There are many misconceptions about how atmospheric processes work, and one that often seems to present a mental barrier is the idea of How much work can one molecule do?
This idea – presented in many ways – has been a regular occurence in comments here and it also appears in many blogs with eloquent essays on the “real role” of CO2 in the atmosphere, usually unencumbered by any actual knowledge of the scientific discipline known as physics.
Well, we all need mental images of how invisible or microscopic stuff really works.
When we consider CO2 (or any trace gas) absorbing longwave radiation the mental picture is first of trying to find a needle in a haystack.
And second, we found it, but it’s so tiny and insignificant it can’t possibly do all this work itself?
How much can one man or woman really do?
This article is really about the second mental picture, but a quick concept for the first mental picture for new readers of this blog..
Finding a Needle in a Haystack
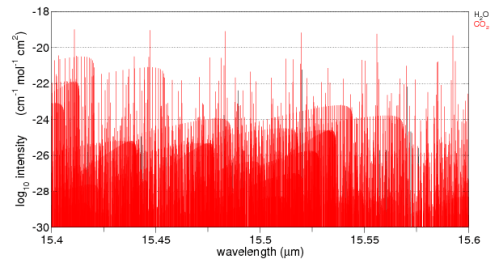
Think of a beam of energy around 15.5μm. Here is the graph of CO2 absorption around this wavelength. It’s a linear plot so as not to confuse people less familiar with log plots. Water vapor is also plotted on this graph but you can’t see it because the absorption ability of water vapor in this band is so much lower than CO2.
The vertical axis down the side has some meaning but just think of it for now as a relative measure of how effective CO2 is at each specific wavelength.
Here’s the log plot of both water vapor and CO2. You can see some black vertical lines – water vapor – further down in the graph. Remember as you move down each black horizontal grid line on the graph the absorption ability is dropping by a factor of 100. Move down two black grid lines and the absorption ability has dropped by a factor of 10,000.
Now, I’ll add in the absorption ability of O2 and N2 – the gases that make up most of the atmosphere – check out the difference:
Spectralcalc wouldn’t churn anything out – nothing in the database.
15.5μm photons go right through O2 and N2 as if they didn’t exist. They are transparent at this wavelength.
So, on our needle in the haystack idea, picture a field – a very very long field. The haystacks are just one after the other going on for miles. Each haystack has one needle. You crouch down and look along the line of sight of all these haystacks – of course you can only see the hay right in front of you in the first one.
Some magic happens and suddenly you can see through hay.
Picture it.. Hay is now invisible.
Will you be able to see any needles?
That’s the world of a 15.5μm photon travelling up through the atmosphere. Even though CO2 is only 380ppm, or around 0.04% of the atmosphere, CO2 is all that exists for this photon and the chances of this 15.5μm photon being absorbed by a CO2 molecule, before leaving this world for a better place, is quite high.
In fact, there is a mathematical equation which tells us exactly the proportion of radiation of any wavelength being absorbed, but we’ll stay away from maths in this post. You can see the equation in CO2 – An Insignificant Trace Gas? Part Three. And if you see any “analysis” of the effectiveness of CO2 or any trace gas which concludes it’s insignificant, but doesn’t mention this equation, you will know that it is more of a poem than science. Nothing wrong with a bit of poetry, if it’s well written..
Anyway, it’s just a mental picture I wanted to create. It’s not a perfect mental picture and it’s just an analogy – a poem, if you will. If you want real science, check out the CO2 – An Insignificant Trace Gas Series.
CO2 – The Stakhanovite of the Atmospheric World?
Back in the heady days of Stalinist Russia a mythological figure was created (like most myths, probably from some grain of truth) when Aleksei Stakhanov allegedly mined 14 times his quote of coal in one shift. And so the rest of the workforce was called upon to make his or her real contribution to the movement. To become Stakhanovites.
This appears to be the picture of the atmospheric gases.
Most molecules are just hanging around doing little, perhaps like working for the _____ (mentally insert name of least favorite and laziest organization but don’t share – we try not to offend people here, except for poor science)
So there’s a large organization with little being done, and now we bring in the Stakhanovites – these champions of the work ethic. Well, even if they do 14x or 100x the work of their colleagues, how can it really make much difference?
After all, they only make up 0.04% of the workforce.
But this is not what the real atmosphere is like..
Let’s try and explain how the atmosphere really works, and to aid that process..
A Thought Experiment
For everyone thinking, “there’s only so much one molecule can do”, let’s consider a small “parcel” of the atmosphere at 0°C.
We shine 15.5μm radiation through this parcel of the atmosphere and gradually wind up the intensity. Because it’s a thought experiment all of the molecules involved just stay around and don’t drift off downwind.
The CO2 molecules are absorbing energy – more and more. The O2 and N2 molecules are just ignoring it, they don’t know why the CO2 molecules are getting so worked up.
What is your mental picture? What’s happening with these CO2 molecules?
a) they are just getting hotter and hotter? So the O2 and N2 molecules are still at 0°C and CO2 is at first 10°C, then 100°C, then 1000°C?
b) they get to a certain temperature and just put up a “time out” signal so the photons “back off”?
c) other suggestions?
The Real Atmosphere – From Each According to His Ability, To Each According to His Need
What is the everyday life of a molecule like?
It very much depends on temperature. The absolute temperature of a molecule (in K) is proportional to the kinetic energy of the molecule. Kinetic energy is all about speed and mass. Molecules zing around very fast if they are at any typical atmospheric temperature.
Here’s a nice illustration of the idea (from http://www.chem.ufl.edu/~itl/2045/lectures/lec_d.html).
At sea level, a typical molecule will experience around 1010 (10 billion) collisions with other molecules every second. The numbers vary with temperature and molecule.
Think of another way – at sea level 8×1023 molecules hit every cm2 of surface per second.
Every time molecules collide they effectively “share” energy.
Therefore, if a CO2 molecule starts getting a huge amount of energy from photons that “hit the spot” (are the right wavelength) then it will heat up, move even faster, and before it’s had time to say “¤” it will have collided with other molecules and shared out its energy.
This section of the atmosphere heats up together. CO2 can keep absorbing energy all day long even as a tiny proportion of the molecular population. It takes in the energy and it shares the energy.
If we can calculate how much energy CO2 absorbs in a given volume of the atmosphere we know that will be the energy absorbed by that whole volume of atmosphere. And therefore we can apply other well-known principles:
- heating rates will be determined by the specific heat capacity of that whole volume of atmosphere
- re-radiation of energy will be determined by the new temperature and ability of each molecule to radiate energy at wavelengths corresponding to those temperatures
Conclusion
The ability of a CO2 molecule to be “effective” in the atmosphere isn’t dependent on its specific heat capacity.
Molecules have embraced “communism” – they share totally, and extremely quickly.
Update – New post on the related topic of understanding the various heat transfer components at the earth’s surface – Sensible Heat, Latent Heat and Radiation




I wish CO2 were on the chart with the other gases just for completeness.
Ok, thought experiment-
The average kinetic energy of all molecules at the same temperature being the same means that no molecule actually possesses more heat than any other species in the same lot, correct? All gases are equal, just more massive diffuse slower?
Mystery gas absorbs on a certain spectral line in the IR band.
If a molecule gets hit by this color photon, it is absorbed.
If the molecule emits the same color photon, there is no net transfer of energy and it’s therefore functionally identical to reflection.
If the molecule does not emit its signature photon, it immediately emits blackbody IR or bumps into another molecule and shares the energy until it has the same kinetic average as any other molecule.
Thus, any heat ‘trapped’ is not held within that molecule. It keeps reacting with regular kinetic and radiative transfers the same as every other molecule does.
If this molecule were not there then the portion of the spectrum it absorbs would either be absorbed by different molecules that can or debit the energy balance.
Flawz?
trivia:
cellulose calorific value 17520 kJ/kg
Therefore, whenever it’s not interacting with photons of a particular color, the mystery gas is perfectly neutral with respect to any other thermal transfer functions it is performing.
It is only when acting as a functional reflector of its particular wavelength that any difference in outcome may result.
Trapping a photon of its characteristic color and sharing it is but a picosecond delay because once it’s distributed such that it ends up as any other color IR, it’s not reflectable any more.
Any of the characteristic color that is emitted downward is functionally reflectance.
Floz?
Dave McK:
On average it’s true. If you look at the graph above there’s a distribution of speeds, so it’s a statistical thing.
If this happens it’s quite close to reflection. Close enough since we are talking about the basics.
The rest (1st comment) is all good from my first reading. No flawz.
The 2nd comment I didn’t totally understand. It sounds right from “the vibe”.
Hi,
I just came across this site. It looks pretty good. 🙂
You write:
[ Even though CO2 is only 380ppm, or around 0.04% of the atmosphere, CO2 is all that exists for this photon and the chances of this 15.5μm photon being absorbed by a CO2 molecule, before leaving this world for a better place, is quite high. ]
Doesn’t this support the ‘absorption to extintion’ argument which claims that all the absorbable LW radiation is already being absorbed and additional CO2 will only result in the heat transfer occurring closer to the ground?
Sorry if you’ve covered this elsewhere previously.
Thank you.
I was making a model in my mind where colored molecules are considered the same as particulate reflectors of a particular portion of spectrum.
This is supposed to help me conceptually isolate purely radiative properties of the circulation of energy, which I think was the thrust of the lesson.
It’s because my experience with gases is with distillation at various pressures, which is all about phase change. Studying the radiative heat transfer properties is basically new to me.
trivia: immerse fresh concrete in liquid CO2 and it becomes dense limestone almost immediately.
trivia2: the mortar in the middle of the great wall of china is not fully cured yet.
Mostly Harmless:
It’s a good question, no need to apologize for asking a legitimate question.
If that was the end of subject and that wavelength of 15.5um was the complete story, possibly.
The subject is a little bigger, this post was just, “how to visualize the fact the CO2 might be significant” rather than the complete picture. As always, getting the complete picture requires some time..
Take a look at the CO2 – An Insignificant Trace Gas? series. Seven parts.
The end result is that the equations which govern the processes of absorption and re-radiation across all wavelengths and through a vertical column in the atmosphere can be solved to calculate the effect of any change in any trace gas. These are the radiative transfer equations.
The calculated results are not intuitively obvious but match the observed values.
Without solving these equations, the result is an unknown. Many people claim to have solved the problem with no recourse to the maths. It would be like somehow getting a spaceship to Mars “by eye”…
This is a very good post. Thanks,
I will go back and review your CO2 gas series.
Now if I recall, energy transfers, whether kinetic or radiative, come at a cost.
That seems to me to be a significant issue.
Another would be that in your haystack analogy, there would be a point where haystacks would make an effective barrier.
On analogies..
Analogies can help – and hinder and distract.
It’s just a caricature that has been drawn. Not the real thing.
If there was a perfect analogy for CO2, everyone would “get it” and the CO2 series in Seven Parts would not really need to exist.
scienceofdoom,
Good point. AGW has certainly gotten a lot of mileage from caricatures.
To answer your question, I guess a molecule can do as much work as there energy to absorb.
Mr. Science of Doom,
I’m not sure if you know me from WUWT but I know you, and I was curious if you might consider a write-up I’ve been working on. You should have my e-mail now if you’re interested.
Best Regards
Sorry make that “consider reviewing a write-up I have been working on”
Typing on a mobile phone… RAWR 😦
I have always been confused about something … I thought it was the case that a CO2 molecule can only absorb a photon if the energy of the photon (which relates to its wavelength or frequency) matches one of the vibrational states of the molecule or promotes an electron to a higher excited state.
Have I got this part of the story wrong?
This would seem to indicate that CO2 can absorb one photon and then it has to sit out the dance until it gets rid of that quanta of energy. Is that the case?
So, how can it get rid of that quanta? It would seem that only the right sort of collisions can do that?
I’ll follow scienceofdoom’s answer on this one. I got interested too.
My non-expert intuintion says it does not depend on collisions, though. It’s just the emission of electromagnetic energy, that can happen in the vacuum. Let’s see.
Richard Sharpe:
I really like the way things are explained here:
http://www.electro-optical.com/eoi_page.asp?h=Education
Everything radiates in the blackbody spectrum all the time via ‘vibrational modes’.
Spectral lines relate to electron shells and are quantized.
That color property is independent of temperature property.
Richard Sharpe:
On your first part you are correct. On the second part, I think you are right but I’m not 100% sure, it’s a very in-depth subject.
How does quantized vibrational/rotational energy translate into kinetic energy of molecules it collides with?
I would expect – from quantum theory – that it would need to translate this quantized energy into the same energy in another molecule in a collision.
There is a distribution of energies for the molecules it is mixing with (see graph above). If my expectation is correct, then it there would be a statistical calculation of how long before such a collision occurred at any given temperature and pressure.
However, in my next trip to the university library I will see if I can dig out something specific from some weighty tome.
I expect that some elegant experiments have been done to isolate these exact points – probably 50-100 years ago..
Watch this space..
It would be very interesting to know what fraction of monochromatic absorption could be angular momentum vs kinetic energy.
It is my understanding that in CO2 lasers N2 is excited via electrical discharge to a level where they can transfer a quanta of energy to CO2 (one of the vibrational modes of CO2, I believe), and these excited CO2 molecules can then be induced to emit coherent light …
So, it would seem that CO2 can transfer its energy to N2 in the atmosphere, of which there is plenty …
However, I wonder about fractionating, since H2O is lighter than N2 which is lighter than C02 … and I wonder if H2O can accept energy from excited C02 molecules …
That led to a fun tour through the books.
I didn’t know N2 had any part in CO2 lasing and also learned that the laser transitions are actually on vibration-rotation bands, not the electron.angular.momentum kind of photons. (surely there’s a proper word for that but I don’t know it)
You can vaporise water with a CO2 laser. I have a 25W that I don’t get to play with enough.
Richard Sharpe:
I started doing some calculations of energy of a transition (1×10^20J) and what molecular speed change that would mean in a H20 molecule and then realized you appear to have already answered most of the question.
N2 and O2 make up the bulk of the atmosphere. If CO2 can transfer rotational/vibrational quantized energy to kinetic energy of O2 and N2 then you have demonstrated that this process takes place. H2O, as 1% of the atmosphere, will be at the same temperature as the surrounding molecules of O2 and N2.
Still it would be nice to know the gritty details – a few equations and tidy results – showing e.g. % CO2 molecules at sea level, at 200mbar, at 1mbar that share energy thermally vs re-emit (before sharing thermally) – and how the molecular weight of recipient molecule affects that.
I expect it is just tied into the Maxwell-Boltzmann distribution of energy of gases at the relevant temperature, along with probabilities of collisions (dependent on pressure). But I will see if I can find out more – unless visiting experts to this blog can lay it all out..
I’m on an island, so it’s really not laziness that keeps me from a library to find proper references, but If I recall the basics, a photon that is absorbed in such a way as to change an electron’s orbit to a higher state adds strictly to potential energy and the energy so stored can only be re-emitted in the same way (quantized, monochromatic).
Thus it is not measurable as temperature in the same way that latent heat of vaporisation exists as potential energy in a water molecule but does not change the temperature.
That’s why I was finding the ‘reflection’ model useful.
Thus, apart from those re-emitted photons of CO2’s spectral signature, CO2 is just another molecule except for specific heat, which has raised a question for me:
Does the specific heat of a molecule hide as latent potential too?
Alexandre:
If that was the case then energy could not be shared. The reason is that a CO2 molecule, for example, can only emit radiation at specific wavelengths. And O2 and N2 for example are transparent at these wavelengths so they would have no way to absorb any energy.
Here’s one explanation from someone who appears to know what he’s talking about.
I don’t know how to link to the actual comment where it’s explained – http://rabett.blogspot.com/2009/08/getting-rudys-back-next-latest-issue-of.html – but the actual comment is about halfway down – EliRabett said… and the time of 3:01 PM.
That was a nice discussion by EliRabett of a condition known as Local Thermodynamic Equilibrium. He introduces this concept in this post.
—
A minor quibble about a comment above re. the inability of O2 to absorb or emit radiation in the optical/IR. Unlike N2, O2 has a permanent magnetic dipole moment. Quantum mechanically speaking, these are much less probable and therefore much weaker transitions than the electric dipole transitions which characterize the vast majority of the IR molecular transitions. (The problem with molecules like N2 and O2 is that they are homonuclear diatomic molecules — too danged symmetric in charge distribution.)
There is a vibrational-rotational band near 6.4 microns mediated by electric quadrupole transitions, and a the vast forest of (weak) transitions lying beyond 30 microns. Astronomers who do ground based optical/near-IR spectroscopy are intimately acquainted with the weak O2 bands between ~0.6-0.8 microns, especially the twin nuisances: “B-band” near 0.687 microns and the “A-band” near 0.765 microns. These result from vib-rot transitions atop electronic transitions (I think).
Nevertheless, O2 is most certainly a minuscule player in the opt/IR. The electronic transitions of O2 in the UV are much more important sources of atmospheric opacity.
So, we have a CO2 molecule. It has just absorbed a 15.5um photon and that has moved it to an excited vibrational/rotational state (I am not sure what that 15.5um quanta does, so could use some illumination 🙂
Now, how many ways can it give up that energy?
1. It can re-radiate it, if the mean free path is longer than the mean time to re-radiate.
2. It can transfer that energy to N2, it seems, and boost and electron in that N2 to some excited state above the ground state. Perhaps I have it wrong here.
However, can it transfer that energy to another molecule it meets as velocity so that that the temperature of the gas the CO2 is embedded in increases? I dunno, which is why I am asking.
I read Rabett’s explanation (thanks for the link), but I’m sorry to say that it’s largely beyond my reach.
I understood the time the molecule takes to collide is usually less than the time it take to re-emit the photon. That was interesting, I did not imagine that.
However, it’s not clear to me how a vibrating molecule transmits more kinetic energy to others.
The process where vibrational energy is transferred as kinetic energy is called V-T collisional relaxation. It is not particularly efficient, maybe 1 in 1000 collisions, but the average collision time at atmospheric pressure is ~ 0.1 ns. so you get a time of ~ 10 microseconds.
If you want a model of what happens, think of the collision partner approaching one end of a vibration molecule, the atom on that end which is moving because it is vibrationally excited “strikes” the other molecule and the energy is transferred to kinetic energy. Think billiards.
“I understood the time the molecule takes to collide is usually less than the time it take to re-emit the photon. That was interesting, I did not imagine that.”
I didn’t imagine it either- but I’ve found literature that says transfers of energy are primariy radiative and that conduction is not the dominant means.
With various opposing assertions, I’m stuck on confusion and can’t progress until certain of these details are resolved.
Might have to drop the class until I’m better prepared.
On Conduction:
Molecules transferring energy by collision doesn’t mean that conduction of heat is taking place.
Conduction of heat would be taking place if there was a net transfer of heat between two points. (Think of molecular collision as microscopic while conduction is a larger scale process).
Gases are very inefficient at conducting heat compared with the other methods of heat transfer of radiation, convection and latent heat.
The exception for conduction is at the boundary layer of the earth’s surface where the transfer of heat to the very lowest layer of the atmosphere is by conduction and is significant. Convection then ensures that heat is moved up through the atmosphere.
Energy Modes of a Molecule:
At the molecular level an individual molecule has energy from kinetic energy and also vibrational and rotational energy.
All of these modes together make the energy of one molecule.
Exactly how we picture the process of energy transfer (vibrational energy state being converted to kinetic energy of another molecule) taking place I don’t know. Picturing molecules with quantum energy states (in vibrational and rotational modes) isn’t particularly easy either, although I’m more comfortable with that “mental picture” from studying some quantum mechanics.
I found a few books that simply state that this is what happens without attempting to provide the appropriate mental picture.
“Handbook of Atmospheric Science” by Hewitt and Jackson (2003) –
Thermodynamics books just describe the internal energy of a molecule as the “translational motion” plus internal rotational and vibrational motions – then state that “during collisions a transfer of energy takes place”.
– If I find something which describes lays it out in more detail I will share it.
Thanks, ScienceofDoom.
“Molecules transferring energy by collision doesn’t mean that conduction of heat is taking place.
Conduction of heat would be taking place if there was a net transfer of heat between two points.”
This helped – to realize that collisions can take place where there is no net transfer (similar to reflection, in that regard). Any transfer of kinetic energy, though, seems to belong included in the definition of conduction, still, and the measurement ‘temperature’ refers only to that.
My private lexicon must have no self contradictory elements and each definition must refer to an objective entity, so I have some chores to do before I’m ready to approach the topic of weather or climate again. My cognitive tools are inadequate at this time. I can detect elisions but not resolve them.
heh – it all comes back to that freakin double slit, doesn’t it?
Maybe making a little headway. I have to work with what I already know to hook things in-
so I’m experimenting with electonic analogies- see what you think:
temperature is like voltage
specific heat is like capacitor with a resistor.
latent energy of phase change is like a capacitor with a forward diode and reverse diode that drops the voltage so it can’t discharge while the rest of the system is at higher voltage.
opacity is radiative resistance
absorption of a photon into an electron orbital is a quantum capacitor with back to back zener diode
absorption of a photon by rotational or vibrational modes is like raising the voltage
transfer of energy is conduction
conductivity is conductivity
heat we feel is the work done is the watts.
Woot! It’s always a glorious moment when I finally make sense of something. It’s like that last turn of a rubik’s cube where everything is suddenly where everything should be.
Thanks ScienceofDoom for providing the atmosphere. I had to bmob to quench my thirst, but it was a pleasant venue to do it.
May you all find your rosetta and make plenty sense – otherwise there will continue nothing but expense.
Arrivederci.
Oh- this article was inappropriately titled, I think, as it doesn’t deal with what is classically known as ‘work’. Amirite?
Ciao.
A question!
A molecule absorbs a photon
It is now more energetic/warmer – is this causing it to vibrate or have electrons jumped a level?
If vibration; then energy can be shared with other molecules. If this is the case then doesn’t re-radiation occur at longer and longer wavelengths as the blackbody temperature has been reduced by the sharing of energy?
If electrons have jumped then their return should emit a photon of the same wavelength – no energy loss? This will be emitted in any direction at random. Is it true to assume that further collisions will occur to those emitted in directions close to the horizontal until after enough collissions 50% go up and 50% go down (on a disc world)?
If it is not a flat earth then a larger proportion of the horizontal photons will go up to space is this correct?
Or am I totally confused?
Sorry more than 1 question
Thanks
Mike
thefordprefect:
I feel like I am the more I think about it.
The standard works just state that the transfer of energy takes place via collision – with no qualifications.
When a molecule which has been excited to a higher energy state (whether vibrational or rotational) experiences a collision (or the right kind of collision) the energy must be transferred into kinetic energy without a photon emission. Otherwise the process would be that the molecule returned to its original energy state via re-emission of exactly the same wavelength photon as it originally received. And there would be no thermal effect on the atmosphere.
Sounds like strictly speaking it is true, someone who enjoys integrals of solid angles can tell us the exact answer.
The complete treatment of the radiative transfer equations includes integration of emissions over all solid angles (rather than just assuming 1/2 up and 1/2 down).
It is some comfort to see that I’m not the only one to have trouble understanding the vibrational-kinetic energy transfer between molecules.
It looks like that’s how it works – you can make house experiments observing the warming of an air bottle with more CO2. If this vibration did not affect its speed (directly or indirectly) this would not happen. But how it works seems to be more complicated.
One question: does a vibrating molecule have more kinetic energy? Does it move faster than otherwise?
About the sphere-disc issue.
Does gravity play a role there? If yes, then the sphere curve could be offset by it… just an amateur speculating.
CO2 and radiative forcing….
Hello, it’s me again, catching up on you commendable posts, having been away for a while…..
Maybe you’re leading up to this. One thing I have not been able to get any light on, since I first came across it in IPCC 2007 Technical Summary, is “HOW DO THEY WORK OUT THE VALUE OF RADIATIVE FORCING ?” Can it be calculated from first principles and the concentration and properties of the gas involved, or is it determined empirically, or a bit of both?
The second question is of course the one I raised with you before, and that is, “HOW IS RADIATIVE FORCING RELATED TO CHANGES IN TEMPERATURE?” I have always presumed that there was some simple relationship, (for if not, what use is it ?! ) but as I pointed out earlier, the obvious calculation (to me) gave a delta-T quite a bit lower than seems to have happened.
A further one I would be much interested in, if you can get around to it, is the vexed question of feedbacks.
Thanks again for you informative postings!
James McC:
From the radiative transfer equations – see
CO2 – Part Three – for the absorption and emission equations
Part Five – how the radiative convective model is constructed, which uses these equations for a known temperature profile.
These equations are mostly derived from first principles. The properties of the gases (absorptivity and emissivity) are empirically determined – although apparently can also be derived from first principles via quantum mechanics.
The convective structure of the atmosphere is not derived from 1st principles but observed.
I made up a method which didn’t work as you pointed out. The real calculation relies on the radiative convective model – as described in Part Five but I believe that the right way to do the “simple” calculation is to use the top of atmosphere (troposphere) radiative forcing compared with top of atmosphere temperature. This is a reasonable approximation because there is a reasonably constant “lapse” rate (temperature profile) in the troposphere. Therefore, an increase in the top of atmosphere temperature causes a similar temperature change at the surface.
So (following Part Seven) – Tnew^4/Told^4 = (239 + 3.7)/239, where Told=255. Then Tnew = 256K – a 1K, 1’C increase.
This is just a rough and ready method and I haven’t spent the time to find out how good a method it is…
Keep getting sidetracked from this popular subject. Soon..
Alexandre:
No. Kinetic energy is separate from internal energy states.
I don’t understand the question.
Scienceofdoom,
I was speculating about fordprefect´s question. He said that since the world is a sphere, and not a disc, more than 50% of re-emited energy would go “up”. Perfectly horizontal, tangential emissions would be directed towards space, instead of the ground. Subtle downward angles would also escape to space because of the Earth´s curvature.
My amateur question was: does gravity have any influence there? Would more than 50% of re-emissions occur downwards because of gravity, somewhat compensating the loss of energy because of this spheric curvature?
Actually, my own guess is no. Earth´s gravity is too weak for that kind of effect.
And my other guess is that the curvature issue is moot, too, since each photon won´t travel too far to meet another CO2 molecule. Over short distances the statistical relevance of this curvature would be negligible.
Articulating the question better made me think of it in a clearer way, too…
CO2 and radiative forcing…
Thanks for your comments! You new calculation make sense to me, as now does the reference to the tropopause in the definition of radiative forcing … i.e. we calculate the change in mean terrestial radiation temperature. There will then be the same change to the surface temperature, if the lapse rate is constant. Of course it is the broadest possible approximation, but it hangs together at that level.
I look forward to your postings on feedback.
Richard Sharp:
There is a 3rd way that the molecule can give up the energy called Stimulated Emission. This is where an electron will drop energy levels and emit a photon when in the presence of the electromagnetic radiation (photon) of the same frequency.
This is how lasers generate monochromatic coherent waves.
I have never seen it’s role discussed in conjunction with atmospheric CO2 absorption/emission. But it would definitely occur.
Scienceofdoom:
“When a molecule which has been excited to a higher energy state (whether vibrational or rotational) experiences a collision (or the right kind of collision) the energy must be transferred into kinetic energy without a photon emission. Otherwise the process would be that the molecule returned to its original energy state via re-emission of exactly the same wavelength photon as it originally received. And there would be no thermal effect on the atmosphere.”
This is where I get stuck as well. Molecular collisions are consideration inelastic, as long as blackbody radiation is not allowed to carry energy away from the system. If blackbody radiation is allowed then all the collisions become inelastic.
The use of the elastic collision in calculations is really just an average of the system – half are inelastic, and the other half are “super-elastic” to varying degrees.
To me this posses a question: if a molecule absorbs a photon, increasing its energy level, is involved in a inelastic collision, losing some energy (but not the full amount equal to the energy of a photon at its characteristic lines) how can it absorb any more photons, until such time as it has returned to the proper (photon absorbing) energy level?
Thus, no more photons can be absorbed (shoulders included) while the molecule is in the partial energy state.
The only way a photon can be absorbed is if the energy jump of the molecule matches the energy level of the incident photon. And these levels are well defined for atmospheric molecules.
However, the energy transfer of elastic collisions between disparate molecules – to my knowledge – does not adhere to these levels.
Another tentative question:
When I expose CO2 to longwave radiation, do the molecules vibrate only or do they accelerate as well?
When you expose a bottle of CO2-rich air to heat, it warms more. So it should accelerate the molecules, right?
Is this relevant?
“Effect of multiple encouters to vibrational to tranlational energy transfer”
The title looks suggestive, but I can’t find the full text or even abstract.
a few typos corrected:
“Effect of multiple encounters on vibrational to translational energy transfer”
Snider 1994 – Canadian Journal of Chemistry
This one is available online, although not specific to V-T energy transfer:
Vibrational Energy Transfer
Flynn et al. 1996
Hi SoD.
No one seems to have added a calculation so here goes.
The wavelength I have chosen is around 4um why?
Well the 4um one is one that co2 does not share with H2O and so can be used as a kind of co2 identifying flag.
Photon energy E=hf with the usual meanings
E =4.97×10^-20J
The translational KE of a molecule at 300K is 6.21×10^-20J
Increase in KE of co2 after absorption = 8 fold
I know its a massive jump and when I first saw the result I was surprised.
The rotational and vibrational modes would be triggered by the absorption.
So the co2 molecule is now like someone who has just won the lottery.
He finds that he has lots of “new friends”.
At 10^10 collisions per second has excess energy is soon shared out by equipartition of energy and co2 molecule returns to average value.
So we see that co2 is very good at transferring heat to other gas molecules.
How is it at cooling in the reverse process?
Well to get enough energy to emit a 4um photon the co2 will have to accumulate eight times the average KE of its neighbours.
Not very likely.
So the “backradiation” from this particular wavelength will be almost non existant.
Bryan:
How did you calculate the probability?
scienceofdoom
“How did you calculate the probability?”
Well its just the reverse of the absorption calculation.
The co2 molecule would have to have eight times the kinetic energy of the average molecule at that temperature to emit 4um photon.
A more sophisticated method would be maxwell Boltzman distribution curve.
The speed would have to be 8^0.5 of RMS speed =2.83V(rms)
If you look at a curve you will see its getting remote!
Bryan:
And what’s the probability per collision?
And the probability of it taking place in 1 second below 60km in the atmosphere?
A similar but less extreme results occur for the 15um band that co2 shares with H2O.
Here the emission requires just over twice the average KE or a speed of 1.46 V(rms).
taken altogether, emission is a lot less likely than absorption.
Further there is the fact that the emission can go in any direction in a x,y,z three dimensional space then we would expect “backradiation” to be a lot less than surface up radiation.
Bryan:
So, are you saying that emissivity of CO2 is not equal to absorptivity at 15um?
And what does “less likely” mean in numbers?
scienceofdoom
“So, are you saying that emissivity of CO2 is not equal to absorptivity at 15um?”
Emissivity<<absorvancy
If they were equal the net result would be zero.
For the 4um emission the chances are less than 0.1%
Effectivily it just does not happen.
There will be a tendency for co2 and H2O to radiate (if at all) at the longer wavelengths.
This means that the so called "backradiation" has almost no effect on the surface on the planet.
This is why this radiation cannot be ulilised to do work.
Therefore it should not be described as heat.
This is the central point that G&T make.
Bryan, Bryan, Bryan what are you thinking?
According to Kirchoff’s law, emissivity = absorptivity for a given substance at the same wavelength (and for some surfaces direction needs to be defined also).
So you don’t agree with Kirchhoff’s law. Foundational thermodynamics.
And even when you claim that Kirchhoff is wrong you are mixing up your arguments and concepts.
If you claim, contrary to Kirchoff and basic thermodynamics that the emissivity<< absorptivity of a particular trace gas at a given wavelength, that doesn't solve the problem you have.
Because we can measure the downward longwave radiation from the atmosphere, e.g.:
So you need to explain how it happened, seeing as emissivity of CO2 is so low. Why is the 15um band so strong? (Wavenumber 667cm-1 = 15um band for those not using to seeing wavenumbers)
(By the way, of course the 4um band for CO2 from the atmosphere will be so weak because the Planck function for a 288K surface has less than 0.2% of energy below 4um even for a blackbody. And much less for the colder atmosphere.)
And now it has happened – 15um radiation radiated downward at the surface of the earth – you have to explain why the ground doesn’t absorb it.
So far:
a) in discussions on another blog you claimed that, the radiation “reaches” the ground – but never explained why it wasn’t absorbed
b) suggested that this downward longwave radiation wasn’t radiated by the atmosphere, but instead reflected – but couldn’t explain why there was so much energy in the 15um band – so apparently dropped that line of argument
c) now claiming that Kirchhoff went wrong and emissivity doesn’t equal absorptivity to dig yourself out of the hole – not realizing that it only shows you can’t explain where this radiation comes from
d) now claiming that “this is why” the radiation has no effect on the surface of the earth – the reason appears to be because it can’t have been generated in the first place, even though it is..
I don’t even think G&T will be proud of you.
But you are doing fantastic work explaining to everyone reading this blog how weak your argument is.
Now you have surely led up to the grand finale where you explain why this downward longwave radiation doesn’t get absorbed by the earth’s surface.
Don’t keep us in suspense any longer. Now is the time.
scienceofdoom
“According to Kirchoff’s law, emissivity = absorptivity for a given substance at the same wavelength (and for some surfaces direction needs to be defined also).”
You have a particularly naive view of heat transfer.
When a green plant absorbs some em radiation to make starch does it simultaneously emit the same radiation?
You have not grasped the fact that if the absorption of 4um radiation was exactly balanced by the emission of 4um radiation the net effect would be zero.
Which would shoot a massive hole in the case for AGW.
If the absorption of the radiation can lead to other energy changes, like increase in KE which can then be transferred as in example above.
The lengthening longwave radiation is absorbed/emitted by the surface as well as being scattered from the surface and so on.
The net effect of the absorption is for all practical purposes zero.
“(By the way, of course the 4um band for CO2 from the atmosphere will be so weak because the Planck function for a 288K surface has less than 0.2% of energy below 4um even for a blackbody. And much less for the colder atmosphere.)”
You have made the case for G&T.
The surface emits 4um but the atmosphere cannot recipricate.
When a green plant absorbs some em radiation to make starch does it simultaneously emit the same radiation?
Aren´t you maybe confusing black body radiation with a molecule absorption/radiation?
A green plant absorbs sunlight and emits at other frequencies, depending on its temperature (Planck function, muddied up by the emissivity curve).
The GHG radiation happens at specific wavelengths, being absorbed and re-emited by the molecules in the same frequencies. I guess you could say the net effect is zero here (the molecule does not retain that energy for long, as is soon back to its original state), but that does not mean this does not change the way that energy moves through the atmosphere.
Alexandre
The fact that the energy of 4um em photon absorber by CO2 molecule is changed into KE is generally agreed by both sides in the AGW debate.
The KE is then shares out via collisions is also agreed.
My point is that it is very unlikely that the CO2 molecule will ever again build up eight times the average KE required to emit a 4um photon.
Bryan
The fact that the energy of 4um em photon absorber by CO2 molecule is changed into KE is generally agreed by both sides in the AGW debate.
The KE is then shares out via collisions is also agreed.
I don´t know about the sides of this debate, but it´s fine that we agree about this here.
My point is that it is very unlikely that the CO2 molecule will ever again build up eight times the average KE required to emit a 4um photon.
Sorry, I don´t think I get it. As I understand it, the molecule won´t have to “build it up” again. It will only re-emit the photon whenever the molecule absorbs one again, be it a 15um photon coming from the heated planet surface, or a “second-hand” re-emited photon from another molecule.
(I chose 15um because that´s where OLR is mostly absorbed by CO2. The same should apply to 4um if CO2 absorbs any there.)
(And I´m just a layman trying to think along.)
Alexandre
The reason I focused on 4um is that it is not shared by H2O and hence can be used as a marker for CO2.
This can then be examined for in a composite spectrograph.
The co2 will not store the photon as such, if that what you mean.
This idea comes from thinking electrons move to a higher energy level.
This is not the mechanism here.
The co2 will be more likely to absorb a 15um photon but H2O is the big player at that frequency.
What we see generally in any heat transfer process is relatively high quality thermal energy moving in such a way that entropy is increasing.
High temperature surface shedding heat to lower temperature surfaces.
High energy EM radiation moving to lower energy EM radiation.
All this in accordance with 2nd law of thermodynamics.
This is G&Ts central point.
Bryan
The co2 will be more likely to absorb a 15um photon but H2O is the big player at that frequency.
Modtran seems to show something different. The CO2 “bite” at the middle of the OLR spectrum (around 15um) remains very obvious no matter how much H2O you add to it.
So does Hitran too in its spectral data…
Bryan
Emissivity = absorptivity for a given material at the same wavelength and direction.
Emission does not equal absorption in most cases because the temperature of the source body is not equal to the temperature of the receiving body.
Emissivity is not the same as emission.
Emissivity is a property of the material and a function of wavelength and direction.
Emission is the actual thermal radiation leaving the body.
But your confusion over these basics suggests the need for a new article on these topics. Watch out for it.
Don’t forget to update us on what happens to that 15um downward longwave radiation that reaches the earth’s surface.
scienceofdoom
“Emission does not equal absorption in most cases because the temperature of the source body is not equal to the temperature of the receiving body.”
Yes so the 4um em from surface can heat up the atmosphere.
The chance that a 4um from the atmosphere reaching the surface is vanishingly small.
Just as G&T state and you have agreed above.
There is a better chance of the co2 emitting 15um wave but the vast majority of 15um is from H2O.
So we see statistically at each radiative transfer there is a tendency to degrade the radiation i.e. to move to longer and longer wavelengths.
When these longer wavelengths such as 15um( and longer) are absorbed by the earth the effect is so small it can be ignored.
I don’t expect to convince bryan sweeney of anything, but for those passers-by..
Bryan’s “explanation” isn’t a clear one and mostly he never actually deals with particular problems or questions but just “moves on”.
I have asked numerous questions which have been ignored.
1. Kirchhoff’s law states that emissivity = absorptivity at a given wavelength and direction but Bryan hasn’t actually finished up to explain whether Kirchhoff was wrong – just accused me of “thermodynamic naivety”.
Most people believe Kirchhoff is right. Including naive people like myself.
2. Bryan didn’t understand the different between emissivity and emission but doesn’t stop to admit this. It’s very basic thermodynamics.
What’s important for passers-by is to note the lack of engagement.
3. Bryan hasn’t explained why, if his “explanation” of absorptivity/absorption and emissivity/emission by trace gases in the atmosphere is correct (which it’s not), why it is that downward longwave radiation matches the standard climate science conclusion (from solution of the radiative transfer equations).
(Note: Bryan is totally wrong about water vapor having a higher absorptivity/emissivity than CO2 at 15um but what’s the point of engaging on this one).
4. Bryan hasn’t explained what happens to this downward longwave radiation when it “reaches” the earth’s surface. Why isn’t it absorbed? This question has been asked many times. But never answered.
Not much point trying to “engage”. But probably worth a follow up post on the basics for the silent visitors.
Sod says
Bryan hasn’t explained what happens to this downward longwave radiation when it “reaches” the earth’s surface. Why isn’t it absorbed? This question has been asked many times. But never answered.
I said
When these longer wavelengths such as 15um( and longer) are absorbed by the earth the effect is so small it can be ignored.
SoD says
(Note: Bryan is totally wrong about water vapor having a higher absorptivity/emissivity than CO2 at 15um but what’s the point of engaging on this one).
I say
Well why is it that in Antarctica when water vapour tends to be very small and yet CO2 is still there at normal levels the backradiation tend to zero
Click to access TownEtAl_2005.pdf
Bryan:
Claims to have answered the question by saying..
Here’s the downward longwave radiation for one iconic example:
And the reason why 15um radiation, when absorbed is “so small” is?
“Bryan said so“?
Perhaps a longer explanation would be in order.
Of course, note that the explanation already contravenes what Bryan has explained of the G&T hypothesis. Which is that “thermal radiation from the colder to the hotter would be in contravention of the 2nd law”
Even a “small enough to be ignored” amount would be surely in contravention of the imaginary 2nd law of thermodynamics
scienceofdoom
The second law of thermodynamics is really based on statistics.
A simple example.
An small ice cube is placed in a much larger beaker of hot water.
The ice cube melts and the water temperature drops.
The resultant water temperature can be very accurately calculated if initial temperatures and masses are know.
The use of the first law of thermodynamics is used here.
The first law would not be violated if the reverse procedure happened.
In other words if the ice cube reformed, but as we all know this is unlikely in the extreme.
This is the domain of the second law.
Odd situations arise where the 2nd law might seem to be in danger such as the Radiation Frost example I gave you in another thread.
The air temperature sometimes being higher that the ground temperature but its thermal capacity is not enough to affect ground conditions.
In the case of conduction from a hot surface to a colder surface there must be situations when a particular outlier from the more energetic end of the colder surface meets an outlier from the less energetic end of the hotter surface and energy is transferred from cold to hot.
Warm winds from the south will heat up the atmosphere of Britain.
Cold winds from the north will do the reverse.
When we get down to practical situations the Second Law is not violated.
Statistical example again.
If a can of coke is poured into an Ocean there will be a theoretical rise in the depth of the Ocean.
The rise however will be so small that it is not capable of being measured and can be ignored.
Karl Popper would go further and say if it cannot be measured we cannot say it even exists.
I would not go as far as Karl with this one.
Alexandre
“The co2 will be more likely to absorb a 15um photon but H2O is the big player at that frequency.
Modtran seems to show something different. The CO2 “bite” at the middle of the OLR spectrum (around 15um) remains very obvious no matter how much H2O you add to it.
So does Hitran too in its spectral data…”
I say
Read the paper below.
In Antarctica when water vapour tends to be very small and yet CO2 is still there at normal levels the backradiation tend to zero
Click to access TownEtAl_2005.pdf
This is the result found by real researchers carrying out experiments.
I think your sources are really computer programs.
I know which source I would rely on.
Bryan
Read the paper below.
In Antarctica when water vapour tends to be very small and yet CO2 is still there at normal levels the backradiation tend to zero
I did not read the entire paper. It´s quite long, I don´t have all that time available and I´m quite sure it would be much of a wasted effort, since my background is not that good. As I said, I´m just an interested layman.
I read the abstract and conclusion, though, and paid due attention to graphs and tables. I did not find anything that would support the claim “In Antarctica when water vapour tends to be very small and yet CO2 is still there at normal levels the backradiation tend to zero”.
Quite the opposite. The graphs match the spectral data from the models I linked (not surprisingly), and the abstract and conclusion say CO2 backradiation (which I take is the same as Longwave Downwelling Flux) stays pretty constant throughout the year.
Table 7, for example, shows 23 W/m2 in winter and 35 W/m2 in summer. The conclusion has this sentence:
“The decrease in atmospheric temperature from summer to winter explains nearly all the decrease in CO2 flux and the remaining flux due to water vapor.”
Do you have any other reason to believe these widely and commercially used models are just a product of fiction?
Alexandre
The amount of water vapour in the air is very small in the Antarctic, hence the fall off in “backradiation”.
The brief summary on page 4235 states that even for “clear skies” (that is no cloud) the ratio of water vapour/co2 is 2:1
If there were clouds and by implication more water vapour the ratio would increase significantly.
The computer programs (of which there are several) could not predict the climate for the next year never mind the next 50.
The paper by G&T go into the topic of computer programs dealing with climate models.
People need to get out into the real world with instruments to find out what’s really happening.
Bryan
The brief summary on page 4235 states that even for “clear skies” (that is no cloud) the ratio of water vapour/co2 is 2:1
“No cloud” (droplets) dos not mean “no H2O” (the gas, the part that does the GHE as molecular absorption and scattering).
If there were clouds and by implication more water vapour the ratio would increase significantly.
No, that´s different than no H2O, as I mentioned above. And your assertion by no means explain the largely remaining CO2 LDF in winter, or the paper´s own conclusion that it´s largely explained by the drop of temperature itself. What do you think about this?
The computer programs (of which there are several) could not predict the climate for the next year never mind the next 50.
Ok, now it got confusing. You´re dismissing “computer programs” as a whole, mixing up climate models, radiative/convective models, and programs that only generate a graph with the known, empiricaly achieved absorption coefficients.
Even the paper you link resorts to “modeled” radiation (Fig. 2). Does that mean we should dismiss it too?
On top of that, you seem unaware of the difference between initial value problems and boundary condition problems. No one is trying to predict if the year 2058 will be an El Niño year, for example, but just the overall trend.
Indeed, people need to get out into the real world with instruments to find out what’s really happening. When they do that, what they write down and conclude afterwards also deserve attention.
Alexandre
The statement should read
The radiative flux ratio of water vapour/co2 is 2:1
Alexandre
“Even the paper you link resorts to “modeled” radiation (Fig. 2). Does that mean we should dismiss it too?”
Short answer – Yes
Only trust real experiments.
This topic is riddled with backers of one particular climate model or another.
The climate models each have at least one computer program to make projections of the future climate.
Most of these programs have been developed over several years.
If three years ago the funding agencies had stipulated that the programs should make testable predictions and if they failed their funds would be cut off.
Not one program would survive.
G&T prove by mathematical means that the task is beyond the capability of the programmers and computers.
So far G&T have been proved correct.
Bryan:
I have special rules for people who post non-stop repetitive comments or questions without reading answers or responding to them.
So far only one commenter has fallen foul of this arbitrarily-applied rule but I think you might be the second.
The rule generally gets applied when the bulk of the posts are coming from one person.
Most of mine are questions which you never actually answer. It doesn’t seem to bother you. You make claims that you don’t back up. It doesn’t seem to bother you. This has been the case on other articles and other blogs.
You make mistakes that you don’t respond to or acknowledge.
This makes it hard for these posts to be a useful resource for others. This is because on the one hand I don’t want to have a non-stop bryan-scienceofdoom counter but also I don’t want to leave your comments there unacknowledged.
To some extent I am happy to have you embarrass yourself this way, but please, have a think about whether you want to be able to post on science of doom.
Answer questions responsibly or you won’t be able to post. Or don’t answer questions responsibly but just post occasionally.
SoD
I am quite content to let readers draw their own conclusions about who is embarrassing themselves.
I have answered your oft repeated questions several times but perhaps you don’t like or understand my answers.
You seem to be slipping into the pattern of attacking the person rather than the comment so frequently adopted by supporters of AGW.
Bryan
The paper you show as reference confirms CO2 downward radiation throughout the year, with a moderate seasonal variability nearly entirely explained by temperature change itself. Table 7 is an easy reference for that.
How does this fact fit into your original idea that CO2 downward radiation “tend to zero” when there’s no H2O? That’s an unanswered question (for one).
G&T prove by mathematical means that the task is beyond the capability of the programmers and computers.
According to your understanding, what’s the difference between a model’s calculations and G&T’s “mathematical means”? A calculation is only reliable if done by hand?
Alexandre
….”How does this fact fit into your original idea that CO2 downward radiation “tend to zero” when there’s no H2O? That’s an unanswered question (for one).”
Backradiation figure on worldwide basis around 342 W/m2
Figures from quoted paper go as low as 23W/m2
See source below
….G&T prove by mathematical means that the task is beyond the capability of the programmers and computers.
According to your understanding, what’s the difference between a model’s calculations and G&T’s “mathematical means”? A calculation is only reliable if done by hand?….
You should read the G&T paper it is available as a free download.
Basically the main problem seems to be that the climate throws up several non linear differential equations which don’t have specified boundary conditions
Bryan
Backradiation figure on worldwide basis around 342 W/m2
Figures from quoted paper go as low as 23W/m2
Not exactly. This worldwide average figure is the total LDF, including all GHG and probably also clouds. Te 23 W/m2 figure is just CO2, clear-sky. Let’s compare apples with apples here.
Total clear-sky backradiation in Antarctica is “in the range of 110-125 W/m2 during summer (December–January) and 60–80 W/m2 during winter (April–September)” (your reference, in the abstract).
This paper analyses LDR in Ontario Canada, finding rather similar results: 24.8 W/m2 for CO2. I guess ScienceofDoom has a post on this paper.
So, again: no support for the claim that when H2O drops, CO2 backradiation “tends to zero”.
Basically the main problem seems to be that the climate throws up several non linear differential equations which don’t have specified boundary conditions
I’m sure there’s a lot of the real world that is oversimplified in climate models. They still do a poor job predicting regional climate change and decadal prediction, for example (remember: just an interested layman speaking).
Initial value input is still insufficient, and computing capacity is an important limitation in this kind of job.
But they’ve already achieved some pretty good results predicting global patterns like the polar amplification (Arrhenius said that already in the 19th century) and the overall temperature trend (Rahmstorf 2007).
So it’s not like there’s any support to throw the whole science out claiming CO2 does not obstruct IR (which is a reasonable implication of your “tend to zero” claim). This kind of basics always gets confirmed in observations.
Alexandre
“So it’s not like there’s any support to throw the whole science out claiming CO2 does not obstruct IR (which is a reasonable implication of your “tend to zero” claim). This kind of basics always gets confirmed in observations.”
To clarify my reply was meant to show that H20 was the major player in the radiative flux downwelling radiation.
The paper from Antarctica shows that even when there is low humidity and in clear skies conditions, the flux from H2O is twice that of CO2.
Also the total backradition readings (for instance August of 41.6W/m2) is a small fraction of the average global 342W/m2.
Also the authors say that the uncertainty in the readings is 8W/m2 so the reading might have been as low as 33.6W/m2
I did not intend that impression that I had meant that the readings would ever be actually zero.
Quite apart from H2O and CO2 radiance there will also be the effects of elastic scattering and Moonlight to be considered
Bryan
I think it’s clearer now, thanks.
The difference made by man-enhanced atmospheric CO2 is just a few W/m2 (the difference, not the total backradiation). It is a known fact that CO2 is just the second place in the most important greenhouse gases, being H2O the first. Here’s a reference. Even so, changing the second-biggest GHG is not innocuous.
If I remember right, doubled CO2 forcing alone would raise global temperatures in 1ºC. The accepted overall sensitivty (i.e. including feedbacks) is calculated to be around 3ºC. So the direct role of CO2 is known to be a small part of the whole.
There’s a very extensive peer-reviewed literature about quantifying this sensitivity, starting in the late 19th century and continuing until today, through different lines of evidence, reaching similar results.
Click to access EngrCritique.AGW.Science.3.pdf
I know it’s not peer-reviewed (might be a good thing), but it does cast doubts on current levels of co2 being lower now than during the past 2000 years.
justcherrypicked
Good post interesting comments on computer models page31=>37 and the hockey stick page 60
[…] Note 2 – The subject of molecules of CO2 and water vapor absorbing energy and sharing this energy by collision with other molecules close by was covered to a limited extent in How Much Work Can One Molecule Do? […]
Alexandre
Thanks for the reference Ive printed it off.
I find it easier to read that way.
Alexandre
I have now had a chance to read the paper reading by Kiehl and Trenberth.
Click to access KiehlTrenbBAMS97.pdf
On page 200 we find “The emissivity of low and middle level clouds is assumed to be 1.
..for high level clouds emissivity is set at 0.6″
Note these figures seem to be plucked out of (pardon the pun) ‘thin air’.
Instead of using real experiments this pair seem largely to be using computer models and tweaking the parameters to achieve some preconceived structure.
It is little wonder that none of these models stand up to any reality check.
On page 206 we have the famous diagram of the energy budget.
Notice that the values given are mostly given to three significant figures.
The figures in the diagram have since been revised and for example the Earth surface radiation now is given as 396.1 W/m2 that is to four significant figures.
Now any experimenter from an exact science will know that you should not claim any more accuracy in your final result than was used to calculate that result.
Which in the case of the diagram(page 206) should be to one significant figure.
The emissivity numbers I quoted above from the article are to one significant figure.
The other emissivity numbers I have seen are to two significant figures (Science od Doom examples).
This rule if applied makes the diagram unusably crude.
Yet that is the blunt reality of the situation.
This report would not be acceptable if presented in a first year physics lab.
One other problem with the Earth Surface Radiation figure when used in the Stefan Boltzmann equation for 15 degree Celsius temperature.
It gives an emissivity of well over one which is physically impossible.
Bryan
First of all, I’d like you to become aware of how closely you scrutinized this paper, and in contrast how easily you accepted the “engineer’s critique” (it’s so superficial you can’t even call it a “study” or “paper”, be it on the first or last year of a graduation). Attention is good, just use it when analysing contrarian claims as well.
About the cloud emissivity: he assumed two figures, one for low and another one for high cirrus. It’s an explicit assumption, and you’re right to question whether these are valid figures.
I found this reference about that, and figures seem to range from 0.4 to around 0.8 (although I don’t trust my own judgment on the subject. My knowledge is not enough to interpret it safely. There may be some missed underlying assumption that is relevant)
But the figures we were talking about were clear sky ones. So I would think cloud emissivity is irrelevant on this issue.
Then you do a quick digression on their motives “tweaking the parameters to achieve some preconceived structure”. It would be more scientific to say “these assumptions are not valid because of this or this”. Or you could show some other paper that finds something different with more plausible assumptions. But you did not do this. (oh, and again: cloud emissivity seems to be irrelevant to clear-sky figures)
About the Earth Surface Calculation: my amateur calculations show 15ºC is indeed the temperature that yields 390 W/m2 irradiation. Could you please ellaborate on your claim?
Alexandre
Thank you for your reference and once again I will print it off and get back to you.
“tweaking the parameters to achieve some preconceived structure”. It would be more scientific to say “these assumptions are not valid because of this or this”
If we look at any reference to emissivity figures(even SODs) we quickly find that real figures are less than one.
There is a need for the utmost accuracy in trying to prove one way or another if AGW exists.
As Lindzen ha pointed out over the last weekend;
There has been a rise of about 0.7 degrees C over the last 150 years.
So any rounding up of the figures could give the impression that a problem existed when a more accurate figure might prove the opposite.
“About the Earth Surface Calculation: my amateur calculations show 15ºC is indeed the temperature that yields 390 W/m2 irradiation. Could you please ellaborate on your claim?”
Yes your calculation is correct and to get that figure you had to use the “perfect black body” figure of e=1.
Now use the same equation to find out what value of emissivity corresponds to the new revised figure of 396.1W/m2
[…] of joy because in three other articles on this blog (The Imaginary Second Law of Thermodynamics, How Much Work Can One Molecule Do? and On the Miseducation of the Uninformed by Gerlich and Tscheuschner (2009)) and one on another […]
Alexandre
I think you have come to the same much the same conclusion as myself about the cloud emissivities.
I would put them in a range of 0.8,0.6,0.5,0.3.
I f we compare these figures with the Keihl, Trenberth paper we have;
1,1,0.6.
A glance tells you that these two sets are widely different.
The K-T numbers appear to be guesses.
However this paper features at the top of the IPCC case and I feel its just not good enough.
On computer models I have further concerns.
Equations and laws are used that are themselves not entirely reliable.
I think if you read the paper below on the robustness of Kirchhoff’s Law you will have some concerns.
At the end of the day only experiments will lead us to the physical processes underpinning the climate.
Click to access PP-19-01.PDF
[…] with… I’m not going to go into detail on that. There’s a ton of places, like here, where you can look up a detailed explanation. But when you consider the scale issues, it should be […]