In How the “Greenhouse” Effect Works – A Guest Post and Discussion there was considerable discussion about the temperature profile in the atmosphere and how it might change with more “greenhouse” gases. The temperature profile is also known as the lapse rate.
The lapse rate has already been covered in Potential Temperature and for those new to the subject Density, Stability and Motion in Fluids is also worth a read.
Some Basics
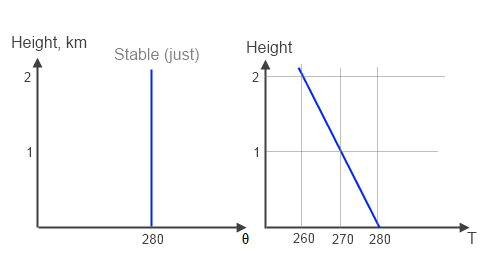
Let’s take a look at a stable (dry) atmospheric temperature profile:
Figure 1 – Just Stable
The graph on the left is the potential temperature, θ, and on the right the “real temperature”, T. The temperature declines by 10°C per km (and this value is not affected by any “greenhouse” gases). The potential temperature is constant. Remember that for stable atmospheres the potential temperature cannot reduce with height.
A quick recap from Potential Temperature:
- “potential temperature” stays constant when a parcel of air is displaced “quickly” to a new height (note 1)
- potential temperature is the actual temperature of a parcel of air once it is moved “quickly” to the ground
- in dry atmospheres the actual temperature change is about 10°C per km
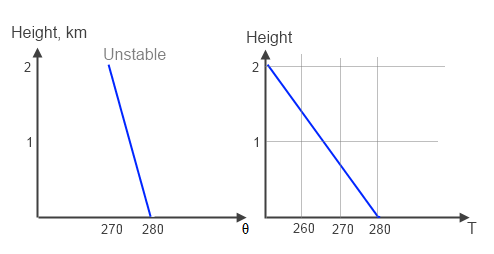
Now an unstable atmosphere:
Figure 2 – Unstable
Because the temperature at a given altitude is “too cold”, when any air is displaced from the surface it will of course cool, but finish warmer at 1km and 2km than the environment and so keep rising. This situation is unstable – leading to convection until the stable situation in figure 1 is reached.
We can also see that the potential temperature decreases with altitude, which is another way of conveying the same information.
The important comparison between the first two graphs is to understand that figure 2 can never be stable. The atmosphere will always correct this via convection. Exactly how long it takes to revert to figure 1 depends on dynamic considerations.
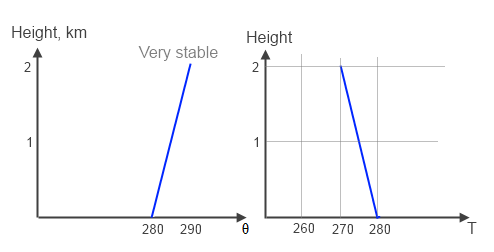
Let’s look at another scenario:
Figure 3 – Very Stable
Now the temperature reduces with height, but not sufficiently to induce convection. So a parcel of air displaced from the surface ends up colder than the surrounding air and sinks back down.
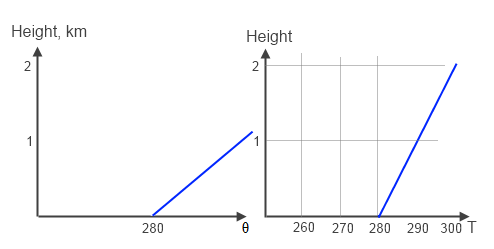
And we can even get temperature inversions, very popular in polar winter and nighttime in many locations:
Figure 4 – Very very stable
So how do figures 3 & 4 come undone? Surely once the atmosphere is stable to convection then it becomes static and heat can only move radiatively from the surface into the atmosphere?
The basic principle of heat movement in the climate is that the sun warms the surface (because the atmosphere is mostly transparent to solar radiation) and so the atmosphere is continually warmed from underneath.
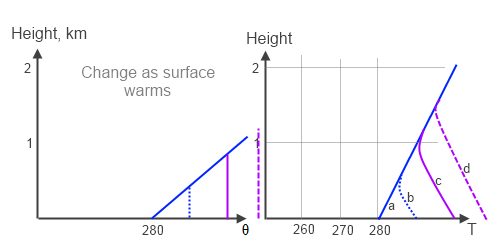
Figure 5 – Atmospheric temperature changes as surface warms
As the surface warms the atmospheric temperature profiles move from a → d. This is a result of convection. But where does all this heat go that was convected from the surface into the atmosphere.
Here is a graphic reproduced from Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Eleven – Heating Rates:
Figure 6 – Radiative Cooling of the Atmosphere
This illustrates that the atmosphere is always cooling via radiation to space – and cooling at all altitudes.
So the atmosphere cools via radiation, the surface warms from solar radiation and when the lapse rate reaches a critical value convection is initiated which moves heat from the surface back into the atmosphere.
As a minor question, how does a temperature inversion ever get created? It’s a temporary thing. In the case of nighttime, the surface can lose heat via radiation more quickly than the atmosphere. The surface is a more effective radiator than the atmosphere. In the case of the polar winter, the same effect takes place over a longer timescale. But eventually, when the sun comes up, the surface gets reheated.
Where Convection Stops – The Tropopause
Actually there are a few different definitions for the tropopause. But let’s save that for another day. There is a point at which convection stops. Why?
Suppose there was no convection, only radiation. If we consider heat transfer via radiation then there is a change as the atmosphere thins out.
Let’s take a massively over-simplistic approach to help newcomers. Suppose a photon of a given wavelength has to normally travel 100 molecules before getting absorbed. In this case, as the atmosphere thins out from 1000 mbar (surface) to 200 mbar (typical tropopause), the same photon would have to travel 400 molecules before getting absorbed. This means that the temperature change vs height reduces the more the atmosphere thins out. As a way of thinking, it’s like the resistance to temperature change reduces as the atmosphere thins out.
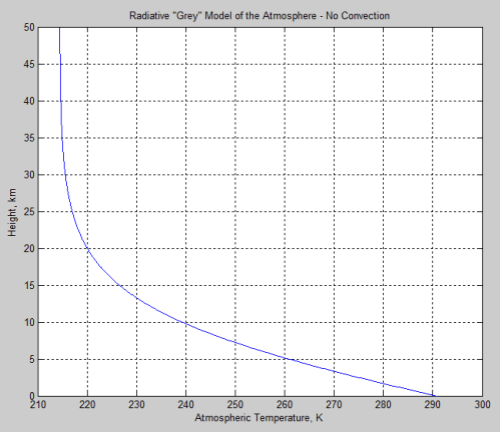
A simple example of radiative equilibrium for gray atmospheres (note 2) is given in Vanishing Nets:
Figure 7 – Radiative Equilibrium
See how the temperature change with height (the lapse rate) reduces the higher we go. So at a certain point the potential temperature always increases with height, making the atmosphere resistant to convection.
The point at which the radiative lapse rate is less than the adiabatic lapse rate is where the atmosphere stops convecting. However, this is not technically the tropopause (note 3).
Another way to think about this for newcomers is that the temperature reduction caused by lifting a parcel of (dry) air 1km is always about 10°C. So if the temperature reduction due to radiative heat transfer is 5°C then the lifted parcel is always cooler than the surrounding air and so sinks back = no convection.
Now the atmosphere is not gray so this is not a simple problem, but it can be solved using the radiative transfer equations with numerical methods.
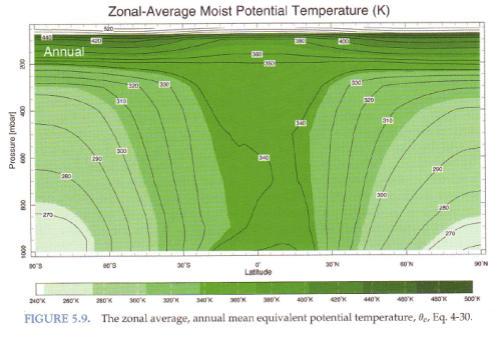
We can see the real (averaged) climate in this graphic of potential temperature:
Figure 8
In the tropics the (moist) potential temperature is close to constant with altitude until about 200 mbar. And at other latitudes the potential temperature increases with height very strongly once we get above about 300 mbar. This shows that the atmosphere is stratified above certain altitudes.
Increasing CO2 – The Simple Aspects
Let’s consider the simple aspects of more CO2. These got a lot of discussion in How the “Greenhouse” Effect Works – A Guest Post and Discussion.
We increase the amount of CO2 in the atmosphere but at the surface the change in downwards longwave radiation (DLR) from the atmosphere is pretty small, perhaps insignificant.
By comparison, at the top of atmosphere (TOA) the radiative effect is significant. The atmosphere becomes more opaque, so the flux from each level to space is reduced by the intervening atmosphere. Therefore, the emission of radiation moves upwards, and “moving upwards” means from a colder part of the atmosphere. Colder atmospheres radiate less brightly and so the TOA flux is reduced.
This reduces the cooling to space and so warms the top of the troposphere. Therefore, there will be less convective flux from the surface into this part of the atmosphere.
As a result the surface warms.
Increasing CO2 – The Complex Aspects
The real world environmental lapse rate is more complex than might be inferred from the earlier descriptions. This is because the large scale circulation of the atmosphere results in environmental temperature profiles that are different from the adiabatic lapse rates.
The environment can never end up with a greater lapse rate than the adiabatic lapse rate but it can easily end up with a smaller one.
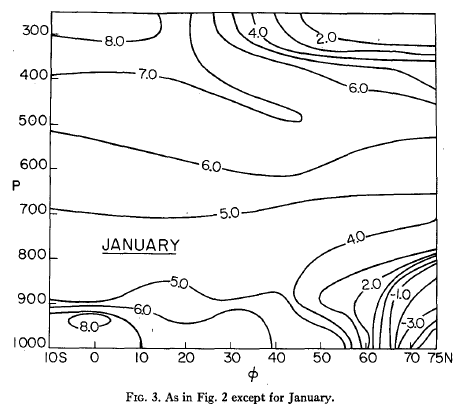
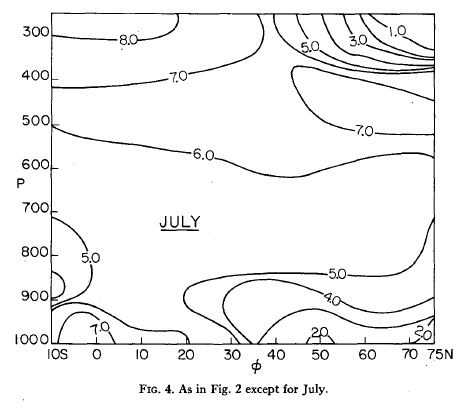
More on this in another article. But as a taster, here are some monthly averaged environmental lapse rates:
Figure 9
Figure 10
And of course, one of the biggest questions in an atmosphere with more CO2 is how water vapor concentration changes in response to surface temperature change. Changes in water vapor have multiple effects, but the one for consideration here is the change to the lapse rate. The dry adiabatic lapse rate is 9.8 °C/km, while the moist adiabatic lapse rate varies from 4 °C/km in the tropics near the surface (where the water vapor concentration is highest).
Consider an atmosphere where the temperature reduces by 15 °C in 2km. Dry air moving upwards reduces in temperature by 20 °C – which is colder than the surrounding air – and so it sinks back. Very moist air moving upwards reduces in temperature by about 10 °C – which is warmer than the surrounding air – and so it keeps rising.
So more moisture reduces the lapse rate, effectively making the atmosphere more prone to convection – moving heat into the upper troposphere more effectively. (Cue tropical hotspot discussion).
References
Atmospheric Lapse Rates and Their Parameterization, Stone & Carlson, Journal of the Atmospheric Sciences (1979) – Free paper
Notes
Note 1: A parcel of air displaced “quickly” to a new height is written for ease of understanding. Technically, potential temperature stays constant if a parcel of air is displaced “adiabatically” – which means no exchange of heat with the surrounding atmosphere.
Note 2: A gray atmosphere is one where the absorption vs wavelength is constant. More technically, this is usually a “semi-gray” atmosphere because the atmosphere is transparent to solar radiation but absorbs terrestrial radiation.
Note 3: The tropopause is usually defined where the lapse rate is at a minimum. In radiative equilibrium the temperature would continue to decrease with height even after the point where convection stops. It is only the presence of radiative gases (ozone) that absorb solar radiation that cause the stratospheric temperatures to increase.



A few questions, and points of disagreement:
1. Why have GHGs no effect on the dry lapse rate?
2. What would be the effect of increased GHGs on figure 6?
3. The atmosphere is not “mostly transparent to solar radiation”. See eg _http://telstar.ote.cmu.edu/environ/m3/s2/04solarad.shtml (you might have oversimplified it there)
4. Under “Increasing CO2 – The Simple Aspects” why don’t you just state that a temperature increase at TOA obviously means a temperature increase at the surface, due to the lapse rate? (analogously, an increase in atmospheric mass would move TOA upwards, once again warming the surface)
5. “more moisture reduces the lapse rate”…but H2O is also a GHG. So what is the actual (total) contribution of water vapor to tropospheric temperature?
6. Not sure what are the basis for Note 3. Stratospheric temperatures increase somewhat on Jupiter for example _http://telstar.ote.cmu.edu/environ/m3/s2/04solarad.shtml and Saturn _http://astronomy.nmsu.edu/tharriso/ast105/Saturn.html – not to mention Venus, Uranus, Neptune. Positive tropospheric lapse rates and negative stratospheric lapse rates appear to be a characteristic of every planetary atmosphere, independent of composition.
As usual, there is no polemics in the above. I have always found this site considerably of a higher quality than most as it just shows what the IPCC could have been if rent-seekers and primadonnas had been isolated from the start 😉
BTW…an old obsession of mine…it shouldn’t be called TOA but TOT=Top of the Troposphere.
The tropopause as level of relevant radiative forcing is apt because the stability means convective transfer of heat can be neglected.
Or can it? There is a certain amount of troposphere/stratosphere exchange each year which remains unquantified. But with that exchange, there is surely energy exchange as well.
Further problems with the tropopause as significant level is that for much of the winter polar regions, no level meets the standard definition and in the tropics, there may be multiple levels meeting the standard def.
It does also raise the question about the significance of upper level radiance.
Omnologos,
1. Because the equations which relate energy conservation and expansion result in a formula: lapse rate, Γ = -cp/g, where cp = heat capacity of air under constant pressure and g = gravitational acceleration.
2. See figure 4 in Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Eleven – Heating Rates
3. It is a handy approximation in providing conceptual understanding.
4. Because it is not obvious to everyone.
5. This is a complex subject which can only be answered with GCMs. The intent of this article is to help readers understand the lapse rate cause and effect better.
6. The basis for note 3 is that the lapse rate minimum is not the same point as where convection ceases.
Omnologos,
More on point 6..
The reason that stratospheric temperatures increase with height above the convective region is due to the absorption in the stratosphere of solar radiation. If there was no absorption of solar radiation in the stratosphere then temperatures would continue to decline with height.
thanks!
SoD,
I think what increases the lapse rate toward the DALR (10 C/km) is interesting, and there’s more to it than you say.
The atmosphere, with GHG, does cool by radiation. But it’s also warmed from below. The more it emits (via GHG conc), the more it absorbs. The nett result is a separate thermal gradient. With radiative transfer in general this is hard to calculate, but in the case of a fairly opaque atmosphere, it follows the Rosseland model. This gives an explicit temperature gradient that incorporates the effect of cooling from above that you describe (for a given upward IR flux). It’s a grey gas model, and not fully accurate for the relatively transparent air, but it’s still useful.
The point is that you can quantify the effect, and I believe it is not enough to bring the lapse rate up to observed values.
There is another effect, described here, which I believe is dominant. You have described how when the DALR is exceeded, convective instability transports heat upward, and brings the LR back down. That effect is proportional to the amount by which the DALR is exceeded. The excess creates a heat engine; the upward transport, from hot to cold, transfers kinetic energy to the gas.
Below the DALR, this runs in reverse. Vertical motion is forced by the wind. Downward moving air becomes warmer than ambient, transporting heat down, and taking energy from the flow to counter buoyancy. Upward moving also takes energy, and “transfers cold” up – ie heat down. The nett result is a heat pump, with energy from wind maintaining the lapse rate. This incidentally works without GHG.
I’ve quantified this for Venus in this post, which also discusses Rosseland.
SoD,
First two comments.
Although I believe that I know what you are talking about the inserted paragraph of massively over-simplistic approach seems difficult to understand. Also the numbers 100, 1000, 200 and 400 appear to conflict (should the last be 500, I’m not sure as I don’t understand the paragraph fully).
The Figure 8 is based on equivalent potential temperature, but is it really relevant at higher latitudes and does the Figure justify fully the conclusion
“.. the potential temperature increases with height very strongly once we get above about 300 mbar. This shows that the atmosphere is stratified above certain altitudes.”
as the equivalent potential temperature is perhaps not the right variable to use for discussing stratification, when the air is far from saturation.
Pekka Pirilä,
The problem with “massively over-simplistic” approaches is that people who understand the subject wonder whether it makes sense.
With any attempt to explain complex stuff to newcomers I may well have missed the mark and any who don’t understand it please throw in a comment. There is no “right answer” when trying to make hard subjects easy..
At 1000 mbar the density is about 1.3 kg/m3 but at 200 mbar the density is about 0.3 kg/m3. I just did the calculation.
Equivalent potential temperature is just like potential temperature but with moisture taken into account. So if there is no moisture Equivalent potential temperature = potential temperature.
θe = θ exp(Lq/cpT)
where q = water vapor, L = latent heat of vaporization, cp = specific heat capacity, T = temperature, θe = equivalent potential temperature and θ = (dry) potential temperature
So if the air is dry, θe → θ as q → 0.
My point on the equivalent potential temperature is that it changes when the moisture level changes at values far from saturation but these changes do not affect the buoyancy. Thus considerations of equivalent potential temperature do not reflect always the local stability conditions.
SoD,
Your link for the Stone – Carlson paper didn’t work for me, but a Google search with “Stone Carlson lapse” gave a link directly to the AMS journal that worked.
Thanks, I updated the article with the correct link.
Pekka Pirilä
“..but these changes do not affect the buoyancy..” ?
The buoyancy consideration is simply the temperature of the parcel of air moved to that altitude vs the surrounding (environmental) air temperature at that altitude.
Maybe I misunderstand your point. Can you explain in a little more detail, perhaps with an example?
SoD,
When the moisture is less than 100% the stability is controlled by dry lapse rate and by the differences in (non-equivalent) potential temperature. If all air that needs to be considered has the same absolute moisture and the moisture remains less than 100% the equivalent potential temperature and the potential temperature have different values but change in unison.
If we must, however, consider air of varying moisture this is not true but the two potential temperatures are not related in such a simple way. This is true when horizontal air motion is taken into account. There may also be problems when temporal averages are considered as the averages do not necessarily follow the same laws at values used in calculation of averages, because the laws may have variable coefficients.
Pekka Pirilä,
So if I understand you, the fact that relative humidity is generally less than 100% in the free troposphere means that moist (aka equivalent) potential temperature should not be used?
For reference for other readers here is the average relative humidity field:
SoD,
I don’t say that it should not be used, but rather than none of available variables does a perfect job. As long as we are guaranteed that relative humidity remains below 100% the standard potential temperature is fine. When the air is everywhere saturated the equivalent potential temperature works well and it works well also in some other cases when it gives the same results as the standard potential temperature. Thus it’s certainly useful in a wide range of situations, but not perfect always.
My understanding is that the equivalent potential temperature may give rather misleading results when winds lead to the situation where the moisture content varies significantly with altitude at relative humidities well below 100%. Similarly calculating averages at locations where the relative humidity varies essentially in time may lead to misleading results.
The plots that you have shown are interesting and informative but not as straightforward to interpret as you wrote. Thus the text should perhaps be reformulated but I’m not expert enough to propose what would be the best formulation. I’m a physicist but not an expert on atmosphere and my comments are based on general knowledge of physics and some simple reasoning.
Pekka Pirilä,
Not really.
(Dry) potential temperature, θ, is conserved in adiabatic processes for dry air and for moist air where no condensation or vaporization occurs.
Equivalent (aka “Moist”) potential temperature, θe, is conserved in adiabatic processes for both dry and moist air.
θe = θ exp(Lq/cpT)
where θe = equivalent (aka “Moist”) potential temperature
θ = (dry) potential temperature
L = latent heat of vaporization
q = specific humidity
cp = specific heat at constant pressure
T = temperature in K
If no condensation (or vaporization) occurs then q is constant.
Note that equivalent potential temperature is a different concept from the saturated adiabatic lapse rate.
So if we take the example of a parcel of moist air moving up from the surface it will follow a dry adiabat until condensation occurs (at the LCL = lifting condensation level), after which it follows the saturated adiabatic lapse rate. [Of course, if it mixes with dry or unsaturated air at any stage this changes things].
But for the entire (adiabatic) raising of this parcel of air, equivalent potential temperature is conserved.
And for reference here is the annual mean dry potential temperature:
c.f. Figure 8, we can see that in the upper troposphere and the stratosphere dry and equivalent potential temperatures are very similar.
SoD,
I don’t understand what you mean by “not really” as nothing in what you write further contradicts my statement.
The equivalent potential temperature is fine when we can follow one particular parcel or air and compare it only with other air that has gone trough the identical history, but it’s not valid when air of differing histories and consequently different moisture levels meet. All the derivations that you refer to are based on the single common history.
My second comment on the problems that we meet in applying the concepts to annual averages are true for all choices of temperature variable. In most regions the temperature profile follows closely either dry or moist adiabat over some altitude range during some periods but not the same adiabat or any adiabat always and not over the same range of altitudes. When that’s the case as it’s at high latitudes and less strongly at mid latitudes the annual average is nothing more than annual average of a variable that is determined by many different processes at different times. Only the tropics can be described fairly well by one approach.
Concerning the similarity of the two plots in the upper troposphere.
The plots must be similar in that region because the saturation absolute moisture is already so low that the energy released in condensation is rather small in comparison with heat capacity of dry air.
Pekka Pirilä,
Earlier you said:
Yet your latest comment (9:17pm) appears to agree that the figure does show it – due to the fact that both dry and equivalent potential temperature are almost the same for the upper troposphere.
SoD,
There must be some misunderstanding between us. In the upper troposphere where temperatures are low and absolute moisture also always low the dry and moist adiabat are almost identical and the potential temperatures also change in unison.
The differences occur in middle and low troposphere where we face as an example the situation that dry downcoming air warms rapidly and has a rather high lapse rate reduced by dissipative processes from the dry adiabat. That goes on until the warm air hits moister air of higher potential temperature near to the surface and cannot continue the downwards motion. This results in very low lapse rate and might result even in temperature inversion. Such situations are not equal over all seasons which leads to all kind of apparent effects in annual averages.
I think I share your concern for SoD’s explanation Pekka Pirila, there just aren’t enough variables in the formula offered. All ‘parcels’ of air are not equal for buoyancy! Without a connection between ‘q’ and the gravity constant, or at least ‘density’, a parcel finds ‘density equilibrium’ at an indeterminate ‘final altitude’ and ‘final temperature’ along the lapse rate curve.
Though, this is a complex problem that SoD may not want to include in this thread, as altitude and decompression also alters the relative humidity and the latent heat of condensation within the parcel. 🙂
Best regards, Ray Dart.
I failed the login test yet again. 😦
Ray Dart, AKA suricat. 🙂
Ray.
The following is the sequence in an ideal model of how the increased CO2 heats the ground and atmosphere.
The process of adding CO2 to the atmosphere occurs from the surface, and convection and mixing distribute it to the rest of the atmosphere over a finite time. However, let us image an addition of an instantaneous well mixed quantity of CO2, small enough to not significantly affect the average CP or total mass of the atmosphere, so that the temperature level and lapse rate are also the same as before, immediately after the addition of the CO2. Also assume solar insolation is the same as before, and only consider solar energy absorption to occur at the ground. The question is: how does the surface and atmosphere heat up more from the greenhouse effect. The approximation will be used that the lapse rate, surface temperature, and solar insolation, are uniformly distributed over location and time to simplify the issue, and clouds and feedback are ignored.
When the CO2 level jumps up, the effective average outgoing radiation altitude to space also instantly increases. The average altitude was about 5 km before addition, due to previous levels of water vapor, CO2, and other greenhouse gases and clouds. An increase from doubling the CO2 has been claimed to cause an eventual increase in temperature of about 1.2 C if all other effects are unchanged. For an average environmental lapse rate of -6.5 C per km, this implies the average outgoing level was raised by about 185 m once new equilibrium was reached.
However, the raise in outgoing level would occur as soon as the CO2 level increases, and is not directly tied to the temperature. It is a radiation absorption issue only. Since the temperature was initially 1.2 C lower at 185 m above the initial 5 km level, the radiation to space is initially lower from the new level. When input and output were balanced, the average temperature of the 5 km level was 255 K, and radiated 239.7 W/m2. Initially at the new altitude (5.185 km), the average temperature is 253.8 K, and radiates 235.3 W/m2. Initially, just after the CO2 level made the step jump, the air temperature is the same as before. However, the increased resistance to radiation heat transfer up, due to the more opaque atmosphere, results is less of the absorbed solar energy being initially removed from the ground by radiation heat transfer (which is a significant, but not necessarily dominant means of heat transfer up). Note the back radiation is not heating the ground; it is just slowing the net radiation heat transfer up. This accumulating solar energy results in the ground heating up, as the excess energy accumulates. Once the ground heats up a small amount, this increases both convective and radiation heat-transfer, compared to just before the ground heated noticeably (but after the CO2 was added). However, at new equilibrium, total heat transfer out is the same as before (equals solar energy input), the convective heat transfer is a larger fraction of total heat transfer up at new equilibrium.
The increased heat transfer does not change the lapse rate at the new final equilibrium from before the CO2 was added. However, since the increased surface heating started immediately at addition of CO2, but the thermal lag of the finite mass of the atmosphere took a while to rebalance by convection and radiation, the lapse rate does increase some during the non-equilibrium stage. In the end, the energy is transmitted by conduction, convection, and radiation up through the atmosphere, driving it toward the same lapse rate as before the CO2 was added, but with the entire temperature level shifted up 1.2 C at final new equilibrium, for corresponding altitudes. This leads to the temperature at 5.185 km to be driven up to 255 K at the new equilibrium. At that point, the outgoing balanced incoming, and no additional heating of the surface or atmosphere occurred.
Leonard,
That’s one of the self-consistent ways of simplifying the description. My main complaint is that the heat capacity of the atmosphere is very small in comparison with the mixed surface layer of oceans. Therefore the adjustment of the atmospheric temperature is essentially instantaneous and the main source of delay is in oceans. Land areas are both smaller and faster to warm up.
Another point that’s not a complaint in the same sense is that I don’t like the emphasis given to the “average radiation level” as that’s not a physical entity but only a numerical value obtained from certain calculations. This is, however, more a matter of taste than disagreement on deeper level.
Pekka,
I agree with your points, but simplification makes the understanding of process easier to discuss. As far as the ocean accumulation, there are currents lasting hundreds to thousands of years, and just tracking a few years at a limited number of sites is by no means clear indication of what is happening.
[…] 2012/08/12: TSoD: Temperature Profile in the Atmosphere – The Lapse Rate […]
SOD,
I have a question that this topic brings to mind.
The average atmosphere has a greenhouse radiative forcing which leads to an auto-convective atmosphere ( as seen in Manabe and Strickler):
Because this is unstable, convection restores the atmosphere to a more stable lapse rate, and in so doing, negates some portion of the radiative imbalance.
By adding additional CO2, presumably the radiative forcing alone leads to an even steeper lapse rate, but convection again opposes.
The Myhre paper indicates what the radiative forcing for a doubling of CO2 should be ( 3.7W/m^2). Much of the analysis assumes this level.
But that is before convection.
Isn’t the forcing overstated because it will be reversed by convection?
Couldn’t ALL of the radiative forcing be reversed by convective motion?
No, that’s after convection. All relevant scientific papers since Manabe and Strickler at least, if not for even longer, are based on an atmosphere whose temperature profile is restricted by convection. That’s always one central starting point.
The Myhre paper I was referring to regards forcing. The IPCC definition of RF does not include convection. The GCMs then model an atmosphere incorporating the RF as a climatic response.
This may be valid for all I know, but it is an asynchronous numerical solution to concurrent physical processes.
The Myhre paper is based on an atmosphere with convection but it needs not consider it explicitly. The calculation of forcing as is done in the Myhre paper assumes that the troposphere is unchanged in all other respects expect CO2 concentration. That affects radiation immediately but changes the temperatures as well as convection with delay. The IPCC definition of forcing refers to the moment before any temperatures have changed and before convection starts to change.
The changes in the temperatures and convection enter the analysis when the consequences of the forcing are considered, not in the forcing as defined by IPCC.
In the above replace “expect” by “except”.
Climate Weenie,
There are competing feedback effects. The forcing is easy to calculate but the feedbacks are the tricky bits.
From a simple perspective the lapse rate doesn’t change due to convection. When the atmosphere becomes more opaque the upper troposphere warms, and this reduces convection. Which is what warms the surface.
If you grasp this important principle then you can start to think about second order effects – the feedbacks.
There are other papers which express this better but as I had just used this graphic in an earlier article, here are the respective feedback effects as calculated by some GCMs from Soden:
As you can see the lapse rate feedback is a negative feedback. But this feedback is usually the increased radiation to space as a result of a warmer atmosphere.
I see what yer sayin’ – the GCMs determine convective response in the from of lapse rate feedback.
Worthwhile for me to re-read Manabe and Soden papers a number of times.
“From a simple perspective the lapse rate doesn’t change due to convection. When the atmosphere becomes more opaque the upper troposphere warms, and this reduces convection. Which is what warms the surface.”
You may wish to revisit this paragraph. Manabe demonstrates that convection warms the upper troposphere and cools the lower troposphere,
opposing the unstable atmosphere that radiation alone causes, which restores stability.
Climate Weenie,
The Manabe & Strickler 1964 paper (recommended to everyone) has a number of topics and the one which I think you are referring to is how the temperature profile under radiative-convective equilibrium differs from that under a solely radiative equilibrium.
In this case, of course the temperature cools more slowly (higher lapse rate) under radiative equilibrium and so convection (by comparison) results in a warmer upper troposphere.
This is a different topic from the mechanism of temperature change in the atmospheric/surface temperature profile due to an instantaneous change in CO2 concentration -which Manabe & Strickler don’t actually cover.
The Myhre paper calculates a net absorption increase of 3.7 W/m^2 per CO2 doubling (i.e an instantaneous reduction in direct surface radiation to space). This entire amount is then arbitrarily applied to the IPCC’s definition of ‘radiative forcing’, for which I argue is not only incorrect, but a rookie or freshman mistake. I maitain there is absolutely no physical or logical basis for which the atmosphere would downward re-emit this entire amount as it is applied to the IPCC’s definition of RF.
Also, despite what apparently most people think, RT simulations, such as what Myhre has done, do not actually calculate the IPCC’s defintion of RF. They calculate net changes in direct surface radiation to space for changes in GHG concentration, such as when CO2 is doubled.
RW,
You have not understood the Myhre paper (by that I refer to the 1998 GRL paper, but his earlier 1997 JGR paper explains some issues better). Myhre uses in his calculation several alternative models which all take as starting point the observed temperature profiles at three latitudes, that this is enough was demonstrated in the 1997 paper. The 1997 paper contains also more discussion on the models used.
The radiative transfer models calculate absorption and emission at all altitudes and are in this dependent on the temperature profiles. They indicate clearly that their calculation is not based on calculating only the net changes in direct surface radiation to space.
All your claims about deficiencies in the calculation are erroneous and must be caused on your ignorance of what was really done.
Pekka,
I’ve sought direct clarification on this specific point from Myhre himself. The physical meaning of the 3.7 W/m^2 is the instantaneous reduction in direct surface radiation to space. That is, when CO2 is doubled the atmosphere absorbs an additional 3.7 W/m^2 of surface radiative power that was previously passing directly from the surface into space the same as if the atmosphere wasn’t even there. Now of course the absorption occurs all the way up at different amounts at different altitudes, but the 3.7 W/m^2 is the total net absorption increase through to the TOA because it includes a stratosphere adjustment (which is very small anyway).
My point is that what is being calculated in RT says and implies absolutely nothing about what happens after absorption, and has been arbitrarily assumed to be all downward directed upon re-emission by the atmosphere via the IPCC’s definition of RF, for which there is no physical or logical basis.
There must have been a misunderstanding between you and Myhre.
Pekka,
email me and I’ll forward you the entire exchange I had with Myhre.
wetmorer @hotmail.com
Pekka,
In other words, what I’m saying is the RT simulations calculate the net absorption increase for changes in GHG concentrations. They do not calculate the IPCC’s definition of RF, i.e. change in net (down minus up) irradiance (solar plus long-wave; in Wm-2).
Click my name and check last lines of “About me” for email address.
Found it and forwarded…
Perhaps a better way to put this is the physical meaning of the 3.7 W/m^2 per CO2 doubling is just plain ‘minus up’.
[…] May Rainfall Above Average – June BelowHeaven is 140 Degrees Hotter Than HellOctober Weather in CaliforniaTemperature Profile in the Atmosphere – The Lapse Rate […]
scienceofdoom,
You appear to not have read my comment Aug 12 at 7:45 pm. The main energy going into the atmosphere comes from the solar heated surface, and direct atmospheric absorption of solar energy by clouds. However increased CO2 does not directly increasing cloud effects. The initial response to increasing CO2 is to make the atmosphere slightly more opaque to long wave radiation, with little effect on incoming solar energy, and raise the average altitude of outgoing long wave radiation. This does not initially result in heating of the upper Troposphere and reduced convection. It reduces surface radiation heat transfer up and increases surface convection, due to the surface heating more from the reduced radiation heat transfer. Total heat transfer up is initially increased over the level before addition of CO2 (with all of the increase due to convection effects, not radiation), but once the entire atmosphere is heated to the new level (with lag due to thermal capacity), due to the raising of the altitude of outgoing radiation, the total heat transfer up is the same as before (and equals absorbed solar energy). At the new level, the radiation heat transfer up is less than before, but convection up increases.
My last sentence is for heat transfer from the surface. Obviously at the outgoing altitude, all escaping energy is radiation, and at equilibrium, equals absorbed solar energy.
Leonard,
I don’t think I agree with you. Finally, we might have a real disagreement!
A more opaque atmosphere will radiate less to space from the mid to upper troposphere which cools it = radiative cooling. Let’s call this Δu.
The surface will experience a much less significant change, an increase in DLR = radiative heating. Let’s call this Δd.
I believe that Δu > Δd.
Agree or disagree? (before I go further)
I disagree with your contention. The convection continually adjusts to maintain the lapse rate (locally warm spots are more buoyant so quickly rise to mix and restore the local environmental lapse rate), so variations in radiation heat transfer does not significantly change the average temperature profile in the atmosphere even at fairly short time scales. However, the increased opaqueness would immediately cut radiation heat transfer from the surface, so it would heat up. There is a significant lag to transfer to the air, especially from the oceans (note most of the energy absorbed in the oceans is at significant depths).
scienceofdoom,
To specifically respond to your question, keep in mind that atmospheric omnidirectional radiation fluxes occur based on local absolute concentration of absorbing and radiating gases and local average temperatures. The outgoing radiation to space occurs from a range of altitudes, but on average is from about 5 km. At this altitude, both lower radiating gas concentrations and significantly lower average atmospheric temperature occurs than from near the surface. Thus the downward amount of atmospheric radiation near the ground, (delta d) is > than (delta u), the radiation upward to space (or upward from any altitude higher than a lower portion of atmosphere). There is no steady state condition that this is not so for real atmospheres. Even for transient conditions, there would not be a hot spot since the convection and reduced radiation flux up from lower levels does not increase local energy concentration faster than the lapse rate at whatever level it as the entire profile shifted up to the slightly warmer temperature at corresponding altitudes.
SOD,
One more comment. The large increase in DLR is due to the greater opaqueness of the atmosphere resulting in DLR coming from location closer to the ground, which are warmer than from the initial higher locations. Since net radiation flux is the difference of the ground up minus the absorbed air down, the net flux up decreases, and solar energy accumulates on the surface but added convection removes it.
Leonard,
To clarify, d > u (that is, the DLR at the surface is greater than the emission to space from the atmosphere)
But I expect that under this dynamic condition Δd < Δu (Scenario A)
The reasons for this can’t be determined “by inspection” because it is a very non-linear problem. I haven’t proven it, I expect it. The reason is primarily about the DLR from water vapor at the surface.
And I could be wrong. I can test it without water vapor because I have built a line by line MATLAB model. But to include water vapor properly I need a continuum model which is especially important for water vapor near the surface because there is a (specific humidity)2 relationship.
In any case, if it turns out that Δd > Δu (Scenario B) then in that case the dynamic description would be a little more complicated but we would come to the same conclusion about the new steady state condition (we have already agreed about the steady state condition).
Under Scenario A, the description should be correct – do you agree? (I realize you don’t accept Scenario A at the moment).
Under Scenario B:
– the surface warms up due to the increased DLR
– the upper atmosphere warms up (but less) due to a reduction in radiative cooling
– what happens next exactly depends on the profile of warming in the atmosphere due to less CO2 radiative cooling:
SOD and others: When I look at the Moist Potential Temperature Plot (latitude vs altitude, Figure 8) and the Relative Humidity Plot, I try to picture them superimposed with the Hadley and other cells. If I understand the situation correctly, parcels of air can move along lines of equal moist potential temperature without any work being done by or on the parcel. The fact that these plots are annual means and zonal averages complicates things.
a) The dark green band from 20S to 20N is obviously associated with the ascending branch of the Hadley circulation which appears to be rising adiabatically since it is all the same potential temperature (340 degK). The plot doesn’t actually tell us if the air is rising or falling, just that it appears to be well-mixed adiabatically from the surface to 200 mb.
b) The descending branch of the Hadley circulation comes down with a potential temperature of 320-330, probably after radiatively cooling at 200 mb. Cooling would require about 5-10 days according to Figure 6. The slight tilt to the lines suggests that the air doesn’t descent perfectly vertically or adiabatically.
c) However, the relative humidity plot shows us that the Hadley circulation is not an adiabatic process. As the Hadley circulation descends from 700 mb to 900 mb, relative humidity nearly doubles despite the rising temperature. Is there some sort of turbulent boundary layer mixing at these altitudes (that brings in moisture from the ocean, but not over land)? Likewise, there is some reduction in the relative humidity of the ascending branch, where one might anticipate 100% relative humidity during adiabatic movement.
d) I can’t really see any signs of the polar or Ferrel cells, suggesting that they may move slowly enough that mixing or radiation make these significantly non-adiabatic circulations.
e) The constant potential temperature at all latitudes just above 200 mb is consistent with, but does not require, the existence of meridional circulation at this altitude. Is there an explanation for the constant potential energy at this altitude? What radiative equilibrium (Figure 7) would look like on a moist potential temperature plot?
Frank,
Good questions. I will be writing some articles about global atmospheric circulation and we can try and address these points along the way.
Leonard,
Is there a way to directly email you?
RW,
You can mail me at the National Institute of Aerospace, where I am currently employed. My e-mail (which could be found on the web for that location) is:
leonard.weinstein@nianet.org
Thanks. I have emailed you.
FWIW, while the lapse rate is independent of temperature, the temperature at any point in the atmosphere depends on the temperature at the surface and THAT depends on the greenhouse effect. The simplest way of visualizing this is that the greenhouse effect shifts the line in Fig1, moving the intercept
Eli,
Who or what are you responding to. Both SOD and I both agree that is true. No one I know said otherwise. If one of my statements made you think I indicated otherwise, then my text was not clear enough, but I don’t know where.
As usual there was lots of the usual triumphant back and forth about how the lapse rate is independent of the greenhouse effect, which is true, but Eli merely pointed out that the greenhouse effect is the major determinant of the actual temperature at all tropospheric altitudes. Otherwise it would be a hell of a lot more like Antarctica in the tropics. Was not directed specifically at thee at all.
Eli Rabett says….. without the greenhouse effect…..
” Otherwise it would be a hell of a lot more like Antarctica in the tropics”
Solar isolation producing a maximum of 123C at the equator .
Underwater volcanic activity keeping some parts of Oceans from freezing up.
These two facts alone should remove any cause for alarm on that score.
Oh right, those mysterious underwater volcanoes. As Fermi said about aliens where are they. OTOH, Eli reads blogs.
Eli,
Again, what are you responding to. I have stated often that the lapse rate is temperature independent, but the level is determined by the solar insolation combined with greenhouse effect (altitude of outgoing radiation).
Leonard – to be picky: the temperature at any point in the troposphere depends (mainly, but not only) on the temperature at TOA (actually, at the top of the troposphere), and that depends (also) on the greenhouse effect.
The surface is not magically changing the temperature at TOA, it’s the other way around.
“the greenhouse effect is the major determinant of the actual temperature at all tropospheric altitudes. Otherwise it would be a hell of a lot more like Antarctica in the tropics”
That is backwards, of course.
Were radiative forcing not countered by convection, the surface would be much hotter than it actually is.
The tropics would be not a hell of a lot more like Antarctica, but a hell of a lot more like hell.
SOD,
All of my comments assumed the water vapor did not increase significantly when the CO2 jumped, so it is not a player at the start of the transition. It would eventually increase with time (it has to significantly heat at the surface, surface water evaporate, and be convected partially up), but then the entire system would go to the new equilibrium probably as fast. There may be some intermediate state where water vapor would allow a hot spot, but not at the start, or at the new equilibrium. Obviously the real world is much more complicated (even the lapse rate is not really short term constant, and day to night and other variations make any transition too complex to realistically model well).
The basic point I want to make is that even though the outgoing radiation flux is decreased at the beginning of the transition, so is the upward radiation net heat transfer feeding the middle and upper atmosphere. i.e, the radiation heat transfer is everywhere (except right at the surface) reduced at first, and convection controls the lapse rate. Please note that radiation heat transfer is smallest at the surface (due to the added component of convective heat transfer), and largest at the altitudes where it goes to space (since convective heat transfer approaches zero there).
Leonard,
I was also assuming no instantaneous change in water vapor.
If we took a dry atmosphere then I agree with your reasoning. My understanding, which I am happy to be proven wrong about, is that in the lower troposphere water vapor radiation is so strong that CO2 concentration changes have less effect than would be expected. I.e., if the atmosphere is already radiating strongly at a certain wavelength then more CO2 has little effect.
It is probably of curiosity value only..
Within limits. At the surface concentrations of CO2 can vary strongly, as can concentrations of H2O and, at least in the CO2 bend region there is not so much overlap (try it with SpectralCalc. Of course, there is also the issue of aerosols.
SOD,
If somehow scenario A ( Δd < Δu) were true for a transient condition, then there could be a transient hot spot. I do not see how it is likely for the reasons I stated. I think we both agree on the final equilibrium state.
SOD,
Keep in mind that in the real world, CO2 presently increases about 2 ppm per YEAR. Atmospheric mixing and shifting elevation is very slow, so no real chance of scenario A seems possible, since the atmospheric lapse rate adjustments would occur much faster than in a year, and the result would be essentially continuous essentially equilibrium states.
Given that you can see the annual cycle in the various CO2 mixing ratio records, it is not so slow. FWIW, in general it takes about 5 years to push a gas molecule from the surface up to the tropopause, so mixing at 1 or 2 km would be just a matter of months. Mixing across the equator would be at about the same order or a bit faster (look at the lags in the CFC mixing ratio measurements.)
Warmer sea surface increases moisture at low latitudes. That’s expected to result in a reduced lapse rate and larger warming at fixed altitude than at the surface. The moisture content of the atmosphere is close to the saturation value and thus determined by the local temperature. At the same temperature the moisture is as high after the warming as it was before. That means further that the emission directly to space from water vapor is close to the same after the warming as it was before.
Considering the situation as was done above we see that there is very little feedback from low altitude water vapor changes. More detailed analysis would certainly find some effect, but from the above we can understand why the water vapor feedback and lapse rate feedback cancel largely at low latitudes. The well known observation that their sum is known more accurately than they are separately has a clear physical reason in the fact that the moisture level is controlled by the local temperature of the air.
Similar considerations do not apply in situations where the moisture is not near saturation as it is guaranteed to be in the raising air over tropical ocean.
Leonard Weinstein writes: “An increase from doubling the CO2 has been claimed to cause an eventual increase in temperature of about 1.2 C if all other effects are unchanged. For an average environmental lapse rate of -6.5 C per km, this implies the average outgoing level was raised by about 185 m once new equilibrium was reached.”
All this makes sense if the lapse rate itself doesn’t react to the increased radiative forcing. But of course we know it must do because any increase in surface temperature leads to enhanced evaporation from the oceans thus reducing the lapse rate. How else did is it possible that life on Earth survived for the last 3 billion years ?
What about CO2 ? The basic argument for the enhanced greenhouse effect depends on a raising of the effective height of the free path length for IR photons of CO2 to colder levels thus reducing heat loss to space. If simultaneously enhanced CO2 forcing leads to an adjustment of the lapse rate caused by induced evaporation from 70% coverage of global oceans then there is negative feedback.
What I tried to explain is that the negative lapse rate feedback that you describe is always and unavoidably linked with an equal positive water vapor feedback that cancels its effect locally. Elsewhere the water vapor feedback is stronger and therefore the net effect of these feedbacks is positive.
Clive,
Your second comment discusses an additional point that leads to a negative feedback of some strength. That brings, however, up the questionable value of the concept of average altitude of emission to space. It must be understood that this value is never calculated as an average of some distribution of altitudes but it’s determined by calculating first the the total emission, determining then the temperature of a black body that emits as much and checking then what is the altitude of that temperature in an averaged atmosphere. Thus any change in atmosphere that influences the outgoing radiation influences also this “average altitude”. Calling it the “average altitude” is really misleading, “effective radiative altitude” might correspond better to the meaning of this value.
With increasing temperature of the tropical ocean and reduced lapse rate the convection reaches higher altitudes and the tropopause is pushed up. The temperature of the tropopause is not likely to change much as the net effect of all changes, what changes more is its altitude as far as I understand. Both before and after the change we’ll have around tropopause a range of altitudes from uppermost troposphere to low stratosphere of a temperature close to the minimum. The change in the CO2 concentration is not the only reason for the apparent change in the altitude of emission at wavelengths dominated by CO2, the change in the temperature profile has also a significant effect. There will always be more CO2 near to the temperature of the tropopause than near the somewhat higher temperatures of the upper troposphere because the thickness of such a layer is inversely proportional to the local lapse rate. That gives always a special importance for the layers around the tropopause. That means further that all mechanisms that raise the altitude of the tropopause are important for the radiative balance, not only the change in CO2 concentration.
I don’t claim that I understand all the details but I’m pretty sure that most of the commonly presented descriptions are significantly incomplete.
SoD,
I’m wondering why my comments end very often in the state of awaiting moderation (as one comment did again a while ago). Not nearly every time but perhaps every third time or so. Do you have any idea of the reason for that?
Pekka,
It must be frustrating. A lot of Frank’s comments end up in moderation for reasons I have never been able to work out.
I’m using hosted WordPress and it comes with a lot of under the hood stuff that I can’t change (and wouldn’t want to try). If it decides there is something not right it puts it into moderation. If it is more sure there is something not right it puts it into spam. Some of the WordPress algorithms are determined by what is happening on other blogs.
Not a perfect world and sorry to all those who get comments held up for a long time.
For anyone who gets a comment disappearing please email me at scienceofdoom – you know what goes here – gmail.com. For comments that say “In moderation” I already will have a WordPress email waiting for me.
Clive Best,
As far as I can tell from the published data, there is a modest increase in very low altitude absolute water vapor level, but not in relative humidity, which is the major cause of change of lapse rate. At higher altitudes, the absolute water vapor level does not show significant change, and relative humidity actually decreases. The net effect of all seems to be no obvious change in average lapse rate.
Clive Best,
“What about CO2 ? The basic argument for the enhanced greenhouse effect depends on a raising of the effective height of the free path length for IR photons of CO2 to colder levels thus reducing heat loss to space. If simultaneously enhanced CO2 forcing leads to an adjustment of the lapse rate caused by induced evaporation from 70% coverage of global oceans then there is negative feedback.”
What do you mean by ‘enhanced greenhouse effect’ in this context? Also, negative feedback in what way? Increased cooling of the surface via increased heat removed from the surface as the latent heat of evaporation? Or something else?
@RW @ Leonard I am referring to AGW (enhanced greenhouse effect) caused by human emissions of CO2. An induced change in energy flux through the atmosphere must surely also be reflected by a change in the lapse rate. Changes in water vapor is one obvious source for this. However I suspect that CO2 has also a role here.
I think the lapse rate will change increases in CO2 because more IR photons emitted from the surface will now be absorbed by CO2 molecules than before. Part of this energy will be transfered to nearby N2 and O2 molecules, and the rest re-emitted as photons randomly. This essentially causes a gradient heat flow through the atmosphere.
If anthropogenic increases in CO2 itself changes the lapse rate by thermalising absorbed IR with adjacent air throughout the atmosphere, then it too would provide negative feedback by reducing the lapse rate. In other words the effective radiation altitude where the CO2 fog clears is now slightly warmer than it was before (due now just to CO2). Perhaps this thermalising effect is already included in radiative transfer models, but I have never seen this stated explicitly.
Pekka Pirilä argued that the H2O negative feedback from the lapse rate is almost exactly balanced by the positive feedback from extra H2O greenhouse effect. If the same turned out to be the case for CO2 then we can all go home !
OK, so by ‘enhanced greenhouse effect’, you are not referring to claimed net positive feedback. That’s what I was asking. You won’t get any argument from me that the case for net positive feedback, let alone net positive feedback of 300% or more coming from the two most dynamic components of the whole atmosphere (clouds and water vapor), is spectacularly flawed.
What I discussed applies to water vapour because water condesates when the temperature drops and very little water is left when the tropopause is reached. Nothing comparable occurs with CO2.
CO2 has very little direct influence on lapse rate because it has little effect on the thermodynamic properties of air at the present or foreseeable concentrations and because the lapse rate is determined by these properties rather than changes in radiative energy transfer as long as we are in troposphere.
Clive Best,
And this is a helpful comment because it shows that you have no idea about the subject at all.
What is an adiabatic process?
Have a read of Potential Temperature.
Those who have not understood physics basics are doomed to perpetual mixing up of the basics.
Have you wondered why not a single atmospheric physics text book agrees with you? Do you even know that not a single atmospheric physics text book agrees with you?
SoD: Lets try and keep the abuse to a minimum! Adiabatic means no heat input into the system. I am simply trying to better understand myself what drives the lapse rate. I am not trying to preach some dogma! If I am wrong then I will simply accept my mistake – for me it is not a problem. A perfect M-B gas without any greenhouse gases – i.e.no H2O or CO2 will result in a DALR – however you derive it. Such a planet will radiate freely to space from the surface but will also loose a small amount of kinetic energy, and IR radiation from ionised gas into space . Some energy balance will eventually be reached maximising entropy. Gravity and Cp(bulk heat transfer) determine the lapse rate.
Adding H2O and CO2 causes IR energy(heat) to be absorbed differentially throughout the atmosphere. H2O has 2 phase transitions at Earth temperatures resulting rapid heat flux responses. CO2 also effects the lapse rate by diffusing heat from up the surface. Any change in either water vapor or CO2 concentration must therefore change the lapse rate.
Punto Basta.
Clive,
Some greenhouse gases are needed to create a convective atmosphere. Without any absorption and emission of radiation the atmosphere would be essentially isothermal and relatively warm in comparison with most of surface, because it would lack all strong mechanisms that could cool it.
As soon as we have convection the lapse rate is determined by it. More CO2 will increase a little the heigth of the troposphere but not the lapse rate within it.
Clive Best (August 20, 2012 at 9:32 pm)
I apologize.
So many people keep confidently disputing physics 101 that I find it difficult to always stay calm.
Clive,
Thermalising absorbed radiation does not affect the lapse rate. Convection adjusts it. The increase in CO2 is far too small alone to affect the average Cp, and thus lapse rate.
Leonard,
Thanks for the clear statement ! Although I still need to think about it !
cheers
Clive
Should read : I think the lapse rate will change as a consequence of increases in CO2
@Pekka Pirilä .
I think you have hit the nail on the head…..
“Some greenhouse gases are needed to create a convective atmosphere. Without any absorption and emission of radiation the atmosphere would be essentially isothermal and relatively warm in comparison with most of surface, because it would lack all strong mechanisms that could cool it.”
If the atmosphere consisted only of nitrogen – would there be a lapse rate ?
Please convince me !
The greenhouse gases emit radiation from the top of atmosphere making it colder than the surface. That’s the reason for any lapse rate. Pure nitrogen emits extremely little and would not have any lapse rate or actually it might be warmer at the top as it absorbs some UV from sun.
With enough greenhouse gases the lapse rate would be higher than is stable. That leads to convection that brings the lapse rate to the limit of stability. This limit is the adiabatic lapse rate. It’s value depends on density and specific heat of the gas and also on moisture and heat of condesation in case of air saturated by water vapor.
Doesn’t gravity comes in here somewhere as well ? Nitrogen molecules must still loose energy rising to the top of the atmosphere. With a fixed surface temperature of ~ 288K and ~ 3K temperature for outer space surely there must still be a temperature gradient even without greenhouse gases ?
I have my umbrella open for more abuse !
Gravity has always one effect: It makes the density of the atmosphere smaller at higher altitudes. There are less molecules that have high velocities and there are less molecules at high altitudes, but the average velocity does not depend on the altitude in an atmosphere where convection is excluded.
Each individual molecule slows down when going up, but that’s exactly canceled by the effect that originally faster molecules are more likely to go up than slow ones. The exact cancellation applies to a atmosphere that totally insulated in all other aspects except that it’s in contact with a surface of constant temperature at bottom. Even nitrogen has some interaction, only extremely weak, and the Earth surface is not at constant temperature. These factors change the situation a little, but even so an atmosphere of pure nitrogen would be close to isothermal. There would be a thin layer of inversion near surface when the atmosphere is warmer. That stops effectively the cooling influence of cold surface, Higher up conduction would win over very long periods and bring the atmosphere close to isothermal. The conduction is a very weak process but even so it would probably be the strongest one.
All the above is of little relevance for understanding the troposphere but it’s closer to what happens in stratosphere of the real Earth atmosphere.
The adiabatic lapse rate is the temperature change (K/km) that a parcel of air has if it is vertically displaced adiabatically.
This means, if you move a parcel of air 1km upwards how much does its temperature change if there is no transfer of energy in or out of the parcel of air.
The temperature changes because the pressure reduces with altitude and so the parcel of air expands. This is doing work against its environment and the first law of thermodynamics says that this energy must come from somewhere. It comes from internal energy of the parcel of air, thus reducing its temperature.
The reason this is a useful concept is because conduction and radiation move heat relatively slowly compared with atmospheric motion (the wind).
We can see that this is a useful concept because the theoretically calculated adiabatic lapse rate matches the actual environment where convection is common.
If the environment creates a temperature drop with altitude which is greater than the adiabatic lapse rate then convection will take place, as explained in Density, Stability and Motion in Fluids.
Regardless of whether any significant convection ever occurs in practice, if you displace a parcel of air adiabatically by 1km it will reduce in temperature by an amount determined by the adiabatic lapse rate.
All of which has been explained in previous articles.
And it doesn’t mean that the atmosphere does not absorb and emit radiation. It is one very small part of the jigsaw puzzle called climate.
This discussion has been very useful for me at any rate, because it has cleared up in my mind what is the basic driver for the lapse rate.
Without any greenhouse gases in air there would be no IR loss to space from the top of the atmosphere. The Earth would then radiate directly from the surface and the average temperature would be -18C. The atmopsphere would become approximately isothermal also with a temperature of ~ -18C
Hydrostatic change of pressure with height remains the same.
There would be no lapse rate and essentially no convection in the atmosphere.
Now we add greenhouse gases so the atmosphere itself absorbs and radiates energy to space. Loosing energy from the top of the atmosphere sets up a temperature gradient. Convection then drives the lapse rate towards the DALR. Nick Stokes has given an analogy with a heat pump where work is expended to maintain the lapse rate, which I think is rather nice.
“The heat pump is provided by vertical air movements (the energy comes from the KE lost). Heat is pumped down, balancing the conduction upward, and this is the mechanism pushing the lapse rate toward the DALR. Pump efficiency is proportional to the deviation from the DALR. So the actual lapse rate settles to a point below the DALR where the heat pump balances the conductive losses (including IR transport). Adding GHGs pushes that balance point further below.”
Clive,
All known atmospheres contain some “greenhouse gases”, even if in relatively small concentration. Since thermal conductivity in the absence of convection is incredibly slow (it would take from many thousands to millions of years to conduct energy from the ground to the top of the atmosphere in the absence of convection), even a small amount of convection would totally dominate the mixed state of any atmosphere. Except for conditions where radiation cooling make the ground cooler than the air above it (night or comparable), the mixing due to wind from day/night variation and different latitude temperatures, and buoyancy of ground heated air would tend to maintain a lapse rate close to adiabatic (with possible modification due to evaporation and condensation). The adiabatic lapse rate is only dependent on specific heat of the air and level of gravity. The actual level of temperature in the profile is only dependent on absorbed solar energy, lapse rate, and average altitude of outgoing long wave radiation. Even with very little absorbing gas, so that the outgoing altitude is near the ground, the lapse rate will establish as near the “wet adiabatic level”. The condition for an average isothermal atmosphere is not realistic with real atmospheres.
Leonard,
I like to think in the way Clive presented the logic. That the one alternative is purely theoretical does not take anything off from the value it has in understanding what’s going on.
Furthermore the case of only a little greenhouse gases would not be so different from that in the sense that the troposphere with it’s adiabatic lapse rate would then be only a small part of the atmosphere. The tropopause would be at much lower altitude and most of the atmosphere would be in stratosphere even by air mass.
Clive,
If the absorbing gases were very small (say no water vapor or methane, and CO2 at only 10 ppm) so that the level of outgoing radiation was very near the ground, and assuming the same absorbed solar insolation, the ground would only be slightly above 255 K, but the atmosphere would have a lapse rate close to 10 K per km.
Yes, the lapse rate would be the same but extend only to very low altitudes.
Pekka,
Heating in the Stratosphere is due to UV dissociation of O2 and formation of Ozone, which absorbs solar energy to heat. Reducing greenhouse gases would not lower the altitude this occurs at. While a much lower average altitude of outgoing radiation from ground absorbed radiation would result in much lower mixing at higher altitude (but still some from spill over from lower level winds), conductivity is still a MUCH lower speed process, so even the small residual spill over mixing combined with the small amount of thermal conduction downward, and small residual radiation (some CO2 would exist all the way up) would not reduce the location of the Tropopause as much as you seem to indicate, even though it would lower some. The result would likely be a slow deviation from lapse rate over a large distance, and a much broader Tropopause. The devil is in the details.
Leonard,
I admit my error.
The absolute temperature of the tropopause is 16% lower than the surface temperature in case of an atmosphere with very weak absorption and emission of all radiation as long as there’s enough radiative heat transfer to dominate over conduction. Only when the radiative heat transfer is so extremely weak that conduction gets important the temperature of the tropopause gets close to the surface and it’s altitude correspondingly low.
Nitrogen does absorb far ultraviolet and that has also some lowering influence on the altitude of tropopause, but the solar radiation has very little energy at those wavelengths. Thus the effect is rather weak.
I have to make a further correction.
The value of 16% that I mention above is derived in Pierrehumbert’s book on Planetary Climates for a atmosphere that’s gray over the LWIR range of wavelengths and transparent for incoming solar radiation. Furthermore an emittivity of 100% is assumed for the surface. It’s based on the following simple argument:
When absorptivity/emissivity is very small practically all LW radiation absorbed by the atmospheric gases originates at the surface. The gas emits equal amounts downwards and upwards but the radiation from the surface is only the upwards radiation. For radiative balance that is the signature of the tropopause the upwards radiation from the gas must be one half of the radiation it absorbs from that coming from the surface. For gray gas the Stefan-Boltzmann formula is valid and radiation is proportional to the fourth power of the temperature. Fourth root of 0.5 is 0.84 or 16% less than 1.0. This is the 16% I mentioned.
If the only significant GHG is CO2, the 15 um band dominates both absorption and emission. Thus we must determine the temperature from the requirement that Planck’s law gives a emittive power of one half for the radiation at that particular wavelength in comparison to that obtained at the temperature of the surface. Taking for the surface temperature the value of 278K the corresponding temperature is 232K. As another example choosing 254K as the surface temperature the temperature of the tropopause turns out to be 215K. Temperature ratios are for these cases 0.83 and 0.85. Thus the value of 0.84 is a good approximation for the 15um band of CO2 but the result would be quite different if the only important wavelength would be 5um as an example (ratios 0.94 and 0.94). Here we see that the temperature dependence at a single wavelength may differ strongly from that of a gray or black body.
SOD and LW
I must be missing something; for this and the previous article and comment all seem more complicated than need be.
The slope of the dry adiabat (the lapse rate) varies inversely with specific heat at constant pressure. H2O has a higher specific heat than dry air and the moist lapse rate is steeper than the dry one. CO2 has a lower specific heat than dry air and moves the adiabat back towards its original slope. Neither change in the slope of the adiabat affects the surface temperature. Why, then, do we expect increasing quantities of CO2 in the atmosphere to warm the surface?
A warmer surface requires an outwards shift of the adiabat. Adding CO2, the heaviest of the atmospheric gases, would do this. By contrast, adding the lighter-than-air H2O gas would shift the adiabat inwards and reduce surface temperature.
But change in atmospheric density is not among the explanations usually given for a warming surface. One of these is that adding CO2 forces atmospheric radiation to space higher and weaker resulting in the atmosphere losing energy to space at a lesser rate. Supplied with energy from below at an unchanged rate, the atmosphere thus stores energy and warms. A warmer atmosphere relative to the surface steepens the adiabat (reduces the lapse rate). It doesn’t outward shift the adiabat and warm the surface, as Leonard Weinstein’s explanation implies. The warming of the atmosphere carries a self-regulating negative feedback – its rate of energy loss to space increases and stored energy and temperature reduce. With reference to the adiabat, the addition of CO2 moves the radiating co-ordinate up the adiabat and rotates the adiabat clockwise about its origin at the surface; while the negative feedback reverses these movements. But wherever the adiabat and the radiating co-ordinate on it settle at the new equilibrium, the surface temperature remains unchanged.
Another explanation involves the warmer atmosphere radiating to the surface at greater intensity causing it to warm. This is the contentious “back- radiation” which follows Prevost’s theory of radiative exchanges (1792) but contravenes Clausius’s 2LoT (which forbids energy transport from colder to warmer regions- 1850). SOD explains away the apparent theoretical conflict with his credulity-stretching “imaginary vs real 2LoT”. LW on the other hand, though recognising back-radiation, disallows its direct surface warming quality, instead attributing to it a quality of resisting energy loss from the surface which translates, via an ill-defined mechanism, to a warming surface due to storage of solar energy.
Hoping that my interpretations of these analyses are reasonable, evidently both can’t be right and, I would submit, both are flawed. The models employed by both analysts ignore the ubiquitous presence of radiation and direct interaction with bodies it surrounds. Absorption and emission of radiation involving the surface or atmospheric GHGs would then result in changes in the energy density of the surrounding radiation medium, absorption reducing it, emission increasing it. This would obviate the general need for complicating view factors between bodies mutually exchanging radiation and for atmospheric back-radiation specifically. It would also obviate the cognitive disjunct between K&T’s assessment of the relative energy supply to the surface from the atmosphere and from the sun, 2:1 in favour of the atmosphere, and everyday experience.
SOD, if I might be so bold as to venture to suggest a theme for a future article it would be: Experimental provenance of mutually exclusive theories: radiative exchanges which prescribes atmospheric back-radiation and 2LoT which proscribes it.
John..
..Comment moved to the The Amazing Case of “Back-Radiation” article..
John Millett,
The moist adiabat is steeper than the dry one not because of the specific heat capacity of water vapor vs dry air. The moist adiabat is steeper because the latent heat term appears in the conservation of energy equation that is used to derive the lapse rate.
Do the calculation for yourself of the difference in heat capacity of dry air vs moist air at 15g/kg and report back with the answer. Likewise for dry air with CO2 at 280ppm vs 560ppm.
Perhaps a reformulation on the role of latent heat release is helpful for some people. (There’s nothing new in this except in the formulation.)
When a parcel of air moves up it expands, which means that it pushes surrounding air outwards. In that it uses it’s internal thermal energy to do work. That means that the air cools. How much it cools is determined by the temperature change that is needed to release the required amount of energy.
In dry air the ratio of energy released and temperature change is the specific heat. When the air is saturated by water vapor the decrease in temperature leads to condensation of part of the moisture because the saturation value of absolute moisture decreases rapidly with decreasing temperature. This adds to the the energy released when the temperature decreases and has an effect similar to an addition to the specific heat to create a higher “effective specific heat”. The moist adiabat is obtained when the calculation is determined using this “effective specific heat”.
There are some additional effects related to the expansion and to the reduction in the number of molecules in gas phase, but these effects are much smaller under atmospheric conditions and they are taken into account when the calculation is actually done.
In lower and middle troposphere the release of latent heat is a very significant effect. In highest troposphere it a small effect because the saturation moisture is so small at lowest tropospheric temperatures that further reductions in moisture cannot release much energy any more. The influence of the specific heat of CO2 is always negligible and that of water vapor is also small but not totally negligible in the warm moist air of lower troposphere at low latitudes where around 5% of air is water vapor.
One further difference is that the above is presented for the upwards motion of the air parcel. For the downwards motion the adiabat is typically the dry adiabat as the water droplets or ice particles may have fallen down or reached large enough size to prevent the rapid evaporation that would be needed to maintain saturation in warming air.
Pekka Pirila
That was useful, thankyou. A couple of points:
“When a parcel of air moves up it expands, which means that it pushes surrounding air outwards. In that it uses it’s internal thermal energy to do work. That means that the air cools. How much it cools is determined by the temperature change that is needed to release the required amount of energy”.
Why does the air parcel move upwards?
It is common knowledge that “hot air rises”. However, a hot air balloon rises, not proximately because the air inside it is hot, rather because the resultant higher energy density raises the pressure inside above that outside; and the outside pressure is lower at the top than at the bottom. I think the same mechanism applies in the natural air parcel, resulting in a vertical elongation of the parcel as observed in cloud formation.
The phrase “release the required amount of energy” asks “released to where?”. It can’t be to the surroundings, it can only be internally. I find it more satisfactory to think in terms of reducing energy density which occurs as volume increases as the higher internal pressure pushes the parcel boundary outwards (mainly upwards). The specific heat quantifies the relationship between changes in energy density and temperature. Moist air’s higher specific heat explains its steeper adiabat.
John Millet,
The energy released is used to do the work inherent in the expansion of the volume of the parcel, i.e. in pushing the rest of the atmosphere out of the extra volume taken by the parcel when it expands.
The reason for the rising motion of a parcel is often in its temperature being higher than the surrounding atmosphere has at the same altitude. Going further back to the reason of this temperature difference the reason is typically in the temperature of the surface at that location. There might, e.g., be a dark spot that absorbs more solar radiation and gets hotter than the surrounding areas. Another common reason for the rising movement is in larger scale air flows that may force the parcel to go up while its temperature is not different from the surrounding air at the same altitude.
Scienceofdoom
“The moist adiabat is steeper because the latent heat term appears in the conservation of energy equation that is used to derive the lapse rate”.
I take your point: insignificant GHGs can have only minuscule effects on the lapse rate through variations in either specific heat or density. Something else is needed to explain the large difference between dry and moist lapse rates. I think I see the link between lapse rate (an atmospheric temperature profile) and atmospheric energy balance (the time rate of change of atmospheric energy content). An increase in the latter implies the atmosphere cooling faster relative to the surface, that is, an increase in the lapse rate, a flattening of the adiabat.
John Millett,
I will copy your comment to the The Amazing Case of “Back-Radiation” article and you can discuss your amazing theories there. Readers please respond on the topic of back radiation in that article. John Millett has already explained his theory in the comments there.
Thanks, Pekka. Your description is clearer to me when I mentally substitute the words “expend” or “consume” for the word “release”.
John Millett,
Air is heated by conduction and convection from the solar heated ground. As air heats at constant pressure it expands to lower density (pv=RT). Buoyancy then lifts the lower density hotter air up where it expands and cools as has been previously stated. It also mixes with and carries some surrounding air up. There is also cooled air in the upper Troposphere (cooled by radiation to space) that falls back downward, since it is now more dense than surrounding air that has not yet lost as much energy by radiation. Thus there are continual up and down currents carrying the heated ground energy to radiate to space, as well as some direct radiated energy from the ground, and some radiation heat transfer due to local absorption and radiation.
Got it, though ” mixes with and carries some surrounding air up” looks at odds with the assumed adiabatic process.
The bit in your explanation of the greenhouse effect that I don’t get is how raising the radiating altitude translates into a warmer surface, implying an outward shift of the adiabat. Rather, the additional surface energy stored in the atmosphere, that increasing the mass of the absorber induces, creates a temperature gradient between the atmosphere and the surface, implying a rotation, not a shift, of the adiabat.
Leonard Weinstein
Two equations presented by two authorities relating surface and effective radiating temperatures perplex me greatly:
Hartmann……….Ts = (n+1)^0.25 * Te where “n” is the number of atmospheric slices.
Pierrehumbert……..Ts = (Ps/Prad)^(R/Cp) * Trad
Recognising that radiative equilibrium is not a sound basis for determining the relationship between effective radiating temperature and actual surface temperature, nevertheless, how reliable is a theory (radiative transfer) in which the value of the objective variable (surface temperature) depends on the arbitrary choice of the number of atmospheric slices which pass on radiation from one to the next?
The choice is not without constraint: the layer must not be so thick as to contravene the isothermal assumption nor so thin as to inhibit the molecular collision that supports that assumption and the accompanying blackbody assumption. If, for current purposes, half a degree gradient within the layer is assumed to approximate local thermal equilibrium, 13 layers per km would be necessary in an atmosphere with a lapse rate of 6.5 degrees per km or 200 layers in total. These numbers would imply a surface temperature 3.76 times the effective radiating temperature compared with the actual 1.13 times. They also imply that the non-radiative fluxes cool the surface by an incredible 700 degrees C. Alternatively, obviously no fewer than one slice, or the whole atmosphere, may be chosen, in which case the non-radiative fluxes cool the surface by 15 degrees. Two slices is the popular choice (forget the constraining assumptions) whence non-radiative fluxes cool the surface by about 50 degrees.
Hartman, using the 2-layer model, shows DLR at 89% of pre-albedo solar flux (compared with 50% absorbed by the surface directly) proclaiming it to be the key to our benign climate. Without it, he maintains, not only would the surface be much colder on average but also the diurnal range would widen owing to both colder minima and (surely illogically) hotter maxima.
Pierrehumbert provides this formula linking surface and effective radiating temperatures:
Ts = (Ps/Prad)^(R/Cp) * Trad
A similar eqn could be written, with the variables primed, for an atmosphere with a higher concentration of CO2 which raises the radiating altitude. That is, T’rad and P’rad less than Trad and Prad, respectively. The ratio of the two eqns would be:
(T’s/Ts) = (P’rad/Prad)^(R/Cp) * (T’rad/Trad)
Unless my rusty math has failed, because all RHS terms are less than unity, so must be the LHS. That is, apparently perversely, increasing CO2 concentration reduces surface temperature.
John,
You got the first factor inverted. PRad is in the denominator in the first formula and should switch to the numerator.
I had realised the error. But thanks all the same
John Millett,
The example is simply to illustrate the effect that a high concentration of “greenhouse” gases can have using a simple set of equations that (almost) anyone can understand. As we increase the number of “demonstration layers” we reach a point for that atmosphere where the layer becomes optically thin and so does not emit like a blackbody. This means the number of layers cannot be arbitrarily increased.
In any case the real calculation for such an atmosphere uses the radiative transfer equations. This becomes a pair of differential equations with boundary conditions and the result has become known as the semi-gray model.
You can see it explained in plenty of detail in The Mystery of Tau – Miskolczi – Part Five – Equation Soufflé, with the full derivation in this comment.
SoD,
Is there a way to email you or the site?
Email is scienceofdoom – you know what goes here – gmail.com
Pekka,
“You got the first factor inverted. PRad is in the denominator in the first formula and should switch to the numerator.”
As indicated, in a contemplative corner I realised the error. Still in that corner, there is more to this, isn’t there?
Via the gas laws, the ratio Prad/P’rad would be the reciprocal of the ratio T’rad/Trad.
(Prad/P’rad)^(R/Cp) would be less than (Prad/P’rad) because the exponent is less than unity and the product with (T’rad/Trad) would be less than unity, leading to the overall result – a warmer surface induced by adding CO2 (if, as Leonard argues, adding CO2 pushes radiation to space higher in the atmosphere).
John,
Using the gas law to conclude that the ratio Prad/P’rad is the reciprocal of the ratio T’rad/Trad requires knowing that the volume is unchanged, i.e. the conclusion is right for gas in a bottle, but not for gas in atmosphere where the volume is changing. Without the change in the volume the temperature would also be constant. All derivations related to adiabatic processes for rising air are specifically about adiabatic expansion.
One place, where you can find the derivations is in the lecture notes of Rodrigo Caballero on atmospheric physics. Although the material is originally based on a graduate level course, the chapter 2 is relatively accessible with only little background in thermodynamics.
John Millet,
The balance of energy in and out is the source of an equilibrium temperature value (I am not talking of the intermediate process, but final long term average value at equilibrium). The average effective location of the outgoing temperature is the altitude where that calculated temperature value occurs (it is not this simple, since the actual outgoing altitudes vary from the surface to very high, but an effective average can be found to simplify the problem, and does a fairly good job). The mechanism of moving the effective average altitude upward with increase in absorbing gases is simply that it is the partial pressure of absorbing gases, not total atmospheric pressure, that mainly determines the amount of absorption at any location, and thus the average location where the radiation finely leaves the atmosphere moves to a higher altitude with increased partial pressure. The mechanism that drives the lapse rate is not directly dependent on the optical absorption or value of absolute temperature (for fixed Cp and sufficient mixing of the atmosphere), so if the average altitude of outgoing radiation is raised, the surface will heat up due to the integrated value of the lapse rate over the greater distance. The average actual lapse rate is actually fairly constant over the distance to the average altitude of outgoing radiation (-6.5 C/km), so the estimation is simple. Ratios of temperature and level of back and forward radiation adjust to these facts, and not the other way around.
“…..the surface will heat up due to the integrated value of the lapse rate over the greater distance”.
The only way the surface will heat up is by storing energy; it won’t respond to a computation. The question is how does increasing CO2 cause the surface to store energy? That wouldn’t increase the solar supply, You reject increase in supply from “back-radiation”, favouring reduction in upward radiative flux as the cause. This flux would reduce if the surface surroundings warmed; but this would involve the rejected”back-radiation”. The question posed has no obvious answer. Resolution of the conundrum could be to recognise that increasing CO2 raises not only the effective radiating plane but also emissive power (overlooked as a result of the over-simplifying blackbody assumption), thus maintaining flux intensity despite the lower radiating temperature and eliminating the need for the elusive warmer surface.
John,
You are certainly right in saying that the surface warms only when energy is stored at (and below) the surface. From the point of view of the surface the only immediate effect of a sudden addition in CO2 is to increase the downwelling radiation. Nothing else affects surface before warming has had time to proceed.
Gradually the surface warms and that leads towards a new equilibrium with more convection and latent heat transfer. Also the emission from the surface increases but not as much as the downwelling radiation which is increased both as a direct consequence of additional CO2 and because of warmer troposphere.
The changes at the surface are complex and difficult to calculate quantitatively, only the qualitative description given above is easy to produce. Therefore most people prefer to look at the energy balance at some altitude above the troposphere. The heat capacity of the troposphere is small in comparison with the top mixed layer of the oceans and also in comparison with the annual energy fluxes. Therefore the net energy flux at the surface must be very close to the net energy flux at any level above the troposphere when considered on annual or multiannual time scales.
In a warmer troposphere there’s more IR both up and down and there’s also more convection and transport of latent heat. Above the troposphere we are bound by the requirement that OLR must balance solar SW that is absorbed below that altitude. When we are high enough to be sure that downwelling LWIR is very small we know that upwards LWIR cannot increase with warmer surface any more. The higher up those levels are the warmer the surface is. To calculate quantitatively the change in surface temperature we need more detailed knowledge about the changes in altitudes, temperatures at those altitudes and lapse rates to relate the temperature of the top of troposphere to the surface temperature.
Pekka,
“From the point of view of the surface the only immediate effect of a sudden addition in CO2 is to increase the downwelling radiation. Nothing else affects surface before warming has had time to proceed.
Gradually the surface warms…..”
A surface warming due to storage of atmospheric down-welling radiation requires spontaneous flow of energy from a cold region to a hot one, a violation of 2LoT. It also exposes the flaw in SOD’s explaining away the violation provided outflow exceeds inflow – in that case the surface would be cooling, not warming. Leonard Weinstein recognises the problem but, in my opinion, hasn’t been able to demonstrate how the warming surface is due to storage of solar, not atmospheric, radiation. A third explanation – there is no surface warming – would obviate the impasse. Might this result if adding CO2 raised not only the radiating plane (as Leonard argues and SOD I think would agree – although DeWitt Payne, using MODTRAN, shows a warmer radiating plane) but also emissive power which all three assume to be the theoretically maximum at the outset? Emissive power would be enhanced by both increased emitter mass and longer path, an offset to reduced outward flux density due to colder emitting temperature. Under DeWitt’s finding, the offset would be less as the effect of path length would be reversed.
Taking a different approach: the atmosphere’s radiative balance is in deficit – more energy is lost to space than is gained from the sun and from the surface. The radiative deficit is balanced by non-radiative fluxes from the surface. Adding CO2 would increase the solar gain, that from the surface being unaffected – an overall reduction in the deficit. The atmosphere’s extra solar gain is the surface’s loss. The surface stores less solar energy and its temperature rises less than before. A cooler surface supplies less non-radiative flux, matching the reduced demand due to the lesser radiative deficit. A cooler surface implies less evaporation and a drier climate. This condition in turn implies less cloud, lower albedo, more solar energy storage at the surface and rising surface temperature – automatic negative feedback stabilising surface temperature.
John Millett,
Not much point having this discussion on each and every article. Clearly you have zero chance of ever understanding radiative heat transfer. Please can you instead go ahead and raise your unique concepts – that are contrary to all heat transfer textbooks – in The Amazing Case of Back Radiation.
Or in the article where the extracts from the six heat transfer textbooks are shown. Perhaps in that article you can explain why they are all wrong.
John Millett says
“A surface warming due to storage of atmospheric down-welling radiation requires spontaneous flow of energy from a cold region to a hot one, a violation of 2LoT..”
What the second law states is that HEAT cannot flow spontaneously from a lower to a higher temperature.
In the case of a purely radiative exchange, the HEAT would be the net radiative flux.
Heat has the ability to do thermodynamic work.
I have come up with a neat logical proof that a warmer surface can absorb energy from a colder surface.
Three objects separated by a vacuum can ‘see’ each other.
Object A is at 320K
Object B is at 300K
Object C is at 280K
All three objects will emit 10um photons as part of their BB spectrum.
All would agree that object B will absorb a 10um photon from A.
However a 10um photon from C is identical in all respects.
Photons are also Bosons so it is impossible to differentiate them.
So by logic if B absorbs a 10um photon from A it must absorb one from C.
Some Climate Science experts including SoD think its rather tiresome to differentiate between the terms heat, infra red radiation, energy and work.
They quote from textbooks that are rather careless in the use of thermodynamic terminology.
So on and on the confusion spreads.
The angels in heaven will rejoice when SoD recommends his readers to study the Carnot Cycle and banish forever the pointless confusion.
Bryan,
The word “heat” is used in perfectly good thermodynamic texts in many different ways. When the only thermodynamics that we had was classical thermodynamics without any statistical theory or micro level physical explanations to back it up it was essential that the definitions were taken literately. Thermodynamics of that era was an axiomatic mathematically formulated theory that could not be explained by deeper understanding but that had been found to be true wherever it had been properly tested.
For more than 150 years we have had a deeper understanding based on some simple properties of micro level physics and statistical analysis. There were originally some serious problems in the statistical theory but they have been resolved by quantum mechanics. Thus we can now discuss the physical processes as you did with the example of photons. Having the understanding of what kind of energy can be classified as heat energy we are not any more limited by the axiomatic approach. Some of the ways that are presently natural contradict the classical view that heat can be identified only in the flow of heat as it’s now possible to determine the absolute value of heat energy in some body or some volume of gas.
Similarly we can discuss the two components of energy transfer whose difference was the heat flow of axiomatic thermodynamics. Sticking to the definitions of past centuries is now rather a burden than the only acceptable choice. Discussing the two components separately is very useful in case of radiative heat transfer but does not make much sense in case of heat conduction.
It’s good to know the different ways the word “heat” is used but it’s pointless to claim that only one of the ways is correct.
Pekka Pirilä says
“It’s good to know the different ways the word “heat” is used but it’s pointless to claim that only one of the ways is correct.”
Perhaps I’m old fashioned!
Maybe you can give me an example from the thermodynamics section of a modern University Physics Textbook where heat is said to flow spontaneously from a colder to a warmer surface.
Until then I cannot help thinking that a little ‘wriggle room’ is required to help the IPCC current greenhouse theory evade the second law of thermodynamics
For readers’ benefit I will collect mine and Bryan’s contribution to the debate on this blog over the last two years or so.
Readers will see what a shining light Bryan has been in explaining the simple thermodynamic truth that the colder atmosphere causes a warmer surface than would have occurred without the colder atmosphere.
We will see what a contribution he has made and how consistently and how clearly he has articulated these thermodynamic basics. And, of course, how quickly he clarified his position on this confused topic when any reader requested it.
With a few plain sentences Bryan has forever banished confusion and everyone has been clear on whether or not energy from the colder atmosphere is or isn’t absorbed by the earth’s surface and does or doesn’t change its temperature.
His clarity on the use of the word heat is of course at the center of his clear exposition of cause and effect.
Alternatively readers might realize that Bryan has taken two years to come to terms with the basic fact that a colder atmosphere can increase the temperature of a warmer surface, and attempts to conceal his embarrassment on this topic by repeating a mantra. We will see. The blog tells a tale.
Unfortunately the moderator has decided that such an investigation would be a breach of Blog Etiquette so interested readers will need to review the past discussions for themselves.
SoD
DeWitt Payne and Pekka Pirilä have both recommend the lecture notes by
Rodrigo Caballero.
Rodrigo’s use of the term ‘heat’ is exactly the same as my own.
You were correct to pose the question of what happens to radiation from a colder source on arrival at a warmer surface.
In my early replies I said something like…. ‘I don’t know’.
I think that this is the correct answer if you are uncertain, unless you are on some kind of ego trip.
In my defence I would point out that not a lot of attention is paid to the question in thermodynamics text books.
The major point of their writing was to find the efficiency of converting thermal energy into work.
The back radiation plays no part in this so it is almost totally ignored.
On further investigation I have concluded that this ‘colder’ radiation is absorbed subject to the constraints of absorbency of the warmer material.
However the ‘colder’ radiation cannot of itself ‘up-convert’ its nature to increase the temperature of the warmer surface.
Nor can radiative energy be continually recycled without entropy gain.
Rather the effect is to reduce the net radiative flux from the warmer surface making its heat loss less.
If the warmer surface’s internal production of thermal energy continues unaltered its temperature will increase.
None of this is controversial.
I can give you a long list of Climate Science professionals who use the Carnot Cycle as a structure for the vertical temperature behavior of the tropopause.
As Rodrigo shows , there is nothing inconsistent about using technically correct thermodynamic terminology and expressing views about atmospheric science.
Bryan,
The axiomatic definition of heat is certainly consistent, but that’s not the only meaning of the word “heat”.
People, who have never learned any thermodynamics know the word “heat” and have a rather good understanding of what that is. This common language meaning of the word is used by physicists as well. A more precise definition of stored heat energy can be given although textbooks may tell that such a concept does not exist in classical thermodynamics.
Basically we can divide the energy of any body or other system that has some limits to two parts: “organized energy” and “disorganized energy”. The organized energy includes things like the kinetic energy of the motion of a body or a macroscopic parcel of gas as a whole, the potential energy of those bodies or parcels and also energy related to the pressure of a parcel of gas. The disorganized energy is energy related to the motion of and interactions of individual molecules relative to their surrounding. This disorganized energy can typically be characterized by statistical distributions of the energies of each molecule and each degree of freedom of those molecules. These statistical distributions include the Maxwell-Boltzmann distribution of molecular velocities in gas and many other distributions related to rotations, vibrations etc. This disorganized energy is heat energy. There’s typically a local thermal equilibrium characterized by local temperature that determines the form of each of these statistical distributions.
Radiation can get its energy from heat energy of the neighborhood of the point of emission and it can deliver its energy to the heat energy at another location. That can happen both from warmer to colder and from colder to warmer. According to Kirchoff’s law the former is always stronger when radiation between two bodies or parcels of gas is considered excluding the role of all radiation that does not involve both bodies. The classical thermodynamics doesn’t discuss these details, because it’s an extremely limited way of looking at the physics. It’s a very valuable way but it’s looking at the system from outside the “black box” where everything really happens in all its detail. The more modern approaches to physics can look also inside the box and tell in detail, what’s going on there.
The concept of heat in classical thermodynamics remains correct for classical thermodynamics, but that does not allow for condemning different uses of the word when we are looking at things classical thermodynamics cannot discuss. Radiative heat transfer is one of those things because classical thermodynamics does not know what radiation or radiative heat transfer is. Classical thermodynamics knows only the net sum of all components of heat transfer, not anything of its division nor absolute values for heat that’s stored in a volume at any moment.
The second law does not tell that net radiative heat transfer is always from warmer to colder, it tells only that the total net heat transfer including all mechanisms goes that way. We need something more, the Kirchoff’s law, to make the statement for the radiative energy transfer separately.
Pekka Pirilä,
If both the hot and cold areas have absorption and emission coefficients that are not significantly temperature dependent, the radiation energy transfer equation does tell us that net radiative heat transfer is always from warmer to colder independent of the conduction and convection. You do not need anything else.
Leonard,
That’s exactly what I meant in saying that Kirchoff’s law guarantees that the net transfer is from warmer to colder. Or actually Kirchoff’s law in its modern form tells more. It tells that for every wavelength separately there’s more radiative energy transfer from the warmed body to the colder one than in the opposite direction. I don’t know any other (independent) law that could tell either that detailed information or in a generally valid way tell that the total net radiative heat transfer is always from the hot to the cold. (Again including only the radiation that is emitted from one of the bodies and absorbed by the other excluding all radiation that’s emitted or absorbed by other matter or escapes to free space.)
I didn’t mean to contradict any standard physical knowledge, but I wanted to tell that the classical thermodynamics cannot say anything about the subprocesses. The Second law is a statement on the sum of all subprocesses and tells nothing on the further details. I made this statement to emphasize, how much more we know when we have also the micro level physical knowledge and the statistical derivation of thermodynamics.
Pekka Pirilä,
Pardon, but my comment also need to say that the coefficients are the same for the two regions. There are possibilities that unequal one would require additional information.
Hi SoD, sorry to be late to the party but I have a question about something you said:
“We increase the amount of CO2 in the atmosphere but at the surface the change in downwards longwave radiation (DLR) from the atmosphere is pretty small, perhaps insignificant.
By comparison, at the top of atmosphere (TOA) the radiative effect is significant. The atmosphere becomes more opaque, so the flux from each level to space is reduced by the intervening atmosphere. Therefore, the emission of radiation moves upwards, and “moving upwards” means from a colder part of the atmosphere. Colder atmospheres radiate less brightly and so the TOA flux is reduced.
This reduces the cooling to space”
Well we and nature have added quite a lot more co2, but rather than the cooling to space being reduced, OLR seems to have increased by around 1.4W/m^2 since 1950 (radiosondes), and by around 0.5W/m^2 since 1980 (satellites).
Why is that?
Tallbloke,
On your own blog you said:
It’s great to see such high standards of ethics from you and certainly your laudable approach ensured that I stopped posting any questions on your blog.
But it does have me wondering, not for the first time, why you are posting a question here.
If you seek answers you should seek them elsewhere.
SoD, You have some really important, well written and informative science on your blog. I find it a source of reference and is much appreciated.
However i think you may be doing your self a disservice by over-reacting to perceived criticism.
Clivebest,
Tallbloke accused me of altering other peoples’ comments on my blog to overcome my difficulty with their “excellent arguments”.
By any standard that I know it is an accusation of dishonesty.
My “over-reaction” to actual libel was to no longer post comments on tallblokes’ blog and to question why on earth he has posted comments on this blog given his claim.
I could do over-reaction. It would look a lot different, believe me.
Clive: SoD and I are well on the way to resolving this ancient dispute and hopefully normal friendly scientific discourse will be resumed shortly.
SoD,
You say:
“And of course, one of the biggest questions in an atmosphere with more CO2 is how water vapor concentration changes in response to surface temperature change. Changes in water vapor have multiple effects, but the one for consideration here is the change to the lapse rate. The dry adiabatic lapse rate is 9.8 °C/km, while the moist adiabatic lapse rate varies from 4 °C/km in the tropics near the surface (where the water vapor concentration is highest).
Consider an atmosphere where the temperature reduces by 15 °C in 2km. Dry air moving upwards reduces in temperature by 20 °C – which is colder than the surrounding air – and so it sinks back. Very moist air moving upwards reduces in temperature by about 10 °C – which is warmer than the surrounding air – and so it keeps rising.
So more moisture reduces the lapse rate, effectively making the atmosphere more prone to convection – moving heat into the upper troposphere more effectively.”
Very interesting, but is it fair to ask how this behavior is consistent with positive water vapor feedback? It would seem to be consistent with the opposite given the process of convection primarily accelerates radiative cooling by moving energy non-radiatively higher up in the atmosphere where when radiated it has a greater chance of passing through the remaining atmosphere into space.
RW,
You are correct. This is a negative feedback.
If you take a look at this graphic, which was shown in Visualizing the Climate Response summarizing the results of various climate models:
you can see that the lapse rate feedback is a negative feedback, the water vapor (radiative) feedback is a positive feedback and combined they still result in a positive feedback.
Do you know if these calculations include increased cooling as a result of increased removal of energy as latent heat from the surface as more water evaporates?
Increased evaporation is included. It’s closely related to the reduced lapse rate in the atmosphere as both are parts of enhanced transport of energy from surface to atmosphere as latent heat. Evaporation takes energy from the surface and condensation releases that heat higher up in the atmosphere.
When evaporation takes energy from the surface the temperature difference of the surface and the lowest atmosphere is decreased and that leads to a reduction in both net radiative heat transfer and convection near surface. Thus the total energy transfer which includes all three forms of heat transfer does not change nearly as much as the latent heat transfer alone. Their sum is controlled by the energy balance of the surface and the share of each in the sum depends on the strength of evaporation.
Looking at the graphic, I guess not. It seems to me this would be equally as significant (if not more so) as the lapse rate feedback.
RW,
There’s no way you can see anything of that in the graphic, but it’s there.
Pekka,
OK, I think I understand. Thanks for the explanation.
Pekka Pirilä
In your post above you say:
This would only be true if the planetary surface were isothermal, but it’s not. The combination of poles colder than the equator and planetary rotation will create circulation and that circulation will create a lapse rate close to the adiabatic rate pretty much everywhere even if the atmosphere were perfectly transparent at all wavelengths. We hashed this out a long time ago at Nick Stokes site and here. See also SoD’s next post: Atmospheric Circulation-Part One
I’m a little surprised no one called you on this, although I only skimmed the thread comments so I may have missed something.
DeWitt Payne,
The point you make is familiar to me but I believe it’s not correct. The reason is in the fact that without radiative heat transfer the atmosphere can be heated rather efficiently by the hottest areas of the surface but there are no efficient mechanisms that can cool the atmosphere anywhere on the Earth. Thus practically the whole atmosphere would warm up to a temperature close to the warmest surface areas.
That’s a little oversimplified, but close to what would result as far as I can see. In areas where the surface is colder than the atmosphere a very thin layer of strong temperature inversion would develop. Some circulation would be maintained in the stationary state but only at very low altitudes and mainly around the highest temperature spot on the surface. The rest of the atmosphere would be warmer than the surface below, stratified and thus prevents convection effectively.
It’s true that conduction is extremely weak. As conduction is the process that would ultimately lead to the isothermal state, it’s possible that my argument fails. It’s possible that I overlook some other very weak mechanism that would still be stronger than conduction. It’s possible that my view that the low altitude circulation that involves convective heating from the hottest spot and conductive heat transfer through the thin inversion layer in other areas would not leave the rest of the atmosphere fully stratified.
Even so I have not seen any real arguments against my reasoning, all the counterarguments have been presented by people who have not yet gone through all steps of my argumentation or by people who don’t state specific enough reasons for not agreeing with my view on the ultimate outcome in that imaginary situation.
I have had some discussion on these issues on my own site under “random topics” but not with anyone with strong physics background. Thus I have not felt strongly contested.
Both N2 and O2 have a tiny but still finite emissivity. So the atmosphere would never reach an isothermal temperature of say T=255K, since as the atmosphere warms the N2,O2 radiation loss to space would increase as T^4. Convection would be able to maintain some lapse rate to balance the N2 O2 radiation loss.
My comment was for an imaginary situation with exactly zero emissivity. When we start to discuss counterfactual situations it’s common that people make differing assumptions.
It’s been modeled and apparently conduction is enough because the surface boundary layer would be thinned by winds from the convection cells. I don’t have the link for the article on modeling a planet with a transparent atmosphere and can’t find it in a quick search.
When I asked Gavin Schmidt, who knows a lot more than I do about the subject, he specifically rejected the idea that the atmosphere would be isothermal vertically. The details would depend on other properties of the atmosphere, whether the conditions for baroclinic instability are achieved in the mid-latitudes for example.
Let’s look at it another way. Suppose we postulate that the atmosphere is so viscous that convection is impossible. Would the atmosphere be isothermal? I don’t think so. The surface at the poles would be much colder than the equator and vertical conduction would be faster than horizontal conduction because the distance is shorter. If a non-convecting atmosphere isn’t isothermal, then it is even less likely that an atmosphere with low viscosity would be isothermal because any vertical movement the atmosphere will create a temperature gradient. And there will be vertical movement driven by the horizontal flow created by the pressure gradient force.
Your proposed isothermal atmosphere might, in fact, be stable. But I don’t see how you could get there. Formation of circulation cells would act to reduce heat transfer from the equator to the poles.
DeWitt Payne,
As should be clear from my above comments my proposal is based on intuition of one physicist. We seem to agree that it’s validity is determined by quantitative factors. The situation is so extremely different from the real atmosphere that I have doubts on everybody’s intuition including experienced climate modelers – as I have doubts also on my own intuition.
I thought that no-one would have been interested enough in this kind of unrealistic case to construct a model that’s really applicable to it. (I don’t think that any standard atmospheric model would have been built having the applicability to this case in mind.) Perhaps someone has. If so it would be intellectually interesting to learn about that.
Whatever the right answer is, I don’t see how it would have anything more than a curiosity interest. Trying to figure out what could happen may be educative – and essentially as educative when the outcome is false than when the outcome is right as long as the physics is taken qualitatively correctly into account.
Pekka,
I suspect this is a thought experiment perhaps taken too far. How high above the planet would the temperature remain isothermal ? If such a thermal boundary were to exist then above it there would be a temperature discontinuity with outer space @ 3K. This would surely violate the 2nd law of thermodynamics at that boundary.
Clivebest,
That’s not a relevant consideration. The temperature at the outer edge of the atmosphere may have any value without problems of that nature. That’s true for the real atmosphere as well. The temperature of the Earth atmosphere is actually very high as far out as defining a temperature for it is meaningful.
Pekka,
OK – so the bottom line is:
There can be no greenhouse effect without a lapse rate, and there can be no lapse rate without a greenhouse effect.
Do you agree ?
Clivebest,
I agree on that, but i add that there may be a lapse rate without any significant GHE.
My proposal is that the atmosphere would be significantly warmer than the average surface temperature. It would be coupled effectively only to the warmest spot of the surface and have very little effect on the surface temperature which would be essentially the same as without atmosphere. That would apply to all atmosphere except for a very thin layer near surface. That atmosphere would have zero lapse rate almost everywhere.
The other alternative that DeWitt Payne considers likely has a dry adiabatic lapse rate over a significant range that could be almost as high as the present troposphere because the theory of optically thin atmosphere tells about such a troposphere. That atmosphere would, however, not have any greenhouse effect either. The surface temperature would be essentially the same as in my alternative, but the upper troposphere would be much colder.
I must add that it’s not clear what the height of the troposphere would be in absence of all radiative heat transfer. The theory of optically thin atmosphere is based on the assumption that there’s some radiative heat transfer but the absorptivity is so small that almost all radiation of all wavelengths passes through the atmosphere without being absorbed. At the same time it’s, however, assumed that radiative heat transfer is much stronger than conduction which is not true when the absorptivity/emissivity is exactly zero. This is one reason for my conclusion that the height of the troposphere would be very small. We would have only a isothermal stratosphere above a very thin layer of temperature inversion.
[…] temperature increase. Then it checks between each layer to see if the lapse rate is exceeded (see Temperature Profile in the Atmosphere – The Lapse Rate). This means the atmosphere would be unstable, resulting in […]
Your style is unique in comparison to other folks I have read stuff
from. Thanks for posting when you’ve got the opportunity, Guess I’ll just bookmark this page.
Having read this I thought it was rather informative.
I appreciate you spending some time and effort to put this information
together. I once again find myself spending a lot of time
both reading and leaving comments. But so what, it was still worthwhile!
Je suis arrivé sur votre log ppar hasard puis je ne le regrette nullemkent !!!
I’ve got to go with Clive and DeWitt on the pure nitrogen weather system .. and I was just as surprised to see how long that went unnoticed. Conduction will trigger convection, once their both going, its a nice little conveyor belt. Whack in the differential warming/cooling rates of land/sea, some Coriolis, not to mention un-smooth surface whirling around, etc (adding in a bit of geology would also do it) … and Bob’s your uncle, a nitrogen weather system (or should that be a whether system:-).
I am not sure if I understood Pekka’s comment on the point of the atmosphere must always be warmer than the surface (or did I miss read that?). Anyway, quick trip to the Arctic and then to the Sarah would soon sort that.
However, it was correct of him to point out that thermodynamic heat transfer can’t be considered on the basis of a single photon (I don’t think he actually phrased it like that, so my bad if I’ve miss paraphrased), since Thigh -> Tlow is always right in the “ensemble” (not to be confused with that nutty “ensemble model averaging”). Indeed, save for some conceptual issues, trying to measure a single atom or photon is, tautologically, meaningless (at least according Heisenberg et al), and why Boltzmann’s bits are called “statistical” thermodynamics etc.
However, and again not sure if I miss-gathered this, but is the generally thrust of some these discussions that the (A or not)GW means all radiation from surface leaves at TOA (or where ever sufficient transparency exist)? If so, have I missed something or does that not crush the entire GHG proposition. After all, if there is warming it is because the temperature has gone up, which means not all the energy leaving the surface can be leaving at TOA
in – out +sink/source = accumulation = a dT/dt /=0, assume no sinks etc
in – out = a dT/dt /= 0, so in cannot = out
Also, if the satellite data say that pretty much across the IR all the bands that can absorb (GHG) are fully or mostly saturated, then what (GHG) radiation/energy is actually leaving the planet?
If you put a few telephone poles into a river, and they all stick out at the top, then sure, you have held back some of the river, and the “temp” (i.e. height) of the river will increase on average, but after that it makes no difference how much taller the poles are. The extra height at the poles produce some dP, and so the water between the poles flows faster (e.g. non absorb frequencies), and everything comes to steady state (not be pedantic, but “equilibrium” means something else).
I am also not sure about the adiabatic argument for a rising/expanding “blob” of air. It is not quite the same thing as yanking the plunger on a syringe, or the cooling of an aerosol can on emptying. For example, as a balloon rises, it expands, that may change its temp, but the expansion is doing work on its surroundings, so some energy is passed to the nearby (i.e. not adiabatic in the full energy sense). A blob of air, in a more complicated/mixing manner will also do work on its surrounding on expansion, and also by virtue of its motion, etc. But, the motion does not stop there, as one blob rises, another sinks as part of the conveyor belt, where the energy balance is in the reverse direction. Just the existence of the conveyor belt requires energy, etc.
Also, while the blob would cool on rising, why would the TOA not get a tiny bit warmer, etc on GW.
This point is further muddled in the GCM’s since some tend to keep the pressure at the surface fixed (now that would be weather free environment).
Also, the blob of air etc story, as I was happy to see someone mention, cannot really be that simple since at the tropo there is the good old 200-300 mph (ok 400 km/h) jet stream, very high altitude clouds (ok not too many, but still), etc. which surely indicate that the matter is rather more complex.
BTW, isn’t there a sure-fire way to end many of these uncertainties. I don’t know enough about some parts of this story, but has anybody ever performed something like (or equivalent to):
1) rent a satellite with good instruments, three of four AWACs with spectrometers top&bottom plus all the usual gas/weather instrumentation, and throw in a few balloons for good measure.
2) Then put and circle the AWACs at, say, every 3km directly above each other, and of course, the satellite directly above them, and pop the balloons also. Then, ok it’s just for some hours, and it’s just one spot, but one would then have altitude specific absorption and relate GHG etc data.
Repeat that a few times, shake well, and serve.
Would that not help resolve much of this guessing? Or has somebody already got that type of data?
… given the many many billions already spent, would that be tiny drop well spent?
I’ll apologise in advance for being on the math-geek side, and suggesting something that may be laughable to the experienced experimental physicists et al 🙂
DrOli,
Standard understanding is that earlier warming leads to increased emission at TOA but at the level that would cancel all imbalance. The remaining imbalance drives warming. IPCC AR5 gives the estimate 2.3±1.2 W/m^2 (at 90% level) for total forcing up to 2011. Typical estimate for the remaining imbalance is 0.9 W/m^2, i.e. close to half of the total. Both numbers are highly uncertain, thus the ratio is also very uncertain.
Dr. Oli,
You think that something like this hasn’t already been done? Try looking up ERBE, SURFRAD, or arm.gov. The HITRAN database of molecular spectroscopy was started by the Air Force and used to design heat seeking missiles and other imaging devices. The problem is precision. Satellite spectrophotometers don’t have the spectral range and measurement precision go close the radiation balance to less than 1 W/m². Even the AERI FT-IR spectrophotometer used to measure atmospheric spectra from the surface cuts off at 3300cm-1. But when you do have the vertical temperature and humidity profile available, it’s possible to calculate the expected atmospheric emission spectrum using high resolution line-by-line approach that differs by only a small amount from the observed spectrum. Atmospheric radiative transfer is well understood science.
Closing the energy balance at the surface to within less than 1 W/m² is also problematic because determining heat transfer by convection, particularly horizontal convection (advection) is difficult.
Dear Mr Payne
Please accept this as a response to both of your recent posts, and only as a matter of professional courtesy:
1) Your posts are certainly self-consistent, and also “Frank/SoD consistent”. Amongst other things, you don’t seem to know difference between a question and a statement. Curiously, you don’t seem to know this difference even after it has been a central theme in the posts you are contributing to, so to speak.
Often (as in these cases), when I ask a question, it has nothing to do with my understanding of (science/math etc) facts, it is my attempt to gather the views of others … a point raised repeatedly in the posts, but it seems conveniently absent from your thoughts.
Apparently, your interpretation of a question asked is an invitation for abuse.
As such we differ fundamentally in our approach.
2) As an illustration, perhaps the links you provided do somewhere have altitude data, but not so as I could find, and I don’t have an unlimited budget.
Bizarrely, if data of that type actually existed in the sense you seem to shout, then that data would put an end to many of the to’s and fro’s in this blog and many discussions on the minutia relating TOA, adiabatic this and that, etc etc.
… which was the source of the original post, though again it seems to suit you to ignore that.
That would seem like a very great leap forward, but in spite of the force of your rhetoric you have chosen not to take that great leap, or help others to take that great leap. Not sure what to make of that, but again we differ fundamentally in values.
I simply have no interest in commenting on your other excursions, as that is the correct value to assign to those.
Many thanks for the advice on the “‘tude”, please be sure to accept that I put the same value to that as your other indulgences, and I am impressed with your comments as they show the same degree of attention to facts as before.
I had already noted that I would not respond to SoD and Frank, and with this note please accept that my cost/benefit ratio for this blog has gone singular, so I have terminated all connections and will not receive or respond to anything in this connection.
I expect this will please you as you too seem determined to get the last word in, so please feel free to vent and concoct anything you like. You are welcome to insult me and fabricate more stories, and posture as much as you like to be that legend in your mind.
PS. Can’t remember who it was, but I recall a digression attempting to use inflation and cross-border inflation as a metaphor … be a dear, and let that person know they need to look up Purchasing Power Parity, and Interest Rate Parity just for starters, and if they really want to know what actually happens in the real world, learn about basis swap trading, FXA trading, etc.
All the best
Dr Oli,
You have “miss-gathered this”.
I don’t want to figure out whose incorrect comment you correctly understood, or whose correct comment you misunderstood, but you can cite a specific point if you want it clarified.
On your specific question I extracted – some of the surface radiation leaves without being absorbed, most of the surface radiation is absorbed and re-radiated from higher up in the atmosphere, and lots of heat moves from the surface into the troposphere via convection.
I did some specific calculations to help readers understand the emission and absorption ratios better, and you can see this in Visualizing Atmospheric Radiation – Part Three – Average Height of Emission and other articles in that series.
This post is to explain some more about why the atmospheric temperature profile is the way it is. It is quite uncontroversial fluid mechanics.
You can see nice simple demos of this kind of behaviour in simple fluid experiments and dynamic considerations always complicate a matter, but despite the complexity of the dynamic considerations the actual average lapse rate in the tropics ends up being close to the theoretical value.
You can do some back of the envelope calculations to see how close to adiabatic a process is for a parcel of air moving up at typical atmospheric speeds. No one claims rising air is exactly adiabatic, but it’s like the ideal gas laws. They are ideal laws and not actually correct at all – the question is in each case how useful the formula is and where and how far reality departs from the equations being used. I try to write these articles less technically than the textbooks that most people haven’t read and aren’t going to start reading.
I think from your comments you haven’t understood what potential temperature is and why the concept is an important one. If a parcel of air moves adiabatically up OR down its potential temperature will stay constant. If a parcel of air moves almost adiabatically up or down its potential temperature will stay almost constant. The inevitable result of rising air is a cooling, the inevitable result of sinking air is a heating and this is fundamental physics from the first law of thermodynamics.
The conveyor belt is being driven by solar radiation heating the ground through the (mostly) transparent atmosphere.
In fact this article attempted to explain these very points.
And on your experimental suggestions your last point is exactly on the money. Experimental atmospheric physics is very far ahead of where you appear to think the questions are.
You have not stated what specifically you disagree with in the heat balance equation I provided, and I can’t see how anywhere in the universe that equation could be anything but correct.
I would be grateful for a demonstration of how/when/where:
d(in) – d(out) + q = d/dt (contents)
fails.
I had made no comment nor had any intention to comment on potential temperature. Perhaps your claim that I don’t understand PT can wait until I actually remark on it.
If you take exception with the law of conservation of energy etc … OK, that is your prerogative. Which, incidentally, is also one of the laws of thermodynamics, but which you seem to rely on only on a selective basis (see below).
Your statement on the laws of thermodynamics are used and predicated on the just the opposite of what I actually said. My statements were precisely the point that there is up AND down and the implications of that, since some (possibly yourself) had been posting purely with just the “up” part of the story, and omitting many many other factors also.
Also, it is a little, well, annoying, that you took a snippet as you did and then whinged about my “snippet” on issues that I clearly addressed just a couple lines below your choice of snippet. For example, does my “telephone pole” metaphor not say that “some frequencies” get through, etc etc?
At the very least, we are agreeing, but I am confused as to why your “careful” extraction is used to imply the opposite.
… if you wish to deter my submissions, there is no need for trickery, just let me know …
As for the altitude/absorption relationship, and forgive me if this is critical, but your response is engaging in exactly the sort of problem I was seeking to overcome.
My clear and repeated points included:
1) The dynamics are much more complicated compared to some of the simplifications, and there is much to and fro since there is no actual data or proof provided with many of the submissions.
2) My question then was, has anybody ever anywhere bothered to get actual real factual altitude based data (especially absorption data). As I said, it need not be via my AWAC’s joke, so long as it is real data.
If I understand you response correctly:
1) I can find absolutely no reference or even claim that such data exists.
2) Your kind contribution of the “back of the envelope” calculation BOEC is all very well and good, but its just that … yet another model, without real data (at least not that I could tell).
… yes the TOA absorption spectra is real data, but your BOEC is not (apparently), combining the TOA with modelled z-axis. So the altitude component of those BOEC charts is, apparently, model not data.
… which brings us back to the initial question
I am not sure if it was intended, but your commentary on my interest in real data seems to be critical for a lack of understanding. I would be grateful to know how an interest in real data (especially when there is theoretical dispute) is in some sense a “lack of understanding”.
If on the other-hand, I have misunderstood your statements, and there is the sort of data that would resolve all this speculation, then I would be grateful for the references.
For example, is there actual data that show absorption spectra at various altitudes (hopefully along with related measures), etc? Not sure why somebody should get slammed for asking that question, especially when that type of data would resolve many of the issues in the posts.
It is rather unfair and uncalled for to say “Experimental atmospheric physics is very far ahead of where you appear to think the questions are”, when what I asked for was just for that data. Why not just say if that exists and what it says? Why not just point to a link as you are usually so quick to point to model based links? … why not just answer the question (or not) instead of casting aspersions?
I’ll skip the other issues for brevity … though it would be nice if criticism of my comments would be based on what I actually said … and I don’t see how ad homonym style attacks help resolve data/fact issues.
Dr Oli,
It’s a shame that you have got upset. In your comment of October 25, 2013 at 7:57 pm you said:
I said in my response that this is correct. I said that not all the energy leaving the surface is leaving at TOA.
Therefore, there is no need to correct the equation you cited. It is correct.
The point being, climate science does not say “all the energy leaving the surface can be leaving at TOA”. This is the bit you have misunderstood.
Hopefully that is clear. If you feel this is a personal attack on you then you are visiting the wrong blog.
Hmm, let’s see, you provided strong criticism and aspersions based on completely false concocted claims about my statements, and did so repeatedly. Frank too seems to be in that habit
You’ve just done it again. Too weird.
I did NOT say climate science has this or another view. What I was responding to was some of the posts that DID say something specific.
Do you understand that difference? I wonder.
Repeatedly, pretty much in every post (with one exception for a special point), I have something to the effect
in – out + q = a d/dt of contents
that is an absolute truth, and there is no way around it, why you keep saying that I don’t understand this or that is beyond me. A conservation equation does not imply any misunderstanding of any of the bits you had thrown into the muddle (e.g. potential temp, etc).
Do you actually understand what these equations mean? Have you ever derived the system of coupled PDE’s at the core of these issues? Do you have any idea how difficult (perhaps intractable) it is to get q right?
On the many occasions you claim I didn’t understand this or that with the equations, you made no statement with equations or facts. I then asked you to be specific about the bit in the balances you condemned. So far, nothing.
Indeed, if you get that equation right, you’ve got the lot and everybody can go home (though almost surely still not forecast the climate reliably).
I notice that in spite of your ridiculous aspersions on the data/experimental issues, not once have you actually responded to that with anything other than more ambiguities and misdirection.
Many times I am simply asking questions, NOT because of any lack of understanding of the maths or physics, but because I am interested in the positions taken by some people who have their own take on these issues.
I have repeated that a number of times also, as you are aware.
… yet, here we are again.
Personally, had I got it as badly wrong as you (or Frank), I would have apologised for MY errors unabashedly, not spun into something like “It’s a shame that you have got upset”, as if somehow it’s somebody else’s failing, when it’s yours.
… but that’s just my way
As yet another example of fanciful fabrication, my criticism of your bad errors is not an “up set” thing, it just a statement of fact.
I guess your pattern continues.
How about this, feel free to fabricate anything, feel free to insult/abuse, I won’t bother with a reply. You can have the last word, as that seems important to you. I’ll just call it day … perhaps I can find some place with grown ups.
Cheers
Pot, kettle, black. Try losing the ‘tude’ dude. Otherwise your experience here is likely to be repeated elsewhere.
Howdy! I just want to give a huge thumbs up for the good information
you have got here on this post. I can be coming again to
your blog for extra soon.
I like to share information that will I have accumulated through the calendar year to assist improve group overall performance.
[…] lapse rate (yes, I realise that the environmental lapse rate can differ from this – see Science of Doom’s post, for example). One can then estimate the surface temperature by working from the effective emission […]
Very good blog post. I absolutely appreciate thbis website.
Stick with it!
I hope this is still the right place to ask this!
I thought I understood everything about the lapse rate, but now I am at a loss to explain the following effect. Can anyone else (Pekka?) explain why the Ranque-Hilsch vortex tube works ? see: http://en.wikipedia.org/wiki/Vortex_tube
A gas injected at high pressure into a tube producing vortex flow causes a strong temperature gradient between the outer wall and the centre. The centrifugal force effectively produces an enormous gravitational acceleration ~ 10^6g at the outside of the tube. Perhaps the “lapse-rate” is measured because the gas never reaches thermal equilibrium before escaping. Despite this I think this proves that there really is a gravito-thermal effect – at least transiently.
So if you released an inert gas without GHGs on the surface of a planet at temperature T, for example the moon, what would happen ? I think the vortex tube experiment shows that a lapse rate would be immediately produced as molecules lost kinetic energy against potential energy diffusing upwards with height. Without any greenhouse gases the atmosphere could not radiate to space so the surface remains at temperature T. The atmosphere should then slowly become isothermal as heat is conducted upwards from the surface – but does it ? The boundary with space is interesting because molecules with velocities above the escape velocity are lost to space. This is a loss of heat from the top of the atmosphere. Is this perhaps enough to maintain the lapse rate indefinitely ?
Clive,
This was discussed at length on the Venus atmosphere threads starting here and continuing here and here.
The loss from the top of the atmosphere would depend on the temperature, the magnitude of the gravitational field and the composition of the atmosphere. High temperature, low gravity and low molecular or atomic weight would all increase the rate of loss. For an atmosphere like the Earth’s, I don’t think the energy loss rate would have a significant effect. OTOH, conduction is a really slow process and loss to space could possibly keep an isolated, open topped column from becoming isothermal.
The big problem for a planet, though, is that the surface, assuming a transparent atmosphere, will have a latitudinal temperature gradient. That will force latitudinal mass flow which will in turn create turbulence. I am of the opinion that conduction from turbulence will overwhelm conduction by diffusion and the result will be a lapse rate close to adiabatic, i.e. constant potential temperature rather than constant real temperature. Pekka disagrees with this, at least for the case of a non-rotating sphere with a transparent atmosphere.
Our disagreement concerns the quantitative outcome as far as I have understood.
My view is that the warming of the atmosphere would lead to a situation where the residual circulation is so weak that the whole atmosphere above some rather low altitude is stratified and free of all convective or turbulent mixing. A very weak circulation would persist near the surface maintained by warming of air at the warmest part of the surface and conductive cooling by all cooler parts of the surface.
The lowest layer near the surface would have temperature inversion at every point outside the (moving) hotspot. That layer would be thin near the hotspot and reach maximum height in polar winter. The profile of this layer would lead to some airflow towards the hotspot while the opposite part of the circulation would take place just above the inversion layer.
What would really take place can be determined by a full calculation.
If the planet has major mountain ranges those might have a significant influence on the outcome as they would break through the inversion layer, add to the cooling, and through that make the circulation stronger.
The stratification I mention in the second para would very slowly lead to isothermal atmosphere in total absence of radiative heat transfer, but even an extremely weak radiative heat transfer would maintain a lapse rate. The related convection would, however, be very weak and it would not change the energy balance of the surface or the lowest atmosphere significantly. The inversion described above would remain essentially unmodified as long as the radiative heat transfer is as weak as it would be for a pure nitrogen and oxygen atmosphere.
Pekka,
I don’t think you understand how hard it is to maintain diffusion only transport over a distance as short as 1 cm in water with a viscosity two orders of magnitude larger than air. That’s why I don’t believe that you could have diffusion dominate over distances of thousands of km at a pressure that was anything close to 100,000 Pa.
I would also like to point out that an isothermal atmosphere would not be homogeneous if it were composed of gases with different molecular or atomic weights. It’s turbulence that keeps the oxygen, nitrogen, argon and CO2 well mixed and at the same volumetric mixing ratios with altitude. Diffusion can only dominate in the total absence of turbulence. The turbopause in the Earth’s atmosphere is at an altitude of ~100 km. The stratosphere in the Earth’s atmosphere is still turbulent and well mixed in spite of the very strong temperature inversion that prevents vertical convection. As I remember, Pierrehumbert said that the modeled transparent atmosphere planet would have a constant potential temperature above the mixed layer, not a constant temperature. And that’s for a non-rotating sphere.
DeWitt,
I agree that a diffusive atmosphere is not well mixed. Each of the molecular types has its own height profile. The ratio of O2/N2 would decrease with the altitude. That observation does not validate or disprove any of the other arguments, it’s just a related fact.
Clive,
The following should explain partially, what’s going on. If the empirical observations would be fully explained by that, there were no reasons for more complex explanations. Thus what follows cannot be a full explanation, but it does explain the phenomenon qualitatively.
Gas cools when it expands adiabatically or “partially adiabatically” by which I mean that heat transfer is not efficient in comparison with the rate of cooling due to expansion. Fast rotation maintains a strong radial pressure gradient in the tube. Gas that enters the tube goes to the high pressure side near the surface. Part of that gas exits from the other end at high pressure and temperature from the neighborhood of the outer surface, while part part is removed at much lower pressure near the axis from the other end (In some variations from the same end separated only by a cone that has outlets both at the axis and near edge).
As all gas enters at high pressure that part that exits at low pressure has gone trough the expansion and cooled in that. Whit suitable pressure levels at the inlet and at both outlets the expansion is largely adiabatic. Therefore the cooling is strong enough to make the device work.
There’s a lot of turbulence in such a tube. Part of the work done in forcing gas to the tube is converted first to turbulence and from that to heat. That makes the exiting hot gas warmer then the incoming gas.
The reason of cooling is exactly the same as in the ascending flow in the atmosphere. In both cases it’s do to cooling in adiabatic expansion.
Some other cooling processes are based on the same principle, most clearly the cooling of gas in expansion turbine.
===
Then on the release of gas on the moon.
A lapse rate would not be immediately at the beginning of the release, when the gas pressure is extremely low and the mean free path of the molecules much larger than the average height of the atmosphere. In that early phase the gas would expand freely and almost without molecular collisions. Free expansion is not isentropic, but conserves internal energy and is therefore isothermal. Thus the initial atmosphere would be isothermal at the temperature the gas has immediate after release. (Depending the way the release occurs it may cool in the release. Thus the temperature may be considerably less than in the container, but need not be,)
An exponential density distribution with height would be created immediately. The molecules that have a low speed at the release would remain in the low part of that atmosphere, while the fast molecules would reach higher altitudes. The average velocity of molecules would be the same at all altitudes because those molecules that reach high altitudes have as much velocity left there as those that don’t reach high altitudes have at their position. This can be calculated explicitly from the Maxwell-Boltzmann distribution of the velocities at the point of release, when the assumption of no molecular collisions is made.
Pekka,
In your explanation you are really saying that in the vortex tube high pressure = high temperature and low pressure = low temperature. So something like the perfect gas law PV=RT applies in the vortex tube. However that large pressure is the result of the strong rotation which by the equivalence principal is the same as a strong gravitational field.
Let me try one last thought experiment on you. Imagine that the atmosphere on earth is compressed into a shell 100m thick. Lets ignore all other effects like differential heating etc. or if you prefer it simply take a flat plane with constant surface temperature T. The compressed atmosphere also reaches the same temperature T. Now release the atmosphere and a lapse rate is generated immediately. There are two ways to view what happens.
1) Macroscopic: Adiabatic expansion against gravity generates both barometric pressure and a lapse rate.
2) Microscopic. Molecules reaching a height h in a gravtitational field have on average lost kinetic energy – mgh. So 5/2kT’ = 5/2kT – mgh
Are both views equivalent ?
Clive,
There’s no general rule that high pressure is connected with higher temperatures, but there’s the law of cooling in adiabatic expansion. That’s the point here, and that’s the point in the atmosphere.
In the release you describe, no lapse rate will be generated, but the atmosphere will expand without cooling. To get the cooling the expansion must take place against a counterpressure a do work by forcing the dividing surface move.
From the point of view of kinetic theory this is related to the same affect that a ball that hits a wall moving away will bounce back with a smaller velocity. Without that wall it will keep on moving with the same velocity. This is the reason for the cooling in adiabatic expansion, and lack of cooling in free expansion.
The deceleration in upwards motion under gravity is a totally different issue, which does not lead to cooling but to a reduction in the density with altitude.
In the vortex tube the setup is such that there is a counterpressure, and therefore there’s cooling in the expansion. In convective flows in the Earth atmosphere there’s also counterpressure. Therefore there’s cooling in expansion (and warming in compression).
Thanks Pekka,
You are of course right !
I was wrongly thinking of work done by the gas against gravity, but gravity is instead the cause of the height density distribution ~No*exp(-mgh/kT). However, I think the thought experiment does kind of show why you need GHGs to maintain a lapse rate. On release, radiative losses begin to cool the top of the atmosphere and block radiative losses from the surface. This generates a temperature gradient, which is then maintained long term by convection at the adiabatic lapse rate. Heat must flow through the atmosphere.
Clive,
Your latest comment is correct.
It’s essential to understand that
1) Radiative processes drive towards larger lapse rate in the troposphere. These radiative processes include
– heating of the surface by solar SW,
– IR emission from the troposphere to the space as well as
– radiative heat transfer between the surface and the atmosphere and between various parts of the atmopshere
2) The influence of convection and transport of sensible and latent heat is towards lesser lapse rate. Convection limits the lapse rate, it does not create the lapse rate in typical conditions of the Earth atmosphere. Forced convection may produce a lapse rate under some conditions. That may be true in polar winter where the surface is not heated by the sun. The circulation of the polar winter is driven by circulation of lower latitudes, and the low latitude circulation by solar heating.
Maintaining convection consumes free energy (energy capable of doing work). The source of this free energy is the solar radiation, but the emission of IR from lower temperatures than the heated surface is also essential for the supply of free energy. Emission of IR from the upper troposphere drives circulation much more efficiently than emission from colder parts of the surface.
Clive, DeWitt, Peeka: While trying to follow a recent discussion from someone who will remain nameless at Lucia’s, I also began to wonder about what fractionation by molecular weight tells us about any spontaneous temperature gradients that are set up in the atmosphere. Intuitively, it seems that very high in the atmosphere where lighter gases are enriched, kinetic energy is being converted to potential energy and could cause a spontaneous temperature gradient might develop for this reason. Since there is no enrichment in the troposphere (due to turbulent mixing?), the lapse rate there can’t be due to this mechanism. Is the mathematics of enrichment by molecular weight with altitude available online anywhere? (I checked Caballero?)
Whether thermodynamic equilibrium exists in a very thin atmosphere where collisions are infrequent and the gas doesn’t have a well-defined temperature raises significant questions about the meaning of any gradient in the kinetic energy (not necessarily temperature) that might exist. However, if a very thin atmosphere far from a planet has such a gradient in kinetic energy (ignoring heating from the sun for the moment), then there should be a transition as more moves lower to regions where collisions are more frequent and thermodynamic temperature is meaningful.
Frank,
See above on the turbopause in the Earth’s atmosphere.
Fractionation by atomic or molecular weight due to gravity has nothing to do with kinetic energy, only mass. An isothermal column of mixed gases would indeed have different scale heights for the different components with the scale height inversely proportional to the atomic or molecular weight of the component. Isotopic enrichment by centrifugation wouldn’t work otherwise. In that case the acceleration gradient is so high that it overwhelms turbulent mixing.
Frank,
In extremely thin atmosphere the local energy transfer processes do not dominate over those that transfer energy between more distant points that have different temperatures to the extent the temperature can be defined for such situations. Under such circumstances it’s common that different constituents have their separate equilibriums. The “temperature” of ozone may differ substantially from the temperature of CO2 at the same location. I used parentheses, because the temperature is not well defined, but it’s still well enough defined to make such a statement meaningful.
(My first scientific paper was on a related issue. It was on the coupling of the temperature of the nuclear spin system with the temperature of the temperature of electrons and atoms at very low temperatures obtainable with best cryostats of the time.)
DeWitt and Pekka: Thanks for the replies. Let’s see if I’ve got this right. If I pop a balloon filled with equal numbers of hydrogen and nitrogen molecules at 300 degK in the middle of a empty 10 km vertical cylinder on the moon, all the molecules and continue moving in the direction they were going at the instant the balloon was popped with a Boltzmann distribution of speeds. Those that happened to be going “straight up” will climb to a height determined by initial KE = final PE = mgh, with the average H2 rising 14 times the average N2, assuming no turbulent mixing or collisions or radiation. That will conveniently producing a Boltzmann distribution of maximum heights (exp(-mgh/RT)?) in the gravitational field (but not for the height of all molecules in the cylinder?) Eventually all of the molecules will fall and independently bounce off the bottom of the cylinder “forever” until I add some intramolecular collisions. About half were “falling” when the balloon popped.
(#1) From DeWitt’s reply, I gather that at equilibrium (with collisions), fractionation by height is controlled only by the Boltzmann distribution with respect to the gravitational field (proportional to exp(-mgh/RT), with mgh/RT being one scale height). If there is no turbulent mixing, fractionation occurs by using a different m for each gas; but if turbulent mixing exists, then the average value of m controls all components. So how is KE distributed at each height? (#2) Does the standard Boltzmann distribution of molecular speeds exist at each altitude independent of altitude/PE? This would make the total energy (KE+PE) not follow the Boltzmann distribution. (#3) The alternative is that total energy (PE+KE) is distributed according to the Boltzmann distribution. Since PE already has that distribution, all molecules would have the same KE. Not likely. (#4) Another possibility (requiring my misunderstanding DeWitt) is that total energy is allocated by the Boltzmann distribution and partitioned in PE and KE components by some mechanism that I don’t understand. (#5) In all cases, temperature depends only on mean KE, but I’d assume the concept of temperature begins to lose it normal meaning when significant changes in PE are occurring between collisions.
So my intuition has mislead me and the existence of fractionation doesn’t prove anything. Fractionation can occur purely by a Boltzmann distribution of PE or by a Boltzmann distribution of total energy (as suggested by … ?)
This leaves the problem of getting from the initial conditions of paragraph 1 (where KE + PE is conserved) to the equilibrium conditions of paragraph 2 – obviously through collisions. Obviously v_x and v_y can be changed by random collisions and should follow the Boltzmann distribution. But collisions that change v_z could to be permanently linked to the Boltzmann distribution of PE. So I’m now guessing that hypothesis (#4) above is correct, v_x and v_y are controlled by the own Boltzmann distributions while PE is controlled by its Boltzmann distribution and v_z by conservation of energy (and momentum?). This would explain why Pekka hints that fractionating gases at the same height can have “different temperatures”.
(There remains a problem defining the Boltzmann distribution with respect to height/PE when the temperature isn’t isothermal and varies with height, but I don’t want to think about that now. And there are probably some parallels between how chemical and gravitational potential energy are treated. I’m supposed to know something about the former.)
Frank,
In a mixture of gas with a high rate of collisions every continuous degree of freedom has the same average energy. Thus the kinetic energy related to the x-component of velocity has the same average for each kind of molecule. The average velocity is smaller for the heavier molecules. The value is the same for the other components (y and z). This average is fully determined by the temperature and the energy distribution is exponential (proportional to exp(-E/kT)).
For a single kind of molecules the distribution in the potential energy is also proportional to exp(-E/kT) in an isothermal situation, but this distribution applies to the whole isothermal column, not locally. The potential energy is also a degree of freedom and takes an equal amount of energy as the other continuous degrees of freedom. (For a mixture this applies for the average mass of the molecules.)
The discrete degrees of freedom follow the proportionality to exp(-E/kT) as well, but that does not lead to exactly the same average. The difference may be large, if there are only few energy states as there are for the vibrational modes. The rotational modes are so dense and numerous that they have essentially the same average energy as the continuous degrees of freedom.
The rule is not that the sum of energies of various degrees of freedom is the same in different locations (like at different altitudes), but that each degree of freedom has its energy independent of the others. Thus molecules at higher altitude have more energy in thermodynamic equilibrium. All potential energy is extra to the kinetic energy, not taken off it.
With more degrees of freedom the same total internal energy leads to a lower temperature, and that affects all degrees of freedom.
I hope that you get some additional input for your thinking from the above.
Pekka: Thanks for your informative reply. Your key statement appears to be that four degrees of freedom are distributed by the Boltzmann distribution, velocity in three dimensions and PE. (I called this possibility #2.) Why doesn’t conservation of energy and linkage between v_z and PE reduce the apparent four to three degrees of freedom? They are linked at the molecular level. Even when turbulent mixing is responsible for changing the PE of large numbers of molecules without collision, the energy must come from somewhere.
Frank,
A larger number of degrees of freedom means that the specific heat is larger. The best known case is the influence of rotational degrees of freedom on the specific heat of gases. The influence of the energy related to the altitude in under gravity is similar, but not exactly the same. Two new degrees of freedom raise the molar specific heat at constant volume of two-atomic gas from 3R/2 to 5R/2, while the influence of the altitude is related to the difference between specific heat at constant pressure and specific heat at constant volume. This difference is R or as large as the influence of two rotational degrees of freedom although there’s only one component in altitude. The absence of the factor of 1/2 is related to the linear rather than quadratic dependence of energy on the free variable.
A Google search with the words equipartition theorem altitude should find some texts, where this issue is discussed, but I don’t propose any particular as a good source.
A larger specific heat means that more energy is needed to raise the temperature, but it does not influence the equilibrium temperature reached when heating and cooling act for long as heating by sun and cooling by emission of IR have done on the Earth.
Pekka: Thanks again for another reply, but it appears I used misused the term degrees of freedom. I understand how degrees of freedom effects effect heat capacity, but I was asking about something different. So let’s restrict the discussion to a monoatomic gas.
In the absence of a gravitational field, v_x, v_y, and v_z for a molecule will be distributed by collisions with an average of 1/2kT in each dimension and a Boltzmann distribution of speed (proportional to exp(-mv2/2kT). Individually and overall those collisions will be constrained by conservation of momentum and energy. Now we add a gravitational field. The earlier discussion (and everything else I have seen) indicates that the PE of the molecules is distributed proportionately to exp(-mgh/RT), which I also think of a Boltzmann distribution. Does the result produce four INDEPENDENT modes over which energy can be distributed, or does the law of conservation of energy link the distribution of v_z to the distribution of PE, as my intuition suggests. Any linkage would result would be three independent modes.
Viewed from the molecular level, a linkage is provided through conservation of momentum: any collision increasing one molecule’s p_z must decrease another molecule’s p_z. Upward-moving molecules must be slowing (cooling?) between collisions and downward-moving molecules must be accelerating (warming?).
Frank,
I tried to answer that question already, but added at the same time something else, which may have been confusing. My purpose was to answer at the same time also some additional questions that may now arise.
Altitude and related gravitational energy enters as an additional degree of freedom, but with an average energy of kT per molecule rather than kT/2. That’s because the dependence of energy on the altitude is linear rather than quadratic as it’s for components of velocity.
That’s valid for thermodynamic equilibrium which means isothermal and with a separate density profile for each type of molecule.
In a real atmosphere with convective mixing and with only local rather than global thermodynamic equilibrium the situation is different.
Thanks, Pekka. I’ve tried to understand this from both a macro and molecular perspective, but I’m not getting very far.
Frank
Equipartition of energy breaks down for polyatomic gases.
The classic Kinetic Theory of Gases works for monatomic gases since this involves only translational degrees of freedom
To explain the increase in the heat capacity of polyatomic gases it is necessary to involve quantum mechanics.
A brief introduction is given below
http://hyperphysics.phy-astr.gsu.edu/hbase/kinetic/eqpar.html
Bryan,
The equipartition applies mostly also to polyatomic gases. As I wrote above, it applies rather accurately to translational and rotational degrees of freedom. Where it does break down is with vibratioanal excitations. Therefore the specific heats of H2O and CO2 are temperature dependent to a significant degree, while those of N2, O2, Ar etc. are not at normal temperatures.
Quantum mechanics is needed to understand the way discrete energy levels contribute to specific heat. (The rotational energy levels are also discrete, but the quantitative effect of that is very small due to the large density of rotational energy levels.)
Bryan and Pekka: I did understand about the equipartition of energy across various degrees of freedom, including internal vibration and rotation. My question was whether there were separate degrees of freedom for potential energy and v_z. The kinetic theory of gases usually doesn’t include a gravitational field; but if one is included, v_z and PE might be linked. Furthermore, all collisions between molecules conserve momentum in the z-direction. I wondered how PE and v_z could be independently distributed in a gas given the requirement to conserve momentum and energy. If one considers a single molecule bouncing inside a container in a gravitational field, v_z and PE don’t vary independently.
I suddenly realized that momentum of gas molecules is not conserved when they collide with the walls (especially top and bottom) of the container. This provides a mechanism to independently distribute kinetic and potential energy in the vertical direction that didn’t appear to be possible when considering only collisions between molecules.
Frank,
Your observation is essential as it alone explains totally both the vertical density profile and the isothermal temperature of the gas in the case where all collisions between molecules can be disregarded. This case is discussed in chapter II.B.1 of this paper by Berberan-Santos et al.
I have written some related notes on the interacting case here.
DeWitt,
Your argument is that very small disturbances win over diffusion. My argument is that the disturbances are, indeed so minimal that this will be the case. We are discussing an extreme scenario, therefore arguments based on regular situations do not apply. As long as we do not have access to a real careful analysis we may keep on believing our own intuition and neglect that of the other.
On part of the question I’m, however, very confident. I feel sure that with absolutely no radiative heat transfer, most of the atmosphere will have a lapse rate less than adiabatic. That would immediately mean that it’s stratified. From that follows that no convective vertical mixing can take place. Here the point is that anything less than adiabatic lapse rate will lead to this conclusion. I’m really confident that such a stratified situation would dominate the atmosphere in that case. Breaking the stratification requires a strong disturbance, weak ones mean nothing. That makes me so confident on the conclusion.
Switching to the case of very weak radiative heat transfer the situation is different as that would be sufficient for maintaining the adiabatic lapse rate. When the lapse rate is adiabatic, very weak disturbances may lead to convective circulation. Thus the whole troposphere could contribute to the transfer of heat from the hotspot to high latitudes. Still the heat loss from the atmosphere remains small. Still that prevents also a large flux from the surface to the atmosphere. Still that means that the temperature just above the inversion layer must be high, with a potential temperature very close to that of the hotspot of the surface. The circulation pattern of the troposphere may, however, be very different from the case of a stratified atmosphere.
Pekka,
No vertical movement does not mean no movement unless the surface is also isothermal. In which case the atmosphere would indeed become isothermal over time. But a sphere exposed to radiation does not have an isothermal surface. There will be horizontal energy transfer. That horizontal air movement will create shear and hence turbulence. Given the low viscosity of air, that turbulence will propagate vertically. There is horizontal air movement, turbulence and mixing in the Earth’s stratosphere where the temperature actually increases with altitude.
According to Pierrehumbert’s simple atmospheric model, the height of the tropopause in an optically thin atmosphere is inversely proportional to the emissivity/absorptivity, which controls the temperature of the stratosphere. Therefore in an optically transparent atmosphere, there would be no stratosphere as the height of the tropopause is undefined.
DeWitt,
I have discussed that horizontal movement in a couple of comments. My conclusion is that it’s so weak that it does not lead to significant turbulence. In particular it cannot break the stratification.
Optically thin atmosphere of discussed by Pierrehumbert is such that
1) Almost all radiation from the surface passes trough the atmosphere, ant that’s true for all wavelengths. That’s needed as the calculation assumes that the intensity of radiation from below is independent of the altitude.
2) Conduction can be neglected relative to absorption of radiation from surface and emission from the atmosphere.
Making absorptivity so small that conduction must be taken into account changes the energy balance near tropopause. At the tropopause heating includes both radiation from the surface and conduction, while cooling is by emission and conduction. The conductive cooling is weaker than conductive heating as the lapse rate is much smaller above the tropopause. That raises the tropopause temperature leading to a lower altitude for the tropopause. The higher tropopause temperature is the reason for non-isothermal stratosphere as the upper stratosphere will have the essentially same temperature as in the standard case of optically thin atmosphere.
Pekka,
You’re neglecting the pressure gradient force. With a latitudinal surface temperature gradient, the height at constant pressure must decrease with latitude because the surface layer at high latitude, in the absence of fairly massive circulation, will be cooler than the surface layer at the equator. The resulting pressure gradient at constant altitude will cause circulation at altitude as well as at the surface. The stratification will be near the surface, caused by the temperature inversion needed to transfer energy by conduction from the warmer atmosphere to the colder surface as latitude increases. For a non-rotating sphere, there will be longitudinal pressure gradients as well. For a rotating sphere, things are even worse for your point of view as you get the Coriolis force at play as well as the diurnal cycle of the surface layer.
I keep at this because I’m willing to be convinced that I’m wrong. You haven’t done it so far.
DeWitt,
I have realized all the points you mention, and I have given some thought on them. That has all, however, been on the qualitative level. Some calculations would be needed to have more trust in the conclusions.
If my ideas are correct, all wind velocities are low. Thus the Coriolis effect would not be strong either. The first calculation would be a two-dimensional calculation of laminar flow and conduction. That should tell, whether the assumptions of that calculation are internally consistent. Such a calculation might be easy enough to do based on the curiosity value alone.
Pekka: The references you provided above seem to suggest that a fully rigorous derivation is impractical because too many interacting variables are changing. (See p 408 column 2 about the error of pi/2.) In chemistry, molecular dynamics is used to study/calculate the behavior of molecules as they evolve under the laws of physics. See http://www.youtube.com/watch?v=lLFEqKl3sm4 if you aren’t familiar with this field.
Applying the “strategy of molecular dynamics” to the kinetic theory of gases in a gravitational field might produce useful answers to questions that can’t be addressed by purely mathematical derivations. Imagine doing calculations on an isolated column of gas and following what happens when a gravitational field is suddenly turned on. In theory, at least, one could monitor all of the changes in pressure (impulse transferred to the wall at any particular height), temperature (mean kinetic energy at that height), density, PE, etc. It would be interesting to follow the temperature gradients that develop (some PE is converted to KE, so the gas will warm). Presumably the column will gradually return to an isothermal state with the expected pressure gradient and distribution of KE and PE. Since a post by Clive suggested that some work is actually being done in this field, there might be some value to seeing such a simulation and knowing that every individual energy transfer was carried out following the laws of physics.
One would need a massive gravitation field to created a significant gradient in potential energy in a box of gas molecules small enough to follow individually. Given the importance of enrichment of uranium by gaseous diffusion, such simulations presumably have been carried out for other purposes.
Frank,
The paper of Berberan-Santos et al is written to present some rather simple examples. The case of non-interacting molecules can be described easily without further simplifications, but the case with the error of pi/2 does involve simplifications. I don’t think that correcting for that would be difficult, but it would have made the description of the case lengthier and a more complex.
Kinetic theory can be used to explain in a different way phenomena described fully by formal thermodynamics. In that the rigor is not essential. The relationship between statistical description of molecular dynamics and thermodynamics is understood rigorously enough from the point of view of physics while some issues are still argued upon on the more fundamental level of mathematical physics.
Where molecular dynamics remains useful is in derivation of quantities thermodynamics cannot tell like diffusivity or thermal conductivity. It enters also in the determination of the equations of state (van der Waals equation or virial expansion). These calculation require also Quantum Mechanics. One interesting and difficult issue of that nature is the description of skin layer phenomena in air near the surface of water and in collisions of gas molecules with those in liquid.
All statistical thermodynamics can be derived from the following basic ingredients:
1) a list of free variables (or degrees of freedom)
2) density of states as function of free variables
3) formula that tells the energy of the state in terms of the free variables
4) the principle that the likelihood of each state is proportional to exp(-E/kT)
The most often disregarded ingredient is the density of states. For monatomic ideal gas it’s assumed to be constant when the free variables are the Cartesian components of position and velocity (or momentum) of each molecule. This assumption leads to the Maxwell-Boltzmann distribution and other familiar results.
Adding interaction or further free variables like rotation changes the formula (3) and possibly also (2). What exactly happens for (2) may require totally new postulates in classical statistical mechanics, but Quantum Mechanics is more useful in that. QM tells that rotational states are discrete and evenly spaced. it tells also the complex structure of vibrational and electronic states etc.
Standard thermodynamics describes only equilibrium states. That allows for studying simple enough transitions from one equilibrium state to another, but not any dynamical effects that enter in transition. Isentropic processes are an example that can be studied, but assuming isentropicity implies assuming absence of any dynamical effects. Some simple non-isentropic transitions like free expansion to empty space can also be handled partially, but your example of introducing suddenly gravity probably not. Molecular dynamics does not involve that same limitation, but may be too complex in practice.
===
One more comment on vertical velocity and altitude. As I mentioned in the chapter following the numbered list, the components of position and the components of velocity are independent variables. The position does not enter in standard calculation of the ideal gas, because no energy is related to it. With gravity that changes for the vertical component of position. All components of position enter in a complex way, when molecules interact.
Frank,
There is actually a fairly straightforward experiment which can simulate exactly this. Take a gas in thermal equilibrium and put it inside a gas centrifuge of radius R and bore radius a.
See diagram here.
Now suddenly spin the centrifuge at angular velocity ω The effective gravitational acceleration at a distance z inwards from the rim is
For a gas with specific heat Cp the “lapse rate” would be
I think even a Dyson motor from a vacuum cleaner would be enough to test whether a temperature gradient formed in the centrifuge or not ! For a 10cm radius spinning at 100 cycles/sec I calculate the following temperature profiles
see graph here
My current opinion is that there would indeed be an initial temperature gradient – just like that observed in the vortex tube. However this would quickly decay to isothermal with a remaining barometric pressure gradient. The experiment is so easy that I can’t believe it hasn’t already been done !
Clive,
Results of that kind of experiments are sensitive to the detailed setup. Turning gravity suddenly on is a different case from accelerating a centrifuge even, when it takes place rapidly, because speeding up the centrifuge involves a strong tangential acceleration. Thinking on that I realized that a different centrifuge would do exactly the right experiment.
What would be needed is a centrifuge where the experimental setup is placed in a tube that can rotate around an axis parallel to the axis of the centrifuge, but located at the and of an arm. When the rate of rotation is increased according to an appropriate formula the acceleration perceived by the gas inside the tube is always nearly constant and parallel to the tube.
I wrote this to indicate what kind of details must be considered to make two different experiments correspond closely to each other.
Cyclone type setup is quite different from this and leads probably to very different outcome.
My above explanation may be difficult to follow. This picture may help in that. My proposal differs from that by having longer arms and by having the tubes always horizontal. Initially they are tangential, but during the acceleration turn closer and closer to radial.
Pekka,
I agree that your proposed set up would be even better. You could imagine having 6 horizontal tubes rotating together on 6 radial arms. Then we can fill each tube with a different gas – air, CO2, N2, Ar, H2O etc. and compare results.
In addition the outer surface of each tube in the radial position could be held at a constant temperature to represent the earth’s surface. The inner surface would ideally be transparent to IR with the whole apparatus placed within a vacuum.
Of course all this may not be possible but it would be extremely cool to be able to demonstrate and test the greenhouse effect within a laboratory.
Clive & Pekka: Real world experiments are great, but they can be difficult. I’m interested in the ideal world of kinetic gas theory, where the collisions are between elastic hard spheres, there is no turbulence or radiative energy transfer, the system is perfectly isolated, and the walls have no heat capacity. I think it would be enlightening to see a molecular dynamics simulation of: a) a temperature/pressure/density gradient developing with time after gravity is turned on and b) the temperature gradient gradually dispersing with time.
I would be extremely surprised if a temperature gradient didn’t develop when gravity is turned on. The center of gravity of the gas will fall, converting some PE to KE. I sure looks like PdV work is being done on the gas at the bottom by the weight of the gas above.
Briefly on the experiments.
For the development of physics the most successful approach has been designing experiments that isolate as far as possible individual features or effects, and that can by that be compared with minimal uncertainties with specific equations.
System level experiments where the behavior of a more realistic system is compared with results of complex calculations based on several equations are also important and can reveal errors or gaps in the way the equations are used. Any experiment may do that, but experiments that mimic closely the real world case are typically most interesting.
Doing the kind of experiment discussed by Clive and myself at a level of accuracy that has a significant change of revealing anything new is probably too costly and cumbersome. Even then it would be very unlikely that anything useful would come up. The basic equations have been verified thoroughly enough, while problems of calculation are most likely to be case specific and teach little essential to other cases including the real world situation that has been mimicked.
Frank,
Your thought experiment can be fully solved as combination of evolving adiabatic compression and expansion and the very much slower heat conduction. No molecular dynamics is needed for that calculation.
The situation can be considered in two equivalent ways. One is the original one, where gravity is switched on, the other is considering a tube that is accelerated with constant acceleration in the direction of the tube starting from time = 0.
Initially adiabatic compression starts at one end of the tube while adiabatic expansion starts at the other end. Both these deepen and expand towards a new hydrostatic equilibrium of exponential density profile. Temperature profile is given by the formulas of adiabatic compression/expansion. As heat conduction is extremely slow that can be forgotten in that calculation and considered separately on a totally different time scale.
Pekka,
You are essentially agreeing with Frank that his thought experiment does indeed produce a lapse rate. That lapse rate is essentially -g/Cp ! Yes, without GHG conduction the lapse rate would then eventually decay resulting in isothermal equilibrium.
Your paper on the kinetic gas theory in a gravitational field includes a term with dT/dz. The isothermal case dT/dz = 0 is just one solution. A sufficient condition to maintain a lapse rate would be that IR radiative losses from TOA are greater then heat conduction from the surface.
Clive,
My note is specifically on the case with no radiative heat exchange. Radiative heat exchange with exterior makes the case non-adiabatic (by definition of adiabatic).
This paragraph is really a fastidious one it helps new web visitors, who are
wishing for blogging.
After exploring a few of the blog posts on your blog, I really like your technique of blogging.
I bookmarked it to my bookmark webpage list and will be checking back in the near future.
Take a look at my web site as well and let me know your opinion.
Awesome post.
[…] Temperature Profile in the Atmosphere – The Lapse … – Figure 8. In the tropics the (moist) potential temperature is close to constant with altitude until about 200 mbar. And at other latitudes the potential temperature … […]
[…] Science of Doom. The “Greenhouse” Effect Explained in Simple Terms ; Earth’s Energy Budget ; CO2 – An Insignificant Trace Gas? ; Atmospheric Radiation and the “Greenhouse” Effect ; Back Radiation ; Theory and Experiment – Atmospheric Radiation ; Tropospheric Basics ; Temperature Profile in the Atmosphere – The Lapse Rate […]
[…] Temperature Profile in the Atmosphere – The Lapse Rate … – A few questions, and points of disagreement: 1. Why have GHGs no effect on the dry lapse rate? 2. What would be the effect of increased GHGs on figure 6? […]
[…] Temperature Profile in the Atmosphere – The Lapse Rate … – A few questions, and points of disagreement: 1. Why have GHGs no effect on the dry lapse rate? 2. What would be the effect of increased GHGs on figure 6? […]
I’m with Clive on this… that there would be a lapse rate of 9.8ºC in an atmosphere without radiative gases ( e.g. Sahara desert with a dry lapse rate and therefore no potential temperature increase with altitude ).
Warmed air in contact with hot desert will always be less dense and so rise.
There will always be a density gradient in an atmosphere within a gravitational field.
All that turbulence can do in a non radiative atmosphere is to return cooler air from above back to the surface at the same rate of warming on the descent as
it cooled on the ascent.
Diffusion would very slowly spread the warm air from the tropics polewards but not fast enough to counterbalance the increased heating under the now uninterrupted tropical sun.
Eventually Earth’s AVERAGE effective temperature of -18ºC would be reached at 5km and via the dry lapse to an AVERAGE surface temp. of 32ºC. with any surplus thereafter being radiated straight to space.
Equilibrium would be restored.
At first glance it might seem that a non radiative atmosphere would be hotter at the surface. In reality I would imagine it to be a world of huge tropical to polar gradient, large seasonal differences and big diurnal variation but with the same average .
My take on it all, is that water in all its forms is a moderator of extremes not an amplifier.
Furthermore the 33ºC of surface warming attributed by NASA to (mostly) water vapour is actually 10km up at the tropopause due to the Potential temperature increase of 3.3/Km throughout the troposphere due to the average reduced lapse rate from the dry at 9.8ºC to 6.5ºC. (10 x 3.3 = 33).
The only thing that winds and turbulence can do on a planetary scale is to mix the warmer air above with the cooler to below to become the average throughout resulting in a rise of 16.5ºC. at the surface.*
Whether this extra warming of the atmosphere of 3.3ºC/ km is due to latent heat from a cooling of the surface somewhere else or a direct heating via radiative/thermalisation processes from the interception of direct solar energy to prevent the surface getting as hot, is up for discussion.
*A rise of 16.5ºC from what ? …you may ask.
With the oceans covering 71% of the Earth’s surface, having 1200 times the heat capacity of the atmosphere and being above freezing most of the time, this would provide the base temperature for the planet of above zero.
I don’t think this is right. If there were no greenhouse gases, then the effective radiating altitude would be at the surface, not 5km up. So the surface would be effectively -18C.
This follows from the heat flows; calculate the temperature of the surface when all heat flows in or out of the Earth’s system from there.
I’m with Windchasers on this one. But the average temperature would be lower. That’s because radiance is proportional to the fourth power of temperature. A larger diurnal temperature range for the same total radiated power will give you a lower average temperature. The lower limit for this sort for this sort of thing is a sphere tide locked to the sun. The average temperature for the entire sphere is less than -100 degrees C. Or look at the Earth’s moon.
DeWItt, WIndchasers, Clive and others: FWIW, I had new insight on lapse rate that may be useful: The observed lapse rate is determined by the nature of the mechanism that transports heat the fastest.
1) Adiabatic expansion/contraction during convection produces a lapse rate of -Cp/g WITHOUT enrichment of lighter gases at higher altitude. This mechanisms is fastest in the lower atmosphere.
2) Thermal diffusion by molecular collisions produces an isothermal lapse rate. (As in Feynman’s Lectures). The same sort of thermal diffusion occurs in liquids and solids, where a change in potential energy is negligible. In the lower atmosphere, there is negligible change in potential energy in between collisions. Under normal circumstances, this mechanism (2) would dominate over mechanism 3.
3) Molecular diffusion (conserving the sum of molecular potential and kinetic energy) also produces a lapse rate of -Cp/g, but WITH enrichment of lighter gases at higher altitude. This happens above the turbopause. The rational in Feynman’s Lectures no longer applies because a Boltzmann distribution of molecular speeds no longer exists. (This mechanism is in at least one paper and advocated by the infamous DC and others skeptics.)
When gravity is “turned on” in 2-D molecular dynamics simulations with many molecules in a box, mechanism 2 is faster than mechanism 3 and the temperature gradient is transient. If you put in a few molecules, the opposite is true. Somewhere between is a transition to a system where a stable temperature exists proportional to the average kinetic energy. (IIRC, you can also see a transition in simulations of non-adiabatic expansion into a vacuum, which doesn’t produce cooling when only a few molecules are involved. I don’t understand enough statistical mechanics to explain what is happening.)
I suspect that if you agree what heat transfer process is dominates, it will be easier to agree upon what “should” happen.
Frank,
Molecular diffusion is slow compared to turbulent mixing, sometimes known as eddy diffusion. It’s actually quite hard to have pure molecular diffusion even in a lab setting. In any large system, turbulence will be present and dominate mixing. That’s why gas molecules below the turbopause are well mixed and do not have scale heights based on molecular or atomic weight.
https://en.wikipedia.org/wiki/Eddy_diffusion
DeWitt: Exactly. Both mechanisms 1 and 3 both create the same lapse rate (-Cp/g), but your facts and lack of fractionation explain why the mechanism 3 is not responsible for the lapse rate in the lower atmosphere.
When convection/bulk motion doesn’t occur, mechanism 2 is faster than mechanism 3. I can see it empirically in 2D molecular dynamics simulations. The laws of physics argue for the same result: Feynman Lectures, your arguments based on COE.* However, those laws don’t apply in very thin atmosphere where the concept of a thermodynamically-defined temperature can’t be applied. 2D molecular dynamics simulations also explore this regime.
FWIW, my “insight” is that all three mechanisms are operating at the same time, and the lapse rate we observe is determined by the fastest mechanism. The “correct answer” for “alternative planets” depends on what processes you believe dominate there. For the Earth, the observed lapse rate and the lack of enrichment by MW prove which process is operating fastest in the lower atmosphere.
* I’ve written an outline of a proof that dT/dz = 0 in an isolated column of gas based on the kinetic theory of gases. I have derived
P = ρv2
dP/dz = 2ρv*(dv/dz) + v2*(dρ/dz)
-2ρv*(dv/dz) = v2*(dρ/dz) + ρg
If dv/dz is zero, there is no temperature gradient. So far, equilibrium has not been assumed. I need to apply a constraint that the number of ascending molecules equals the number of descending molecules. That determines the dρ/dz term. Acceleration makes the average velocity of downward molecule slightly faster than upward – until a collision occurs. I can get both zero and non-zero answers for dv/dz and can’t convince myself of the right answer – if there is one. I’d be glad to share it with you, if you are interested. (As you may remember, “the truth shall make you free”.)
Frank,
I don’t know what you mean when you distinguish between thermal diffusion and molecular diffusion. I would say they are the same process.
As DeWitt points out, eddy diffusion produces a uniform mixing ratio and molecular diffusion produces gravitational separation, so that each gas obeys the barometric law individually.
The temperature profile is not a direct result of mixing processes, although it is influenced by mixing. It is the result of where the fluid is heated and cooled. The adiabatic lapse rate is only a limit to the lapse rate. On average, the lapse rate in the troposphere is significantly less than adiabatic even though turbulent mixing dominates everywhere.
You wrote: “Molecular diffusion (conserving the sum of molecular potential and kinetic energy) also produces a lapse rate of -Cp/g … a Boltzmann distribution of molecular speeds no longer exists.”
If you don’t have a Boltzmann distribution, you can’t define temperature, so you can’t define a lapse rate.
A non-radiative atmosphere over an isothermal surface would be isothermal. It would make no difference if the mixing mechanism was molecular diffusion or turbulent diffusion. A non-isothermal surface would induce large scale convection that would create temperature gradients in the atmosphere. I am not sure what the result would look like.
Mike M: In a solid, there is thermal diffusion, without molecular diffusion. In a 2D molecular dynamics simulation of a dense atmosphere, I believe I see kinetic energy diffusing via collisions much faster than molecules diffuse.
http://physics.weber.edu/schroeder/md/InteractiveMD.html
This teaching website allows you to view a realistic molecular dynamics simulation of a gas in a 2D box in the presence or absence of gravity at various temperatures and densities. You can halt a simulation, turn on the gravity, watch the molecules fall, picking up kinetic energy (creating a “temperature gradient”) and almost immediately see the temperature gradient disappear as collisions redistribute kinetic energy – leaving behind only a density gradient. The redistribution seems to be much faster than any molecule moves from the bottom to the top of the box. Thus there is a difference between molecular diffusion and thermal diffusion.
If you put only a few molecules in the simulation, they behave differently. As best I can tell, the difference in behavior depends on whether collisions are frequent enough so that a well-defined temperature and a Boltzmann distribution of molecular speeds exists (especially in the vertical direction). The correct answer may be: it depends. One Earth, enrichment by MW only exists above the turbopause.
You can also simulate expansion into a vacuum by changing the size of the box with different numbers of molecules present. That behavior also SEEMED to depend on the number of molecules. For someone like me (with an empirical orientation, who has forgotten everything he knew about statistical mechanics and whose thermodynamics is rusty), these molecular dynamics simulations provide useful information about systems too complicated to understand from first principles … especially when first principles such as a Boltzmann distribution may not apply. You may prefer theory.
In the lower atmosphere, the potential energy change between collisions is negligible compared with the amount of kinetic energy being exchanged by collision. A Boltzmann distribution of molecular speeds in the vertical direction is a reasonable approximation.
Mike wrote: “If you don’t have a Boltzmann distribution, you can’t define temperature, so you can’t define a lapse rate.”
You are correct, but a group of skeptics believe that Earth’s lapse rate is produced by gravitation. They derive a lapse rate of -Cp/g by assuming the sum of kinetic and potential energy is conserved at different altitudes in the atmosphere. I hate to introduce you to this swamp of misinformation, but you can look up thermogravitation. Clive Best has an intelligent presentation of this material.
Or you can see traditional science here:
https://www.researchgate.net/publication/304347731_Loschmidts_Temperature_Gradient_Paradox_-_A_Quantum_Mechanical_Resolution
Frank,
You wrote: “The redistribution seems to be much faster than any molecule moves from the bottom to the top of the box. Thus there is a difference between molecular diffusion and thermal diffusion.”
As you say, in the condensed phase, heat diffuses much faster than molecules, but in a low pressure gas heat and molecules diffuse at essentially the same rate. The reason is that in the condensed phase, molecules are “trapped” by the surrounding molecules, but energy can still be readily transferred. In a gas at high density (much higher than one atm.), molecular diffusion should slow down relative to diffusion of heat. I think that a 2D simulation would have effectively a much higher density than in 3D, which may contribute to what you see.
You wrote: “If you put only a few molecules in the simulation, they behave differently.”
If I understand the units at the Weber State site, the default box size of 250 corresponds to about 80 nm. That is somewhat less than the mean free path at one atmosphere. So I think that the few molecules case likely corresponds to gases at atmospheric pressure. If there is no energy flow, you should get a Boltzmann distribution even at low pressure, unless the simulation is doing something wonky.
You wrote: “simulate expansion into a vacuum … That behavior also SEEMED to depend on the number of molecules.”
I am not surprised. A Lennard-Jones potential will give non-ideal behavior at high pressure and that will change what happens in a Joule expansion.
You wrote: “simulations provide useful information … You may prefer theory.”
Simulations can certainly be helpful, but they can also be misleading. Simulations are theory. I do prefer first principles.
You wrote: “In the lower atmosphere, … A Boltzmann distribution of molecular speeds in the vertical direction is a reasonable approximation.”
In a non-radiative atmosphere, that should apply at all altitudes. What perturbs the distribution is not gravity, but energy flux.
You wrote: “a group of skeptics believe that Earth’s lapse rate is produced by gravitation”.
I have encountered them and argued with them, to no effect. Try asking them why the deep ocean is cold.
Frank wrote: “The redistribution seems to be much faster than any molecule moves from the bottom to the top of the box. Thus there is a difference between molecular diffusion and thermal diffusion.”
Mike replied: As you say, in the condensed phase, heat diffuses much faster than molecules, but in a low pressure gas heat and molecules diffuse at essentially the same rate. The reason is that in the condensed phase, molecules are “trapped” by the surrounding molecules, but energy can still be readily transferred. In a gas at high density (much higher than one atm.), molecular diffusion should slow down relative to diffusion of heat. I think that a 2D simulation would have effectively a much higher density than in 3D, which may contribute to what you see.
Frank continues: I read the Wikipedia articles on thermal diffusion and molecular diffusion without understanding how to write a formula that calculates the ratio of these two processes (even in one dimension). Does the ratio depend on density?
Collisions temporarily block molecular diffusion in one directions and transfer kinetic energy that same direction. Intuitively the ratio may depend on density.
Frank wrote: “If you put only a few molecules in the simulation, they behave differently.”
Mike replied: If I understand the units at the Weber State site, the default box size of 250 corresponds to about 80 nm. That is somewhat less than the mean free path at one atmosphere. So I think that the few molecules case likely corresponds to gases at atmospheric pressure. If there is no energy flow, you should get a Boltzmann distribution even at low pressure, unless the simulation is doing something wonky.
Frank replies: Real gases expand about 1000-fold (in three dimensions) going from liquid to gas at atmospheric temperature. I’ve usually chosen to do simulations where the molecules fill about 10% of the area, which is modestly higher “density”. Simulated gravity is obviously much stronger than normal. I’ve been forgetting about the Leonnard Jones potential distorting behavior at temperatures where differences in kinetic energy produce different colors. Thank you for pointing this out.
Frank wrote: “simulations provide useful information … You may prefer theory.”
Mike replied: Simulations can certainly be helpful, but they can also be misleading. Simulations are theory. I do prefer first principles.
Frank continued: Since different people have reach different conclusions based on first principles, I think it is useful for anyone to see gravity turned on and a density gradient produced without a temperature gradient. The simulation is applying first principles – Newton’s Laws.
Frank wrote: “In the lower atmosphere, … A Boltzmann distribution of molecular speeds in the vertical direction is a reasonable approximation.”
Mike replied: In a non-radiative atmosphere, that should apply at all altitudes. What perturbs the distribution is not gravity, but energy flux.
Frank continues: I respectfully suggest you may be wrong here. If a gravitational field changes the kinetic energy of widely spaced (non-radiating) gas molecules faster than collisions can re-establish a local thermodynamic equilibrium, then a Boltzmann distribution of molecular speeds no longer exists. In fact, gravity perturbs the Boltzmann distribution for all molecules between collisions at any pressure, but the mean free path in the lower atmosphere is so short that the perturbation is negligible. This perturbation may be relevant above the turbopause and in 2D simulations with few molecules. It is also the first step in the derivation of thermogravity.
Frank wrote: “a group of skeptics believe that Earth’s lapse rate is produced by gravitation”.
Mike wrote: I have encountered them and argued with them, to no effect. Try asking them why the deep ocean is cold.
Frank replies: The links I provided above include a real scientific publication and an open minded blog post. They were chosen to be inoffensive.
The “insight” I expressed above about “Which mechanism is fastest?” is one way for [reasonable] people to resolve their differences. (For example, the deep ocean is cold because the MOC is faster than thermal or molecular diffusion.) Seeing a simulation is another. Recognizing that the acceleration of gravity doesn’t produce a significant change between collisions is another.
Frank wrote: “how to write a formula that calculates the ratio of these two processes (even in one dimension).”
For a low pressure gas, thermal diffusivity = diffusion coefficient, at least to a good approximation. I don’t think there is simple relation for liquids and I am quite sure there is no such relation for solids.
Frank wrote: “Collisions temporarily block molecular diffusion in one directions and transfer kinetic energy that same direction. Intuitively the ratio may depend on density.”
That is correct, once the density is high enough to “trap” a molecule in a particular location. That effect is negligible in a a low pressure gas and is ignored in the corresponding derivations found in physical chemistry textbooks.
Frank wrote: “people have reach different conclusions based on first principles … The simulation is applying first principles”.
Yes, but if people reach different conclusions, at most one is correct. Good simulations start from first principles, but they need to make assumptions and approximations that, along with possible errors, can be hard to spot. Both general reasoning and simulations have their uses.
I wrote: “What perturbs the distribution is not gravity, but energy flux.”
Frank replied: “I respectfully suggest you may be wrong here.”
I am confident that I am not wrong. A non-Boltzmann distribution will result in entropy production. Absent an input of energy, the system will evolve to a Boltzmann distribution. A gravitational field does not provide an input of energy, unless there is net vertical motion.
Frank wrote: “Frank replies: The links I provided above include a real scientific publication and an open minded blog post.”
Yes, and they both dispute the sky-dragon version. The usual derivation is not specific to low pressure gases (unlike the correct derivation of the adiabatic lapse rate) and implies a lapse rate of 77 K/km in solid lead. That would provide a much easier measurement than the centrifuge experiment proposed in the blog post.
Frank wrote: “Which mechanism is fastest?” is one way for [reasonable] people to resolve their differences. (For example, the deep ocean is cold because the MOC is faster than thermal or molecular diffusion.)”
Yes, but that won’t help if people think that thermal diffusion can produce a non-uniform temperature at equilibrium.
Frank wrote: “In the lower atmosphere, … A Boltzmann distribution of molecular speeds in the vertical direction is a reasonable approximation.”
Mike replied: In a non-radiative atmosphere, that should apply at ALL ALTITUDES. [my emphasis] What perturbs the distribution is not gravity, but energy flux.
Frank continued: I respectfully suggest you may be wrong here. If a gravitational field changes the kinetic energy of widely spaced (non-radiating) gas molecules faster than collisions can re-establish a local thermodynamic equilibrium, then a Boltzmann distribution of molecular speeds no longer exists. In fact, gravity perturbs the Boltzmann distribution for all molecules between collisions at any pressure, but the mean free path in the lower atmosphere is so short that the perturbation is negligible. This perturbation may be relevant above the turbopause and in 2D simulations with few molecules. It is also the first step in the derivation of thermogravity.
Mike wrote: I am confident that I am not wrong. A non-Boltzmann distribution will result in entropy production. Absent an input of energy, the system will evolve to a Boltzmann distribution. A gravitational field does not provide an input of energy, unless there is net vertical motion.
Frank now continues: Above the turbopause, gases are fractionated by MW. Doesn’t that reduce entropy? Actually, it doesn’t. The traditional concepts of temperature and entropy don’t apply (or are difficult to apply*) where local thermodynamic equilibrium doesn’t exist. Local thermodynamic equilibrium doesn’t exist in the upper atmosphere, so isn’t it inappropriate to use entropy to justify what happens at “all altitudes”???
(*In the upper atmosphere, some people refer to a “thermodynamic temperature and a blackbody temperature”. Perhaps the concept of entropy also has been extended, but that is not within my capabilities.)
Simple Newtonian mechanics in the real world and in 2D molecular dynamics simulations don’t “know” about the Boltzmann distribution: That is an emergent behavior of large numbers of rapidly colliding gas molecules. (It is probably derived mathematically in statistical mechanics.) If I simulate the behavior of hundreds of molecules in a gravitational field, those in the top half of the box appear to be moving at the same average speed as those in the bottom half. When I put a few molecules in the same simulation, those in the top half of the box appear to be moving more slowly than those in the bottom half. (thermogravitation?) However, slower-moving molecules are not “colder”.
Those who believe in thermogravitation don’t understand this emergent behavior of systems in LTE. Likewise, those who talk about re-emission of absorbed photons don’t understand another consequence of LTE, thermalization. I helps to remember what they don’t understand.
Frank wrote: “Above the turbopause, gases are fractionated by MW. Doesn’t that reduce entropy? Actually, it doesn’t. The traditional concepts of temperature and entropy don’t apply (or are difficult to apply*) where local thermodynamic equilibrium doesn’t exist. Local thermodynamic equilibrium doesn’t exist in the upper atmosphere, so isn’t it inappropriate to use entropy to justify what happens at “all altitudes”???”
At thermodynamic equilibrium, gases fractionate according to molecular weight. That can be sustained with no entropy production and no input of energy. Mixing ratios that are independent of altitude, as observed below about 100 km, is a non-equilibrium situation that results in continuous production of entropy and requires a continuing input of energy (in the form of turbulence, for the atmosphere).
I suspect that the simulation may give misleading results due to the fact that no molecules are crossing the upper boundary in either direction.
LTE does not exist very high in the atmosphere, but that is a result of absorption and emission of radiation (and maybe interaction with the solar wind, if high enough). I see no reason why a hypothetical non-radiating atmosphere would not be in LTE, even at very low pressure. The rate at which equilibrium is restored is irrelevant if nothing is perturbing the equilibrium.
Mike: Thanks for taking the time to discuss this stuff.
You said: I suspect that the simulation may give misleading results due to the fact that no molecules are crossing the upper boundary in either direction.
FWIW, you can turn the gravity up so high, that few molecules reach above the middle of the box and “none” bounce off of top of the box (at modest temperature). So the situation can be much like our atmosphere, with an undefined top.
DeWitt: Axioms are important for mathematics, but science relies on experiments. However, you once discussed a thought experiment involving two identical isolated columns filled with different gases or working fluids. If they spontaneously developed a temperature gradient in a gravitational field, that difference could power a heat engine and perpetual motion. I haven’t forgotten the clearest explanation I have read about this subject.
Nevertheless, there might be some value in showing that the existence of a spontaneous temperature gradient is incompatible with the kinetic theory of gases.
I don’t know if you noticed my comment to DeWitt above about trying to prove that the kinetic theory of gases requires that an isolated column of gas at equilibrium must be isothermal.
P = ρv2 (kinetic theory)
dP/dz = 2ρv*(dv/dz) + v2*(dρ/dz)
dP/dz = -ρg (hydrostatic eq)
-2ρv*(dv/dz) = v2*(dρ/dz) + ρg
The above equation is true at all times. If dv/dz = 0, then dT/dz must(?) be zero. At equilibrium, we have the additional constraint that the net vertical flux at all altitudes must be zero. If dρ/dz is – ρg/v2, I’m home If gravity causes a slight difference in the speed of upward and downward molecules – between collisions
Frank,
Thermodynamic equilibrium, i.e. no temperature gradient, is an axiomatic concept of thermodynamics.
https://en.wikipedia.org/wiki/Thermodynamic_equilibrium
Other intensive properties may not be uniform in the presence of a long range, unchanging force field, gravity e.g. Since this is axiomatic, it’s not subject to proof. Rather, any equation that does not give isothermality is wrong by definition.
Frank wrote: “When I put a few molecules in the same simulation, those in the top half of the box appear to be moving more slowly than those in the bottom half.”
What I see is similar speeds independent of height, but things are a lot “busier” near the bottom, since there are more molecules there. I don’t see an easy way to get enough data for a good statistical analysis. I did a test with 500 molecules in the largest possible box, took an “all atoms” snapshot and did a quick analysis; there seems to be no dependence of kinetic energy on height. As should be the case.
Mike M.,
Kinetic energy ≡ temperature. If it’s isothermal, then the average kinetic energy is the same for all altitudes. The total kinetic energy is lower because there are fewer molecules. But molecules at higher altitude have greater gravitational potential energy. The Virial Theorem comes in here somewhere. There’s a discussion of this in one of the Miskolczi threads.
For a gravitationally bound cloud of gas, a loss of total energy results in a higher temperature, i.e. a negative heat capacity.
MIke wrote: What I see is similar speeds independent of height, but things are a lot “busier” near the bottom, since there are more molecules there. I don’t see an easy way to get enough data for a good statistical analysis. I did a test with 500 molecules in the largest possible box, took an “all atoms” snapshot and did a quick analysis; there seems to be no dependence of kinetic energy on height. As should be the case.
This is what I see too, without a proper analysis of the data. (I think there is a way to export data at any time point.)
If you pause a simulation without gravity, turn on the gravity, and resume, you will see many molecules falling and heating up the bottom of the box and leaving colder molecules at the top. Then collisions quickly diffuse heat throughout the box, leaving a density gradient, but no temperature/kinetic energy gradient. At least, this is what I perceive.
If you use 5 molecules in the simulation, I see something different. When kinetic energy is converted into potential energy faster than collisions can redistribute it, there appears to be no LTE and no Boltzmann distribution. 5 Molecules in a large box isn’t a very realistic scenario, but I think this is a situation where the thermogravitation advocates may be right. And it may be impossible to create a Maxwell’s demon capable of exploiting this situation (if I am correct.). More than a century of experience suggests it can’t be done. But some are trying.
Mike: The other thing I should note is that these calculations are not “perfect”: They involve a time step of finite size, which is adjustable. Just because something happens in a simulation doesn’t prove that it would happen in a more sophisticated calculation. By chance, I encountered a simulation on Youtube that looked very jerky (and probably wrong) from someone promoting Maxwell’s Demons.
DeWitt wrote: “Kinetic energy ≡ temperature. If it’s isothermal, then the average kinetic energy is the same for all altitudes. The total kinetic energy is lower because there are fewer molecules. But molecules at higher altitude have greater gravitational potential energy. The Virial Theorem comes in here somewhere. There’s a discussion of this in one of the Miskolczi threads.”
I think we all agree that isothermal means uniform average kinetic energy. The issue is whether there are conditions for which kinetic energy, and therefore temperature, varies with altitude at equilibrium. I say no, in agreement with you, Feynman, and many others but in disagreement with the sky dragon types. I think that Frank also mostly agrees, but is wondering if there might be extreme conditions where that breaks down and would like to see a kinetic calculation that shows it is so.
I think the Virial Theorem is for an isolated system, so does not apply here. I am not convinced that the negative heat capacity in gravitational bound systems is really a heat capacity since it is unclear to me that such systems are at thermodynamic equilibrium, with a thermodynamic temperature.
Frank wrote: “Just because something happens in a simulation doesn’t prove that it would happen in a more sophisticated calculation.”
That is why I am skeptical of drawing conclusions from a simulation unless they agree with more general arguments.
5 molecules. Hmm. With 1 molecule you will obviously get KE + PE = constant, if I understand what the Weber State simulation does. So there might be a small number effect; thermodynamics requires very large numbers of molecules. That is not the same thing as a number density effect. If it is a small number effect, it will go away with a bigger box and a proportionately bigger number of molecules.
Let’s see … If I get the data for one molecule at many time steps, that should be like a sample from a very large system. Looks like the site lets me do that, but it might take a while.
Mike: For LTE to exist, collisions need to redistribute energy within the local group of molecules faster than energy enters or leaves the group by any other mechanism. When that happens, we can apply the Boltzmann distribution. Under special circumstances, radiation can be absorbed and emitted faster than collisions redistribute it. In a gravitational field, can upward moving molecules become slower-moving and downward moving molecules become faster moving, disruption LTE if collisions are rare.
At 1 atm and 288 K and MW 29 amu and diameter 3E-10 m, average velocity equals 458 m/s and the mfp is 1E-7 (Hyperphysics). The ratio of kinetic energy to potential energy change between collisions:
KE/PE = [mv^2/2] / mgh = 1E11
So, the potential energy change between collisions is about 1E11 times smaller than the kinetic energy that can be exchanged when molecules collide. Obviously this is negligible almost anywhere in the atmosphere. A massive gravitation field is needed to create a density gradient in the tiny box used in 2D simulations. With only 5 molecules in the box, changes in potential energy are faster than energy exchange by collisions,
The thermogravity crowd pays attention to the potential energy change (“molecular diffusion”) and ignores energy transfer by collision (“thermal diffusion”).
All of this would be a picky waste of time, if I didn’t want to apply it to my kinetic theory of gases derivation. There is a density gradient in an isolated column of gas at equilibrium. Also the upward and downward fluxes at any height must be equal. If equilibrium implies isothermal, then the speed of upward-moving molecules at height z must be the same as the speed of downward moving molecules height z+dz. However the density gradient appears to mean that there will be more upward moving molecules than downward moving ones. At least this may be true when the mean free path is bigger than dz. And I think this dilemma is solved by gravitation acceleration of the downward moving molecules by gravity. (:)) On a molecular scale (as opposed to the macroscopic PV=nRT scale), this may be the “origin” of the density gradient.
Frank wrote: “For LTE to exist, collisions need to redistribute energy within the local group of molecules faster than energy enters or leaves the group by any other mechanism.”
That is correct. But for a non-radiative gas, there is no flow of energy. A gravitational field can not induce a flow of energy in an equilibrium system. That is contrary to your argument. If there was a flow of energy, there would be a temperature gradient, as you conclude. If there was a temperature gradient, there would be a flow of energy. In either case, you could use the flow of energy to do work. But you can not get work out of a system at equilibrium.
Frank,
I am getting close to giving up on this. One last try.
For equal mass hard spheres, a collision will, on average, change the energy difference by 1/2. I will use that in the following derivation of the lapse rate according to your theory, but we will see that it is only a detail.
Assume that the average vertical mean free path is L. Molecules lose an average kinetic energy of mgL between collisions while traveling upward. A collision restores, on average, (1/2)mgL. So the average drop in energy per upward step is (1/2)mgL. Collisions in the horizontal plane might make the fraction smaller, but will not change the form. For a vertical distance H, the number of steps, N, is H/L. So over that distance, the change in potential energy is N*(1/2)mgL = (1/2)mgH. The mean free path does not matter.
One can quibble about the details in the above, but those do not change the important result that the mean free path does not matter. That result is what we should expect from general principles: In a system at equilibrium, macroscopic thermodynamic properties, such as temperature, can not depend on microscopic details, such as the size of molecules or even the existence of molecules. So at thermodynamic equilibrium (which means, among other things, no radiative transfer or horizontal temperature gradients), there will be no vertical temperature gradient, no matter how low the pressure.
Mike M: Thank you for sharing your expertise. Please don’t “waste” time on me if you don’t get some reward from it.
You wrote: “Assume that the average vertical mean free path is L. Molecules lose an average kinetic energy of mgL between collisions while traveling upward. A collision restores, on average, (1/2)mgL. So the average drop in energy per upward step is (1/2)mgL. Collisions in the horizontal plane might make the fraction smaller, but will not change the form. For a vertical distance H, the number of steps, N, is H/L. So over that distance, the change in potential energy is N*(1/2)mgL = (1/2)mgH. The mean free path does not matter.”
Almost perfect. The mean free path matters because with each collision kinetic energy is redistributed many orders of magnitude faster than it is converted into potential energy. Thus LTE exists and one can apply all the rules of classical thermodynamics (which you understand far better than I do). However, if a molecule travels far enough in a gravitational field between collisions, LTE will no longer exist. Molecular dynamics simulations allow one to see the transition from a “world” with a few molecules where E = KE + PE, to a “world” of many rapidly colliding molecules where PdV and TdS play critical roles (and changes in PE are negligible).
The advocates of thermogravitation get lost in this transition. They don’t recognize that collisions (thermal diffusion) are redistributing kinetic energy between molecules many orders of magnitude faster than molecular motion (molecular diffusion) interconverts PE and KE. Unfortunately, they calculate a lapse rate of -Cp/g when they ignore heat transfer by collision/thermal diffusion.
(When trying to apply the kinetic theory of gases to molecules in a gravitational field, I’m also struggling with this transition.)
At the beginning of this conversation, I hypothesized that the lapse rate we observe is determined by the process that transfers heat/kinetic energy the FASTEST. In Feynman’s isolated column of gas, transfer of heat by collision/thermal diffusion is many orders magnitude faster than kinetic energy is exchanged with potential energy (by molecular diffusion in a gravitational field). So we should observe no temperature gradient (and fractionation by MW, according to you), at least where LTE exists. In the troposphere, however, bulk convection redistributes heat much faster than thermal diffusion, so we have a DALR of -Cp/g (modified by humidity) and no fractionation by MW. (Similarly, the ocean is not isothermal because other processes are much faster than thermal conduction.) And with GHGs, we need to consider the competition between convective and radiative heat transfer. From my perspective, all modes of heat transfer at competing with each other.
DeWitt and you are applying what DeWitt called axioms that are appropriate in most common circumstances (such as “isothermal at equilibrium”). I’m suggesting you also consider which heat transfer process is fastest. Should there be a temperature gradient on a planet with anon-radiative atmosphere? First, let’s agree on which heat transfer process is operating fastest on this planet.
“First, let’s agree on which heat transfer process is operating fastest on this planet.”
Heat from a surface will use convection if it has the possibility. Right at the surface the energy leaves as emission which has to equal the speed of light, colder air is present nearly always, and it moves away from the hotter surface as soon as it is heated, being replaced by cold air immediately. Radiation combined with convection at the surface is the most effective heat transfer by far. Heat transfer by radiation combined with transfer of heated mass. From twodimensional emission to threedimensional emission, that is a massive difference.
If heat from the sun enters the system as radiation at light speed, and OLR leaving at the same speed, all heat transfer inside the system must equal them both in effectiveness. The surface, as inner boundary, must also emit its heat at the same rate as radiation even though it might be mostly convection
In the application of the physics in heat transfer when used in a furnace or similar device with enclosed combustion, there are good examples for heat, surfaces and gas. If you add a steelplate inside it, you increase the surface area for heat transfer to the gas inside. When the plate has reached equilibrium it radiates according to its temperature and there is more heat transferred. Adding a gas to a surface that reach the same temperature, is similar. You now have both a surface and a volume of gas at the same temperature, and they both transfer heat in any direction where there is lower temperature simoultaneously. Between two bodies of the same temperature, there is no transfer of heat. But the surface below the equal temperature gas doesn’t stop or reduce its emission and it still transfer heat to any point above with lower temperature.
The surface radiates according to it’s internal state and it transfers energy according to the mean troposphere internal state, at the same time in any given moment.
[…] Temperature Profile in the Atmosphere – The Lapse Rate. Science of Doom. […]
My question involves output from MODTRAN Infrared Light in the Atmosphere (MILA).
Google this and choose U.S. Standard Atmosphere, but all other settings default. Notice not the intensity versus wave number graph, but the altitude versus temperature plot to the right. This graph gives the usual structure of the U.S. Standard, i.e. a lapse rate in the Troposphere of ~ 6.4 K per km, and then a constant temperature of 216.7 K in the Tropopause between 11 km and 20 km.
Now leave everything the same and put in a 10 K positive ground temperature offset. Note the change in the shape of Altitude versus Temperature. Where does this shape come from? The same procedure produces even more complex distortions using the other model atmospheres and larger offsets.
The one thing that I found these distortions have in common is that if you go to the point at which the temperature just starts to INCREASE with altitude, that point always stays at the same temperature when you input a temperature offset or a different temperature offset. And other points above the base of the Stratosphere also are unaffected by the offset. The initial lapse rate tends to remain constant, which is consistent with the pressure versus altitude in the Troposphere remaining the same since the relevant version of the barometric formula – the one that does not depend on the absolute Temp value but only the lapse rate – is applicable.
But does anyone have an idea where such shapes in the altitude versus temperature are actually computed? Is there a model someplace?
curiosdp wrote: “But does anyone have an idea where such shapes in the altitude versus temperature are actually computed? Is there a model someplace?”
Do you mean how the U.S. Standard Atmosphere is arrived at? If so, see https://en.wikipedia.org/wiki/U.S._Standard_Atmosphere#References and references and links therein.
Or do you mean how MODTRAN calculates the change in profile when you change surface T? That is probably as simple minded as it looks: A constant lapse rate in the troposphere and no change in the stratosphere. The former is justified by the vertical heat flux being the same, so mixing stays the same if humidity stays the same. More water vapor would reduce the lapse rate. The latter is justified by the fact that the stratosphere is heated mostly from above, so the T profile there is not very dependent on surface temperature.
Mike.M, you wrote: “A constant lapse rate in the troposphere … is justified by the vertical heat flux being the same,”
I’m curious. Given that the temperature profile in the atmosphere has to change if the surface temperature changes, why do you think that a constant vertical heat flux justifies no change in the lapse rate?
willb01 wrote: “I’m curious. Given that the temperature profile in the atmosphere has to change if the surface temperature changes, why do you think that a constant vertical heat flux justifies no change in the lapse rate?”
I would expect a change in gradient to produce a change in heat flux.
Mike M, wrote: “I would expect a change in gradient to produce a change in heat flux.”
I realize you are making this assumption, but I’m curious as to why. It seems to me that the gradient is going to be mostly a function of the heat flux and the surface temperature. If the heat flux is held constant, then the gradient will be determined by the surface temperature. I think you must be assuming something about the surface temperature that’s not clear to me.
willb01,
The gradient is the heat flux when compared to the adiabatic lapse rate. The heat transfer characteristics of the atmosphere aren’t going to change with relatively small changes in temperature. Humidity is a different story because a lot of the heat flux is latent, i.e. involves the evaporation and condensation of water vapor. Changing specific humidity changes the adiabatic lapse rate.
If you change the surface temperature and don’t change the temperature of the atmosphere above it, the heat transfer rate will change.
You need to learn some fundamentals about meteorology. Try this:
http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.180.4815&rep=rep1&type=pdf
DeWitt,
The heat flux has to more or less balance the W/m^2 from the sun absorbed at the surface, does it not? The incoming radiation from the sun is the only significant factor affecting heat flux. How can the gradient alter this?
willb01 wrote: “The heat flux has to more or less balance the W/m^2 from the sun absorbed at the surface, does it not?”
Correct Averaged over the whole surface of the Earth and a reasonably long time, like a year, they balance to within a fraction of a percent.
willb01 wrote: “The incoming radiation from the sun is the only significant factor affecting heat flux. How can the gradient alter this?”
It can’t. Which is why it makes sense that the gradient is fixed by the heat flux, barring some other change, such as absolute humidity.
Note that changing the surface temperature does not change the steady state heat flux. So the gradient does not depend on surface temperature.
Mike M., thanks for the response. I’m still a bit fuzzy on why you make this statement:
“Which is why it makes sense that the gradient is fixed by the heat flux,…”.
Caballero theoretically derives temperature gradient values by moving parcels of air around within the volume of the atmosphere. The implication (at least for me) is that the gradient is dominated by turbulence and convection. If heat flux is playing a significant role wrt the gradient, then I would also expect the thermal conductivity of the air to play a role. No where does Caballero mention thermal conductivity.
DeWitt, you wrote: “The heat transfer characteristics of the atmosphere aren’t going to change with relatively small changes in temperature.”
Doesn’t this depend on what caused the temperature change in the first place? If the change is caused by an increasing GHG concentration, isn’t this exactly what’s happening – a change to the heat transfer characteristics of the atmosphere?
willb01,
Thermal conductivity is orders of magnitude less rapid than turbulent mixing from eddy diffusion. That’s why Cabellero ignores it. The only place thermal conductivity matters is at boundaries. Even then, the thickness of the stagnant boundary layer where heat transfer is only by diffusion will vary with wind speed outside the boundary. The rate of heat transfer by diffusion is inversely proportional to the thickness. A sufficiently large temperature difference across a boundary will cause turbulent mixing.
Not really. Doubling CO2, for example, actually represents a quite small change in the overall radiative balance, on the order of 1%. And we’re not making a step change.
In fact, even mixing is ignored in the basic calculations of lapse rate.
Willb01 asks: Does the gradient control the heat flux or does the heat flux control the gradient? Both. It called radiative-convective equilibrium.
Remember, for the surface and atmosphere to maintain a constant average temperature, the rate at which heat enters and leaves must be the same at altitudes. For simplicity. let’s consider a 1-dimensional model, pretend that no SWR is absorbed by the atmosphere, and that 240 W/m2 (post albedo) arrives at the surface. That means that the upward flux at the surface and at all altitudes would need to be 240 W/m2.
Our current atmosphere is too opaque to thermal infrared to let 240 W/m2 of radiation alone leave the surface. Currently OLR is about 390 W/m2 and DLR is about 333 W/m2, a net radiative cooling of only about 57 W/m2. Simple models predict that a surface temperature of about 350 K would be needed to balance inward and outward fluxes by radiative cooling alone
So, imagine “turning on” the sun and watching surface temperature rise towards 350 K. At a certain surface temperature, the lapse rate becomes unstable. Then the rest of the 240 W/m2 can be removed from the surface by convection instead of radiation – as long as that heat can escape to space fast enough so it does not build up. So, surface temperature is controlled by the maximum stable lapse rate – starting from the average altitude from which photons escaping to space are emitted, aka the characteristic emission level. When the lapse rate is unstable, convection is removing enough heat from the surface to send a total of 240 W/m2 upward no matter how opaque the atmosphere is.
As you move higher in the atmosphere, the density of GHGs (especially water vapor) decreases. More of the 240 W/m2 is transported upward by radiation and less by convection. By the time you reach the tropopause, radiation does the whole job and temperature depends on radiative equilibrium, and no longer the lapse rate.
In the real world, about 1/3 of incoming SWR is absorbed as it passes through the atmosphere. So, an upward flux of only about 160 W/m2 (convection plus radiation) needs to leave the surface. but that figure rises to 240 W/m2 (radiation only) by the tropopause.
When we add more GHGs to the atmosphere, 240 W/m2 no longer escapes from the characteristic emission level, so that level must warm. The lapse rate transmits that warming to the surface. Warming puts more water vapor in the atmosphere, which lowers the lapse rate. So more warming occurs in the upper troposphere than at the surface – a negative lapse rate feedback compared with a model where the lapse rate doesn’t change. In theory, rising humidity reduces the flux to space by about 2 W/m2/K, while increased warming in the atmosphere at higher altitudes (lower lapse rate) increases the flux to space by 1 W/m2/K (where K is the change in surface temperature).
Frank,
Thanks for the lucid and informative response. I would like to ponder it first and then perhaps ask a few questions if you don’t mind. In the meantime I have a comment which I’m hoping you might consider:
In your description, you discuss heat transfer in the form of radiation and convection at the surface. I’m not sure exactly what you mean by ‘the surface’. I tend to think of it as composed only of:
– the physical ground
– vegetation
– liquid water (lakes, rivers, seas, oceans)
– ice (glaciers, ice sheets, ice floating on water).
In your 1-dimensional model, this I believe is the surface absorbing SWR from the sun. Heat transfer across the corresponding surface-atmosphere boundary won’t then include convection. My heat transfer list for this boundary would instead be the following:
– Radiation.
DLR and OLR
– Conduction.
The wind blowing through vegetation and across ground, water and ice
– Phase changes.
Evaporation, condensation, freezing, melting, sublimation
– Mass transfer.
Rain, dust, micro-organisms such as pollen and spores
Net cooling of the surface would be the sum of all these mechanisms.
willb01,
Sure it will. You will have both packets of air warmed by the surface rising because it’s less dense than the air above it and packets of moist air that is also warmer and less dense than the air above it with some of the water vapor condensing to liquid and releasing energy as the air rises and cools. Moist air does not cool as fast when it rises as dry air. No horizontal wind is involved, just air rising and falling.
willb01: The surface is everything that is not atmosphere, but touches the atmosphere. The flux from the surface to the atmosphere is mostly radiation and latent heat. Since I’m trying to present the big picture, I don’t want to go into detail about the small fraction that is not. The atmosphere is even simpler. You can calculate latent heat flux from the average annual rainfall of 1 m (1 m^3/m^2/yr) and guess at a small correction for precipitation that falls as snow or ice rather than liquid water. Radiation transfer calculations are well understood, but tedious. The greatest uncertainty arises from clouds, which I included as part of the atmosphere.
When water vapor is convected upward, that is a heat flux equivalent to radiation. At some altitude, it condenses and falls as precipitation. We measure NET upward convection of latent heat by measuring precipitation. For both LWR radiation and convection, I’m presenting net fluxes. DLR is fairly small compared with OLR in the upper troposphere and the importance of DLR grows as you get lower in the atmosphere. Physics allows us to calculate radiative fluxes, but not convective fluxes. Physics only tells us when the atmosphere is unstable towards buoyancy-driven convection (aka an unstable lapse). However, most of these complications disappear when you realize that radiative-convective equilibrium means that convection must be delivering whatever upward heat flux is needed. (Otherwise the atmosphere will be cooling and the increased lapse rate will increase convection). It also may not be obvious that the upper troposphere loses far more heat by radiation than it gains.
Thank you for saying that my comments were informative and lucid. I usually learn far more here from SOD, DeWItt, Mike M and others than I give back. SOD has discussed all of the subjects I briefly presented far more effectively than I have in the early years of this blog. Those posts are gems, if you have the time to read them. You can find a roadmap to them here:
DeWitt Payne:
No it won’t.
Warmed by the surface, but not via convection.
You are describing heat transfer within the atmosphere and not across the surface-atmosphere boundary.
I agree that convection helps cool the ground surface by constantly moving heated air upwards, away from the surface, and allowing cooler air to replace it. But heat transfer across the atmosphere-surface boundary is via conduction and radiation, not convection.
I probably shouldn’t have mentioned wind in relation to conduction. What I was trying to convey was the concept of air coming in contact with the surface, resulting in heat transfer due to that contact.
willb01 wrote: “Heat transfer across the corresponding surface-atmosphere boundary won’t then include convection.”
That is correct, since right at the surface there can not be any air motion. As you get closer to the surface, convection gets less and less important. Close enough to the surface convection becomes small compared to conduction (i.e., molecular diffusion). That “diffusive layer” is typically only of the order of 1 mm thick. The slow rate of diffusive (conductive) transport across that layer can be important for mass and momentum transfer, but does not seem to be a significant limitation for heat transfer. The reason, I think, is that there is also radiative and latent heat transfer across the diffusive layer.
willb01 wrote: “My heat transfer list for this boundary would instead be the following:
– Radiation.
DLR and OLR
– Conduction.
The wind blowing through vegetation and across ground, water and ice
– Phase changes.
Evaporation, condensation, freezing, melting, sublimation
– Mass transfer.
Rain, dust, micro-organisms such as pollen and spores
Net cooling of the surface would be the sum of all these mechanisms.”
That is the right idea, but it needs a bit of tweaking. Conduction is due to the random motion of molecules; wind is part of convective heat transfer. What you call “phase changes” is normally called “latent heat transfer”. The heat transfer is not due to the phase change itself, but is due to the mass transfer of a substance (water vapor) that undergo a phase change. I don’t think that mass transfer represents any significant heat transfer other than latent heat transfer.
Willb01: SOD discusses heat transfer by conduction at the link below. The flux per unit area is proportional to the temperature difference and inversely proportional to the distance the heat travels solely by conduction. Conduction can transfer a 1000-fold bigger flux across 1 mm compared with 1 m for a given temperature difference. The hot sand on a beach on a sunny day (or a football field with artificial turf in a stadium) illustrates how large the temperature difference can grow when turbulent winds (convection) don’t reach down to the surface and thermal diffusion must transfer heat much further away from the surface than normal. The difference between these surfaces and nearby grass illustrates the large role vegetation and transpiration.
Fortunately, over land “surface temperature” is defined as air temperature 2 m above the ground. And it is always measured when sheltered from sunlight. So the above complications can be ignored.
Turbulent mixing is also critical for transporting water vapor away from a thin layer of air over the ocean that is saturated with water vapor. The rate of evaporation is proportional to wind speed and “undersaturation” of the air near the surface.
As I pointed out above, radiative-convective equilibrium ensures that when too little heat is being transferred upwards and the surface gets too warm, the lapse rate becomes unstable and convection removes the necessary amount of heat. This is true as long as heat escapes fast enough from the upper troposphere to space by radiation. In the tropics, convection must transfer heat nearly twice as high as in the temperate zone before it can escape fast enough to space as radiation, thus the tropopause is higher there.
For me – but not many others – it helps to think about Venus where radiative cooling is totally ineffective through much of the atmosphere. (DLR differs little from OLR). The “troposphere” or convective region with a constant lapse rate (about 10 K/km due to lack of humidity) is about 50 km thick before the atmosphere is thin enough to let to permit adequate radiative cooling to space. Thus the surface is about 450 K warmer than on Earth, where the convective region is only about 10 km thick. If you object to this over-simplified explanation (and many do), let’s not argue and just worry about Earth.
Curious: The other profiles might be more satisfying than the US Standard Atmosphere. I suspect they arose from the work of Myhre et al (or something similar) that came up with the 3.7 W/m2 accepted values for the forcing from 2X CO2. Myhre started with re-analysis data and created atmospheric profiles from different regions of the globe in difference seasons. You may or may not find more sophisticated temperature off-sets associated with these profiles.
The atmosphere temperature is only changed up to the 10km level. Everything above that is held constant. Any large change in temperature will be a crude approximation as a result. A surface temperature change should have an effect on pressure vs altitude as well, i.e. the atmosphere should expand or contract with increase or decrease in temperature. It doesn’t. MILA is a very simple model.