In Part One I said:
In the next part we will consider more advanced aspects of this subject.
However, although that is where we are heading, first I want to look at a very simple model and progressively increase its complexity. It may be illuminating.
The discussions on the Radiative Transfer Equations at the very informative blog of Judith Curry’s were very interesting. Many comments showed an appreciation for the very basics with a misunderstanding of how they fit together, for example:
IPCC declares that infrared absorption is proportional to the logarithm of GHG concentration. It is not. A logarithm might be fit to the actual curve over a small region, but it is not valid for calculations much beyond that region like IPCC’s projections. The physics governing gas absorption is the Beer-Lambert Law, which IPCC never mentions nor uses. The Beer-Lambert Law provides saturation as the gas concentration increases. IPCC’s logarithmic relation never saturates, but quickly gets silly, going out of bounds as it begins its growth to infinity.
There have also been many questions asked at this blog about the various aspects of radiation and atmosphere, so I thought some examples – via a model – might be beneficial.
The Model
The main purpose of this model is to demonstrate the effect of the basic absorption and emission processes on top of atmosphere fluxes. It is not designed to work out what troubles may lie ahead for the climate.
This model is designed to demonstrate some basic ideas about what are known as the radiative transfer equations – how radiation is absorbed and emitted as it travels from the surface to the top of atmosphere (and also from the top of atmosphere to the surface).
I aim to bridge a gap between the “blackbody shell” models and the line-by-line climate models – in a way that people can understand. Ambitious aims.
The advantage of this model should be that we can try different ideas. And the code is available for inspection.
Absorption and Beer-Lambert
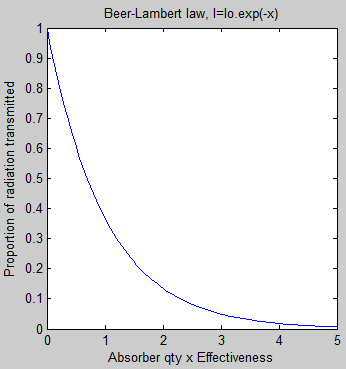
The equation of absorption is known as the Beer-Lambert law – you can see more about it in CO2 – An Insignificant Trace Gas? Part Three.
In essence the Beer-Lambert law demonstrates that absorption of radiation of any wavelength is dependent on the amount of the molecule in the path of the radiation & how effective it is at absorbing at that wavelength (note 1):
Figure 1 – Beer-Lambert Law
Simple Model – v0.2 – Saturation
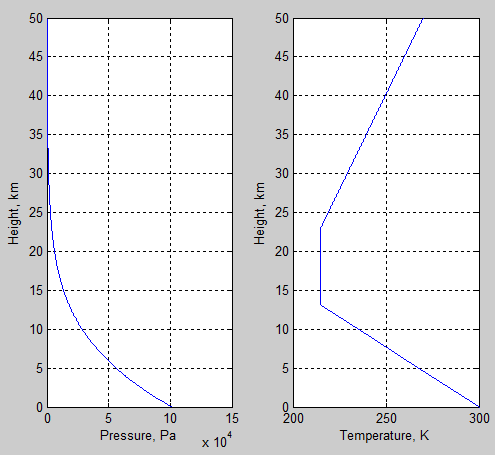
The simple model (v0.2) starts by creating an atmosphere. First of all, atmospheric temperatures, “created”, by definition. Second, using the temperatures plus the hydrostatic equation to calculate the pressure vs height. Third, the density (not shown) is derived.
The surface temperature is 300K.
Figure 2
In this first version of the model (v0.2), we introduce two radiatively-active molecules (=”greenhouse” gases):
- pH2O – pretend H2O
- pCO2 – pretend CO2
They have some vague relationship with the real molecules of similar names, but the model objective is simplicity, so of course, these molecules aren’t as complex as the real-life water vapor and CO2. And their absorption characteristics are in no way intended to be the same as the real molecules.
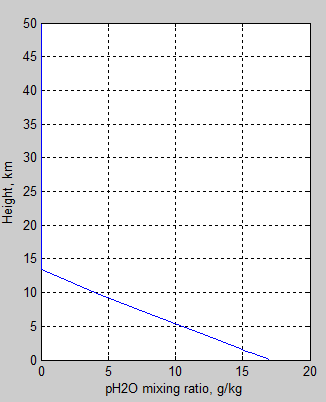
The mixing ratio of pH2O:
Figure 3 – water vapor mixing ratio
The pCO2 mixing ratio is constant with height, but the ppm value is varied from model run to model run.
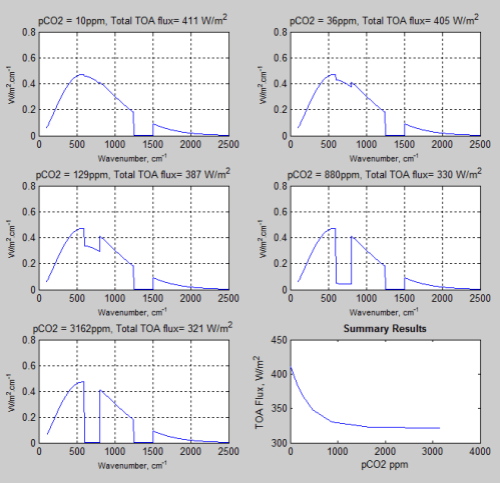
So onto our top of atmosphere results. I’ll refer to “top of atmosphere” as TOA from now on. These results are at 20km, a fairly arbitrary choice.
The results are shown against wavenumber, which is more usual in spectroscopy, but less easy to visualize than wavelength. Wavenumber = 10,000/wavelength (when wavelength is in μm and wavenumber is in cm-1.
- 15 μm = 667 cm-1
- 10 μm = 1,000 cm-1
- 4 μm = 2,500 cm-1
The “notch” that starts to appear around 600-700 cm-1 is from pCO2 as we increase its concentration. The effect of pH2O is held constant. You can see the effect of pH2O as the “notch” around wavenumbers 1250-1500.
Note that the final graph summarizes the results – TOA Flux vs pCO2 concentration:
Figure 4
What we see is that eventually we reach a point where more pCO2 has no more effect – saturation!
We see that as the concentration keeps on increasing we get a reduction in the TOA flux down to a “saturated” value.
And when we plot the Summary Results we get something that looks very like the Beer-Lambert law of absorption! Nothing like the IPCC result shown in CO2 – An Insignificant Trace Gas? Part Seven – The Boring Numbers:
Figure 5
So, case closed then?
Flaws in the Model
This model is very simplistic. Most importantly, it only includes absorption of radiation and does not include emission.
Simplifications:
- no emission from any layers in the atmosphere
- no dependence of line width on pressure and temperature
- no weaker “wings” of an absorption band
- no overlapping of absorption (absorption by 2 molecules in the same band)
In fact, the model only represents the Beer Lambert law of absorption and therefore is incomplete. If the atmosphere is absorbing energy from radiation, where is this energy going?
The next step is to add emission.
Another important point is that the total number of people involved in creating, testing and checking this model = 1.
Surely there will be mistakes.. I welcome comments from sharp-eyed observers pointing out the mistakes.
Other articles:
Part One – a bit of a re-introduction to the subject
Part Three – the simple model extended to emission and absorption, showing what a difference an emitting atmosphere makes. Also very easy to see that the “IPCC logarithmic graph” is not at odds with the Beer-Lambert law.
Part Four – the effect of changing lapse rates (atmospheric temperature profile) and of overlapping the pH2O and pCO2 bands. Why surface radiation is not a mirror image of top of atmosphere radiation.
Part Five – a bit of a wrap up so far as well as an explanation of how the stratospheric temperature profile can affect “saturation”
Part Six – The Equations – the equations of radiative transfer including the plane parallel assumption and it’s nothing to do with blackbodies
Part Seven – changing the shape of the pCO2 band to see how it affects “saturation” – the wings of the band pick up the slack, in a manner of speaking
And Also –
Theory and Experiment – Atmospheric Radiation – real values of total flux and spectra compared with the theory.
Notes
Note 1 – The Beer-Lambert law is written in a number of slightly different forms. The transmission of incident monochromatic radiation is reduced like this:
I(λ) = I0(λ).exp(-σ(λ)nz)
where I(λ) = transmitted radiation of wavelength λ, I0(λ) = the incident radiation, σ(λ) = “capture cross section” at wavelength λ, n = number of absorbers per unit volume, z = length of path. And this equation relies on the number of absorbers remaining constant per unit volume. The equation can be written differently to deal with the case when the density of absorbers through the path changes.
Note 2 – If there was no radiatively-active atmosphere, then the top of atmosphere flux would be the same as the surface. With an emissivity of 0.98 and a temperature of 300K, the emission of radiation from the surface = 450 W/m².
It is actually calculated in the program by numerically integrating the spectrum of surface radiation, with a result of 447 W/m² (due to not integrating the waveform with enough precision at high wavelengths/low wavenumbers).
Note 3 – The Matlab program used to create this data:
and two functions called from RTE:
==== RTE v0.2 =========
% RTE = Radiative transfer equations in atmosphere
% Objective – allow progressively more complex applications
% to help people see what happens in practice in the atmosphere
% v0.2 allow iterations of one (or more) parameter to find the TOA flux vs
% changed parameter
% Define standard atmosphere against height
% zr = height, pr = pressure, Tr = temperature, rhor = density, all in SI units
Ts=300; % define surface temperature
ps=1.013e5; % define surface pressure
nmv=2.079e25; % nmv x rho = total number of molecules per m^3
maxzr=50e3; % height of atmosphere
numzr=1000; % number of points used to define real atmosphere
zr=linspace(0,maxzr,numzr); % height vector from sea level to maxzr
[pr Tr rhor ztropo] = define_atmos(zr,Ts,ps); % function to determine (or lookup) p, T & rho
% Consider atmosphere in a coarser resolution than used for calculation
% z, p,T,rho; subset of values used for RTE calcs
numz=20; % number of layers to consider
maxz=20e3; % height of atmosphere to consider in meters
zi=round(logspace(0.3,log10(numzr),numz)); % zi = lookup vector to “select” heights, pressures etc
% approximately logarithmically as pressure is logarithmic
z=zr(zi);
p=pr(zi);
T=Tr(zi);
rho=rhor(zi);
% various values
lapse=6.5; % environmental lapse rate ** note potential conflict with temp profile already determined
% could be max lapse rate for convective adjustment
ems=0.98; % emissivity of surface
% work in wavenumber, cm^-1
dv=5;
v=100:dv:2500; % wavenumber (=50um – 4um)
numv=length(v);
rads=ems.*planckmv(v,Ts); % surface spectral intensity against frequency v
% ====== Introducing the molecules ============
% need % mixing in the atmosphere vs height, % capture cross section per
% number per frequency, pressure & temperature broadening function
nummol=2; % number of radiatively-active gases
mz=ones(nummol,numz); % initialize mixing ratios of the gases
% specific concentrations
% pH2O = pretend H2O
emax=17e-3; % max mixing ratio (surface) of 17g/kg
mz(1,:)=(ztropo-z).*emax./ztropo; % straight line reduction from surface to tropopause
mz(1,(mz(1,:)<0))=5e-6; % replace negative values with 5ppm, ie, for heights above tropopause
% pCO2 = pretend CO2
mz(2,:)=3.6e-4; % 360ppm
% absorption coefficients
k1=.3; % arbitrary pick – in m2/kg while we use rho
k2=.3; % likewise
a=zeros(nummol,length(v)); % initialize absorption coefficients
a(1,(v>=1250 & v<=1500))=k1; % wavelength dependent absorption
a(2,(v>=600 & v<=800))=k2;
% === Loop to change key parameter for which we want to see the effect
%
nres=10; % number of results to calculate
flux=zeros(1,nres); % TOA flux for each change in parameter
par=zeros(1,nres); % parameter we will alter
plotix=[1 3 5 8 10]; % graphs to plot
nplot=length(plotix)+1; % plot the “plotix” graphs plus the summary
% work out the location of subplots
if nplot==1
subr=1;subc=1; % 1 row, 1 column
elseif nplot==2
subr=2;subc=1; % 2 rows, 1 columns
elseif nplot==3 || nplot==4
subr=2;subc=2; % 2 rows, 2 columns
elseif nplot==5 || nplot==6
subr=3;subc=2; % 3 rows, 2 columns
else
subr=3;subc=3; % 3 rows, 3 columns
end
% parameter values to try – this section has to be changed depending on the
% run
% CO2 concentration
par=logspace(-5,-2.5,nres);
for n=1:nres % each complete run with a new parameter to try
% — this line below has to change depending on parameter chosen
mz(2,:)=par(n);
% === Main loops to calculate TOA spectrum & flux =====
% first we work through each layer
% then through each wavenumber
% currently calculating surface radiation absorption up to TOA
rad=zeros(numz,numv);
rad(1,:)=rads; % surface radiation vs wavenumber
for i=2:numz % each layer
for j=1:numv % each wavenumber interval
abs=1; % initialize the amount of absorption within the wavenumber interval
for k=1:nummol % each absorbing molecule
% for each absorber: exp(-density x mixing ratio x
% absorption coefficient x thickness of layer)
abs=abs*exp(-rho(i)*mz(k,i)*a(k,j)*(z(i)-z(i-1))); % calculate absorption
end
rad(i,j)=rad(i-1,j)*abs; %
end
end
flux(n)=sum(rad(end,:)*dv); % calculate the TOA flux
% decide if and where to plot
ploc=find(plotix==n); % is this one of the results we want to plot?
if not(isempty(ploc)) % then plot. “Ploc” is the location within all the plots
subplot(subr,subc,ploc),plot(v,rad(end,:)) % plot wavenumber against TOA emissive power
xlabel(‘Wavenumber, cm^-^1′,’FontSize’,8)
ylabel(‘W/m^2.cm^-^1′,’FontSize’,8)
title([‘pCO2 = ‘ num2str(round(par(n)*1e6)) ‘ppm, Total TOA flux= ‘ num2str(round(flux(n))) ‘ W/m^2’])
grid on
end
end
% final plot – TOA flux vs changed parameter
subplot(subr,subc,nplot),plot(par*1e6,flux)
title(‘Summary Results’,’FontWeight’,’Bold’)
ylabel(‘TOA Flux, W/m^2’)
xlabel(‘pCO2 ppm’) % this x label is up for changing
====== end of RTE v0.2 =========
====== define_atmos ============
function [pr Tr rhor ztropo] = define_atmos(zr, Ts, ps)
% Calculate pressure, pr; temperature, Tr; density, rhor; from height, zr;
% surface temperature, Ts; and surface pressure, ps
% By assuming a standard temperature profile to a fixed tropopause temp
% zr is a linear vector and starts at the surface
pr=zeros(1,length(zr)); % allocate space
Tr=zeros(1,length(zr)); % allocate space
rhor=zeros(1,length(zr)); % allocate space
% first calculate temperature profile
lapset=6.5/1000; % lapse rate in K/m
Ttropo=215; % temperature of tropopause
htropo=10000; % height of tropopause
Tstrato=270; % temperature of stratopause
zstrato=50000; % height of stratopause
ztropo=((Ts-Ttropo)/lapset); % height of bottom of tropopause in m
for i=1:length(zr)
if zr(i)<ztropo
Tr(i)=Ts-(zr(i)*lapset); % the troposphere
elseif (zr(i)>=ztropo && zr(i)<=(ztropo+htropo))
Tr(i)=Ttropo; % the tropopause
elseif (zr(i)>(ztropo+htropo) && zr(i)<zstrato)
Tr(i)=Ttropo+(zr(i)-(ztropo+htropo))*(Tstrato-Ttropo)/(zstrato-(ztropo+htropo)); % stratosphere
else
Tr(i)=Tstrato; % haven’t yet defined temperature above stratopause
% but this prevents an error condition
end
end
% pressure, p = ps * exp(-mg/R*integral(1/T)dz) from 0-z
mr=28.57e-3; % molar mass of air
R=8.31; % gas constant
g=9.8; % gravity constant
intzt=0; % sum the integral of dz/T in each iteration
dzr=zr(2)-zr(1); % dzr is linear so delta z is constant
pr(1)=ps; % surface condition
for i=2:length(zr)
intzt=intzt+(dzr/Tr(i));
pr(i)=ps*exp(-mr*g*intzt/R);
end
% density, rhor = mr.p/RT
rhor=mr.*pr./(R.*Tr);
end
======= end of define_atmos =================
======= planckmv ==================
function ri = planckmv( v,t )
% planck function for matrix of wavenumber, v (1/cm) and temp, t (K)
% result in spectral emissive power (pi * intensity)
% in W/(m^2.cm^-1)
% from
% http://pds-atmospheres.nmsu.edu/education_and_outreach/encyclopedia/planck_function.htm
[vv tt]= meshgrid(v,t);
ri = 3.7418e-8.*v.^3./(exp(vv.*1.4388./tt)-1);
end
============ end of planckmv =================


[…] Update – Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Two […]
Just a minor query. Why the non SI derived units? Wavelengths per cm sounds like it was derived from some ancient system – perhaps CGS?
Jerry:
It has become the standard in spectroscopy. I’m not a fan, but the HITRANS database for example is all in cm^-1.
According to an apocryphal source:
According to the 5.35ln(c2/c1) formula used by the IPCC et al. the post-industrial increase in atmospheric CO2 concentration from 280 to the current value of 390 ppm caused an increase of about 1.8 w/sq m in TOA radiative forcing. Yet your “summary results” graph seems to show a change of at least 10 w/sq m over this range, and it’s a decrease, not an increase. I’m probably just displaying my ignorance, but could you please explain?
SOD
You should check “Climate Change Reexamined” by Joel M. Kauffman available at:
Click to access climate-change-re-examined.pdf
If link does not work, use Google scholar
In Fig. 7 note the absorption of IR by CO2 and water vapor in the 700-400 wavenumber range. If the peaks were integrated water vapor would be absorbing most all of IR energy.
At and slightly above (e.g, a few thousand feet) the surface of the earth, where we live, the greenhouse effect due entirely to water vapor. The specific humidty would have to be near 0 for the heavy slug CO2 molecules to compete 50-50 with with the light and frisky water molecules for IR energy.
You should aslo check Table 2. Alice Springs is in the desert and note there has been no change annual mean termp since 1879. The temp data for the cities cleary show the UHI effect.
In real air there is no uniform distribution of mass as shown by weather maps. High pressure cells have more regional mass and drier air than do low pressure cells which usually have higher humidity and which bring weather like rain, snow, cyclones, etc.
The assumption that GHG’s are well-mixed in the atmosphere is incorrect and is a fatal flaw in climate models. The statement that GHG’s are well-mixed only applies to purified dry air which is comprised of nitrogen, oxygen, the inert gases and CO2 and whose composition is independent of site, temperature, pressure, and humidity except for minor local variation in particular with respect to CO2. Purified dry air does not occur in the earth’s atmosphere.
Based on the results (i.e., speculations, conjectures and scenarios) of climate model calculations and the recommendations of the IPPC and the climate scientists, I am now paying a 20% carbon tax on the cost nat gas (ca $5 per gigajoule) in BC. I don’t like at all. I pay a carbon tax of 5 cents per liter of gasoline. Diesel has a carbon tax of 5.5 cent per liter.
Unless you want to eventually start paying hefty carbon taxes on fossil fuels, you should devote you efforts to showing that CO2 does nor cause global warming.
Some background info:
http://www.usciences.edu/academics/collegesDepts/chemistry/Faculty/kauffman.aspx
Wonder how much time he had to spend on the ‘essay’ compared to his normal work?
Roger:
It’s good to ask questions.
First of all, the second question – “why a decrease”?
Imagine if all of the surface radiation was emitted unchanged at the top of atmosphere. (No “greenhouse” effect).
Let’s say 450 W/m² emitted from the surface and, therefore, 450 W/m² emitted into space from TOA
Now we add a “greenhouse” gas and the radiation leaving from TOA = (e.g.) 440 W/m².
The energy leaving the planet has reduced by 10 440 W/m². This means more heating of the planet, therefore, (by convention), a radiative forcing.
So it’s more about convention. If less energy leaves the planet, the planet must warm (at least in the short term).
So a reduction in radiation leaving is an increase in radiative forcing.
Secondly, the actual values I show will of course be arbitrary. I am using “pretend CO2” and “pretend H2O”. The idea is to make the subject clearer by having less complications.
The real models used pull many 100,000s of spectral lines from the HITRANS database. Or use complex band models.
We want to understand how different effects cause different results. To do that we need simplicity.
Thank you. I guess I’ve become conditioned to think of radiative forcing in terms of an increase in heat retained rather than a decrease in heat escaping.
Harold Pierce Jr:
Yet another person happily unacquainted with the real work on the subject.
What has he measured? What has he calculated?
Has he confirmed the reduction in top of atmosphere flux due to different gases? Has he explained the relationship between surface forcing and top of atmosphere fluxes?
Which one has most relevance for the climate?
Would you like to explain exactly what his absorbance graph is and how it varies across the world? Would you like to explain how it affects the top of atmosphere results?
I hope as we progress through this series to give more people the insight needed to easily identify poorly constructed and argued points.
The Beer-Lambert Law is only strictly true for monochromatic radiation. For this example it doesn’t matter since the absorption ‘line’ is rectangular. But it does matter for real lines.
DeWitt Payne:
I’ll make that clearer by adding a note.
That is how the model does the calculation, taking 5cm^-1 at a time.
As you say, it doesn’t matter while the absorption “line” is an extremely wide “block” with no variation.
And as spectral variation is added to the model I expect to have to change the spectral resolution accordingly.
If using the diagram posted by ”science on doom” in https://scienceofdoom.files.wordpress.com/2010/10/goody-1989-clear-sky-spectrum1-499px.png
then the conclusion might be drawn that the additional emission of CO2 into the atmosphere can still give some contribution to the increase of the temperature of the atmosphere. This is because the absorption of upward radiation in the rotational band of CO2 is not to hundred percent, according to this diagram.
On the other hand, if the diagram given in http://en.wikipedia.org/wiki/File:Atmospheric_Transmission.png
is used then the addition of CO2 to the atmosphere will be of no importance as to the temperature of the atmosphere since the diagram shows the absorption in the band of CO2 to one hundred percent, see the specific diagram for CO2 presented below the diagram for H2O.
We can simply estimate the upper limit for how much CO2 can contribute to the temperature of the climate system of the Earth. This is done by integrating the Planck’s distribution function for radiation over the relevant absorption band of CO2 (this has been done millions of times so here comes the million plus one).
We assume thus that all the upward radiation lying within the rotational band of CO2 is absorbed and thermalized to hundred percent, which leads to the increase of the temperature of the air. If now assuming additionally that all this extra energy is prohibited to leave the climate system then we will get the values for the upper limit.
The absorption region is determined by the half width of the absorption band line (as in the case of the Fraunhofer lines), which gives the range from 13um to 18um. The temperature entering the Planck function is that of the surface emitting radiation, for example, 288K, corresponding to 390W/m^2. This gives the maximum contribution from CO2 to be around 87W/m^2. The concentration of CO2 does not enter these calculations. The only important requirement is that all radiation within the band of interest is absorbed before the upward radiation leaves the Earth system.
The similar calculations in the case of water vapor give 246W/m^2. The rotational bands of interest in the case of H2O vapor range from 4.5um to 9um and from 15um to >70um.
The total contribution from both CO2 and H2O is, thus, 87 + 246 = 333W/m^2. And since the average power density for the upward radiation when being emitted by the surface is about 390W/m^2, then we get 333/390 = 0.85, which means that 85 percent of the upward radiation is absorbed and 15%, i.e. 59W/m^2, leave the system. This would be in consistence with 15 – 30 percent for the transmitted upward radiation after the passage through the atmosphere as given in

However, it is most probable that one half of the radiation absorbed by CO2 will be emitted (after the thermalizing) into the space. This lowers the radiative contribution from both CO2 and H2O to 333/2=166.5W/m^2. This gives 166.5/390=0.43, i.e. 43% for the absorbed part of the upward radiation and 57% for the transmitted one, or, in other word, 222W/m^2 emitted out from the system. The climate science operates with the value of 239W/m^2 emitted out into the space, which is close to the roughly estimated value of 222W/m^2 above.
Anyhow, I draw the conclusion that the additional emission of CO2 into the atmosphere has not the potential to increase additionally the total temperature of the climate system since CO2 has already reached its maximal limit of the influence. In such a case, the increase of CO2 in the atmosphere might only increase the temperature in the lower part of the troposphere but on the behalf of the decreasing the temperature in the upper part of the atmosphere.
Wrong answer. By setting restrictions on the wavelength range of CO2 absorption and neglecting the effect of decreasing temperature with altitude, you’ve made the absorption rectangular. It isn’t. I suggest you spend $25 to subscribe to SpectralCalc for a month and do some real atmospheric radiative transfer modeling with a true line-by-line program. If you’re too cheap to do that, MODTRAN will give you similar results for free. Here’s a set of MODTRAN results for 10, 100, 1000 and 10,000 ppmv CO2. There is no decrease in the size of the effect with increasing CO2 concentration that I can see.
DeWitt Payne.
You are right that I made absorption rectangular. I was interested to find how much CO2 can contribute to the climate at best, i.e. to find an upper limit. And this limit will be obtained if CO2 stops all the radiation within the band of absorption of CO2 and the absorbed energy is thermalized, that is it goes to increase kinetic energy of the surrounding molecules.
I have tried the different temperatures of the air and the contribution due to CO2 followed the increase of temperature. But in such a case you must also take into account that the increase of temperature of the atmosphere will also increase the radiation out from the atmosphere to the space.
Ernest: You wrote: “However, it is most probable that one half of the radiation absorbed by CO2 will be emitted (after the thermalizing) into the space.” How does half of the energy absorbed by CO2 get emitted to space? This energy is in the form of kinetic energy/temperature of the molecules of the atmosphere. This energy can’t get to space by conduction or convection, so it must get escape as radiation. Nitrogen and oxygen at 210-310 degK have negligible emission at infrared wavelengths. The only molecules that can emit energy to space (thereby radiatively cooling the atmosphere) are GHGs, mainly water and CO2. They only emit at the wavelengths where they also absorb. Once LW energy is absorbed by the atmosphere, it has trouble escaping the atmosphere.
We can calculate how difficult it is to escape. There are probably better sources of information about the fraction of upward radiation that is transmitted to space than the wikipedia file you cite. If you read the details, the author (not a climate scientist) notes that he only considered the fate of photons emitted in a vertical direction. He doesn’t mention clouds. The standard KT model of the earth’s energy balance indicates that only 40 W/m2 of total 396 W/m2 of upward radiation escapes directly from the earth’s surface into space. 356 W/m2 are absorbed by the atmosphere and clouds, which radiate 333 W/m2 back towards the surface of the earth (DLR). From these numbers, NET upward radiation from the earth’s LOWER atmosphere (ie radiative cooling of the lower atmosphere) occurs at a rate of only 23 W/m2. (Some skeptic scientists believe that even 23 W/m2 may be too high.) Your calculation showing that 222 W/m2 escapes is about right for the UPPER atmosphere. However, essentially all of the water vapor and most of the carbon dioxide are found (and absorb radiation) in the troposphere. Once LW energy absorbed by a GHG, escape to space is much slower than you calculated.
KT model: http://www.cgd.ucar.edu/cas/Trenberth/trenberth.papers/TFK_bams09.pdf
Skeptics: http://www.drroyspencer.com/2010/08/comments-on-miskolczi’s-2010-controversial-greenhouse-theory/
Despite the above issues, you are ALMOST correct when you say that additional CO2 “has not the potential to increase additionally the total temperature of the climate system since CO2 has already reached its maximal limit of the influence.” If you replace the phrase “increase the temperature” with “decrease the radiative cooling of the lower atmosphere” or “decrease net upward radiative flux near the surface”, you would be accurate. Doubling CO2 decreases the upward flux of radiation near the earth’s surface by only about 1 W/m2. (See Figure 2 of Ramanathan 1981; journals.ametsoc.org/doi/pdf/10.1175/1520-0469%281981%29038%3C0918%3ATROOAI%3E2.0.CO%3B2) DeWitt posted similar results from HITRAN at SOD. 1 W/m2 certainly isn’t much compared with the 396 W/m2 leaving the earth’s surface. HOWEVER, we need to consider how ALL energy fluxes change – not just one – before we can calculate a change in temperature.
The temperature of the lower troposphere is not determined by radiative cooling – an process which we have already calculated is far too inefficient in the presence of so much water vapor and CO2. Temperature in most of the troposphere is controlled buoyancy-driven convection, which starts and persists anytime the local temperature gradient becomes too steep (steeper than the lapse rate). If buoyancy-driven convection didn’t occur, surface temperature would increase by about 50 degK.
The only place where the rate of absorption and emission of radiation controls the local temperature is at and above the tropopause. At these altitudes, if radiative cooling is reduced by increasing GHGs, the temperature certainly will rise. There is little water vapor here, so CO2 becomes a more effective GHG because it no longer must compete with water for photons. At the tropopause, doubling CO2 is calculated to reduce the net upward flux of radiation by about 4 W/m2 (see Ramanathan’s Figure 2 again), raising the local temperature by about 1 degC. As long as the lapse rate remains constant, surface temperature will rise by roughly the same 1 degC. When doubling CO2 raises the temperature at the tropopause, the surface can warm further before convection begins.
The person who made the Wikipedia figure you cited and the people who calculate radiative forcing for the IPCC are using the same radiation transfer database, HITRAN. Ramanathan used an earlier version. You might be interested in a different Wikipedia figure that shows how the spectrum of upward radiation at 20 kilometers is changed by 2X CO2. As you anticipate, the center of the CO2 band doesn’t change, but the edges do: http://en.wikipedia.org/wiki/File:ModtranRadiativeForcingDoubleCO2.png
Frank.
Thank you for your comments. Maybe I am wrong but I took it for granted that any object of temperature above 0K emits radiation in accordance to the Planck radiation law. This is due to the collisions between the atoms/molecules, which lead to the so called thermal radiation effect. Certainly, a collision might lead to the excitation of molecules of a given sort to the rotational states if the collisions deliver sufficient amount of energy. In such a case, the result will be emission of photons of a specific energy when (and if) the molecules returns to the initial energy state giving thus a higher intensity of radiation in the specific wavelength as compared to the thermal radiation given by Planck.
In the actual case, the thermalizing of the energy absorbed by CO2 from the upward radiation will increase the kinetic energy of the surrounding molecules (that is increase temperature of the given volume of air). This, in turn, will increase the thermal radiation from this volume to the surrounding and change the net energy balance between this volume and the surrounding ones. The change of the balance is such that the net difference between the energy emitted in the direction to the adjacent volume below and received from this adjacent volume will now be somewhat smaller while the net difference between the energy emitted in the direction to the adjacent layer above and received from this layer will now be somewhat greater. This will represent only a distortion if the event of absorption is the occasional one. But if we have a continuous absorption by CO2 contained in the given volume then this will lead with time to a new temperature profile across the atmosphere.
In my calculations, I have tried to cover both possibilities, one that the energy absorbed by CO2 is not transported to the outer space and the other that 50 % of the absorbed energy by CO2 is radiated down and 50 % up which is the maximum that one can require.
Frank,
Maybe you can help solve what for me is a puzzle. The climate system sheds the heat that it absorbs from the sun in two stages: first, mainly bulk transport by the non-radiative 99% of the atmosphere from the surface to the tropopause; second, radiative transfer from the tropopause to space solely through the other 1%. This requires the 99% to transfer its heat load to the 1%, presumably by molecular collision. In fact, water vapour, the predominant GHG, being practically absent at the tropopause, heat transfer by collision would be from the 99% to CO2’s 0.04%. This looks like a large, naturally inelegant bottleneck in the cooling system. It looks even bigger considering that CO2 can emit only in narrow bands the heat it receives in a broad spectrum of wavelengths. The bottleneck begs the question whether a natural phenomenon (electrical storms?) could render N2 and O2 temporarily radiatively active? Independently of such speculation, increasing CO2 concentration would reduce the effects of the bottleneck, enhancing the cooling system’s performance?
John: What you call “a large, naturally inelegant bottleneck in the cooling system” is what others call “the greenhouse effect”. Both descriptions have strengths and weaknesses. Life has evolved on our planet that is compatible with your “bottleneck”. Perhaps we should look upon all planetary atmospheres are elegant examples of how the laws of physics produce complex phenomena.
How does the “bottleneck” change temperature? If you put more GHG in one “special location” in the atmosphere, you can’t predict a priori whether the GHGs will make that special location warmer or cooler. You need to know if that special location is warmer or cooler than the average location that is exchanging photons with the special location. More GHGs (more emitters and absorbers) will speed up the energy flow from warmer to cooler. Ozone makes the temperature of the stratosphere higher than the atmosphere above and below it, so increasing GHG’s cools the stratosphere (and warms the neighboring air). In most of the troposphere, the air below is warmer and the air above is cooler, but more photons come from warmer regions, so adding GHGs causes warming.
N2 and O2 are symmetrical molecules without a permanent dipole to interact with electromagnetic radiation. Anything that changes this situation can make them absorb and emit radiation. You probably already know about photodissociation of O2 to make O atoms and then O3. Thunderstorms apparently produce NH3, presumably by first splitting N2 into N atoms. There is a weak emission from oxygen that I don’t understand. In theory, however, we know should about all of the significant radiating species in our atmosphere from observing the emission spectrum of the atmosphere and identifying the lines. Astronomers have identified the presence of many (hundreds?) molecules elsewhere in the universe by this technique. Helium was first identified in the sun.
Lightning produces nitrogen oxides not ammonia, well, maybe some ammonia too, but mostly oxides.
Oxygen has a permanent magnetic dipole moment due to unpaired spins of its outermost two electrons which makes it paramagnetic and allows it to directly interact with EM radiation for rotational energy transitions. The 60 GHz band that is used for satellite remote temperature profile measurement is from oxygen molecular absorption/emission.
Ernest wrote: “Maybe I am wrong but I took it for granted that any object of temperature above 0K emits radiation in accordance to the Planck radiation law.” This statement is, of course, correct only for blackbodies. Adding an emissivity term can help, but emissivity is NOT a constant for gases, as it can be for liquids and solids. The use of black- or graybody radiation to describe the interaction between radiation and gases initially caused me an immense amount of confusion (and still does). My confusion ended when SOD introduced me to the Schwartzschild eqn, which describes the fundamental physics of how radiation (I) of a given wavelength is changed (dI) by emission and absorption as it passes a short distance (ds) through a greenhouse gas of density (r):
dI = -(Ikr)ds + [B(T)kr]ds
where B(T) is the Planck function and k is the absorption coefficient for that wavelength. The density of GHG (r) can be expressed in a variety of ways, often in terms of pressure, mixing ratio, and/or density (changing the units used with k). Note that the same absorption and emission constant, k, is used in both the absorption and emission terms. (From a quantum mechanical point of view, emission is simply absorption with time running in the opposite direction, so the same k applies to both.)
When one uses a spectrometer to pass an beam of light through a sample and obtain its absorption spectrum, emission is occurring in all directions and only a tiny fraction of that emission ends up in the detector. The spectrometer’s light source also puts out more intense radiation than the sample. Beer’s Law arises from integrating only the -(Ikr)ds term from Schwartzschild’s eqn. When combined with the concept of blackbody radiation, we obtain the misleading impression that absorption and emission are independent phenomena.
When radiation has passed through a homogeneous, isothermal gas for a long enough distance, dI/ds approaches zero (equilibrium) and I approaches B(T). In this case, we say the gas is “optically thick” and the radiation leaving such gas has a blackbody spectrum (at least everywhere that k is not zero). In climate science, you can find lots of misleading models showing black- or greybody radiation being emitted from a slab of atmosphere. The technically correct models include the phrase “optically thick slab” somewhere – usually without bothering to explain what it means. They rarely explain that the earth’s atmosphere is optically thick at some wavelengths, but not others, so the model can’t be applied to the earth.
Schwartschild’s eqn presumably applies to solids and liquids, as well as gases. Blackbody radiation isn’t really emitted from the surface of solids and liquids, it is emitted by atoms or molecules at and below the surface. To emit blackbody radiation, a solid or liquid surface also needs to be optically thick, just like a gas. For many materials and wavelengths, optically thick may require only a few microns or millimeters of material. (20 um of water is optically thick for IR, 2 mm is optically thick for microwaves and tens of meters for visible.) Furthermore, the vibrational and rotational states of molecules and atoms in solids and liquids are broadened by intramolecular interactions, making emissivity change slowly with wavelength and appear constant.
http://www.barrettbellamyclimate.com/page16.htm shows what emission from the top of the earth’s atmosphere is predicted to look like when HITRAN use a database of k’s for all relevant wavelengths and a “standard atmosphere” to numerically integrate the Schwartzschild eqn for radiation emitted upward from the earth’s surface to space. Blackbody emission curves have been added for various temperatures, perpetuating the MYTH that our atmosphere emits like a blackbody. Most of the SURFACE of the earth does emit like a blackbody with an average temperature of 288 degK. The wavelengths (750-1000, 1080-1250 cm-1) where emission matches that blackbody radiation at 288 degK, indicate wavelengths where the absorption/emission coefficient for all gases in the atmosphere is effectively zero. Most/all of the radiation emitted by the surface at these wavelengths escapes directly to space. If we temporarily ignore the problem that GHG density varies with altitude, integrating Schwartzschild’s eqn produces terms involving exp(-krs), from which we can define “one optical depth unit” or as the distance s for which the term krs (often called tau?) is equal to 1. Average photons escaping to space are emitted from roughly one “optical depth” beneath the top of the atmosphere, often called the “characteristic emission level”. The intensity of the radiation at any wavelength reflects the temperature at this altitude through the B(T) term, but the terms “optically thick” and therefore “blackbody” do NOT apply to this situation.
Frank.
Thank you for supplying us with the Schwarzschild equation and the reference to the diagram presenting the MODTRAN spectrum. Both are of crucial importance.
First I would like to explain that my primarily interest was in founding how much contribution can we expect from CO2. This interest was due to the fact that the temperature of the Earth climate has changed considerably during the millions of years and I was curious to know if CO2 had anything to do with this. The answer seems to be that CO2 can contribute to some degree at low concentrations but the addition of CO2 above some given value will stop to be of any importance.
The Schwarzschild equation and the MODTRAN diagram explain why it is so, at least as I am interpreting them.
Namely, the diagram shows that the atmosphere is already optically thick as for the wavelength within the rotational band of CO2. The intensity of the upward radiation from the surface reaches the equality with the Planck function when passing through the troposphere (compare Schwarzschild equatuion) and the outgoing radiation is now determined by the Planck function for the temperature at the end of troposphere, which is about 220K, (compare the MODTRAN diagram).
Thus, the addition of more CO2 to the atmosphere will have no impact on the outgoing radiation in the wavelengths range of interest.
What conclusions have we to draw if it is so? Firstly, we must look for some other source of energy that might be responsible for the observed temperature trends in the atmosphere, lands and oceans. Secondly, the political action concerning the emission of CO2 might be off beam (overzealous) and missing the real source(s) causing the observed effect – this under the assumption that the observed changes are expected to lead to severe consequences and we have a possibility to govern the development.
Ernest: You wrote: “Thank you for supplying us with the Schwarzschild equation and the reference to the diagram presenting the MODTRAN spectrum. Both are of crucial importance.” Later you wrote: “Thus, the addition of more CO2 to the atmosphere will have no impact on the outgoing radiation in the wavelengths range of interest.” These statements appear to be mutually contradictory.
http://en.wikipedia.org/wiki/File:ModtranRadiativeForcingDoubleCO2.png The text on the MODTRAN spectrum explicitly says that the total upward radiation at 20 km decreases from 260.1 W/m2 to 256.7 W/m2 as CO2 is doubled from 200 to 600 ppm. This represents the difference between the green and blue curves, which we all recognize is negligible in the center of the CO2 band. The difference is non-trivial on the wings. Below, SOD has posted a graph showing information equivalent to the difference between the green and blue lines for change in CO2 and other GHG’s. (I place more faith in the peer-reviewed material that SOD has posted than the anonymous material in Wikipedia, but they are both saying the same thing. Wikipedia makes it simpler.)
The 3.4 W/m2 difference caused by doubling CO2 is only a 1.3% difference. If you differentiate W = eoT^4 with respect to T, substitute for T^3 and rearrange the terms; you will get the handy equation: dW/W = 4*(dT/T). A 1.3% change in blackbody radiation is equivalent to a 0.32% change in degK in source temperature.
At altitudes where temperature is controlled by radiative equilibrium (the tropopause and above), if outgoing radiation decreases due to increased CO2, the temperature will need to warm enough to restore balance. To calculated the actual temperature change, one needs the difference between the upward and downward radiation caused by doubling by CO2 (ie radiative forcing) and the temperature at that altitude. 255 degK is the blackbody temperature in equilibrium with the 70% of solar radiation absorbed by the earth. When you apply the IPCC’s value for radiative forcing (3.7 W/m2) to 255 degK, one obtains the “no-feedbacks climate sensitivity” of about 1 degK for 2X CO2 at the tropopause.
There is plenty of room to be skeptical about what happens after the tropopause warms 1 degC.
Some aspects of these graphs can be misleading. The 220 degK curve and the plateau at roughly 620-640 cm-1 do not indicate saturation! The vertical axis shows us the number of photons reaching a particular altitude at a particular wavelength. The average photon originated about one optical density unit below this altitude. The number of photons depends on the temperature at the altitudes where these photons were emitted (via the Planck function). In the BarrettBellamy version, you can see a small upward spike in the very center of the CO2 band where absorption is strongest and the average photon at that wavelength is emitted from the warmer stratosphere rather than the cooler tropopause. B&B’s graph shows upward radiation at 70 km, rather than Wikipedia’s 20 km. There is supposed to be a similar spike in the ozone band.) http://www.barrettbellamyclimate.com/page16.htm
Eli thinks he sees where you are going and eagerly awaits the next post. three minor points
1. the reason everyone likes wavenumbers is that they are very convenient for everything from the deep vacuum UV (100 nm is 100,000 cm-1) to the far IR (100 cm-1). When you get more energetic (below 100 nm) people start using eV, and when you get to longer wavelengths (radio, microwave) they use Hz or THz or MHz.
2. It would be more “accurate” to put a second box absorption for H2O between 100 and 1000 cm-1. Most of the absorption relevant to the greenhouse effect is from rotational transitions, however, this would weaken the didactical value of the post
3. By eyeball it looks like your water vapor concentration does not fall off fast enough, and it would be better to have it in ppm or a ratio of number of molecules rather than mass units
and a major point. Where this is going to come out when you add emission is that the net effect of the absorption and emission cycles is to move the level at which emission to space in the box is 5-10 km up in the atmosphere where it is below 250K, and emission is weaker. Since the rate of energy being pumped out at those temperatures is much slower, the energy loss per unit time to space will decrease, which has made up for by warming the surface.
A very interesting webpage, really making an effort to explain difficult things in a simple way. Thanks!
I hope I can get an answer to a question I have been pondering over for a while. Maybe it has already been explained in one of the postings.
All discussions I have seen concern radiation transfer, and that the GHG molecules transfer the extra energy to the rest of the molecules in the air through collisions.
My question: in an atmosphere totally void of GHGs, wouldn’t the O2 and N2 molecules still absorb energy from the surface through molecular collisions and then spread it through the atmosphere and also back to the surface through “back-collisions”? How much heat could be trapped through this process? Has anyone calculated this?
There are posts on that topic:
Basically, the atmosphere would be approximately the temperature of the surface at the surface and decrease with altitude. For a spherical planet, the temperature would decrease as latitude increased so there would be circulation from the equator to the poles too. But the surface would be cooler than if there were ghg’s because the surface would see only solar radiation and only the surface would lose energy to space by radiation. So once the atmosphere reached steady state there would be very little net transfer of heat to or from the atmosphere.
Ernest:
Take a look at CO2 – An Insignificant Trace Gas? – Part Eight – Saturation, and especially this graph:
Scienceofdoom”.
Thanks for ”CO2 – An Insignificant trace gas? – Part Eight – Saturation”. I do have notified previously the peculiar form of the absorption band (with the strange line at 15um) when looking on the MODTRAN diagram, so now I know why it is so.
What I observe when studying the MODTRAN diagram is that the minima of both of the wings are already touching the 220K line. I interpret this as the indication that the condition for the optical thickness is already achieved for the radiation within the wings. And the energy emitted to the space (area under the “tooth” curve) is matching the energy input from the Sun. So?
Of course, the adding of CO2 might act as the adding of the extra insulation to the wall of the house without the change of the power input into the house and without the changing of the temperature outside the wall. This will certainly rise the temperature of the room. However, I am not sure that this analogue is working in the case of the radiation transfer.
Did you ever tell us what the correct units were for the vertical axis? Did they provide integration across all wavelengths?
My memory is that the y axis is actually dimensionless because the difference was divided by the Planck function for some temperature (220K), but I could be wrong about that. Supporting evidence is that the peak height for the short wavelength side would be higher if the result were just the difference in forcing.
Propagation of the upward radiation from the surface can, in fact, be compared with the spectroscopic experiment when measuring the absorption of radiation by a sample. We have a source of radiation, the surface of the Earth, a sample, the atmosphere, and the detector, infrared spectrometer placed on the satellite. The experiment comes up with the “Fraunhofer lines”, here with the the absorption bands. The intensity of the detected radiation is described by the Schwarzschild equation.
In the actual case we get a typical absorption spectrum indicating the presence of molecules scattering the radiation from the source. The radiation that does not make out from the sample is heating the sample, and might also influence additionally the temperature of the radiation source.
The dispute is, however about: “What happens if we increase the concentration of the scattering agents in the sample, in this case that of CO2?”. I can suggest the answer: “Since we are not increasing the intensity of radiation within the rotational band of CO2 molecules, the further increase of CO2 will either not give any additional effect or the additional effect will be small and not follow the linear dependence on the further increase of concentration of CO2”. I think that Figure 3 in Part Three of Understanding …. points in this direction.
Actually you are, increasing the optical depth both on the wings and in hot bands. See Ray P (link available at Real Climate). In this model what you are doing is increasing the level at which the atmosphere radiates to a colder point which is another effect.
It might be also mentioned that the effects of the increasing concentration of CO2 in the air can be easily studied inside the greenhouses. The concentration of CO2 is often increased there artificially in order to increase the production of vegetables and fruits. Why not to use this and check experimentally how the enriching of greenhouses by the greenhouse gases will influence the greenhouse effect?
Ernest
Two effects by increasing CO2 concentration.
1. Radiative effect – however the experiment carried out by R W Wood suggests this is pretty marginal.
2. Chemical Effect – photosynthesis – CO2 as plant food this has been shown to have a significant positive effect.
There are two requirements for a greenhouse effect to manifest.
1. The atmosphere has to contain molecular gases that absorb and emit in the thermal IR for the temperature range of interest.
2. There has to be a temperature gradient with the temperature at the effective emission altitude a lot colder than the surface.
A greenhouse fails point 2. You would need a greenhouse on the order of 10 km tall to see an effect.
DeWitt Payne.
And what effect do you expect if we create a temperature gradient along the greenhouse, for example by cooling the roof of the greenhouse to the lower temperature and absorbing all the radiation leaving the greenhouse through the roof?
DeWitt Payne
….” You would need a greenhouse on the order of 10 km tall to see an effect.”…..
This suggests that you are in near agreement with Leonard Weinsteins view that the Greenhouse Effect is only of practical significance above the troposphere.
Bryan, Ernest:
Here is the model results in an isothermal atmosphere (an atmosphere all at the same temperature):
You can see that changing the concentration of pCO2 has no effect on the TOA flux (well a tiny effect, which is just due to numerical issues of finite element analysis).
This is because the atmosphere is absorbing surface radiation (“taking it out of the picture”), then re-emitting radiation (“putting it back in the picture”) at the same temperature – and, therefore, at the same intensity.
In the interests of tedious accuracy:
Reviewing my model results it might be that the tiny 0.3W/m^2 difference in TOA flux resulting from massive changes in pCO2 is caused by the 0.1’C temperature drop over the 16km height in this model.
The “isothermal” model was created by having a convective adjustment of a very small lapse rate.
A better illustration might be a plot of the difference between surface and TOA flux as a function of lapse rate for a fixed concentration of CO2.
After formulating the question on February 1, 2011 at 8:03 am, I have started to think, looking for the answer.
Thus, we start with a situation without atmosphere. Earth is losing energy to the sink (i.e. to the outer space), these energy losses being compensated by the irradiation from the Sun. The result is that the temperature of the Earth and its surface is kept at some given temperature T. The heat losses compensated by the Sun are given by the Stefan – Boltzmann law P = oT^4. If using the convention that the heat flow from the system is positive then the heat losses by radiation are given by P and the heat inflow from the Sun is –P.
We add now the atmosphere. During the transition period, a part of heat delivered by the Sun will go for raising the temperature of the air mass to the temperature of the surface. If ignoring the contribution from the radiation, the heating of the air will be done by heat conduction and convection through the air mass. After the equilibrium is reached, the temperature of the surface will become slightly lower (in accordance to the calorimetric relations if the system had been closed). In the actual case a gradient through the air mass will be established since the air mass is placed between the heater on one side and the sink on the other one losing energy to the sink by radiation. This means that the heat conduction and convection will continue to work striving to cancel the temperature gradient in accordance to the second law of thermodynamics. If we denote the heat losses due to the heat conduction and convection by A then the total net flow out of the system will be now given by P + A. This will tend to decrease the temperature of the surface furthermore.
But this is only a part of the story. There are several missing parts.
One of them is that any object of some given temperature above 0K is radiating energy in all directions,. This includes gases and, hence, also the air mass. This means in particular that a part of the heat delivered to the air by conduction and convection (and evaporation, which is also a missing part but it might be taken into consideration later) will be radiated down to the surface (and not only to the sink as it has been mentioned above).
The other missing factor is that also a part of the radiation P, emitted by the surface, will be absorbed by some specific kinds of molecules in the air. This absorbed energy will raise the temperature of the air and thus raise also its radiation in the direction both to the sink and to the surface.
The heat losses from the surface are thus now not (P + A) W/m^2 but (P + A – B – C) W/m^2 where -B is the energy returned to the surface due to the radiation generated by the heat conduction and convection while -C is the heat returned to the surface due to the absorption of a part of radiation P emitted by the surface. In particular, -C includes the part of the radiation P that is re-emitted by the air to the surface due to the absorption of the upward radiation by the water vapor and CO2.
We are still missing the other effects related to the evaporation, such as the energy of evaporation (D), energy of condensation (-E) (which goes for the heating of the air and thus for the increase of both the upward and the downward radiation by the air mass), the energy (F) required to increase the temperature of the precipitation when the precipitation is reaching the surface and the energy (-G) corresponding to the reflection of the upward radiation back to the surface. And we should not forget about the geothermal energy if we are expected to give a more complete description, and so on. If so required, also these important contributors to the energy balance might be included into the total balance.
(-B – C) might be treated as the extra energy coming from the Sun (with the upward radiation P passing in such a case through the atmosphere unabsorbed). This means that the temperature of the surface will become higher than without the atmosphere (under the condition that B + C > A plus other factors).
What about the heat radiated by the air to the sink? The increase of the temperature of the air mass due to the absorption of a part of the upward radiation from the surface (as well as the energy of condensation of water vapor and so on) is increasing the temperature of the air causing thus the appearance of -B and -C down to the surface and B’ and C’ up into the sink. -B and -C need not be equal to B’ and C’, respectively, if some mechanisms exist making the radiational heat flow more preferable in the direction down than up. In such a case this difference will additionally contribute to the increase of the temperature of the surface.
It should be observed that the quantity -C is generated by P. If all P is absorbed by the atmosphere then (- C) is more or less equal to -P/2 (C is close to C’). If P passes through the atmosphere unabsorbed then (- C) = 0 independently if there is temperature gradient or not.
After this much too long introduction, I can reformulate my question: Does really the temperature gradient through the atmosphere have any considerable importance for the influence of CO2 on the temperature of the system? The main contribution of CO2 to the system is related to the absorption of upward radiation by CO2, its re-emission and is thus a part of –C. This extra absorbed energy is confined within the volume determined by the optical thickness of the atmosphere for the wavelengths within the rotational band of CO2. After that, all the extra photons in this wavelength range delivered by the upward radiation from the surface are removed and the radiation in this range is described by the Schwarzschild equation and Planck function for the radiation propagating between the temperature interval from the end of the optical thickness region and up to the end of the troposphere.
I have looked at my last sentence in the previous post (2:04 pm) and found that there is more about the problem of CO2.
Namely, what happens above the height (La) at which all the surface photons within the absorption band of CO2 have been absorbed, i.e. between La and Lt where Lt is the end of the troposphere?
Assume first that we have no CO2 molecules along the pass of propagation in the upward direction above La. The observer studying the outgoing spectrum at the level Lt will found that the intensity of the band line of CO2 corresponds to that for the radiation at the temperature at La. This is because the air within this column would allow all the radiation emitted at La to propagate up to Lt unaffected, compare the Schwarzschild equation.
If we now start to put CO2 molecules inte the region between La and Lt, then the intensity of the observed band line at Lt will start to decrease. If we fill the region with the number of CO2 corresponding to the optical thickness within the band of absorption, then the intensity of the measured band line will drop to that corresponding to the intensity at the temperature of Lt. This means that all photons within this band emitted from the level La have now been absorbed, a part of them have been thermalized, which has contributed to the increase of the temperature of the air within the layer between La and Lt. If we continue to increase air by CO2 molecules, then the optical thickness will be reached at La1, where La1<Lt, and we are now back to the same situation as we had at La but now with the start at the temperature at La1 and so on.
Conclusions? The enriching of the atmosphere by CO2 leads to two effects:
A. It moves La down, which means that the energy (within the absorption band of CO2, constituting about 10 % of the total upward radiation) delivered by the upward radiation from the surface will now heat a somewhat lesser air mass, which gives a slightly higher increase of the temperature than previously. 50% of this absorbed energy might be re-emitted down (within the wavelengths of the total spectrum of thermal emission at the new temperature) and 50% up.'
B. 10% of the energy emitted from the (imaginary) surface at La will be absorbed within the next optically thick layer for the rotational band of CO2, i.e. between La and La1, 50% being emitted down and 50% up as in the point A above, and so on until the level Lt is reached.
If this picture is correct then the saturation effect will be reached when all the atmosphere is consisting of CO2.
Frank.
I have missed your comment on January 30. I have caught sight of it this morning. You are of course correct, I have discovered it by myself, too, and have presented a modified picture in my post on February 2 at 9:46 am.
After reading your comment, I have also “googled” to find more information about Schwarzschild equation and found it at
http://www.barrettbellamyclimate.com/page45.htm “Barrett Bellamy Climate, Schwarzschild’s Equation”.
What has really surprised me most was that I have constructed (in 9:46 am) more or less the same picture of the radiative transport process as it is given in Barrett. Therefore, I must conclude that the entire climate subject must be extremely simple if I could come so close to the correct description in view of the extremely limited intellectual capacity of mine. However, even this small achievement of mine would be not possible at all without your kindly mentioning about the Schwarzschild equation (on January 28, at 8:59 pm), which I had no idea about previously (even if I had some intuitive feeling for what one might expect).
If I am allowed to bath in the light of this greatest success in my life, then I can come with some more comments (probably totally irrelevant, as usual).
The contribution of CO2 to the climate is related to the “absorption area” below the 288K Planck curve and down to the 220K one. 50% (plus minus) of the energy corresponding to this area is expected to contribute to the changing in the state of the climate system (the other part will be emitted out of the system). This amount of energy is easy to find, either by computation or by the graphical methods. One might be tempted to claim that such a found value is the evidence as to the “saturation” of the influence of CO2 on the climate, as I stated in one of my previous posts, (the “saturation” means in this connection the no more effect will be observed if adding the additional amount of CO2 to the air). However, this is not the case if the addition of CO2 is resulting in the increase of the downward radiation due to the increase of the temperature of the first optically thick layer (i.e. the layer being closest to the surface), which will increase the temperature of the surface and, hence, refer now the upward radiation to this new temperature. This will raise the “tooth” curve from 288K level to a somewhat higher one. The lower part of the absorption band will be raised, too, but the net result will be still the increase of the “absorption area”. Barrett paper presents the number 1.3K for the doubling of CO2 (with all the other effects being neglected) and is mentioning also 0.07K per decade in the tropic region as due to the increase of CO2, but I am not competent to make any comments on these numbers. How much this increase of the temperature of the air will increase the temperature of the lands and oceans is depending on the heat capacity of water and soils, their thermal conductivity and the convectional and evaporational processes in the oceans. I am tempted, however, to agree with Barrett that the total effect of the increase of CO2 must be relatively small since it is masked effectively by the observed oscillations of the average temperature of the climate system being due to the other agents.
The mentioning of the wings within the absorption band of CO2 would indicate that the enriching of the atmosphere by CO2 is leading to the broadening of the absorption band (which I have difficulty to apprehend since the broadening of the width of the band is mostly the result of the Doppler effect and molecular collisions). Or I am completely out of the track? Anyhow, the increase of the absorption area due to the wing story would certainly increase of the amount of energy being consumed by the atmosphere.
I have some more comments but my post had become already too long (as usual).
[…] you compare it with the IPCC graph in Part Two (or Part Seven of the CO2 series) you will see it has some […]
[…] in Part Two, the concentration of pH2O in the lower atmosphere is much higher than pCO2, even for the highest […]
Frank, January 28:
Thanks for your response; apologies for the delay in mine. I see a fundamental difference between the greenhouse effect and my stylised “bottleneck”. The greenhouse effect as espoused by IPCC reflects purely radiative means of energy transfer – GHGs block outgoing longwave and back-radiate some of it to the surface with the effect that the surface becomes warmer than it would otherwise be. Increasing GHG concentration amplifies this effect.
The bottleneck, on the other hand, reflects energy transport by molecular collision, specifically the relatively low probability of collision and energy transfer between the trace GHGs and the bulk non-radiative elements of the atmosphere. Increasing GHG concentration in this case, in contrast with the greenhouse effect, increases the probability of collision, eases the bottleneck and enhances the system’s cooling performance. Increasing CO2 concentration, the IPCC villain, thus has two opposing effects on surface temperature.
We could add a third effect (which gets a mention in Sod’s most recent article): CO2 also absorbs (and emits) in the shortwave part of the radiative spectrum, reducing the solar flux at the surface, a cooling effect. My hope is that, as Sod rolls out the current suite of articles, how the RTE takes account of these three effects becomes clear. Perhaps it alread has – I haven’t caught up as yet.
Let’s take a look on how the surface layer heated by the irradiation from the Sun and the downward radiation communicates with the adjacent air layer that has reached the condition for the optical thickness for the wavelengths within the absorption band of CO2.
The optical thickness and, hence, the thickness of the air layer, (La), (as well as the number of the consecutive air layers satisfying the condition for the optical thickness) is a function of concentration of CO2 in air. The surface layer loses energy to the adjacent air layer by conduction, convection and radiation. In the case of water we must add also evaporation, but let’s omit this problem here. The adjacent layer transports heat to the next air layer by the same mechanisms. It emits also the radiation in the direction of the surface layer. However, if the temperature of the surface layer is higher than that of the air layer, the net energy flow, F, in W/m^2, will be always in the direction from the surface to the air layer.
In the case when the difference between the temperature, Ts, of the surface layer and that, Ta1, of the adjacent air layer is relatively small, the flow of energy from the surface to the air layer might be approximated by
F = P/A = b1(Ts – Ta1) + o(Ts^4 – Ta1^4) = (b1 + b2)(Ts – Ta1) (1)
where b1, in W/(K*m^2), relates to the heat transfer via convection and conduction while b2 relates to radiation.
The outflow of energy from the surface layer will decrease its temperature Ts by
Ts – dTs =Ts – [F/(cs)(rs)(Ls)]dt (2)
where (cs) is the specific heat, (rs) is density and Ls is thickness of the surface layer. Ls increases with time t proportionally to sqrt(a*t) where a is the thermal diffusivity of the soil or water.
10% of F, ie 0.1F, is absorbed by the air layer due to CO2 molecules, which leads to the increase of the temperature Ta by
Ta1 + dTa1 = Ta1 + [0.1F/(ca)(ra)La]dt (3)
where (ca) is the specific heat at constant pressure and (ra) is density of air.
The similar equation is obtained for the heat flow from the first optically thick air layer to the next one where we have to use Ta1 for the (first) air layer and Ta2 for the second one and so on along the height of the atmosphere. Since Ta1>Ta2, the heat exchange between these layers will lead to the decrease of the temperature of Ta1 by dTa12, so that the temperature of the first optical layer changes by
Ta1 + dTa1 – dTa12 (3)
where dTa12 must take care of the change of Ta2 due to the exchange of heat between layer 2 and 3 and so on.
The increase of Ta1 leads to the increase of the radiation from the layer to the surrounding, which means that 50 % of this increase, that’s
0.5o[(Ta1 + dTa1 – dTa12)^4 – Ta1^4] = 0.5o[4*Ta1^3*(dTa1- dTa12)] (4)
should be added to the downward radiation to the surface.
This means that after time dt, Ts has changed to Ts – dTs, Ta1 has changed to Ta1 + dTa1 – dTa12, (b1 + b2) has reduced by the amount given by (4) and (1) has decreased due to all these changes.
This is clearly a tough problem for the computer enthusiasts. The problem resembles a hound trying to catch its own tail. And while the hound problem will finish when the hound had caught its tail, the heat problem will finish when Ts had “caught” Ta during the nighttime (if one believes that this can happen) or when the Sun has started to shine on our CO2 plagued world and affected the changes of Ts and Ta so that it is now Ta that is trying to “caught” the flying (increasing) Ts.
Can we draw any conclusions without performing all these (still much simplified) calculations? Well, more or less if we know La and its dependence on the increase of CO2.
For instance, we might choose dt = 1s, La = 300 m, Ts – Ta = 2K, b1 + b2 = 15 W/(K*m^2). By applying the standard thermal properties of air and water we get
dTs = 0.019 K for the water layer of thickness Ls = 3*10^(-4) m
dTa1 = 7*10^(-6) K for the air layer
while the increase of the downward radiation is below 2*10^(-5) W/m^2.
One can now compute, if one bathers, the changes during the night time respectively daytime as well as double thus calculated values if the doubling of CO2 leads to the decrease of La1 by a half.
As you yourself state, this is a model of only a part of what is happening at TOA. However, I think it is enough!
We know energy cannot be created or destroyed, therefore we can conclude that the total amount of energy that will be emitted by the GHG in the atmosphere is exactly equal to the amount absorbed.
That is, there is an upper limit on the energy that can be emitted…ever. This implies an upper limit on future temperatures. Right?
Marko:
Take a look at Do Trenberth and Kiehl understand the First Law of Thermodynamics?.
A nice simple example of how internal temperatures can increase way above the external temperature.
[…] is a simple example of why. Readers who followed the series Understanding Atmospheric Radiation and the “Greenhouse” Effect will remember that I introduced a simple atmosphere with two molecules, pCO2 and pH2O. These had a […]
btb, great site… maybe you covered this somewhere, so sorry if you did…
a problem many have is how can 1 CO2 hiding in 2500 molecules absorb all that 15 micron radiation? we know it does, so the q is, how “wide” are the IR photons that are getting absorbed relative to the 2500 molecules in the atmosphere the CO2 molecule is hiding in?
By my rough calculations (using PV = nRT, with P = .5 atm, and T = 300) the 2500 N2s, O2s and others, take up about 2.04*10^-23 cubic centimeters, so should have one side on the order of 10^-8 cm.
So, if the “width” (the magnitude of the E and B fields?) is on the order of 15*10^-6m = 15*10^-4 cm, each IR photon would be 4 orders of magnitude “wider” than the 2500 molecules that each CO2 molecule is hiding in… so the very high absorption of 15 micron IR, as seen from satellites, would make sense…
Yes, I’ve been harassing/harassed by a denier who understands CO2 can absorb IR, but refuses to understand it happens significantly in the atmosphere…
thanks again for the site