The subject of atmospheric heating rates is one which is worth spending time on.
What is a heating rate?
To see the usefulness of a heating rate let’s consider the per capita income of a country.
Per capita income compares the ratio of total $ to the total population. If we compare the total income of China to the total income of Laos we don’t have a useful comparison. If we compare the per capita income of China to the per capita income of Laos.. well, who knows whether we have a meaningful comparison – but at least we have something more useful. Something more relevant.
Energy absorbed in a layer of the atmosphere causes heating at a certain rate. Energy lost from a layer of the atmosphere causes cooling at a certain rate.
Heating rates tell us something different from total energy lost or gained. Suppose a 1m layer of the atmosphere gains 1,000 J/m², what will the temperature change be?
The specific heat capacity of the atmosphere at constant pressure is 1005 J/(K.kg) – which means it takes just over 1,000 J to lift the temperature of 1 kg of the atmosphere by 1K (=1°C).
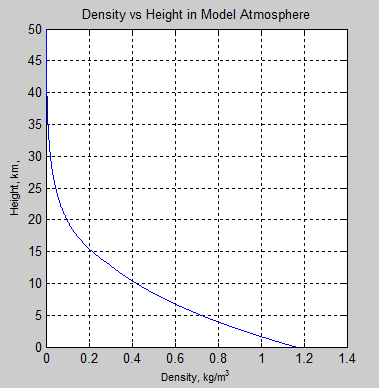
However, the atmospheric density decreases with height:
Figure 1
At the surface, where pressure = 1000 mbar, the density = 1.2 kg/m³.
So 1,000 J/m² lifts the temperature of a 1m layer of the atmosphere at the surface by 0.83 K (calculated by ΔT=1,000/[1.2 x 1005]) .
At the top of the stratosphere, near 50km where the pressure = 1 mbar, the density = 0.0016 kg/m³.
Here, 1,000 J/m² lifts the temperature of a 1 m layer of the atmosphere by 620 K (calculated by ΔT=1,000/[0.0016 x 1005]).
So it’s a bit like sharing out the total income of China among the residents of Laos.
That’s why heating rates are useful – they relate the amount of energy with the amount of atmosphere.
More CO2 equals More Absorption but More CO2 equals More Emission
One of the confusing aspects in atmospheric radiation comes as people start to consider the fact that the atmosphere emits as well as absorbs.
So more radiatively-active gases (=”greenhouse” gases) causes more absorption? Or more emission? Or doesn’t one just balance out the other and so there is no change?
There are legitimate questions to ask.
The only way to answer these questions is to solve the Schwarzschild equation – see Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Six – The Equations.
What we will do is first of all review the heating/cooling rates vs altitude and try and understand some features qualititively.
Here is the right way to think about the problem:
Absorption at any given wavelength depends on:
- the quantity of gases that absorb at that wavelength
- the effectiveness of each gas at absorbing at that wavelength
- the “amount” of radiation travelling through that part of the atmosphere (note 1).
Emission at any wavelength depends on:
- the quantity of gases that absorb at that wavelength (and therefore emit at the same wavelength)
- the effectiveness of each gas at absorbing (and therefore emitting) at that wavelength
- the temperature of the gas
In the case of shortwave (=solar radiation) the atmosphere absorbs but does not emit. This is because the atmosphere is not hot enough to radiate significantly below 4 μm, see The Sun and Max Planck Agree – Part Two.
In the case of longwave (= terrestrial / atmospheric radiation) absorption is from radiation from above and below. But usually the radiation from below is a lot higher than from above. This is because the earth’s surface emits close to blackbody radiation (the surface has a very high emissivity over all wavelengths), and the atmosphere (which doesn’t emit as a blackbody) is hotter closer to the surface.
Doesn’t a Heating or Cooling Rate Mean that the Atmosphere is Heating up or Cooling Down?
No.
The sun heats the atmosphere (a heating rate), but the atmosphere radiates to space (a cooling rate), and also convection moves heat through the troposphere.
We can still have a heating rate, a cooling rate and convective heat transfer while the atmosphere is in approximate energy balance (=not changing in temperature). If the temperature of one part of the atmosphere is not changing then these will sum to zero.
So heating rates vs height give us insight into the strength of these effects, and we can break the effects up between the responsible gases (water vapor, CO2, ozone, etc).
Heating Rates
From the always excellent Grant Petty, A First Course in Atmospheric Radiation, the solar heating of the atmosphere, for a standardized tropical atmosphere:
Figure 2 – Solar heating
If we showed total energy absorbed at each height in the atmosphere, then the troposphere would overwhelm the stratosphere (upper atmosphere). But because we are showing energy absorbed in proportion to the density of the atmosphere, the upper atmosphere appears more important.
We see that ozone causes the highest heating rate in the stratosphere, whereas water vapor causes the highest heating rate in the troposphere, and CO2 has a very small effect.
The water vapor heating rate is – of course – concentrated in the bottom few km of the atmosphere because water vapor is concentrated here.
Most of the absorbed solar radiation is absorbed by the earth’s surface. The surface absorption is not shown in this graph. In turn, the surface heats the atmosphere primarily through convection. The convective heat transfer is also not shown.
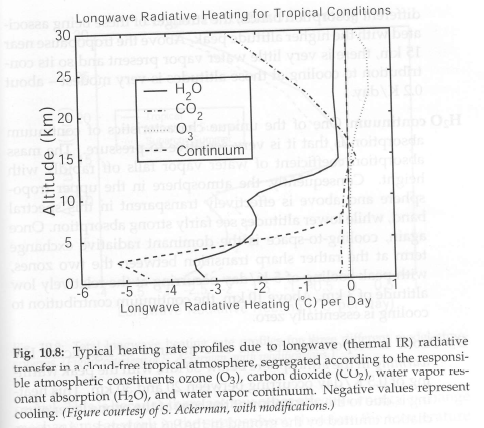
Now let’s look at longwave heating (cooling) rates for a few different regions:
Figure 3
We see that the heating rates are mostly negative, meaning that these are really cooling rates. Most of the atmosphere is cooling via longwave radiation. However, one small part of the atmosphere experiences a heating rate due to longwave radiation – the tropical tropopause.
Why?
The tropopause is the coldest part of the atmosphere – the top of the troposphere and bottom of the stratosphere. And the coldest part of the atmosphere radiates less than it absorbs.
Let’s see the breakdown of cooling rates by individual gases:
Figure 4
We see that water vapor has a peak longwave cooling at around 3 km and another maximum at 10 km. The lower peak is caused by the “continuum” – also shown separately on the graph – which we will return to shortly.
Ozone shows a heating rate in the stratosphere below 30km. If we had the graph extend up to the top of the stratosphere, around 50km, we would see ozone with a cooling to space higher up.
We also see that CO2 has a very small cooling effect until we get into the stratosphere. Generally, each layer experiences a very small heating/cooling effect from CO2 because CO2 has a such a strong absorption that energy is absorbed from layers very close by – which are at very similar temperatures. The tropopause is the coldest part of the atmosphere so absorbs a little more radiation via CO2 than it emits – consequently a small heating effect.
As we get up into the stratosphere we see a progressively stronger cooling to space from CO2. In part, this is because of the reduction in pressure broadening at lower pressures = higher altitudes (see Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Nine). This effect causes the absorptivity of CO2 to reduce at higher altitudes meaning that the radiation from the layer below can “get through” to space.
The Continuum
Water vapor has an unusual absorption profile. In the so-called “atmospheric window” of 8-12 μm where there are no strong absorption bands from any atmospheric molecule (apart from ozone at 9.6 μm), water vapor still absorbs. Here the absorption coefficient is proportional to the atmospheric pressure.
In general, the more we have of a particular gas, the more absorption. This is expressed in the Beer-Lambert law.
But for other gases, the absorption coefficient is a constant for a given wavelength – not proportional to pressure. In the Beer-Lambert law we multiply the mass of absorbing molecules in the path by the absorption coefficient to find the optical thickness (note 2).
For the water vapor continuum the absorption coefficient is proportional to pressure. And absorptivity is a function of the absorption coefficient and the total mass in the path (which is proportional to pressure). Therefore, total absorption in the continuum band is a very strong function of pressure.
Water vapor concentration is concentrated at lower levels in the atmosphere so the total absorption due to the water vapor continuum falls off very quickly with height.
This is why the lower peak cooling rate occurs. The absorption by water vapor due to the continuum above 3 km is very small – so around 3km the atmosphere (in these wavelengths) can very effectively cool to space. Other bands of water vapor absorb more strongly, so effective cooling to space doesn’t really begin until the concentration of water vapor drops to very low values. Hence the second peak at 10 km.
Stratospheric Cooling
A long time we had a look at Stratospheric Cooling. This strange phenonemon is expected from more CO2 in the atmosphere. All other things being equal, the troposphere will warm and the stratosphere will cool.
Radiative-convective models predict this. Once you’ve got to grips with basic radiation in the atmosphere, it is easy to see why the troposphere will warm.
But why will the stratosphere cool?
Some will look at Figure 4 and say “ah ha“. More CO2 will move the CO2 line over the left and so that’s why the stratosphere will cool.
As a cautionary note, the heating rate at level z is equal to:
Figure 5
Where among other terms, the italicized “T” is the band-averaged transmittance between z and z’, and the integrals are (obviously to the mathematicians) for each “level” (note 3) between the surface and z, or between the top of atmosphere (∞) and z..
If we went through this equation we would find that there are competing terms – terms which represent absorption of energy from other parts of the atmosphere (heating), and terms which represent emission of energy from this layer (cooling). Increasing CO2 increases absorption in the stratosphere. Increasing CO2 increases emission from the stratosphere.
Given that radiative-convective models predict stratospheric cooling we can say confidently that more CO2 will move the cooling curve in figure 4 to the left in the stratosphere (note 4). So emission will be higher than absorption.
However, we haven’t developed an intuitive understanding of why. At least, I haven’t.
To develop an intuitive understanding I would need the solution of these equations for a variety of conditions, and after playing around with changed parameters and reviewing results it would all start to make sense. That’s what I would hope.
But that’s just me. Others can perhaps just see it all dance out of the equations in a flash (think – the crazy one in The Hangover in the casino). Or out of the fundamental physics.
Conclusion
Heating rates help give insight into how the atmosphere absorbs and emits radiation from different “greenhouse” gases at different levels.
Generally the peak cooling rates for each band occur when that band “thins out” enough in the layers above to allow significant radiation to space, rather than just to the level immediately above.
Convection is the most important mechanism for moving heat in the troposphere (but not the stratosphere).
This article hasn’t considered convection at all – which just demonstrates the ongoing plot to hide the importance of convection. Once people realize how important convection is, radiative heating and radiative cooling to space will be.. the same.
Other articles in the series:
Part One – a bit of a re-introduction to the subject.
Part Two – introducing a simple model, with molecules pH2O and pCO2 to demonstrate some basic effects in the atmosphere. This part – absorption only.
Part Three – the simple model extended to emission and absorption, showing what a difference an emitting atmosphere makes. Also very easy to see that the “IPCC logarithmic graph” is not at odds with the Beer-Lambert law.
Part Four – the effect of changing lapse rates (atmospheric temperature profile) and of overlapping the pH2O and pCO2 bands. Why surface radiation is not a mirror image of top of atmosphere radiation.
Part Five – a bit of a wrap up so far as well as an explanation of how the stratospheric temperature profile can affect “saturation”
Part Six – The Equations – the equations of radiative transfer including the plane parallel assumption and it’s nothing to do with blackbodies
Part Seven – changing the shape of the pCO2 band to see how it affects “saturation” – the wings of the band pick up the slack, in a manner of speaking
Part Eight – interesting actual absorption values of CO2 in the atmosphere from Grant Petty’s book
Part Nine – calculations of CO2 transmittance vs wavelength in the atmosphere using the 300,000 absorption lines from the HITRAN database
Part Ten – spectral measurements of radiation from the surface looking up, and from 20km up looking down, in a variety of locations, along with explanations of the characteristics
Part Eleven – Heating Rates – the heating and cooling effect of different “greenhouse” gases at different heights in the atmosphere
Part Twelve – The Curve of Growth – how absorptance increases as path length (or mass of molecules in the path) increases, and how much effect is from the “far wings” of the individual CO2 lines compared with the weaker CO2 lines
And Also –
Theory and Experiment – Atmospheric Radiation – real values of total flux and spectra compared with the theory.
Notes
Note 1 – Absorptivity is a different parameter from absorption. Absorptivity is the proportion of radiation absorbed and is dependent on the number of molecules of different radiatively-active gases. Absorption is the total amount of energy absorbed and so depends on the intensity of radiation passing through that part of the atmosphere and the absorptivity.
Note 2 – The Beer-Lambert law can be expressed in a number of different ways. Essentially the units for the amount of the gas (e.g. number of molecules, mass) in the radiation path has to match the units for the absorption coefficient. The same result is obtained.
Note 3 – There are no discrete “layers” in the atmosphere. This is a convenient term for explaining the physics in plainer English (as with many other inexact and non-formal explanations). All the properties of the atmosphere we are considering have continuous change with pressure and, therefore, with height.
Note 4 – The derivation of the equations for heating rates comes from the same equations which are used in radiative-convective models.


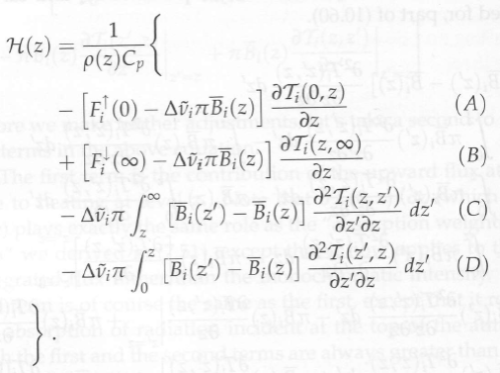


While I enjoy reading your postings, and find them
educational, as I have said
before in my remarks regarding the work of Miskolczi
and the work of Ramanathan, it seems that you
are somewhat missing the big picture, thermodynamics.
Overwhelming, IMHO, all of the details of gas properties,
spectral characteristics, convection flows, etc. is the
unavoidable necessity that the downward radiation
be nearly twice the net solar flux absorbed by the
earth’s surface.
Earth’s climate is driven by heat flows not by the
details of gas properties.
There simply is not enough CO2 in earth’s atmospheric
system for CO2 to significantly change the homeostasis established by the 2nd Law, the solar flux, and by the fact that earth is a water planet. Water vapor will, driven by
thermodynamics, automatically make any very small
adjustments in its total atmospheric presence, and,
in its distribution within the atmosphere,
that may be needed to nearly cancel
the small changes in CO2 that are man caused.
For man made CO2 to have anything close to the
effects that the IPCC has attributed to it, it is
necessary to introduce ad hoc “Positive Feedback
Factors” that are highly unlikely in view of the
body of data and in view of what General System Theory,
and Chaos Theory, have to say about feedback loops
in complex systems.
It seems to me that the starting point for even minded
and objective science is more or less this:
8%, roughly, of the GHE is due to atmospheric CO2
4%, roughly, of atmospheric CO2 is due to man
33 deg. C, roughly, is the total GHE
therefore,
0.1 deg. C, 33x.04x.08, is the approx. agw
This is right in line with the 0.24 deg. C that
Miskolczi gets using his much more complete
and thorough thermodyamics based
analysis.
For stratosphere temperature behavior, changes in ozone have had a much larger effect than changes in CO2. The stratosphere temperature has been nearly constant since about 1995, UAH LS for example. Not coincidentally, that’s when chlorine levels peaked in the stratosphere. Chlorine has been declining slowly since then implying no further reduction or possibly even an increase of ozone. Stratosphere ozone data is very noisy so it’s hard to tell. It’s rather complicated as the rate of ozone destruction increases as temperature decreases for a given chlorine concentration.
james kennedy,
So many errors, so little space. I’ll pick just one:
Wrong. All (to a good approximation) of the increase in CO2 from the pre-industrial level of ~280 ppmv is anthropogenic. That’s 110 ppmv out of 390 ppmv or 28%. Try going here:
http://www.ferdinand-engelbeen.be/klimaat/co2_measurements.html
Miskolczi ignores clouds in his analysis. His calculation of τ is simply wrong.
1. re portion of atmospheric co2 due to man
a. see, for example
b. see, for example, segalstad’s direct measurements
c. Think, for a minute, about the completely specious
and fallacious line of reasoning found in the reference
you gave. It amounts to little more than
asserting that correlation is causation.
Iif the yearly co2 emissions are only 4 % man
caused, if direct measurements of man caused
CO2 agree with my value of 4%, and if the CO2 retention time, by many independent determinations, is around 5 years,
how can the levels
claimed in your reference be remotely possible?
The atmosphere has only a very small ability
to distinguish man caused CO2 emissions from natural
emissions. That being the case, and the retention
times for CO2 being quite short, it is not logically
possible that the accumulation of emissions is
comprised differently than are the emissions
themselves. Think a bit, please.
Your reference amounts to little more than the
simple minded argument that correlation is
causation.
2. Re Miscolczi remark
IMHO, your comments indicate that you have not personally read Miscolczi’s papers. I have.
IMHO, as I understand Miscolczi’s work from direct
personal study, the influence of clouds can only make
Miscolczi’s case stronger not weaker.
IMHO, you do not seem to understand Miscolczi well enough
to express personal opinions on his work.
Read Miscolczi. Think a bit, please.
james kennedy:
If you want to discuss something in the article, please pick a point to start, rather than your sweeping statements.
This is a science blog. Claims require evidence.
You make claims that have been falsified for over 50 years:
Do you disagree with the Schwarzschild equation? This is the equation which allows us to calculate radiative transfer in the atmosphere.
If you are approaching the subject from a scientific point of you will no doubt be well aware of this equation, of its derivation and have some insight into the flaws that no one else has seen, or have some alternative approach to solving the equation.
The alternative would be that you haven’t come to this from a scientific perspective and are just writing whatever seems “right”.
I await with breathless anticipation for verification of your claim, starting with the correct equations of radiative transfer that have eluded everyone from Schwarzschild & Chandrasekhar (Nobel prize winner).
james kennedy:
No one is interested in your opinion. You need to back it up with work.
Perhaps DeWitt Payne does understand the subject better than you. Perhaps you should ask how he reached his conclusion.
That would be an interesting approach. However, it would require you to apply yourself to learning rather than expressing opinions. Expressing opinions is a lot easier but not of interest in this blog.
Please have a read of About this Blog:
james kennedy,
Your 4% figure is out of date. In 2005, combined emissions from fossil fuel burning, cement production and land use changes was 9.4 GtC. If the annual flux between the ocean and biosphere is 120 GtC, then human contribution to the total flux is ~8% and rising. If anthropogenic carbon is such a low fraction of total carbon and the residence time is short, then how do you explain the reduction of the 13C/12C ratio since pre-industrial times? You clearly don’t understand the difference between residence time for a single molecule of CO2 in the atmosphere and residence time in the total surface reservoir consisting of the atmosphere, biosphere and ocean.
I have read Miskolczi (you could at least try to spell his name correctly). You can have a large change in surface temperature with a change in τ that is well within the experimental error of his measurements. (See this comment ) His theory does not, in fact, rule out an increase in surface temperature of 3C from doubling atmospheric CO2. There’s also at least one critical step in the derivation of his theory that’s missing. No one has ever explained how he gets from the ratio of potential to kinetic energy of molecules in the atmosphere to the various ratios of absorption to emission. Miskolczi’s τ is a mathematical construct that has no physical meaning.
Fair enough. You advocate playing by the rules
I follow. Had you actually read what I wrote, you would have
seen this. Instead, you seemed to only
see attacks on your beliefs.
A bare minimum for conducting
a scientific blog is to stay on point, and to follow the
long standing rules for dialectics.
I will spell out what I mean when I use the word
“dialectics”:
A dialectic consists of an exchange of
questions and answers. Ideally, questions
are “well-posed” in that they have “Yes”/”No”
answers. The questions and
answers are intended to form a connected
progression towards
better understandings of the subjects at hand
and hopefully result in resolving differences.
I will demonstrate, using just part of your
last writing.
1. To wit,
you say
”
If you want to discuss something in the article, please pick a point to start, rather than your sweeping statements.
”
I clearly picked a point to start. I even went to the
trouble of explicitly so identifying it with a
“1.”
”
1. re portion of atmospheric co2 due to man….
”
I ask then, a well-posed question:
“Yes” or “No”
Did I pick and indicate a point to start?
2. To wit,
You say,
”
This is a science blog. Claims require evidence
”
I gave the evidence for my value of 5%
man caused yearly emissions.
I repeat it for your convenience, check it out:
I ask then a well-posed question
“Yes” or “No”
Did I give evidence for my “5% assertion”?
I am really at a loss to understand why, instead
of replying to the content of what I wrote
you chose to go off the emotional deep end. No need
in science for such. I stayed with the science.
You diverged from it.
I would much prefer that you be able to stay on
scientific point. Questions such as how much
atmospheric CO2 is man made can be resolved
solely by looking at the data. They are not to
be decided by you refereeing fights between
appeals to authority.
For me to feel you are capable of evenmindedness,
you need to reply to the two well-posed questions above.
I have gone to considerable trouble to make them as
relevant to your accusations as I can. I think, if you
can calm down, you will see that they are in fact
relevant. Each of those only need one word, a “yes” , or,
a “no”.
Did I pick and indicate a point to start?
Did I give evidence for my “5% assertion”?
james kennedy:
This article is nothing to do with the anthropogenic proportion of CO2 in the atmosphere. And makes no claims about it.
I picked up a claim that you made which has at least some relevance to this article.
This article is no 11 in the series about radiative transfer in the atmosphere.
If your only substantive claim is the anthropogenic proportion of CO2 then maybe I might find interest in the totally off-topic discussion or maybe not.
If you want to discuss your claim about radiative transfer then please explain where the Schwarzschild equation is wrong, or how everyone else has miscalculated the effect of CO2 using the correct equation. You can see it explained in detail in Part Six of this series.
This would be “backing up your claim”.
If you don’t want to back up your claim:
– or need time to develop it then please say so.
If you make one specific point with alleged support and 10 sweeping statements with no support don’t start complaining when people pick on the 10 with no support.
If you want people to only stay with one specific point then my advice is just to claim that one specific point.
If it is on-topic for the article you will get to discuss it. If it is off-topic for the article who knows.
OK fine. I will take your lead.
You said
”
If you don’t want to back up your claim:
There simply is not enough CO2 in earth’s atmospheric system for CO2 to significantly change the homeostasis established by the 2nd Law, the solar flux, and by the fact that earth is a water planet.
…………. then please say so.”
Fine. I will take your lead.
First, lets get rid of your that Petitio Principii
Fallacy, and change
“If you don’t want to back up your claim:”
to
“If you want to back up your claim:”
Then, I can back up my claim as follows:
My original email gave the basic and most simple argument against CO2 being a primary cause of global warming. I will give it again
agw < approx. ( .04 X .08 X 33 ) deg. C
agw < approx. (portion CO2 man caused) X
(portion GHE CO2 caused) X
(total GHE)
That's it. I know it seems absurdly simple, but the
reality is that you must show that one of those three
numbers is way off if you are going to show that
CO2 is a major cause of agw.
There can, of course, be debate about those three numbers.
I assert, however, that it is very unlikely that any of
them are so way off that they change the structure
of my argument.
33 deg. C, as I am sure you know, comes directly from
basic radiation physics and is quite solid. It would be
surprising to see T eff. 34.
Would it not?
.08 is open to a lot of debate. Having accumulated
many, many papers on the contribution of CO2 to GW,
I can cite references down to below .05 and up to around
.25. I would say that .08 is good enough to frame
a discussion about the magnitude of the influence
of CO2.
Would you not?
.04 is less widely contentious. I would say that
.05 is probably a bit better. Maybe we could go as
high as .08. I would say that anything in that
.04 to .08 range is compatible with the structure
of my argument.
Would you not?
So there you are. That’s all there is to it.
To make the case that CO2 is a primary factor in
agw, you must show that at least one of the three
numbers, .04, .08, or 33, is way off. Your radiation transfer details, such as the “Schwarzchild Equation”, in order to overrule the thermodynamic reality of “.04X.08X33”, must be part of such a showing; in order for the “Schwarzchild
Equation” to be relevant to CO2 caused GW, it must
be part of such a showing.
Must it not?
o.
james kennedy:
The equations of radiative transfer are not linear.
Ascribing total proportions of the inappropriately-named “greenhouse” effect to CO2 is not so useful, although it might be interesting for some.
What is more interesting is the change in radiative forcing from pre-industrial levels of CO2 to possible future levels of CO2.
All other things being equal, an increase of CO2 from the pre-industrial level of 280ppm to a future value of 560ppm, provides a radiative forcing of almost 4 W/m2.
See CO2 – An Insignificant Trace Gas? Part Seven – The Boring Numbers for the definition of radiative forcing.
This is a pre-feedback value and with no feedbacks and no other changes would be expected to cause a surface temperature increase of 1’C.
The well-proven equations of radiative transfer along with well-proven spectroscopic data on CO2 demonstrate this, and need to be overturned for the result to be changed.
This would be a mountain to climb.
In essence I am saying that the 2nd number in your calculation is inadequate to reach any useful conclusion, even if it was correct.
As to the first number, your main claim, I have never researched it.
And for clarity, your statement in your latest comment:
– should probably read “..against anthropogenic CO2” as you have currently reduced your claim to this.
If you claim that increasing CO2 will have an insignificant impact on the “greenhouse” effect then you will need to demonstrate why the radiative forcing value of 4W/m2 is incorrect. (You can’t do that by a simple multiplication).
Or, of course, demonstrate negative feedbacks for the resulting temperature change, which is a totally different subject.
Great. Good catch. I had noticed the same thing
about my original wording being less explicit than
it should have been. It is confusing. My excuse can only be that the discourse on CO2 around the drunken neighborhood
poker game has come to “CO2” in lieu of
“anthropogenic CO2”. Also, compared to a spoken “CO2”, a spoken “anthropogenic CO2” is something of a tongue twister.
Also, I have also grown tired of the spoken “anthropogenic”
because I almost always have to explain what
“anthropogenic” means. Never mind all that, however, I stand corrected.
To be clear of unnecessary future confusions lets use
acronyms such as
“CO2” => “CO2”
“TACO2” => “total atmospheric CO2”
“ACO2” => “anthropogenic CO2”
“TAACO2″=> “total atmospheric anthropogenic CO2”
etc.
We are talking here, then, about ACO2, and TAACO2,
unless I clearly indicate otherwise.
Having agreed that 4 W/m2 is a right enough number,
can I still assert that AGW due to TAACO2 is insignificant?
“Yes”. “Yes, I think so.”
I’ll get back to you on that.
james kennedy,
If temperature were an extensive thermodynamic state variable, your equation might make some sense. But it’s not. As a result, your equation is nonsense and discussing the relative values of the coefficients is also nonsense.
In reply to your question, I come up with, approx.
0.2 deg. C
Which is to say, I calculate that, if atmospheric CO2
doubles, if such a doubling gives us a total net CO2 forcing
of about 4 W/m2, and if the man-caused portion of that
forcing is about 5%, then, the AGW resulting from
the doubling of atmospheric CO2 will be around
0.2 deg. C.
That is my answer. I realize that it is way, way
lower than many of the numbers that have been
bruited about in the media, both popular and
“scientific”. But….. that is my answer.
I expect more than “Pro Hominen” challenges to
my answer.
I expect more than “Pro Hominem” challenges
to that .05 figure which is a fair mid range of the
many values cited in the litereature for the contribution of anthropogenic CO2 to total atmospheric CO2.
“Yes” or “No”, please
Have I understood your question?
“Yes” or “no” please
Have I answered it?
“Yes” or “No” please
Do you agree with the value given?
“Yes” or “No” please
If you do not agree, can you, without solely
using appeals to various types of authority, explain your own reasons for disagreeing?
If you choose to continue…………
Here’s how I get this .2 deg. C value…………..
Firstly,
The 4 w/m2 value looks to be about right as it
concords with the estimate that the present
forcing value is about 2.5 and the increment
due to doubling CO2 will be about 1.5:
4 approx. = 2.5 approx. + 1.5 approx.
Secondly, how does one convert a forcing value
to a temperature change? While, as is
so often the case in climate science, this is not
a settled matter, we can come up with some
pretty good estimates. I use two:
Estimate 1. .2 deg.K
use 1 K/W/m2
.2 deg. K = 4 W/m2 X 1 K/W/m2 X .05
with the 1 deg. K/W/m2 figure coming from
http://www.physicsforums.com/showthread.php?t=307685&page=5
with the “.05 ” being a midrange of the values cited
for the portion of atmospheric CO2 that is man caused
Estimate 2. .03 deg. K
(I am not comfortable with this estimate in a number of
regards, but it serves as a starting point. My intuition
tells me that it gives too small a result. OTOH, if it
were to turn out to wrong by more than a factor of four
I would be surprised. It should be in the ball park
of being ls th the .2 above.)
…….. use the forcing value as a simple equivalent
to solar input to get, approx., in deg. C,
.03 = ([ (239 + 4)/(239)^.25 ] – 1) X 288 X .05
with 239 being the total outgoing global LWR
288 being the average global temp. deg. K
.05 being mid range of the values cited for
the portion of CO2 that is man caused
You will see, I hope, that estimate 1 and estimate 2
are both compatible with my assertions that man caused CO2 is not a major player in current GW.
Furthermore you see, I hope, that they accord pretty well with what I started out with as a calculation, not using the forcing value of CO2,
.15 deg. C (approx.)
( i.e., .13 deg. C = 33 X .08 X .05 )
Using your 4 w/m2 value for the CO2 forcing,
after atmospheric CO2 has doubled,
I get
about .2 deg. C
as the contribution of anthropogenic CO2.
A separate email elaborates.
james kennedy,
So the doubling of atmospheric CO2 happens by magic? Either the increase in atmospheric CO2 concentration from the pre-industrial level is anthropogenic or it isn’t. If it isn’t , what is the mechanism? Note that the mechanism must also explain why this has never happened during the last 4 or 5 glacial cycles and also causes a decrease in the 13C/12C isotope ratio and a decrease in atmospheric oxygen concentration that closely matches the amounts calculated from converting 13C depleted fossil fuel to CO2 and H2O.
The long wave chart says it is a model are all the charts just models and no real atmospheric readings.
De Witt:
”
So the doubling of atmospheric CO2 happens by magic? Either the increase in atmospheric CO2 concentration from the pre-industrial level is anthropogenic or it isn’t. If it isn’t , what is the mechanism?
”
warming of the earth, especially of the oceans.
Check it out: CO2 lags temperature.
Temperature drives CO2 more than CO2
drives temperature. e.g., the recent rise in CO2 follows the
temperature spike in the Medieval Warm Period
by about 800 years. This value of the amount
CO2 lags temperature, plus or minus a couple of
hundred years, shows up consistently
over data sets of widely ranging time spans.
No magic needed.
Hopefully having learned something in the course
of these various exchanges, I think I can re-do
that last email in a better format where I make it
clearer which voice is being heard at any given time.
De Witt:
”
So the doubling of atmospheric CO2 happens by magic? Either the increase in atmospheric CO2 concentration from the pre-industrial level is anthropogenic or it isn’t. If it isn’t , what is the mechanism?
”
James Kennedy:
”
My answer 1. is
‘ warming of the earth, especially of the oceans, is
the mechanism.
No need for magic.’
.
Here is a really basic citation supporting my answer:
Click to access Natural_Climate_Change.pdf
and, here are some more:
http://www.john-daly.com/oceanco2/oceanco2.htm
Check it out: CO2 lags temperature.
This has to mean that temperature drives CO2 more than CO2
drives temperature.
No magic needed.
.
.
.
My answer 2:
No mechanism needs to be given. All we need to
see is that the data over more than the last hundred
years has consistently shown that the flux of man
caused CO2 is on the order of 5% compared to the total flux
of atmospheric CO2.
e.g. we have
‘…Our atmosphere contains 730 billion tons of carbon as CO2. Each year about 120billion tonnes of carbon are cycled via plants on land and 90billion tonnes via oceans. Human emissions account for about seven billion to 10billion tonnes, or less than 5 per cent, of the annual CO2 flux…’
from
http://www.truenews.org/Global_Warming/climate_change_science_not_settled.html
Imagine that flux A, “FA”, from hose A, and flux B, “FB”, from hose B are both going into a leaky tank.
Call the portion of water accumulated in the tank
that originated from flux A, “AA”.
Call the portion of water accumulated in the tank
that originated from flux B, “AB”.
Now, try to imagine that
AA/AB = FA/FB
is not true.
Neither magic or mechanism needed.
“So the doubling of atmospheric CO2 happens by magic? Either the increase in atmospheric CO2 concentration from the pre-industrial level is anthropogenic or it isn’t. ”
DeWitt Payne. Why so dogmatic? Why the argument from ignorance?
Do you really comprehend every last nook and crannie of the earth system so well that you can say anthropogenic CO2 is the only source of the observed increase?
How about facile oxidation of methane released from clathrate as a candidate? I’m not saying it is a factor, rather I’m illustrating that the system is insufficiently closed to support the inducive argument.
It’s hardly an argument from ignorance. It’s Ockham’s razor. We know for a fact that anthropogenic emissions are about twice as large as the observed increase in atmospheric carbon, 475 Pg carbon emitted and an increase in atmospheric CO2 of 210 PgC. So the simplest explanation is that it’s anthropogenic with the excess being absorbed by the ocean and the biosphere. We know a lot about the kinetics of CO2 absorption from the inadvertent 14C isotope injection from atmospheric bomb tests. The behavior of the atmospheric CO2 concentration is explained fairly well, not perfectly, by those kinetics.
If you invoke another mechanism like clathrate oxidation, you have to explain why it never happened in the recent (geologically speaking) past, and why it’s happening now. And explain why the system is suddenly capable of absorbing massive amounts of carbon so that CO2 hasn’t increased even more rapidly.
Clathrate oxidation has been invoked as a mechanism that keeps the atmospheric CO2 concentration from going to zero. But a sudden increase in oxidation isn’t likely as you would have to have a large increase in deep ocean oxygen concentration or a large increase in temperature or both. There is no evidence this is happening. Not only that, but a large increase in temperature is more likely to cause clathrate decomposition and an injection of methane, not CO2. The carbon isotope injection at the PETM is thought to have been caused by clathrates, but the global average temperature was a lot higher then as was the temperature of the deep ocean.
DeWiit, Thanks for the response. What you say warms the pragmatist in me, but affronts the purist :-). As an engineer, Occam’s Razor is a guiding principle, and because of it, me a mere engineer, never would have considered designing, say, the human blood pressure regulatory system the way it is…… complex systems defy reduction to the simple. I believe the same is true of the climate system.
I agree we can estimate the initial impact of the anthropogenic CO2 load on the atmospheric composition. We know from the physical chemistry of CO2 that it will dissolve and eventually equilibrate with bicarbonate anion in an aqueous solution in contact with it. We also know what happens to this equilibrium if the Na+, H+, Ca++, Cl- concentrations alter. And we know what happens if the temperature and pressure of the system changes.
What else do we know with the same degree of sureity as the above? I’d say not very much. We know that, air temperature, sea surface temperature, atmospheric pressure, sea salinity, and pH all vary geospatially. We know that the distribution of all these parameters is subject to chaotic, quasi-oscillatory, spatio-temporal behaviour. We infer from palaeo studies (getting uncertain now?) that some of the quasi time constants of these processes are very long (millenia?) compared with the modern instrumentation period.
And then, given this chaotic system, ever-weaving about its infinity of stationary states, what can we do other than woefully subsample our parameters of choice, derive spatio/temporal averages (what is the physical meaning of the average temperature of the ocean?), and proceed as if the numbers we derived were from a setup within the laboratory.
I’m not saying that the pursuit of such data and it’s analysis is a waste of time. Far from it. What I am saying is that the simplist explanation doesn’t derive additional authority from being just that. It is a candidate hypothesis, no more, no less.
Thus far I have seen nothing that disproves say “90% of the increase in atmospheric CO2 over the 20th century is anthropogenic”. However, to invert this proposition is to deny the complexity of the system with which we are dealing. To assert the inversion simply because no alternative hypothesis presents itself is indeed an argument from ignorance.
And clathrates….. my hunch is that their impact would be episodic but small due to their being thinly distributed. Having said that I hasten to add that I’m speaking from the point of view that I find it very cool that such substances exist, but don’t really have a clue as to how they might figure in the complexity of the carbon cycle.
Finally, do you know any papers or text books that you consider make a good job of analysing the movement of CO2 between atmosphere and ocean? Thanks.
David Reeve,
I recently purchased The Carbon Cycle edited by Wigley and Schimel. I haven’t had a chance to read much yet, but it seems very thorough.
I also have a post at The Air Vent on carbon emission and atmospheric CO2 concentration:
james kennedy,
That 120 GtC is an annual cyclical flux into and out of the biosphere and oceans. The flux from fossil fuel burning and cement production is new carbon added to the system. When you add something to a system, its concentration goes up. That 120 GtC flux causes only about a 6 ppm change in atmospheric CO2 concentration over the course of a year at MLO.
Ocean temperature has increased nowhere near enough in the last 160 years to explain an increase of 100 ppmv CO2 in the atmosphere. That’s the magnitude of the change from glacial to interglacial conditions, or about a 5 C change in global average temperature. Not only that, but the time constant for evolution of CO2 from the ocean is on the order of 2500 years, which is why CO2 lags temperature as temperature increases from glacial to interglacial.
If it were the ocean, then why don’t we see large CO2 increases for the Roman Warm Period or the Medieval Warm Period. Answer, it isn’t the ocean. It’s us.
For those wanting to find out why many climate scientists believe that CO2 increases are due to human activity I recommend checking the explanations, and then the references, in the IPCC report.
For example, the IPCC 4th report (2007), Chapter 2, p.139:
And then I would recommend reading the first citation, which is the first 50 pages of Chapter 3 of the Third Assessment Report of the IPCC, The Carbon Cycle and Atmospheric Carbon Dioxide
IPCC reports are freely available from ipcc.ch.
Many papers can be found in Google Scholar.
Finding a few starting points is the challenge, as there are thousands of papers on measurements of CO2, and the papers which form basis for determining anthropogenic CO2 (e.g., measurement of isotopic ratios) will date back many decades = hard to find unless you know what you are looking for.
This is why I recommend reading the free IPCC chapter to get a better understanding of the subject. You will also find many references. Pick some old ones and search for them in Google Scholar to see if they are free.
Thanks SOD and DeWitt Payne for the pointers, you’ve motivated me to do some exploring.
An interesting post. SOD wrote about stratospheric cooling by increasing CO2: “However, we haven’t developed an intuitive understanding of why. At least, I haven’t.”
Before SOD was kind enough to introduce the Schwartzschild equation, I developed an intuitive way to think about this problem that may have some value. Imagine two optically thin layers of air of thickness, S: one at lower altitude H1, higher pressure P1 and higher temperature T1 containing n1 GHG molecules with a temperature- and pressure-dependent absorption cross-section of o1; and a second layer at a higher altitude with H2, P2, T2, n2 and o2. What is the energy flux between these layers at a single wavelength? Once emitted, a photon has the same probability (z) of getting from layer 1 to layer 2 or traveling the reverse path – it has to pass through the same number and type of absorbing molecules on either journey.
The flux at layer 2 from layer 1 (I_12) is:
I_12 = zn1o1B(T1)S
and absorption A2 of this incident energy by layer 2 is:
A2 = n2o2*I_12*S = zn1n2o1o2B(T1)*S^2.
For the reverse journey, the absorbed flux is
A1 = zn1n2o1o2B(T2)*S^2
Everything is the same except the B(T) terms! Fortunately, this agrees with the 2LoT, which says that net energy flows from hot to cold and there is no net energy flux without a temperature difference. (It also explains why I insisted that you can’t have net radiant energy transfer through an isothermal, optically thick slab.)
What happens when we double the mixing ratio of a well-mixed GHG? Both n1 and n2 double, but the probability of a photon making the journey, z, drops (to z^2?). So the energy transfer over short distances (z>0.5?) increases with doubled GHG and drops over longer distances (z<0.5?). Doubling GHG concentration quadrupoles the rate of energy transfer, but shortens the distance over which it occurs – which must also shrink the temperature difference.
So far we have only considered the energy flux at a single wavelength. Integrating over all wavelengths, the total flux behaves like the flux at a single wavelength when the temperature difference is small enough that the emission spectrum doesn't shift much going from T1 to T2. This is a reasonable approximation inside the earth's atmosphere.
To predict the temperature change at a particular altitude produced by increasing the GHG mixing ratio, we need to consider where the photons absorbed by the GHG at that particular altitude originated. If the average source location is cooler, that altitude is losing more energy through LWR than it gains. Where increasing GHGs increase the rate of energy loss by radiation, then that altitude will be cooled by increasing GHGs (as long as the average photon – now traveling a shorter path – still originates from a cooler location.) It is easy to see why the stratosphere MIGHT be cooled by increasing CO2; it has cooler air above and below. As long as most of the LWR photons the stratosphere absorbs come from these cooler altitudes, increasing GHGs will cause cooling.
This type of analysis predicts that radiative energy flux in optically thick atmospheres such as Venus or the interior of the sun should be really slow: When photons travel only short distances, there will be negligible temperature difference between the average emitting and absorbing location and negligible NET vertical flux.
What does this predict for the troposphere? At any given altitude, there are more photons arriving from below (where it is warmer) than above (where it is cooler). So I think increasing the rate of energy flux via increasing GHGs should lead to warming.
Without specifying the mean free path of photons and how temperature and therefore B(T) change with altitude, there is a strong element of hand-waving above. However, I think I've got the correct factors: net radiative transfer is driven by differences in T through the B(T) term and the rate photon flow quadrupoles when the mediator of energy transfer, GHGs, doubles. The distance over which the photons travel shrinks, shrinking the difference in T and B(T).
How do we know where the average photon absorbed at a particular altitude was emitted? That's a problem. It's hard to say anything useful about the mean free path of photons since it varies so dramatically with wavelength and altitude. Roy Spencer discusses using an IR thermometer to measure the effective temperature of the sky, but I'm not sure how this device calculates temperature from IR. Perhaps measurements of this type tell us the temperature at which the average LWR photon reaching the device was emitted.
http://www.drroyspencer.com/2010/08/help-back-radiation-has-invaded-my-backyard/
This is obviously quite an interesting blog. However A major factor seems overloked entirely. The question of net global photosynthesis and net global respiration by plants is liklely to have an overiding contribution to the atmospheric CO2 and O2 balance. Before green plants evolved there was supposedly a CO2 rich atmosphere and very litly oxygen. The Q10 of photosynthesis and of respiration are very different. As temperatures rise beyond the photosynthtic optimum or indeed range then the balsmnce shifts ( as it does!) towards CO2. In this manner temperature change leads CO2 change (as it does?). Water vapour likely far more important. Yes a leafy world is “wet” and the evapotranspirasive area from gross leaf area far greater than from plain land or ocean surfaceI. So anthropogenic effects there may be through forest destruction and land use changes that push more precipitation striang back through excellive run-off to ses an oceans. I suggest a reading, caregully of Marco Schmidt’s contibution to the RIO climate change conference. I believe he is largely correct. Colin Lekey ( Visiting Professor of Biology, University of Lincoln UK)r
sorry for obvious typos in above, Easy to read through them I hope. Colin L
Looks like a water problem to me. Our science concerning water is zero. Its here, its great, has some really challenging molecular attachments, so whats next.
AGNs make water. The sun may be burning water., just an idea. Actually I think our sun wants our H2O to be in solid form, less disturbance. Water on the quantum level has been described as a solid crystal, the shape holds but the participants come and go, like square dancers. Water may control the shape of the galaxies, the spirals.
Water does not have a raindrop shape, it falls as round balls. Strangely enough, the first produce of our AGN were the globular clusters. Wouldn’t it be really cool if they are all waterworlds?
You state “This article hasn’t considered convection at all – which just demonstrates the ongoing plot to hide the importance of convection. Once people realize how important convection is, radiative heating and radiative cooling to space will be.. the same.”
Q.1: Please explain how “radiative heating and radiative cooling to space will be.. the same.”
Q. 2 Please explain how O2 and N2 cool
Q. 3 Would the process you describe still allow O2 and N2 to cool if they were the only molecules in the whole atmosphere?
Q. 4 Please point me to where the cooling of O2 and N2 is taken into account in the models used by IPCC..
Q. 5 Referring to NASA 2008 Energy Budget http://earth-climate.com/calculations.jpg
In this net diagram the energy from the surface is split:
53.2% evaporation
10.6% convection
36.2% radiation
Now, when back radiation is added the ground / ocean is warmed more and so more heat comes back out again as it cools. That heat is no different from the original heat, so why is the split so different in the second diagram?
17.0% evaporation
3.4% convection
79.6% radiation.
Doug Cotton:
You just have to read the article.
I said “Absorption at any given wavelength depends on:
-the quantity of gases that absorb at that wavelength
-the effectiveness of each gas at absorbing at that wavelength
-the “amount” of radiation travelling through that part of the atmosphere (note 1).
Emission at any wavelength depends on:
-the quantity of gases that absorb at that wavelength (and therefore emit at the same wavelength)
-the effectiveness of each gas at absorbing (and therefore emitting) at that wavelength the temperature of the gas”
If you think that absorption and emission to space are dependent on other factors why not explain them?
Why don’t you explain what you believe on this subject? I’m not very good at guessing what it is you believe. You ask a vague question. If you ask a precise question I will have a stab at explaining it. If you make a precise claim I can evaluate it.
You have seen my comment following your similar vague claims in another article.
That link references some graphics on your website, not a NASA explanation. I am not a mind-reader. So I have no idea.
[…] is a graphic reproduced from Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Eleven – Heating Rate…: From Petty […]
[…] The atmosphere is not actually transparent to solar radiation and you can find similar graphs of Net Shortwave Heating per Day in many climate science textbooks and papers. The humid lower atmosphere gets a strong solar heating via water vapor. See Atmospheric Radiation and the “Greenhouse” Effect – Part Eleven – Heating Rates. […]
Nice article. I wonder why the water vapor cool has a net cooling effect. However, it absorbs long-wave radiation at many wavelengths except the atmospheric window (8 and 12 um, which cools the atmosphere by allowing the radiation to escape to space). Does the cooling by the atmospheric window outweigh the heating by other infrared bands?
Ahmed4kernel: Your question suggests some confusion. All GHGs both absorb and emit infrared radiation, but the “atmospheric window” constitutes the wavelengths where GHGs don’t significantly absorb or emit. At those wavelengths, thermal IR (LWR) emitted by the surface shines unobstructed through the atmosphere to space, thereby cooling the surface (and there is no downward radiation from the atmosphere warming the surface at these wavelengths.)
At all other wavelengths heating or cooling is the net result of absorption minus emission of radiation. The amount of emission depends on the temperature of the GHG as well as how much GHG is present and its emission cross-section. The amount of absorption depends on the intensity of the radiation passing through the gas, as well as how much GHG is present and its absorption cross-section. (In troposphere and stratosphere where collisions redistribute energy faster than absorption and emission and “local thermodynamic equilibrium” exists, the absorption and emission cross-sections are equal.)
However, the situation is more complicated in the troposphere because the local temperature (that controls emission) depends on vertical convection of heat and not just net radiation (of heat). Whenever, the temperature in the atmosphere drops too rapidly with altitude (the “lapse rate”), the atmosphere is unstable to buoyancy-driven convection of heat and warmer less dense air rises from below (and expands and cools). So, whenever you see negative LWR heating rates (cooling rates), the same amount of heat is being convected upwards to maintain a stable average local temperature.
Above the tropopause, the local temperature is that at which absorption and emission are equal – radiative equilibrium. Below the tropopause, the local temperature is saids to be determined by “radiative-convective” equilibrium, which typically produces a lapse rate of 6.5 K/km (which would be -6.5 K/km except the lapse rate is negative by definition).
Thanks a lot for your reply,
[1] Not sure if the longwave heating rate curves shown above in figures 2 and 3 were based only on the radiative equilibrium (without accounting for the convection currents in the troposphere).
[2] The absorption curve of the water vapor shows stronger absorptivity over a wide range of infrared wavelengths compared to the other GHG (see figure 1 https://www.sciencedirect.com/science/article/pii/S2405844018327415). Yet, strong absorption of LW also means stronger emission of LW, hence the greenhouse effect of the water vapor.
[3] Cooling longwave radiation implies that the net emission of the gases is larger than the net absorption.
Ahmed4kernel asked: [1] Not sure if the longwave heating rate curves shown above in figures 2 and 3 were based only on the radiative equilibrium (without accounting for the convection currents in the troposphere).
Our atmosphere is so opaque to thermal IR (except at window wavelengths) that all of the SWR delivered to the surface can’t escape to space via the thermal IR that the surface emits. The surface warms the nearby air, which expands and rises because it is less dense. However, a risen “parcel” of air has less pressure from the weight of the air above, so it expands and cools. However, if temperature drops fast enough with altitude, the expanded air will still be warmer that the surrounding air (even after cooling from expansion) and that “parcel” of air will continue rising. This is what happens in a thunderstorm. The atmosphere is unstable to such buoyancy-driven vertical convection when the temperature falls 9.8 degC per km when the air is totally dry and about 6.5 degC per km when air is moist and heat is released by condensation of water vapor. SWR delivers an average of about 160 W/m2 of heat to the surface and about 100 W/m2 is removed by vertical convection and 60 W/m2 by thermal IR (mostly through the atmospheric window directly to space). As the atmosphere gets thinner and drier with altitude, a larger fraction of the outward flux of heat can be carried by radiation and less is carried by convection. Convection is no longer needed above the tropopause. In the stratosphere, radiative equilibrium exists, meaning that local temperature is that needed so that emission equals absorption (including absorption of SWR by ozone). Below the tropopause, local temperature falls roughly 6.5 degC/km rise because buoyancy-driven upward convection of heat develops when the “negative LWR heating rates” in these figures cause the temperature to become too low. The negative LWR heating rates are a measure of how much heat needs to be convected from below to maintain a stable local temperature in an atmosphere on the edge of being unstable to buoyancy-driven convection.
We know when the atmosphere is unstable to buoyancy-driven vertical convection, but we can’t calculate how much heat is moved by such convection. However, we can calculate the amount of heat transmitted by radiation GIVEN the temperature and composition of the atmosphere. That is done with the Schwarzschild’s equation, which tells us how much radiation is absorbed and emitted as radiation passes through a layer of atmosphere. Usually only the components of the flux perpendicular to the surface (up and down) need to be considered, since the net horizontal flux is zero. The Schwarzschild equation is a differential that must be numerically integrated from a starting place (the surface for upward flux and the edge of space for downward flux) to an endpoint. This is what is done for all wavelengths by programs such as Modtran:
http://climatemodels.uchicago.edu/modtran/
Using Modtran, I can calculate the change in outward and inward fluxes between 10 and 11 km through a clear tropical atmosphere. The upward flux of LWR is reduced from 317.1 W/m2 at 10 km to 312.4 W/m2 at 11 km by all GHGs (more absorption than emission). The downward flux increases from 30.8 to 44.6 W/m2 traveling from 11 km to 10 km (more emission than absorption in this direction). That means that this layer of atmosphere is losing a net 9.1 W/m2 of LWR heat that on the average is being provided by vertical convection of heat to maintain a constant temperature. Using heat capacity and density, W/m2 can be converted to a temperature change in units of degC/day. If I want, I can deal with just one GHG, say water vapor. However, since absorption bands partially overlap, the effects are not additive.
The output of radiative transfer calculations can be very non-intuitive, but the following may help. Blackbody radiation is radiation where absorption and emission are in equilibrium at the local temperature. Planck’s Law was derived by making this assumption. However, in our atmosphere, the density of absorbing GHGs and their absorption cross-sections are not big enough to produce such an equilibrium everywhere in the atmosphere. Schwarzschild’s equation tell us is how fast the intensity of radiation approaches blackbody intensity (equilibrium between absorption and emission) as it travels through our atmosphere. At 1 km (294 degK), the upward and downward fluxes for all but the atmospheric window are nearly the same (equilibrium, blackbody intensity), but this is only true for the most strongly absorbed/emitted band from CO2 (about 666 cm-1) by the time you reach 10 km (237 degK).
Finally, LWR heating rates must be negative in the atmosphere because the atmosphere is getting rid of the 240 W/m2 of SWR that our planet is absorbing (and not reflecting).