surface has zero conductivity but very high heat capacity.
What is the average temperature as well as max and min temperatures over all the thermometers for conditions 1, 2 and 3 and why? How does that relate to KT97 and TFK09? (Hint: the answer to at least one of these conditions is in G&T).
DeWitt Payne
“Bryan,
This is a golden opportunity for you to prove I’m wrong about you, that you can admit you’re wrong about something.
I’m throwing a concept you raised back to you. The quality of solar radiant energy is not changed by distance from the sun or averaging over time and space. The peak wavelength is still ~0.5 μm. whether the sun is directly overhead or at the horizon (in the absence of an atmosphere).
True or not true?”
Yes you are correct the ‘quality’ of the radiation is maintained particularly if it can be focused.
Solar radiation with its rectilinear propagation is in this category.
Diffuse radiation cannot be properly focused but even then, the individual photons retain their quality.
Once thermalised however all quality is lost.
So I still maintain that the possible experiment I quoted earlier would show that;
One 1368W/m2 is not thermodynamically equivalent to;
342W/m2 + 342W/m2 + 342W/m2 + 342W/m2
I give up. You are hopeless. You’re as bad in your own way as Doug Cotton. You’re apparent failure to grasp the concept of averaging incoming solar flux over time and space now looks like willful disregard meant to provoke controversy rather than simple ignorance. Provide the correct answer to even one of the questions I gave you and I might change my mind. Otherwise you’re on my ‘burn before reading’ list from now on.
I was trained as an electrochemist, btw. Tell me how the Carnot cycle applies to a fuel cell.
DeWitt Payne
Its you who’s hopeless.
The only thing you seem to excel in is insults.
I don’t care to put up any longer with your bad tempered demeanor.
I could roll off a whole catalogue of mistakes you have made but whats the point!
I gave the outlines of a test you could carry out to prove the above point one way or another.
But I forgot you are still busy with your refutation of the Woods experiment.
Yawn!
Bryan,
Why should I do your work and carry out your test? Your assertion that the average of 342 W/m² for the solar flux at the top of the atmosphere in KT97 and TFK09 was equivalent to a shell around the planet at a temperature of 5.5 C was simply ludicrous. Yet you have never retracted it. I provided a more realistic alternative that you completely ignored.
You have yet to provide realistic models equations and calculations to support any of your assertions. Hauling out the Carnot cycle doesn’t count. Your parting shot that you could point out numerous instances where I was mistaken is a classic logical fallacy known as tu quoque. It does precisely nothing to prove your assertions. Nor does it eliminate your own obvious mistakes, none of which you have ever admitted to.
You also always ignore questions the answers to which might cause you to doubt your position even when the solution to a question is provided by G&T in their paper, which you claim is inerrant.
A scientist needs a thick skin. It’s something of a blood sport. Sharp criticism is a fact of life in the field. People call each other idiots all the time. Sometimes, they’re both right. Using perceived insults as an excuse to quit the field and take your ball home is a tad childish, though, don’t you think? Besides, I’ve been far more polite than you actually deserve.
Leonard: The discussion here is looking at the greenhouse effect from at least two different perspectives: 1) A 1LoT perspective or conservation of energy. 2) The 2LoT perspective. Both perspectives should be equally valid.
When we consider all of the energy fluxes (surface energy balance, TOA energy balance), then we looking from the 1LoT perspective. Everything is always cooling by radiation, usually by conduction, and sometimes by convection. Other things are always receiving this energy lost by radiation, conduction and convection. At a molecular level, the ability to lose energy by radiation (individual photons), conduction (collisions) and even convection is inherently reversible, an object can’t be in the business of losing energy by these mechanisms without also being in the business of receiving energy by these mechanisms. However, we can measure/calculate the flux in both directions only in the case of radiation. When external energy fluxes are not in balance, the internal energy/temperature changes in such a way the energy is conserved. When CO2 doubles all of the energy fluxes will change and the system will eventually reach a new equilibrium with different energy fluxes through different mechanisms.
When you ask which body heats (or warms) another, that is the 2LoT perspective. Heat flows even when a system is in equilibrium. The atmosphere is heating space, the earth is heating the atmosphere and space, and the sun is heating all three. Heat flows in only one direction, from hot to cold. Conduction and convection are observed/calculated as one way fluxes and therefore are both energy fluxes and heat transfers. Net radiation, approximately o(T2^4 – T1^4), is heat and possibly your concept of “radiation resistance” is associated with it. When CO2 doubles, the earth will still be heating the atmosphere after and while a new equilibrium is reached.
In this post, you illustrate energy transfer between a ball and an outer shell via conduction, convection and radiation. The first two mechanisms are conventionally described as one-way transfers of HEAT. Some of your readers are uncomfortable with the consequences of replacing them with radiation, which they are more comfortable treating as a two-way transfer of ENERGY.
Another problem with comparing radiative transfer to and from a molecular gas to energy transfer by conduction or convection is that for conduction and convection, one measures only one temperature for a given location. But Teff for radiation from the atmosphere is not the same as the gas kinetic energy temperature, Tk, near the surface. Even though 90% of the radiant energy seen from the surface looking up comes from within a few meters from the surface, Teff is less than Tk because the atmosphere isn’t gray or black. That changes if you put an evacuated opaque shell over the surface because the photon gas temperature in the space between the surface and the shell will always have a blackbody temperature and spectrum unless the shell is perfectly reflecting (or transparent, but then you’re back to what’s effectively a gas, not a shell), which can only be true in a thought experiment.
To put it in chemical reaction terms as Eli suggested above: When you measure pH in pure water (not trivial, btw), you measure the average activity of the hydrogen ion. But the individual hydrogen and hydroxyl ions are continually recombining to form water and other water molecules are dissociating to form pairs of ions. The pH measurement tells you nothing about the rates of dissociation and recombination other than that they are equal if the pH isn’t changing. Similarly, the measurement of a temperature profile in a solid tells you about the net flow of energy. You can’t measure whether there are, in fact flows in both directions, but the theory of diffusion tells you there must, in fact, be fluxes both ways.
There are a lot of comments since my last one. I will only try to answer a few.
If you make a step change in composition, there are clearly transient states that are going to temporarily change lapse rate, etc., due to Cp and storage. I always referred to equilibrium states, so transient states are not what I discussed.
The argument of the difference between solid conduction, gas conduction and radiation is based on the fact that different physical processes and equations represent them. However, all cases have back radiation at some point and to some degree. All can be represented as a net energy flux resistance. The special need for radiation flux information only is important due to the location where radiation to space occurs, since the atmospheric lapse rate is not (on the average) dependent on small changes in gas composition or energy flux from any of the components. The total energy flux in (at equilibrium average) is absorbed solar energy, and the total flux out is exactly the same.The constant values of lapse rate and constant energy flux out means that only the location of outgoing radiation matters for the ground temperature. You throw another blanket over a person, and he warms up. The blanket is not heating the person, the slower transmission at the lower temperature forced the temperature to rise. The persons internal metabolism was the source for the warming there. Changing the boundary condition changes the result. It can change through many transient processes, but I refer to final equilibrium only. Exact same concept for the atmosphere. The absorbed solar energy is the source for atmospheres.
Keep in mind that if CO2 increase, back radiation energy and outward radiation energy both increase, but the difference is SMALLER. Convective energy flux outward compensates to maintain same total energy flux. An electrical analogy is if you have a constant current source with a resistor to a fixed load, and increase the series resistor, the voltage to the resistor increases, but the load sees no (steady state) change.
The question: Do all forms of net energy flux separately have to obey the second law for a system of parallel conductive resistances. YES. All have net energy fluxes from hot to less hot.
What? There is, in fact, no reason to believe that at higher CO2 and surface temperature that the ratio of net surface radiative heat transfer to convective heat transfer must decrease. It might, but that’s a completely separate argument from the one you have been making in this thread. You have provided zero evidence to support this assertion. If it did, that would be a negative feedback and the surface temperature would increase less than the Planck response in the absence of other feedbacks. The window to space would decrease, but that’s because absorption by the atmosphere increases. It says nothing about the net radiative flux from the surface.
DeWitt,
To put some numbers to the case look at the Kiehl & Trenberth (1997) result. They show radiation energy flux from the surface up to be; 390 Wm-2, with 40 through the atmospheric window, and 350 Wm-2 absorbed in the atmosphere. The (average) cooler altitude, where back radiation comes from, emits 324 Wm-2 to the surface. The critical numbers here are the temperature that generated the 390 Wm-2 up, and the 324 Wm-2 down. Emission and absorption coefficients are assumed to be 1, for simplicity. From my eq.2, the temperatures are:
Surface: T= 288.0 K (global long term average)
Atmosphere: T= 274.9 K (average at effective height for back radiation)
From an average lapse rate of -6.5 C/km, the average radiation down altitude is 2.02 km
HOWEVER, doubling the CO2 would almost exactly cut the absorption distance in half. The surface would still be 1.2 C hotter than it was (if the absorption model for doubling CO2 is correct), but the layer radiating to the ground would be only 6.6 C cooler than the surface, since it is only 1.01 km above the surface at doubled concentration. Thus we calculate:
Atmosphere: T=289.2-6.6=282.6
The total radiated energy up now gives: P up = 396.6 Wm-2
Back radiation down gives: P down = 361.6 Wm-2
Delta power per m2 was: 390-324 = 66 Wm-2 (total net radiation energy flux)
With 2 X CO2 it is now: 396.6-361.6 = 35 Wm-2 (total net radiation energy flux)
The result is that doubling CO2 resulted in a hotter ground, but cut the actual radiation net energy flux almost in half. However, total energy transport to the upper atmosphere is exactly the same for fixed solar input and equilibrium. Out of the 168 Wm-2 absorbed solar energy at the surface, we went from 66/168=39.3% of transported (absorbed solar) energy by radiation, to 35/168=20.8% of transported (absorbed solar) energy by radiation.
If you have any questions , please ask. This has nothing to do with negative feedback, it has all to do with the fixed energy, in, fixed energy out and lapse rate, along with the calculations of 1.2 C per doubling of CO2.
Your numbers are bogus. Doubling CO2 would not reduce the effective altitude of emission to the surface by half. It would only reduce the altitude of effective emission by CO2 by half.
Let’s take MODTRAN US 1976 standard atmosphere as an example. We’ll assume that there is energy balance at the default CO2 level of 375 ppmv. MODTRAN only calculates between 100 and 1500cm-1, but the frequencies outside that range don’t change with CO2 concentration significantly, only with temperature so they can be calculated from the Planck equation.
Clear sky, surface looking up, 375 ppmv CO2: 258.673 W/m²
surface looking down; 360.472 W/m²
tropopause (11km) looking down: 266.178 W/m²
surface temperature 288.2
surface emissivity 0.98 (as I said, the flux is only calculated from 100-1500 cm_1, S-B emission would be 383.37 W/m²)
Calculated sky emission for ~0-100 and 1500+ cm-1 = 23.5 W/m²
So the effective temperature of the sky is ((258.673+23.5)/σ)^0.25 = 265.6 K
The effective altitude of emission for a lapse rate of 6.5 K/km is 3.48 km.
Thermal IR emitted – absorbed = 101.2 W/m²
Now we’ll double CO2 to 750 ppmv and increase the surface temperature to a value that returns emission at 11 km looking down to 266.178 W/m² (100-1500cm-1) assuming constant RH.
surface temperature: 289.56 K
surface looking up: 268.501 W/m²
0-100 + 1500+ = 24.4 W/m²
Teff sky = 268.09 K
effective altitude of emission for 6.5 K/km = 3.30 km
difference = 180 m, not 1 km.
Total surface emission: 390.63
Thermal IR emitted – absorbed = 97.7 W/m²
For low cloud cover, the lower surface of the cloud is effectively a black body at very near surface temperature so radiation seen by the surface is almost exactly equal to radiation emitted from the cloud to the surface. The energy absorbed from forwarded scattered solar radiation is returned to the atmosphere by convection only. CO2 makes almost no difference. For cumulus cloud cover with a base at 0.66 km, emission seen by the surface at constant surface temperature increases by ~0.3 W/m² out of 366 W/m² when doubling CO2 from 375 to 750 ppmv. That’s an insignificant change in the average altitude of emission.
The difference in emission and absorption decreases by 3.5W/m² for the 40% of the Earth’s surface that is not covered by clouds and essentially not at all for the rest. The difference on average would be 1.4 W/m². But this is also a very crude calculation that assumes that the lapse rate is constant, that there are no other feedbacks and that the change in surface temperature can be calculated from only the clear sky surface. That’s a lot of assumptions. Given that, I would say that the change is probably not distinguishable from zero in the real world.
You can make the same sort of calculation looking down from the tropopause. The change in the average emission altitude will be measured in meters, not kilometers. The CO2 emission altitude will change by a factor controlled by the scale height, an increase of about 6 km for doubling. But at the center of the CO2 band, you’re already in the stratosphere.
The 30 W/m² to space from cloud covered sky is from the clouds, not through the clouds. Clouds are totally opaque to thermal IR and nearly opaque to near-IR from solar radiation. In fact, that number is the most suspect of all the data, IMO. If you look at actual transmissivity in the thermal IR from the altitude of cloud tops, the percent transmitted to space is quite high, giving a much higher number than 30 W/m² even when corrected for the area covered by clouds. But then I’m only looking at emission from low cloud cover. Mid and high level clouds might make a difference.
DeWitt,
You are correct that I over simplified. I did not include cloud effects, or rain down. Also I assumed all absorbing was done by CO2. However for the moment consider simplifying by neglecting clouds, and consider a grey gas to IR wavelengths. If all absorbing gases doubled, my approach would be correct, in that this would half the distance to absorption, and decrease the relative portion of net radiation transfer of energy up at the same time that the surface heated more, and back radiation increased also, and total energy flux up remained the same. In the extreme case of very large increase in absorbing gas, the absorption distance would approach a very small distance (such as at Venus), but convection and the small radiation heat transfer would still carry the ground absorbed energy up to where it would radiate to space. Only the average altitude of radiation out would determine the surface temperature increase, not the net radiation heat transfer.
“350 Wm-2 absorbed in the atmosphere. The (average) cooler altitude, where back radiation comes from, emits 324 Wm-2 to the surface. ”
Given the absorption coefficient of CO2 and the 400 ppm mixing ratio, pray tell how far the emission in the CO2 bands gets from the surface before it is absorbed? Then work out where the emission from that layer at 324 Wm-2 comes from and how far it gets when emitted.
You are treating a cartoon (an instructive one at that) as a full line by line model and that can only confuse.
Leonard,
Trenberth shows a direct surface to space transmittance of 70 W/m^2 (40 W/m^2 through the clear sky and 30 W/m^2 passing through the clouds). This leaves 320 W/m^2 of the surface radiative flux being absorbed by the atmosphere. The 324 W/m^2 directly radiated to the surface is not all ‘back radiation’ (i.e. defined that which last originated from the surface emitted radiation). Some of this is ‘forward radiation’ from the Sun absorbed in the atmosphere yet to reach the surface, and the remainder is just downward LW radiation sourced from the non-radiative flux of 102 W/m^2 moved from the surface into the atmosphere (mostly via water vapor and clouds).
This is why Trenberth’s picture is confusing to so many and not really completely correct. A more proper depiction would have the total amount of non-radiative energy leaving the surface and have a non-radiative return path from the atmosphere to the surface (i.e. that which returns as the temperature component of precpitation, wind, weather, etc.). Trenberth’s 102 W/m^2 of non-radiative flux from the surface is apparently only that which leaves the surface but is not returned. Moreover, much of the 324 W/m^2 directly radiated to the surface is replacing non-radiative flux from the surface which is not returned, so it’s a net zero flux entering the surface.
My comment above about IR and clouds should have been addressed to you, not Leonard.
You can’t parse the IR radiation by source. Once it gets thermalized (either from the sun or the surface) there is no clue as to its origin, which is why the whole nonsense about the IR radiation being heat or not heat is nonsense. It can be characterized by a temperature. It is thermal. It is heat.
Eli
This is just nonsense.
In the past i have asked you to consult Joel Shore on the difference between ‘heat’ and Infra red radiation.
You will find his views on the topic almost identical with my own.
Physicists all study the Carnot cycle.
For chemists it appears to be optional.
Eli,
IR radiation is not temperature, it is not heat, it is not thermal. It is an energy flux. The net energy flux from all sources is called heat transfer. The net energy gain is called the increased heat content. I dislike using the term heat at all, but the terminology is well established, so it has to be used correctly by the established convention, or avoided by going directly to energy or power or power per area descriptions.
The radiation field DOES have a temperature and for the atmosphere where local thermodynamic equilibrium holds that temperature IS the same as the gas kinetic temperature. Individual photons do NOT have a temperature, any more than an individual gas molecule does, but ensembles of photons and gas molecules do. Inclusion of radiation fields in systems by physicists and decoupling by chemists does lead to much confusion, but in this case it is better to include the thermalized emission and separate the light from the sun which has a temperature of about 5000 K.
Go read some real thermodynamics Bryan.
RW,
Perhaps people commenting on figure 7 of Trenberth & Kiehl 1997 should study the paper itself. It’s a faint possibility that people who find the diagram confusing haven’t read the paper and/or don’t understand climate basics.
Let’s look at the convective energy leaving the surface via latent heat.
The globally averaged rainfall is about 3 mm per day. What comes down must have gone up before, so 3 mm has been evaporated from water on the surface.
Volume per day per unit area, V = 0.003 m3
Density, ρ = 1000 kg/m3
Therefore, mass rate, m’ = 3 kg/day
Latent heat of vaporization, L = 2.3×106 J/kg
Therefore, upwards convective flux, Cup = 2.3×106 x 3 = 6.9×106 J/day
Dividing by number of seconds in a day, Cup = 80 J/s = 80 W.
Note that Cup is a cooling of the surface – heat flows from the hotter surface to the cooler atmosphere. The process is evaporation of water at the surface (energy consumed in the evaporation) and later condensation of water vapor back to water in the atmosphere (energy released in the condensation).
You want to know, Cdown, the amount of heat returned by the rain?
Specific heat capacity of water, cp = 4,200 J/kg.K
ΔT = temperature difference between surface and the rain
Cdown = ΔT.cp.3 = ?
RW, I’ll let you fill in the blank here.
Those who have read the KT97 paper will know the uncertainty around the convective fluxes.
SoD,
Yes; however, my point is really that Trenberth is arbitrarily mixing together two separate energy flows into one depiction:
Sun -> space -> atmosphere -> surface -> atmosphere -> space
as well as
surface -> atmosphere -> surface
This is causing confusion and making it difficult to see and understand fundamentally how the GHE works, and how its ultimately driven by downward re-emitted radiative effects in the atmosphere.
He’s also arbitrarily converting non-radiative forms of energy into radiative forms of energy, which is again misleading and not really correct.
That is, the GHE is not caused by Trenberth’s mythical ‘back radiation’.
RW,
Is this your response to producing the value of Cdown?
You have a value and don’t want to share it?
Kiehl and Trenberth should be ashamed. They claim:
“The purpose of this paper is to put forward a new estimate, in the context of previous assessments, of the annual global mean energy budget. A description is provided of the source of each component to this budget.
The top-of atmosphere shortwave and longwave flux of energy is constrained by satellite observations. Partitioning of the radiative energy throughout the atmosphere is achieved through the use of detailed radiation models for both the longwave and shortwave spectral regions..”
It’s clearly a rubbish paper as it only answers the points posed in the abstract, instead of addressing your conceptual needs.
Actually it’s the atmosphere that does this. The atmosphere is misleading and not really correct?
When you produce your own diagram of surface and TOA fluxes it will be fascinating.
Your self-confidence is boundless so you should give it a go.
And that, readers, is my recommendation to all the many many people, including Bryan and RW, so happy to slate KT97. All for different and marvelously contradictory reasons.
Produce your own diagram and share it with the world.
In fact, if 2 or more readers unhappy with KT97 produce their own version and share it with me (scienceofdoom – you know what goes here – gmail.com) I will be happy to reproduce them here. Unfortunately I will point out the flaws.
I can’t produce a value of C-down, but I know one exists and is of significance (i.e. it can’t be ignored), which is my point.
And yes, Trenberth arbitrarily converts the entire 102 W/m^2 of non-radiative flux into all electro magnetic radiative flux, for which there is no physical basis for doing. To the extent that non-radiative flux is leaving the surface (mostly via latent heat), it’s also being returned in part somewhere else (as the temperature component of precipitation, wind, weather, etc.), and during its circulatory path, after the latent heat is given up as the water condenses, some of the energy is radiated back to the surface and some into space. Some of the energy is replenished by post albedo solar power and surface emitted radiation subsequently absorbed by the water in the atmosphere. The point is the energy largely stays with the water in kinetic form and returns to the surface as the temperature component of preciptiation.
RW,
No, it isn’t being returned anywhere else. The convective energy lost from the surface is the net energy lost from the surface by convection. Any convective transfer from the atmosphere to the surface has already been included in the calculation. The numbers don’t balance otherwise. Not understanding this fact appears to be your fundamental problem with understanding Figure 7.
Say the 97 W/m² from TFK09 were the gross loss from the surface and 50 W/m² were being returned elsewhere. Then the surface would be receiving 161 W/m² from solar energy, 333 W/m² of thermal IR and 50 W/m² of convective energy for a total of 544 W/m². But it would be losing 396 W/m² as thermal IR and 47 W/m² by convection for a total of 443 W/m². That would be an imbalance of 101 W/m² at the surface instead of ~1 W/m². Can you see the problem there?
You claim that conversion of convective energy to radiative energy is somehow arbitrary or forbidden. You’re wrong. Once the energy has been transferred, the molecules in the atmosphere don’t know where that energy came from. It’s all kinetic, rotational and vibrational energy regardless of the source and the photon emission rate and spectrum is determined solely by the molecular physical constants and the local temperature.
Dewitt,
I’m aware that his numbers don’t balance unless the entire 102 W/m^2 of non-radiative energy is converted into electromagnetic radiation. I’m also aware that much of the non-radiative energy is converted into EM radiation. I’m saying you can’t just arbitrarily convert all of it to EM, and this appears to be what he does. Also, some of the post albedo solar energy absorbed in the atmosphere can get to the surface via non-radiative flux from the atmosphere, but this is not depicted.
His ‘back radiation’ of 324 W/m^2 is probably not correct, as some of this power is returned via non-radiative flux from the atmosphere to the surface.
RW,
How much, and where? Prove it. You’re making an unsupported assertion. KT97 and TFK09 are fundamentally measurement based. You’re just winging it.
Why is it you have such a big problem with net vs. gross? All non-radiative fluxes in both directions are contained in the convective flux term. That’s all. 100 percent. Everything. There are no missing unaccounted for convective fluxes. You don’t see the gross convective fluxes because they’re effectively impossible to measure. The ‘back radiation’ is measured, or at least any model calculations have been confirmed wherever they have been measured.
Your other major error is to continue to insist that you can somehow track individual energy flows. You can’t. They all go into one pool. Once there, the energy has no provenance. Joules are just joules. Once latent heat has been released into the atmosphere by water vapor condensing to liquid, the energy is no different than the energy from the absorption of solar radiation or thermal IR from the surface. That energy causes emission of radiation from molecules that have the physical properties that allow them to emit radiation at the local temperature. Period.
HI RW,
I suspect that there are questions that are not immediately obvious. To begin with why are there significant fluxes or latent and sensible heat at all. Or to put it another way; are they the answer to a problem and if so what was that problem?
Here is a possible way of considering this point.
The radiation budget of the atmosphere is immediately determined by the temperature and GHG profiles, and the temperature and emissivity of the surface. The overal tropospheric budget is funded by other processes but the radiation budget is determined by the temperatures and radiative properties.
For Earth these temperatures represent an troposphere that is not in radiative equilibrium. There exists an average radiative energy deficit. Given that this is the case, then without the involvement any other process the troposphere would progress to cool. Such cooling is constrained by the lapse rate. Attempts to cool will tend to produce greater instability and hence greater convection and greater transport of energy from the surface upward.
To that degree the latent and sensible heat fluxes act in response to the radiative deficit of the troposphere.
Were it not for the lapse rate constraint the atmosphere could relax towards a radiative equilibrium. In the presence of a lapse rate constraining the troposphere to a temperature gradient above radiative equilibrium the troposphere enters a regime with convention vigorous enough to close the troposhpere’s energy budget.
The sum of the latent and sensible heat fluxes must equal the energy deficit due to all other tropospheric processes.
Alex
Bryan,
*chirp, chirp*
With 200+ comments in, the discussion has been interesting.
There is value in people attempting to explain a physics process (or combination of physics processes) in different words.
My biggest barrier to understanding any physics subject has always been lining up the maths, the underlying physics fundamentals and “the conceptual idea”. The last one is always difficult. It doesn’t matter if the maths is clearly correct and the equations draw on sound physics principles, there is always a problem getting it in my head if it “doesn’t quite make sense”.
Maybe other readers don’t face that problem?
When stuck I have found that reading 2 or 3 textbooks helps – more than one way of phrasing the problem. Or coming back to the same textbook or paper a month or two later.
Clearly some blog readers have a conceptual problem with the “greenhouse” effect. Any ideas, or rephrasing, that can help those conceptual problems is a good thing. Some have a religious faith that the “greenhouse” effect doesn’t exist. No words or equations will help them with their conceptual problem.
A few comments along the way, in no particular order:
1. The “radiative resistance” idea suggested by Leonard is just another conceptual idea to help students solve heat transfer problems. Most heat transfer textbooks introduce it to help solve some problems. But there is no such thing as “radiative resistance” – it is an artificial construct, a useful way of thinking about heat transfer.
2. Most of the commenters understand the same physics but want to use different explanations to highlight “the key idea”. If the maths are the same for this group then we all understand the same process. Is there only one correct English description of a complex process?
3. Leonard’s comparison of the various simple models he created is instructive. The idea that in the conduction case, radiation in both directions still exists but nets out to zero at each point is another way of highlighting the important of net radiation in the real atmosphere. If that helps some readers understand how atmospheric heat transfer works then it will have been good value.
4. If the discussion had stayed only on the steady state description of an idealized “greenhouse” effect then it would have been less interesting than it has turned out. The discussion has drifted into questions about whether the lapse rate will be constant and the concept of the result of changes in CO2 concentrations.
a) the lapse rate – this cannot be assumed to be constant under changing climate due to “greenhouse” gas changes. There is nothing wrong with working out the changes IF the lapse rate stayed constant as it gives a “no feedback” instructive result. Nothing wrong with that – so long as it is recognized that this is an instructive result only.
b) calculating the effect of CO2 changes in the radiative physics from some linear/log relation assumed from the surface / TOA energy balance in KT97 – this won’t work except by luck. Real calculations (using radiative transfer calculations) of the effect of doubling CO2 concentrations result in a net average increase in effective radiating height of only 150m. Who could predict that from a simple calculation? And the “effective radiating height” is itself a huge oversimplification – a conceptual model to help the student. The climate radiates to space from the surface through to the stratosphere, and as CO2 increases this profile changes in a wavelength dependent way.
And as DeWitt Payne said earlier:
Hi scienceofdoom
“Clearly some blog readers have a conceptual problem with the “greenhouse” effect. Any ideas, or rephrasing, that can help those conceptual problems is a good thing. Some have a religious faith that the “greenhouse” effect doesn’t exist. No words or equations will help them with their conceptual problem.”
I think there is an existential proof for one aspect of the GHE based on something directly observable namely the tropopause. The maintainance of a persistent temperature minimum, higher temperatures both above and below without some process other than conduction, diffusion, and convection, all of which must tend to transfer or transport energy to the tropopause, is highly problematic. No matter how slowly heat is transfered to the tropopause some process must exist that is removing the equivalent amount of energy. It is also a process that must act at a distance as it cannot involve a net transfer to any part of the atmosphere that is at a higher temperature due to the second law. Provided that a tropopause persists at all latitudes there is a problem that cannot be resolved by conduction, diffusion, and convection alone. If path thermal radiance wasn’t known to exist one would have to propose the existence of some process with similar properties.
Secondly, your calculation elsewhere which gives a significant value to the latent heat flux into the troposhere poses a similar and greater challenge without some process whereby an equivalent flux is transfered out of the troposphere.
I suspect that a simple calculation may show that this flux and any reasonalbe estimate of the upward flux from the surface, which is directly observable, will show that their sum greatly exceeds the average total insolation and hence the total average rate of energy loss through TOA. If that is the case there needs be a transfer of energy from a colder atmosphere to a hotter surface which I think is the basis of an existential proof for a transfer with similar properties to backradiation. I suspect the observable surface upward flux rates may be sufficient for an existential proof of “heat trapping” if they exceed known satellite observations of total insolation once adjusted for observable albedo. For if energy is leaving the surface at a greater rate than it is passing through TOA something between the two must be accounting for the discepancy.
I think that most aspects of the GHE can be demonstrated to exist in our troposphere by recourse to simple inequalities based on observables. It is a lot easier to demonstrate the existence of such processes than to quantify them. One only needs to demonstrate an inequality in the flux balance that would be otherwise unresolved. I do not think one needs a lot of equations to prove that the principle aspects of the GHE are evident.
Alex
Hi DeWitt
You wrote:
“Alex,
I agree with Neal. I think you could rephrase your argument in terms of energy and entropy instead of ‘work’. That would avoid using the term ‘heat’ as well.”
You are absolutely right!
Radiance is not well catered for in the verbal forms of the Laws of Thermodynamics. It is not heat and it is not organised work but heat or work are the options that we are offered to pick from.
It seems to occupy a half way house, a disorganised form of work, where we must consider the entropy separately. We must resolve the second law at a higher level in the system not at the level of the interaction.
You are right, the satisfaction of the second law can and arguably should be derived directly from the energy flows and entropy production.
You are right in that if I may not be allowed to argue based on work then I think similarly one should not be allowed to argue based on heat. Neither term is fully appropriate at the level of the physical interaction as we have come to understand it in the centuries since the verbal forms of the law were constructed.
Energy is in transit from a colder to a highr temperature and once absorbed into internal energy at the surface it is oblivious to its source. One cannot proportion the effected rise in temperature to either the Sun or the atmosphere only to their concerted action. Temptation to allocate the effect to the Sun on the basis that we could then associate that component of the radiation as heat can be avoided as we can resolve the second law directly without recourse to heat.
I hope that clears things up. By going down the work route I hope to have drawn attention to the equivalent hazards that beset the heat route.
Alex
Think of it this way. Heat is a form of energy. Heat is emitted or absorbed by thermal objects, thermal objects have positive temperatures. There is no problem considering the Earth or the atmosphere as thermal objects. In the same way the totality of photons emitted/absorbed by the earth or the atmosphere is a thermal system (OK, you have to keep in mind the various changes in temperature from one place to another, but you have to do so for the Earth and the atmosphere also). If you consider the transfer of energy to and from all of the CO2 molecules to the existing collection of photons, that is a transfer of heat.
Eli Rabett
Good for you Eli
Its great to find a man who sticks to his delusions.
Your multiple postings here, there and elsewhere to the effect that heat can spontaneously transfer from a colder object to an object at a higher temperature shows you inhabit a logic free zone.
Is there any point in repeating one more time another rational reason why you are completely wrong?
You would ignore the post as you have ignored all the previous ones.
That’s why I ask you to get Joel Shore to explain the situation to you.
You remember Joel don’t you?
He was the physicist who was a co- author with you in the G&T comment.
Joel now realizes that it was a mistake to imply in the comment that heat transfer goes spontaneously from cold to hot.
Perhaps he can (what nobody else can) explain that this is impossible.
There is no debate in physics about whether or not heat can flow spontaneously from a lower to a higher temperature object.
Thousands of physics textbooks and thousands of physics departments unanimously agree with Clausius that it cannot.
To argue otherwise is to peddle pseudo science.
Bryan,
They should use you as the poster child for the straw man argument. Once again, you’ve made may day more pleasant by your unintentional humor. Or to put in in texting terms: LOL. Not quite ROTFL, but close.
Lulz?
Eli,
I believe lulz is technically the plural of lol.
Hi Frank,
“Surface energy balance will be restored by a combination of increased upward convection and radiation. The latter requires an increase in surface temperature, but the former may not. Evaporation is limited by diffusion/convection of water vapor from surfaces and increases dramatically with wind speed. Assuming that all of the energy from increased DLR is removed by increased radiation allows one to calculate a maximum temperature rise (which will be modified by water vapor and especially cloud feedback).”
There is another energy imbalance that needs to be resolved and arguably acts more directly.
The sum of the latent and sensible fluxes act in response to an equivalent energy deficit in the tropopause, which in its greatest part is a response to the difference between the dynamically resolved lapse rate constrained temperature gradient and the temperatures were the gradient the result of radiative equilibrium alone.
Any increase in the troposphere’s total (considering all LW and SW) radiative deficit needs resolution and that implies an increase in the other available fluxes, i.e latent, sensible fluxes. I believe that all other non-radiative transfers, e.g. surface friction and transport throught the tropopause are small by comparison.
What I mean to say is that the sum of the latent and sensible transfers is not free to take on a wide range of values as might appear to be the case when only considering the surface balance. Their aggregate is constrained by and largely equivalent to the troposhere’s radiative energy deficit. How that sum is apportioned between latent and sensible is open to the detailed effects you mention but the sum is I think well constrained by the radiative processes in the troposhere.
Alex
Alex wrote:
“What I mean to say is that the sum of the latent and sensible transfers is not free to take on a wide range of values as might appear to be the case when only considering the surface balance. Their aggregate is constrained by and largely equivalent to the troposhere’s radiative energy deficit.”
I’m not positive what you mean in the term the “troposphere’s radiation deficit”. I presume that you are referring to the fact that the troposphere is too opaque at thermal infrared frequencies to transmit a large enough upward flux to balance incoming SWR + DLR. So convection makes up this deficit. However, we calculate this radiation deficit by assuming that the surface has a certain temperature and the atmosphere has a given pre-set vertical temperature and humidity profile. In some cases, clouds are considered. To some extent, these inputs are determined by convection and mixing. How does one know how tightly convection is “constrained” by the “troposphere’s radiation deficit” when the radiation deficit is determined by convection.
Suppose one could turn up the speed of the Hadley circulation so that the mean environmental lapse rate changed from 6.5 degC/km to 6.0 degC/km. Following Leonard’s arguments, the temperature of the characteristic emission level (or levels for different wavelengths) is in radiative equilibrium with net incoming SWR, so its temperature is fixed. If it’s 10 km from the characteristic emission level to the surface, turning up the velocity of the Hadley circulation will cool the surface by 5 degC. As best I can tell, the mean environmental lapse rate of 6.5 degC is not derived from any fundamental physics.
Now suppose the radiative forcing from AGHGs mostly goes into increasing the speed of the Hadley circulation, rather than into raising the surface temperature and OLR.
SOD had a post on potential temperature with a figure showing that much of the atmosphere mixes vertically so that the moist potential temperature (aka equivalent potential temperature) is roughly constant overhead (but varies with latitude). If we know the moist potential temperature in one of these well-mixed locations, we have a reasonable estimate of the moist potential temperature overhead. However, any given moist potential temperature can be produced by various combinations of temperature and moisture. (The only fixed point is saturation, but mixing ensures that relative saturation is less than 100% except in the vicinity of the relatively few locations and altitudes where there are clouds. Both unfortunately are determined by convection.
Frank,
Of course the lapse rate is derived from fundamental physics. It’s just not derived from simple fundamental physics. Also Leonard is wrong. Let’s say that with a 6.5K/km lapse rate, the effective height of emission is 5 km and the temperature at that altitude is 255K and the surface temperature is 287.5K. If the lapse rate drops to 6.0 K/km but you fix the temperature at 5 km at 255K, then the surface temperature will be 285K. The average temperature of the troposphere won’t change much, but most of the IR radiation comes from the first 5 km because that’s where 90% the water vapor is. That part of the troposphere will then radiate less and so will the surface. The upper part of the troposphere will radiate more, but not enough to make up for the loss in the lower troposphere. So you won’t have radiative balance at all at any altitude. Not to mention the fundamental contradiction involved in having a lower lapse rate, which is always associated with higher specific humidity, with a lower temperature atmosphere which won’t hold as much water vapor. So in fact, there will probably also be less water vapor to radiate. It’s not at all clear that increasing the Hadley circulation would lower the lapse rate. It would probably raise the lapse rate in the descending branch.
Also, turning up the Hadley circulation will reduce the latitudinal temperature difference. But it’s the latitudinal temperature difference that drives the Hadley circulation. If the Hadley circulation could go faster, it would. There are constraints on that too.
Frank,
This really is classical thermodynamics. I’ve called for Bryan, but if he doesn’t show, I’ll try to argue from what I would think would be his point of view. At the adiabatic lapse rate, parcels of air can be moved up and down without doing work. The potential temperature is constant. When the lapse rate is less than the adiabatic rate, potential temperature increases with altitude and work has to be done to raise and lower parcels of air. That takes free energy. In the atmosphere that can only come from a temperature difference at constant gravitational potential energy, i.e. the meridional temperature gradient, which also causes a pressure gradient force that increases with altitude. There’s an additional factor that Carrick, I think, mentioned over at the Blackboard, in the Hadley circulation but I don’t feel like looking for it right now.
The way I read your post, you’re suggesting that the lapse rate can be decreased, moving it further away from the adiabatic rate, by increasing the circulation rate. That would mean a lot more work being done, higher velocity against an increased force. Where do you suggest this extra free energy will come from?
Oh, and the vast majority of air that moves up due to thermals caused by solar radiation comes back down again locally. Net vertical velocities in the atmosphere are usually measured in units of fractions of a Pa/s. That’s quite slow in m/s. See for example here: http://en.wikipedia.org/wiki/File:Omega-500-july-era40-1979.png
Hi Frank,
I have responded below:
Alex
I think its appropriate to do a reality check here.
There is no debate in physics about whether or not heat can flow spontaneously from a lower to a higher temperature object.
Thousands of physics textbooks and thousands of physics departments unanimously agree with Clausius that it cannot.
To argue otherwise is to peddle pseudo science.
The fact that thermodynamics is a hard subject is not an excuse to ‘dumb down’ the technical definitions of thermodynamics.
Heat is not temperature neither is it infra red radiation.
To argue otherwise is to peddle pseudo science.
Carnot and Clausius are still very relevant their work is studied at every university on the planet.
On the other hand who studies John (who he) Denker?
An idea that utilises the Carnot cycle in a description of the atmosphere is within the orthodox framework of science.
To suggest that this is outdated and that the person should ‘move on’ is groundless nonsense.
To argue otherwise is to peddle pseudo science.
Classical thermodynamics is not in conflict with statistical mechanics.
They are two ways of looking at the same phenomena .
Neither is simple and each has its own strengths.
To argue otherwise is to peddle pseudo science.
Reading this thread reveals again how semantics creates problems in understanding and how semantics is also sometimes used purposefully to mislead.
As DeWitt Payne noticed early in this thread the source of most of the semantic problems is in the concept of heat and in particular in the fact that several people take a extremely formal view that reflects the 19th century classical thermodynamics. Classical thermodynamics was a formal mathematical theory that was found to produce many successful predictions. It was essentially axiomatic as its results were derived from axioms that could not be explained or understood within the theory.
Now we have also statistical thermodynamics which is more powerful as it can be used to derive those axioms from mathematical statistics and a little input from microscopic physics. The limited way of looking at the concept of heat was well justified when the theory was an axiomatic theory, the present deeper understanding of the physics has led to a less formal use of the word.
Thermodynamics is a surprisingly difficult field of physics, surprisingly at least in the sense that very many people believe that they know it much better that they really do. There are also really many engineers who use parts of thermodynamics routinely and correctly as long as they stick to the things they are used in doing but who are likely to fail when required to apply thermodynamics to other problems.
The atmosphere is a complex system where thermodynamics plays a major role. Few people master all the present knowledge on atmosphere and they are likely to know in a complex way: as a collection of very many facts whose interconnections are not very clear. When they or other people with more limited but still fair understanding of the atmosphere try to simplify the picture and tell what’s the real nature of GHE each of them creates a different explanation. They all agree that those explanations are incomplete simplifications but they disagree on what’s most essential to include and what’s better to leave out.
I don’t think that there’s any resolution for choosing the right answer for the question “How GHE effect works?” It’s perhaps easier to agree on questions like: “How GHE manifests itself in the surface energy balance?” or “How the outgoing radiation is affected by GHE? or any number of similar questions on some aspect of GHE.
Another issue discussed in this thread was raised by Omnologos:
Is the “average height where radiation occurs” anything more than a mathematical layer? After all, the adiabat goes up to the tropopause, below which convection is king
In my view this goes really to the core of the matter. To understand the GHE we should discuss in some more details how the altitudes of emission are increasing and how this general increase affects the GHE. The average height is really a result calculated backward when the real results are known. It does not explain anything by itself.
Thus what should we understand in a simplified but more realistic approach?
– First we may calculate the radiative forcing for unchanged troposphere.
– Then we should check how the “no-feedback” warming occurs
– – How the altitude and the temperature of tropopause is determined in our simplified atmosphere?
This point is important as the no-feedback atmosphere is supposed to maintain the lapse rate as closely fixed as possible but keeping the tropopause unchanged would lead to a contradiction. Consistency can in my view be restored in this simplified model only by allowing the troposphere grow a little.
The simplified model is likely to have a precise altitude where radiative heat transfer reaches a balance by itself without any contribution from convection. This point depends on forcing and on CO2 concentration at those latitudes.
This is the altitude where I would fix the connection between the temperature and the altitude and from where I would use lapse rate to calculate the temperature of the surface.
My guess is that this same mechanism is hidden in more detailed atmospheric models and perhaps the closest simple approximation of how the warming will be determined.
In all the above I skip totally the standard feedbacks as considering them is not part of this step but the next one.
HI Pekka,
We possess the collective wit to construct models sufficiently analogous to demonstrate the interplay between some basic atmospheric processes and constraints. Sufficient to demonstrate the tendency for a lapse rate constrained troposhere to develop, to show how that effects the local energy balance and how such changes may be resolved by energy transport by convection.
They would only be crude analogues not to be mistaken for the real thing but sufficient to demonstrate aspects of the roles played by the included processes and their interplay.
Sufficient to allow some of the commonly prescribed constraints to be relaxed, for an atmosphere that expands and contracts according to its temperature, for the height at which the temperature curve changes from being constrained by the lapse rate to be constrainted by radiative balance to move and be resolved according to local energy balance.
Sufficient to demonstrate how the structure could evolve in response to increased IR activity in the atmosphere, or to a change in the lapse rate.
I am pretty confident in this for I am pretty confident that something similar was achieved many years ago.
I ask you whether you think such demonstrators of principle would be a useful enterprise?
Either at the level of actually doing so physically, or simply engaging in the thought necessary to design such an analogue. Sometimes just the necessary directed thinking can be as insightful and arguably less contentious than actually constructing such an analogue.
If either would be a good idea then I am confident that we have the wit but perhaps neither the motivation nor the tolerance to embark on such a project.
Alex
Alex,
What I have been pondering is finding the simplest model that is internally consistent and that describes all essential issues required for understanding the structure of the troposphere quantitatively enough to allow for approximate quantitative calculation of the GHE.
Could one do that with one-dimensional models or does that require some kind of description of the Hadley cells and other similar dominant features of the atmosphere? That’s one of the questions in my mind.
Pekka,
My thinking was more towards what is necessary to demonstrate the origins of atmospheric structure which only requires that a model can get the signs right and the order of magnitude.
As soon as one wants quantitative estimates I suspect one needs a full atmospheric circulation model, and to be fair they struggle to agree.
The type of demonstrator of principle would be aimed below what you wish for.
That said, it would be aimed high enough to demonstrate the following:
That the addition of a uniform ghg concentration would produce an analogue of the troposhpere but would likely get it wrong for the following interesting reasons.
Contrained to produce the correct surface temperature it would:
have too little backradiation, too shallow a tropospere, and too small a requirement for latent/sensible flux.
I am suggesting that such a “dry” atmosphere would never get these things right and stressing the importance of water vapour with its distinctive tendency for its concentration to fall with height/temperature to increase backradiation.
To demonstrate the roles of solar absorption IR emission by stratospheric gases in generating analogues of the stratosphere and the tropopause hence that increasing stratospheric emission results in its cooling, which is not exactly rocket science but worth demonstrating.
To demonstrate that solar absorption below the tropopause is compensated by reducing the requirement for latent and sensible fluxes up to the point where the gradient breaks away from the lapse rate constraint.
I think the above includes a few things that may not be intuitively obvious and perhaps some that I think are obvious but are plain wrong. That would be part of the fun.
I believe that much or all of this could be demonstrated with an atmosphere that is radiatively simple, e.g. various intensities of grey, and computationally simple, e,g, implementable in Excel with Visual Basic macros and hence rather accessible.
My greatest concern would be the getting across the idea that it would be a demonstrator of principle nothing more. People can be quite hostile particularly if it suggests things that they don’t like.
It would get things wrong but that is not a bad thing. It at least raises the issue of how one should try to correct it, e.g. does one tune it, add better science, at what point does meddling to get the right result become cheating. It might also demonstrate some common modelling issues in simple form such as discretisation in space and time and the instabilities and errors that can entail
Given the difficulties with demonstrating the reasonability of the science, I should like to think that a group could form to establish what can be demonstrated by applying the level of thinking typically available on blogs. If this turns out to be well short of reality then that would be a lesson in itself.
Alex
Alex,
I agree that fully quantitative results are an unrealistic goal, but getting reasonably close to the same value for “no-feedback climate sensitivity” as the big models might just be possible. That could perhaps be reached in a way where the agreement applies also to the details of the simple model. For that to be possible those details must have counterparts in the big models, which is not always true for the simple models.
Pekka,
If you mean the 1.2 ºC figure, I think I would struggle to deduce this from any model based on first principles.
Checking with the IPCC it seems to have come originally from Hansen 1984 and Bony 2006, the latter refering to Soden & Held 2006 to get a value for the Planck feedback. The other part, the forcing has been calculated directly but I have no idea how that was done and also from papers by Soden and Held, and doubtless others.
I have always regarded the value for the Planck feedback based on S-B Law going from 255 to 256ºC to be a case of nearly right answer, completely wrong method.
Modtran, clear sky, standard atmosphere gives some value close to 0.8ºC if I recall correctly which is not very close but why should a clear sky estimate be close.
Ultimately it seems to be a based on GCM simulations and happens to be a value that they can mostly agree upon.
From my limited experience no model working from sound physical principles that is simpler than Modtran is likely to give as good an answer except by luck.
Trying to build a demonstration model would I think show that there is a big difference between getting the right basic structure and getting useful values.
Sorry to be negative but I think it is a very difficult question.
Alex
Alex,
By simple I didn’t mean that simple, only that the model is conceptually simple and based on physical principles. Thus I would include MODTRAN as part of the model.
For the one-dimensional case MODTRAN would be used to calculate the radiative part assuming the input profile for the temperatures, then some rather straightforward approach based on lapse rate would be used to calculate a new temperature profile. These two steps would be repeated iteratively until they converge which should not take many steps.
One location cannot, however, represent well the whole atmosphere. Thus the calculation would be repeated at several locations and the results combined.
But then the question arises whether some circulation must be included to make the model self-consistent at the top of the troposphere and at the surface. If this is required then the model starts to be a GCM, perhaps a small GCM but something like that anyway.
Pekka,
I understand better now.
I am not quite sure how I can assist but to begin it may help if I make a few observatins about Modtran.
These are to the best of my recollection:
The way that changing the surface temperature effects the prescribed temperature gradient leads to unphysical behaviour just below the tropopause. The tropopause remains fixed but the troposhere moves with the surface.
The calculation with the addition of clouds is optimised by a repositioning of the vertical layers, it also includes a new prescription for the RH which is sensible but its effect is perhaps not obvious, e.g. the effect of clouds is the effect of clouds including the effect of the changed WV profile.
The calcualtions are doubtlessly done to great accuracy in steradians but the conversion to the output values is achieved by multiplying rounded values by 3.14 as opposed to pi and this results in flux values that move in steps of 0.031 or 0.032.
At the heart of it there should be a general purpose core to which the prescriptions are applied. With 33 vertical layers and 140 bands it is a sophisticated tool. I suspects that it compares well to the radiative code in the GCMs, but I am not certain of that as I don’t know how many bands they use.
Judging from the detailed output which includes a header cards section the prescribed profiles are not input at runtime, they are built in and only selected at runtime.
It doesn’t seem present an easy way to calculate the energy balance for each layer, it doesn’t need to perform this calculation as it relies on a temperature prescription. For the troposhere/tropopause one needs to know how the enerfy balance changes to give some indication of how the tropopause height might change as additional CO2 is introduced. That would still only be half the story as the SW absorption/scattering although it seems included in the package is not active in the LW runs available through the online interface.
I don’t think I have done them any injustice but these are my observations in the absence of any contact with the authors.
The question becomes one of whether this is this enough either as freely available or perhaps if more open access were available. Is this old version (MODTRAN 3 Version 1.3 12/1/95) still propriety software? That I do not know. MODTRAN 5 has ten times better bandwidth resolution and retails at ~US$1000 depending on intended usage.
Alex
Pekka,
The book A Climate Modelling Primer by Kendal McGuffie and Ann Henderson-Sellers comes with a CD containing, among other things, a simple one-dimensional radiative/convective atmosphere model program. Unfortunately, it’s a Fortran program with no graphical user interface for Windows. As a result, unless you’re used to using that sort of thing, it’s not what you would call user friendly. I don’t think it’s quite as bad as LBLRTM, but close. For the results of this sort of calculation see for example Manabe and Strickler, 1964.
Click to access sm6401.pdf
A full line-by-line radiative transfer program is available by subscription from spectralcalc.com. It’s become more flexible over time and now includes the ability to create your own atmosphere. The downside is that the output files can be huge. Since they have an upper limit on file size, calculations may have to be split into several frequency bands.
The MODTRAN version on the web is severely limited for trying to calculate either forcing or temperature change. The pressure vs altitude profile never changes. The lapse rate never changes. Surface temperature changes only propagate up to 13 km. You can only change the CO2 level in the stratosphere. The humidity level in the 1976 US standard atmosphere is relatively low, so it isn’t really representative of the planetary average. But it does give you a decent ball park estimate and the calculated spectra look reasonable when compared to measured spectra.
Ann Henderson-Sellers was also one of those responsible for resurrecting the Wood greenhouse experiment paper. Unfortunately, she and her co-author M.D.H. Jones ( History of the Greenhouse Effect, 1990) apparently didn’t bother to search further issues of the journal to find the rebuttal by Charles Greeley Abbot and now the idea that the radiative transmission characteristics of greenhouse glazing has no effect on greenhouse temperature seems to have achieved unkillable zombie status.
http://ppg.sagepub.com/content/14/1/1.abstract (paywalled).
Yes, you can do it with 1-D models, many have been produced and work. See Yuk Yung’s early iD model of Titan for example
DeWitt
“You can only change the CO2 level in the stratosphere.”
I doubt that you mean that, do you?
Alex
Alex,
Poor phrasing plus I left something else out. I think this is a better statement of what I meant: In the stratosphere, only CO2 and ozone levels can be changed, nothing else. Changing water vapor and temperature only affects the levels in the troposphere.
Thanks for all the responses. They answer much of what I wanted to know. I’m not sure that I would really go on and do the analysis as I was asking the questions to learn what’s possible. I could express the question that I had in mind in one more way:
Radiation codes like MODTRAN allow for calculation of forcing as described for example in the papers of Myhre et al. Typical result: 3.7W/m^2
The forcing can be expressed as an equivalent change in effective radiative temperature of Earth. Typical result: 0.9C.
Next we are told that this corresponds to the change of 1.2C in the surface temperature. My question can now be formulated: What’s needed at the minimum to tell even approximately the ratio of this value to the change in the effective radiative temperature.
Related questions include the question on how the troposphere changes in this warming.
In some sense this questions are not really meaningful as the real atmosphere can never show the no-feedback warming as the feedbacks kick unavoidably in.
One more reason for asking these questions is that there are all too many simplified explanations for the GHE. Being simplified all of them are in a sense wrong and I’m trying to improve my understanding on how one can improve the explanations without the need to refer to large scale models.
Working with old scientific Fortran code is not too bad for me as I have both developed and used such code over 25 years or so (1970-95) and continue to work with C which is not much better.
Since Bryan obviously isn’t going to answer the questions I asked about average temperatures:
For an emissivity/absorptivity over all wavelengths of 1 and S = 1368 W/m²:
1. max=min=average=278.68K
2. max = 394.12 K, min = 0 K (ignoring CMB) average = 157.4 K
3. max = 296.0 K, min = 0 K, average = 275.6 K
For case 2, the maximum temperature occurs at only one place, the point where the sun is directly overhead. Everywhere else has a lower temperature. The minimum temperature is everywhere the sphere is unlit. Case 2 is in G&T where they calculate the analytical solution for the double integral.
For case 3, the max temperature is at the equator for a sphere with axis of rotation perpendicular to the orbital plane and the minimum temperature is at the poles. Note that the average temperature is less than 3.3K lower than for the uniform temperature superconducting sphere.
Here’s 3D plot of the flux at the surface using what amounts to a Mercator projection with longitude on the x axis, latitude on the y axis and flux on the z axis:
Bryan,
Where are you when you could actually make a contribution?
See Frank’s comment above: https://scienceofdoom.com/2012/07/23/how-the-greenhouse-effect-works-a-guest-post-and-discussion/#comment-18367
Atmospheric circulations are heat engines and are therefore constrained by classical thermodynamics. Frank doesn’t seem to think so. He suggests in the linked post that the circulation rate of the Hadley cell and the lapse rate are arbitrary. I disagree and I presume you do as well.
DeWitt: Thanks for taking the time to respond. I am struggling to understand this subject. (Useful refs would be appreciated.)
I’ve been trying to understand GHG warming from a surface energy balance perspective: More GHG’s will produce more DLR and that extra energy can be removed from the surface by increased convection or increased LWR. Increased LWR requires surface warming, but increased convection of latent heat may not, because evaporation increases with wind speed as well as temperature. (Thus my fantasy about cooling the surface by running the Hadley circulation a little faster and producing a lower lapse rate by moving energy to the upper troposphere.) Future temperature rise depends on what fraction of the energy from future increased DLR goes into raising surface temperature/radiation and what fraction goes into latent heat/increasing precipitation.
If one slowly adds GHG to a planet that had none, all of the increased DLR at first will be used to raise the surface temperature because the lapse rate will not be unstable anywhere. At a certain point, however, adding more GHG will produce an unstable lapse rate somewhere and some of the increased flux from DLR will be removed from the surface by convection. As more GHGs are added, I suspect that a larger fraction will be removed by convection. It wouldn’t be surprising if GCM’s did a lousy job of partitioning this increasing upward flux into radiative and convective components.
Unfortunately, one needs to consider more than surface energy balance and moving increased energy from DLR a couple of hundred meters above the surface. Its need to rise kilometers to escape to space by radiative cooling, potentially changing the lapse rate. (Here is where I’m struggling.)
You wrote: “At the adiabatic lapse rate, parcels of air can be moved up and down without doing work.” Very clear. In the case of the Hadley circulation, some packets are going up and some packets are going down. Does this imply that the lapse rate in both regions must be adiabatic? Or can dense sinking air do work and force air to rise somewhere else.
I didn’t mean to say that fundamental physics doesn’t create a 6.5 degC/km mean ENVIRONMENTAL lapse rate. I just haven’t seen anyone DERIVE a 6.5 degC mean environmental lapse rate from any rational combination of dry and moist adiabatic lapse rates (which are calculated from fundamental physics). In ascending regions, the lapse rate theoretically could be the DALR up to the condensation level and the MALR above. In descending regions, it could be the DALR. However, radiation enters into the picture too, especially for slow-moving parcels. In reality, radiosonde profiles are far more complicated that my understanding would suggest, probably due to mixing between parcels. If we can’t say why the mean environmental lapse rate is 6.5 degC/km, how do we know it can’t change with added GHGs.
Once a surface parcel containing additional latent heat rises high enough in the atmosphere, the lapse rate is stable and the parcel will stay there until it radiatively cools enough to sink.
Frank,
Have you read R. Caballero’s Lecture Notes on Physical Meteorology or some other textbook on the subject? If not, you should.
Click to access PhysMetLectNotes.pdf
As I said, the physics of the environmental lapse rate aren’t simple. There won’t be an analytical solution as there is for the adiabatic rate. Basically, you need a full-bore GCM, which doesn’t exist yet. The problem with current GCM’s is that they don’t really do convection because the scale is too coarse. The grid size is 100km. So they readjust the atmosphere profiles in a grid box whenever some stability criterion is exceeded. I’m pretty sure what that amounts to is that they force the lapse rate to the current environmental rate for that part of the planet.
That being said, there are still constraints that can be imposed based on classical thermodynamics and radiative transfer. Which means the lapse rate isn’t arbitrary.
The lapse rate isn’t actually adiabatic anywhere. Dense sinking air can indeed do work, but that’s not the case with the Hadley circulation. Remember that the rising air near the equator starts out with a lot of water vapor. But that condenses and rains out. So the descending air is very dry and will be warmer and less dense than the air around it as it sinks assuming an adiabatic lapse rate. So again, work must be done to bring that air back to the surface. If the rate were adiabatic, then no work would need to be done and circulation would increase. But that circulation will decrease the lapse rate for both the ascending and descending legs until the heat engine reaches something like steady state. If the circulation is too fast, the meridional temperature difference will drop and the lapse rate will decrease and the free energy available to drive the heat engine circulation will drop below the level necessary to maintain that circulation. And conversely, if there were no circulation and the lapse rates were adiabatic, the pressure gradient force would start circulation.
The pressure gradient force is caused by the warmer air at the equator being less dense than the cooler air at higher latitudes. That means that the pressure decreases less rapidly with altitude at the equator. Which means that at a given altitude, there will be a pressure difference that will drive air circulation toward higher latitudes. That will generate a corresponding flow in the other direction at the surface and pretty soon you get a Hadley cell. It’s actually much more complex than that. There are concepts like baroclinic and barotropic stability involved. Read Caballero to at least get a start.
Leonard Weinstein has presented a plausible model for heat transfer through the troposphere.
The model is consistent with the laws of thermodynamics.
Its my interpretation that besides heat absorption at BOA and radiative heat emission at TOA the troposphere is relatively straightforward if we restrict our comments to DALR conditions.
DALR = g/Cp = -9.8 K/km is followed for a neutral troposphere.
The radiative effects are included in the bulk quantity Cp.
For adjacent volumes of air the radiative effects are largely self cancelling.
However gradually higher up the tropopause there is a net radiative loss to space.
The height of the troposphere adjusts to balance BOA and TOA
So the more interesting part is in the region near tropopause and above.
This video clip discusses the implications of ice crystal formation at 80Km.
Hi Frank,
Thanks for replying, I will try to respond to your:
“I’m not positive what you mean in the term the “troposphere’s radiation deficit”. I presume that you are referring to the fact that the troposphere is too opaque at thermal infrared frequencies to transmit a large enough upward flux to balance incoming SWR + DLR. So convection makes up this deficit. However, we calculate this radiation deficit by assuming that the surface has a certain temperature and the atmosphere has a given pre-set vertical temperature and humidity profile. In some cases, clouds are considered. To some extent, these inputs are determined by convection and mixing. How does one know how tightly convection is “constrained” by the “troposphere’s radiation deficit” when the radiation deficit is determined by convection.”
Taking this one bit at a time:
“However, we calculate this radiation deficit by assuming that the surface has a certain temperature and the atmosphere has a given pre-set vertical temperature and humidity profile. In some cases, clouds are considered.”
It is the instantaneous temperature and concentration profiles of gases and condensed water that determines the local emission of IR. The convective energy transport has to play its part in maintaining an energy balance but it does not determine the IR emission so directly.
The local radiative energy balance is the amount of radiation absorbed by a local volume less the amount emitted. For the troposphere this on average has a negative value. On average each volume in the troposphere is a net radiation source. At individual moments volumes could be a source or a sink (e.g. a cloud base).
This may seem to be an apparent paradox, particularly if an energy trapping metaphor is being applied. On average the tropospere acts to convert its internal energy into net radiative energy, it acts to release energy not to trap it. It can be said to do both but the net effect in the troposphere is to convert internal energy to radiance.
In the absence of some constraint, this would cause a local tendency to cool everywhere in the troposhere on average. One constraint is the need, in the convecting troposhere, to adhere to the temperatures implied by the lapse rate in conjunction with the surface temperature. This constrains the temperatures to be above those for radiative equilibrium.
As it is so constrained, the energy deficit has to be made good by some other process and the appropriate process for a lapse rate constraint is to engender instability producing convection and transport of energy from the surface up through the troposhere.
“To some extent, these inputs are determined by convection and mixing. How does one know how tightly convection is “constrained” by the “troposphere’s radiation deficit” when the radiation deficit is determined by convection.”
I do not think I would be splitting hairs to say that the radiation deficit is determined most directly by the radiative field, the temperatures and concentrations, but funded by the convective energy transport. Thereby the convective transport maintains the radiative processes rather than determines them directly.
That is one end of the stick!
You are emphasising the other end of the stick, that the radiation deficit is supported by the net average convective energy transport into the troposphere.
For the whole stick, both are true in combination. I happen to think that a better view is available when both apects are considered in conjunction.
“”I’m not positive what you mean in the term the “troposphere’s radiation deficit”. I presume that you are referring to the fact that the troposphere is too opaque at thermal infrared frequencies to transmit a large enough upward flux to balance incoming SWR + DLR.”
That doesn’t seem to conrespond to what I am trying to say. Your mention of DLR+SWR would imply some statement about the surface budget. So far I have restricted things to the troposheric budget, flows into and out of the troposhere across boundaries at the surface and the tropopause. The tropospheric budget must balance, as must the surface budget, the budget for atmosphere from the tropopause upward, and the one for everything above the atmosphere. For the troposhere we can say that the convective fluxes in must on average balance the radiative losses, providing all other transfers are negligible. I give the view that the convective fluxes are primarily a response to the radiative deficit.
That last point is unlikely to be quite true insofar as it ignores the possiblity that the magnitude of the convective fluxes modifies the effective lapse rate and hence the temperature profile and hence the radiative deficit in quite a direct manner. My understanding is that such an effect must be small, which is equivalent to suggesting that any tendency to increase the magnitude of the lapse rate would trigger an unlimited amount of convective transport. I doubt that is true but it does seem to be considered that quite small variations of the average lapse rate would be compatible with large convective transportation.
The question of what determines the surface temperature remains to be addressed and is I think best viewed in terms of the surface budget but in conjunction with the tropospheric budget. The latter can be viewed as providing a value for the convective fluxes as a residual, or balancing term that can be applied to the surface budget. Imbalance in the surface budget implies a changing surface temperature which in turn effects a change in the troposheric budget by way of changes in the radiative field which implies a change in the convective fluxes and hence a modification to the surface budget. This iteration is resolved towards an overall equilibrium were the various budgets all balance on average.
In this view each process acts in response to other process in compliance to certain restraints and it is not clear if any process should be viewed as more crucial or essential, or would make a better starting point. I think we can say that some linkages are more direct than others and I highlight that the effect of convective fluxes on the troposhere’s radiative budget is not a direct one but a mediated one. Other mediations that I have not mentioned are changes in humidity and the formation of clouds. These seem to be rather direct responses to convection and in turn they are act direct determinants of the radiative budget. The system is complex and interdependent, that is just how it is. Sometimes and for some purposes a particular viewpoint may be more intuitive or insightful than another but I doubt that any particular viewpoint is inherently superior to another for all occassions.
Here I am making a special case for some preeminence for radiative processes. In this case the justification is based on the instantaneous nature of the process. They rely solely on the current snapshot and are not directly affected by the systems prior state. I do not think it is so easy to deduce the current amount of convection based on instantaneous values of other observables but we can deduce that it will be acted upon to assist in the production of a troposphere with a balanced energy budget on average.
Now I may have got things very wrong. It could be that cloudiness responds more effectively, e.g. that there is a faster negative feedback mediated by clouds than by convection. I doubt that is the case but I am very open to being put right on that or similar points.
I am afraid that this all seems overly complicated but it is an attempt to make some progress without unwarranted simplifications and exclusions thus allowing for additional processes to be included and for a new viewpoint to be selected if that seems advantageous. It might be useful to attempt a schematic system’s description of all the interdependent processes in terms of what determines each process, the responses to that process and what those responses determine in turn. Alas I think it would be rather complex but it might be fun.
Alex
Hi Alex,
I’m pretty sure that increasing vertical energy transfer by convection reduces the lapse rate, not increases it. It would have to because it warms the troposphere, making it more stable, while radiation cools it. So there is negative feedback and thus a tendency towards steady state.
The big question is whether increasing ghg’s and surface temperature changes the ratio of radiative to convective net energy loss at the surface. Increased ghg’s mean the tendency of radiation to increase the lapse rate will increase, which will have to be matched by increased vertical convection from the surface. Increased vertical convection means increased advection and a flatter latitudinal temperature profile. But that means less free energy to drive the circulation. That also means a reduced lapse rate, which will increase the average temperature of the troposphere and increase radiation upward and downward. But if the specific humidity increases with the temperature, then the reduced lapse rate will still not be very far from the adiabatic rate.
In other words, it’s complicated. Given the rather simplified convection calculations in current GCM’s, I’m not convinced that results from those calculations are definitive. But whatever the case, there will be a temperature increase. It isn’t possible to increase convection enough to cancel the increase in surface temperature from increased ghg’s completely. If convection could increase above the current level without an increase in surface temperature, it would and the current surface temperature would be lower. I’m probably not phrasing that correctly as I’ve said that before and apparently not been understood.
Increased convection should lead to more dissipation, i.e., further from the adiabatic process. For upwards convection that should reduce the lapse rate but on the other edge of the Hadley shell my impression is that it should increase the lapse rate and thus warm the lowest atmosphere more than without this effect. Do you agree?
According to this link: http://paoc.mit.edu/labweb/notes/chap4.pdf (see Figure 4.16)
What you often get in the sub-tropics is a trade inversion. The descending air follows an adiabatic profile, but stops descending at the top of the surface boundary layer (400m-2km above the surface) creating a temperature inversion. I don’t think that qualifies as reducing the lapse rate.
My proposal applies to the altitudes above the inversion. It might well contribute to the creation of an inversion by adding to the temperature above that level.
Alex, DeWitt & Pekka:
Thanks for your replies.
Dewitt,
You say:
“How much, and where? Prove it. You’re making an unsupported assertion. KT97 and TFK09 are fundamentally measurement based. You’re just winging it.
Why is it you have such a big problem with net vs. gross? All non-radiative fluxes in both directions are contained in the convective flux term. That’s all. 100 percent. Everything. There are no missing unaccounted for convective fluxes. You don’t see the gross convective fluxes because they’re effectively impossible to measure. The ‘back radiation’ is measured, or at least any model calculations have been confirmed wherever they have been measured.”
The problem I have is it doesn’t give a full and accurate picture of what is actually happening. For example, some of the post albedo solar flux absorbed by the atmosphere can get to the surface in non-radiative form. Also, the so-called ‘back radiation’ is not measured, but derived from a lot of assumptions and/or deductions based other assumptions. There is no way to get an accurate measure of this unless you have instruments looking up all over the globe measuring downard LW, which of course there aren’t.
You say:
“Your other major error is to continue to insist that you can somehow track individual energy flows. You can’t. They all go into one pool. Once there, the energy has no provenance. Joules are just joules. Once latent heat has been released into the atmosphere by water vapor condensing to liquid, the energy is no different than the energy from the absorption of solar radiation or thermal IR from the surface. That energy causes emission of radiation from molecules that have the physical properties that allow them to emit radiation at the local temperature. Period.”
I’m aware the separate sources of energy flows cannot be tracked or precisely quantified. The system is far too complex and chaotic. Understanding the distinction between them though is fundamental to understanding what is actually happening in terms is net energy flow. Trenberth’s depiction does a poor job of this is my point.
I believe everybody can agree that the papers KT97 and TFK09 don’t tell much about the net energy flow. Neither do they even try to describe in full detail the components of the energy flows which are very different in different parts of the globe.
What the papers do is to present global totals of certain components of the energy flows and their division to ocean and land areas. For many people, and certainly for me, this is interesting information but by itself it does not help much in determination of the net energy flow, more detailed work and other type of information is needed for that. It’s not surprising that the papers do a poor job in something that don’t even try to do.
But they do set some upper and lower bounds.
RW,
So far all you have offered is your opinion. When you construct your own energy balance, let me know. Then we can discuss whether it’s better than KT97 and TFK09.
How is that going to happen when the atmosphere is colder than the surface almost everywhere? When there is a local temperature inversion, sensible heat transfer has to occur by conduction, which is really slow. That’s because a temperature inversion means there is no convection.
So you don’t believe that the atmospheric radiative transfer equations are valid? You believe that they are deductions based on assumptions rather than solid physics? Then why is it that everywhere that atmospheric radiation has been measured, it is in excellent agreement with the calculated value? See for example SURFRAD and EBEX 2000. I don’t need a million gravitometers spread over the surface of the Earth to know that gravity applies everywhere, not just where it’s measured. Radiative transfer physics is probably better understood than gravity. We still don’t have the slightest clue why, for example, the gravitational force is so much weaker than the other forces. But we can calculate the molecular energy levels of CO2 from first principles, ab initio.
To summarize my position:
The greenhouse effect ‘works’ by creating an imbalance in the energy flows in and out of the atmosphere. One of the manifestations of this in the Earth’s atmosphere is an increase in the effective height of emission of radiation to space. That’s an effect, not a cause.
DeWitt,
It’s easy to agree with that, but by that we have little progress on the question:
“How the Greenhouse Effect Works?”
As has been demonstrated by the failure of innumerable attempts it’s not possible to give a really simple explanation that would not fail in some essential way. To me the next question is: “How complex are we forced to make the answer to avoid similarly serious failures?”
It’s likely that a well selected coherent set of partial answers will be easier to understand than a single simplified model.
Another related point is that there remain a lot of rather bad misunderstandings among people who understand correctly many other features of GHE and that applies both to “warmists” and to “skeptics”. This site is one of the best places for finding answers on such points but I’m sure that there’s still potential to improve on the quality and clarity as well as on the coverage of these answers.
Pekka,
Both DeWitt and I know exactly how an ideal greenhouse effect works. However, in the real world there are complicating factors such as feedbacks, which we do not know all details of. The disagreement between DeWitt and I has only to do with some semantic issues and cause vs effect priority. However, in both of our cases, we agree that the addition of a greenhouse gas increases the resistance of energy transmitting to space without some modification. The modification needed results in an increase in altitude of outgoing radiation, and an increase in surface temperature.
DeWitt,
You stopped too soon. I agree with: “The greenhouse effect ‘works’ by creating an imbalance in the energy flows in and out of the atmosphere. One of the manifestations of this in the Earth’s atmosphere is an increase in the effective height of emission of radiation to space.” The problem is: How does this increase in altitude relate to the increase in temperature? The answer of course is by the lapse rate. This can be seen as a chicken vs egg type of issue, but if the increase in altitude did not result in the temperature increase, what did?
Leonard,
No, I didn’t. If we double the CO2 concentration, the effective height of emission goes up, but the vertical temperature profile hasn’t changed. Simply raising the effective height of emission does precisely nothing to increase the surface temperature. It’s the energy imbalance throughout the atmosphere and at the surface that effects the warming. Period. A new balance is established at the new emission height after all the warming is done.
DeWitt,
The lapse rate is a GRADIENT not a fixed profile. The lapse rate did not change, but the level shifted by the increase. this was due to locking the outgoing level at a higher altitude.
DeWitt,
I think you are again referring to the transient response of a hypothetical case of an instant increase of CO2 and a finite lag of response of the atmosphere. It doesn’t happen that way, so it is not relevant. I am referring to a slow change (increase in CO2) with effective equilibrium at each stage. In all cases, the excess heat at the surface works its way up to re-balance outgoing energy with incoming. Back radiation is not heating (transmitting net average energy down) the surface at any stage.
Sure it happens that way. The size of the increase is unimportant. It’s a finite difference solution to the continuous problem. You can make the step size of concentration and time vanishingly small and it’s still the same problem. I guess you could solve the problem by raising the emission height and temperature in steps too, but the first questions you would get from a lay audience would be: What do you mean by emission and why and how much does the height and temperature increase when you increase CO2? You can’t answer that without getting into radiative transfer. In which case, you might as well solve the radiative transfer case directly.
The other thing is that while most people understand that temperature decreases with altitude, the effective height of emission is a mathematical construct that you can’t actually demonstrate to anyone. The existence of back radiation, OTOH, can be demonstrated by pointing an inexpensive IR thermometer at the sky, preferably with both clear and cloud covered sky. I think it shouldn’t be difficult to explain to most people that if the temperature of the sky increases, so will the temperature of the surface. In fact, when surveyed, something like 70% of the people in the US accept the fact that increasing CO2 causes an increase in surface temperature. They just aren’t willing to spend very much money on the problem because while the consequences may be negative, they’re in the future and uncertain or NFU in quality management terms. People generally don’t do anything much unless the consequences are immediate and certain.
The other thing you can do with the IR thermometer is use it to explain the TFK09 or KT97 energy balance cartoon. You do that by first having the audience imagine that they are in space with their IR thermometers pointing them at the Earth at a distance where the Earth exactly fills the field of view of the IR thermometer. The observed temperature of the Earth is then ~ -18 C averaged over a full year. We know this is approximately true because we have satellites that do almost exactly that. Then we imagine take readings from the surface at many points and many angles to cover the full sky (or use an IR thermometer with a 180 degree field of view, otherwise known as a pyrgeometer, pointed at the sky ) over a year and average them. The observed average sky temperature will be ~ 4 C. We don’t actually do that but we know that where we do measure, the readings agree with calculations. And we’re back to Chris Colose’s explanation of the greenhouse, more radiation down than up. Notice that the effective height of emission plays no part in this demonstration. You could calculate it, but it’s unnecessary.
In fact, most people don’t do all that much if the consequences are certain but in the future. We know this because most people don’t save anywhere near what they ought to for when they retire.
DeWitt Payne
” We know this because most people don’t save anywhere near what they ought to for when they retire.”
How does someone ‘know what is enough’ ?
Retire prior to 2007
Consensus financial advice says , “invest in property”.
Cant lose!
All the rating agencies gave AAA+ ratings to sub prime mortgages, which turned out, to be worthless junk!
Hope you were not sucked into that scam.
Index linked final salary schemes in the 1980’s were thought to be far too conservative.
They did not factor in the then growth of the economy and should be avoided.
However future growth turned out to be far less than anticipated
Today they (final salary schemes) are thought to be ‘gold plated’ and far too expensive for most people.
How is the retirement lifespan factored in?
This is why folk should not exaggerate normal changes in climate.
Britain is lumbering itself with economic hurdles like decarbonising the economy.
Fuel and heating bills are set to rise annually at an alarming rate in a collapsing economy.
The deaths from hypothermia already run into the excess of one hundred thousand.
Windmills and solar cells the politicians say is they only safe way to access energy.
Its a sick joke.
Corrupt or blinkered scientists and politicians push pseudoscience.
The rational way ahead is to spend some resources on a critical review of IPCC science
Bryan,
So your take is since you can’t know how much to save, and you might make bad investment decisions, saving is pointless? The median liquid net worth of retirees is something like $50,000. I think even you would agree that’s nowhere near enough.
I do not think that the Earth’s surface is not both warming and in thernal equilibrium. The disequilibrium is small, probably equal or less than 1W/m^2 but positive.
How might that have come about?
Changes to the surface’s inbound and outbound fluxes.
Has the absorbed insolation increased? I think not.
Has the emitted IR decreased? I think not.
Has the sum of the latent and sensible flux decreased? I think not.
Is there another outbound flux? I think not.
Is there another inbound flux? I think so.
Has it increased? I think so.
Is that increase necessary for the surface warming? I think so.
Is it the sole reason that the surface warmed? I think not..
Is it necessary part of the GHG warming mechanism? I think so.
Is it not the only flux whose variation directly promotes surface warming? I think so.
So why is it not the sole cause of surface warming?
Well, that would be to suggest that the rest of the system played no part.
Yet you insist that the rest of the system does not directly act to promote warming.
Yes, but it also fails to promote a sufficient and counteracting cooling.
So they also serve? I think so.
Alex
Hmmm, to many nots!
I do not think that the Earth’s surface is both warming and in thernal equilibrium.
Alex
Alexander,
The surface has not been warming for over a decade. What are you talking about?
Alex,
Concerning the surface energy balance we may continue to ask what is the most important factor that influences directly the downwelling LW. There’s certainly some direct effect from increased CO2 but that effect is small as far as I can judge. The larger factor is probably the higher temperature of the lower atmosphere not only in absolute terms but also relative to the surface.
Having smaller temperature gradients allows simultaneously for more effective energy transfer trough evaporation and for more DLR to the point where the net LWIR energy transfer is reduced.
DeWitt,
The only thing that heats the ground is solar radiation. Slowing it’s escape is not the same as heating it with back radiation. No system such as the Earth is ever exactly in equilibrium, but on average can be treated as such to see cause and effect. There has been no average heating for over the last decade, while CO2 has made the largest increase. Adding CO2 instantly, would instantly raise the outgoing radiation out level, but that would make cooler atmosphere radiate to space until the excess ground heating was able to heat the atmosphere from the ground up, overcoming the thermal lag. Nowhere does back radiation do anything other than act as a resistance to the net heat flux at the non-equilibrium states. There is back radiation energy flux, but this is not back heat transfer.
When I said raise the outgoing radiation level, I was referring to altitude, not intensity. Also Chris Colose’s idea of cause of greenhouse effect is actually a transient response description, and not relevant to equilibrium, as total energy up equals down for steady state, and convection balances lowered net radiation.
Sometimes I feel a little cold and I turn a heater on. Sometimes the temperature continues to drop.
Do I deduce from this event that the Joule effect has ceased to operate and I might as well turn the heater off? No I don’t.
It is my fortune to have learned the basics of the Greenhouse Effect prior to modern global warming, a time before the modern global temperature datasets.
I believe that the effect applies during decades, years, seasons when the temperature record is rising and when it falls. I believed it applied when there was little or no evidence that it was causing the Earth to warm.
The Earth might have cooled in the last decade and if so I do not find that to be compelling evidence that the GHE has ceased to operate. I certainly wouldn’t like to risk its loss.
If during discussion of “How the Greenhouse Effect Works” we are encounter the critique that an overall rise in temperatures is punctuated by periods were it declines, should we be grateful for that wisdom, or treat it as we find it.
Insofar as the question begs a mechanism, aspects of the processes involved can be discussed. It is possible to conclude that the effect must work by some undescribed mechanism based on some insight, much as it is possible to conclude that a lighter than air craft should rise. That does not inform you as to how it rises, the mechanism invoked, the pressures on the surfaces, the forces in the skin.
Some, but not all, conclusions based on heights and lapse rates are valid as far as a conclusion goes but they are commonly devoid of any described concrete physical mechanism to achieve the real warming that occurs. The end result, a new equilibrium state, is unveiled devoid of any process. That is acceptable as far as it goes and it motivates enquiry as to the processes involved. When precisely does this new found height come into existence, how can it be observed or calculated, how is its existence communicated to the surface below, does this height change whilst the system moves towards a new equilibrium? Many questions that require some knowledge of process, of how the effect works as opposed to that it does work.
For me, adoption of the notion that the Earth’s surface could warm without an imbalance in the inbound at outbound fluxes would entail my being silly or my arguing inconsequential points. So I must confront a real effect with real fluxes and try to represent what real and concrete process may be involved and how they promote or counteract the changes that occur.
I will not be content to look away during the messy process of effecting the change and come back once it is complete nor consider such observation would constitute information as to how the effect works as opposed to that it did work.
Alex
Alex,
It’s sometimes instructive to go back and check what people wrote at the time when the simple models had reached some maturity but everything that was needed to get so far was still fresh in memory. On this basis I looked at the 1978 review paper of Ramanathan and Coakley.
Deriving the basic Radiative-Convective model was done for the horizontally averaged atmosphere. That kind of approach had allowed Wetherald and Manabe to explain the average surface temperature rather accurately but reading the Ramanathan and Coakley article makes one to wonder whether that kind of accuracy was either share luck of based on tuning, so many details were chosen with little justification or even on an almost random basis.
The most serious and questionable physical assumption is neglecting the term that corresponds to the large scale circulation. The circulation does certainly affect the lapse rate and this leads to the important point that could be considered either tuning or a rather random choice, i.e. using a fixed environmental lapse rate. It’s well known that the real lapse rate varies from dry adiabatic to very low values and choosing to use some particular value for the global average cannot be justified directly. The circulation is one major factor that affects the variability of the temperature profiles.
I commented above on the surface balance. What happens at the surface is in a very essential way controlled by the requirement of balance. The overall flow is controlled by the incoming radiation, SW and LW. The shares of outgoing flows are then determined by competition between LWIR, evaporation/latent heat transport and convection. All of them depend on the low altitude temperature gradients and the temperature gradients adjust to make the balance true. One does not need to analyze these processes in further detail to know much of what needs to be known. (The amount of evaporation is certainly also important for knowing the moisture of low atmosphere.)
For most of the troposphere we have two alternatives. Either we just accept some given lapse rate or we switch to using much more complex models.
Then we have the balance at the top of troposphere and the question of the altitude distribution of the source of the LWIR that leaves the troposphere. This is clearly the part of the problem where the role of additional CO2 is important most clearly. That altitude distribution can be integrated to tell the number described somewhat questionably as “the average emission height of outgoing radiation”. Whether we use this average or not estimating the change in the altitude distribution of the points of emission seems to be the best relatively straightforward way of for making the picture in some sense complete and self-consistent.
I have not ignored, and hope to return, to several unanswered comments above. For now I have something else to say about the generality of the debate.
The Use and Abuse of Thermodynamic Concepts and Terms
The notion that the Laws of Thermodynamics are a constraint on our modes of thinking, on our formulation of representations is incorrect. They pertain to the physical system itself. We are not so constrained provided that the system as represented is so constrained. It is the notion that there are ways of thinking about a system that must be forbidden because they break narrow verbal formulations of the Laws yet do not violate the Laws.
To forbid a mode of representation that is superficially unsafe, that embodies apparent paradoxes only resolved at some higher level is the imposition of mind forged manacles.
The Laws are old yet stand the test of time because we have evolved our thinking to meet the challenges that deeping knowledge brings. Quantum Mechanics, General Relativity, Information Theory represent revolutions that have required new ways of thinking about the Laws.
Our understanding of Entropy has undergone many changes and may continue to do so. From narrow clasical thermodynamic formulations, through arguments based on disorder, to ones based on information and ignorance, coarse graining, and perhaps some form of unification at some level with fresh insights from the geometry of phase space, how that relates clasical and quantum views, determinism and information. Some of this is not for my understanding but for younger and hopefully braver souls.
I must argue that little of that progress is compatible with the narrow, arid, but safe representations available to those that bow down to forms of words enscribed a lifetime or more ago.
The abuse of concepts and terms is not a novelty. The use of established yet deliberately narrow scientific views to debilitate thought has a pedigree as a political activity. Science is not pure but can be puritanical. It is a human endeavour not to be divorced from politics. I have borrowed from Arthur Tansley for political and rhetorical effect. Not I think idly but as an example of the political threat once and perhaps still posed by the abuse of ecology once its metaphors take on a life of their own. I am being openly contentious to contrast with its closetted form as when cloaked in the Laws of Thermodynamics.
The Laws of Thermodynamics embody some of our best insights but that does not imply that all representations of those Laws are politically neutral. They can be used to close down debate.
I am by nature a reckless thinker. I will play fast and loose when I am confident that I can build representations whose paradoxes are only apparent and can be resolved in compliance with the constraint imposed by nature which is not the same thing as obedience to dogmatic literalism. My representations may not be the most lucid and if needs be deliberately so, but nor are they the doctrinal mutterings of the dwarfish.
Alex
[…] Comments « How the “Greenhouse” Effect Works – A Guest Post and Discussion […]
May I just say what a relief to uncover a person that genuinely understands what they’re discussing on the internet.
You actually realize how to bring an issue to light and make it
important. More people ought to look at this and
understand this side of your story. I can’t believe you aren’t more popular
because you definitely have the gift.