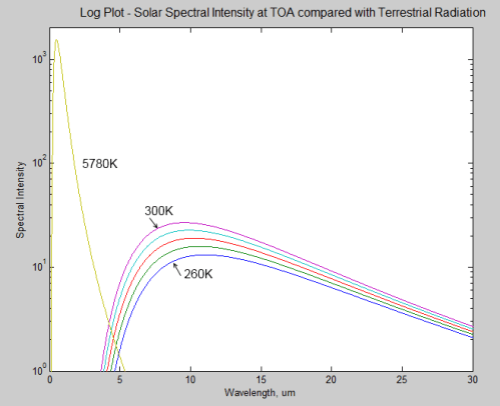
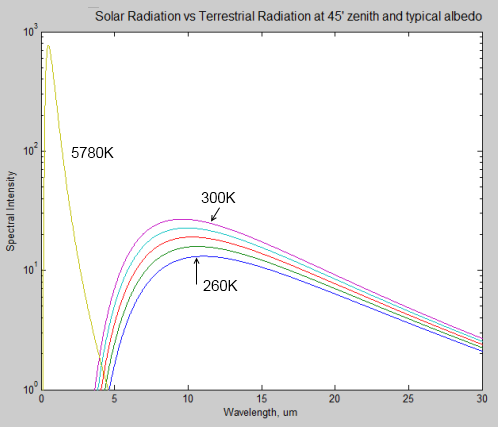
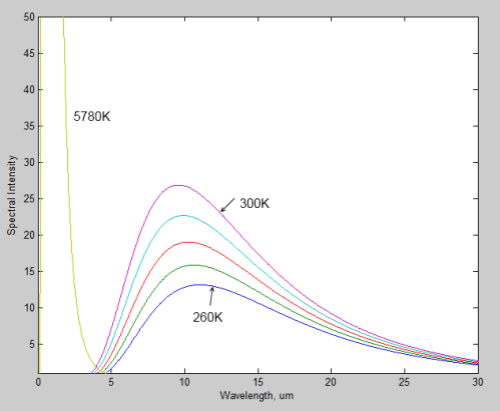
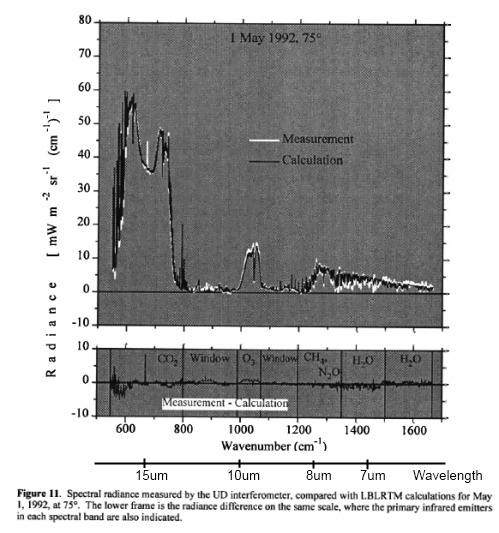
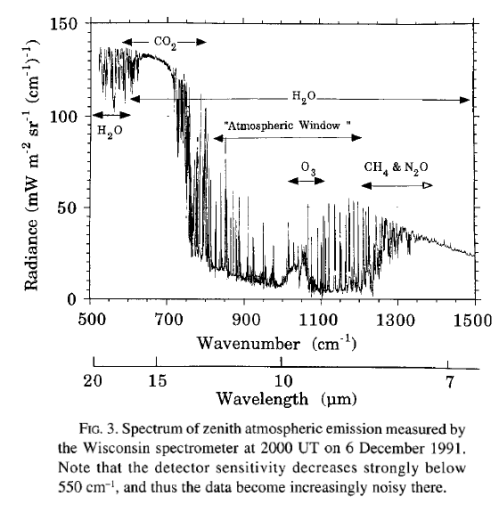
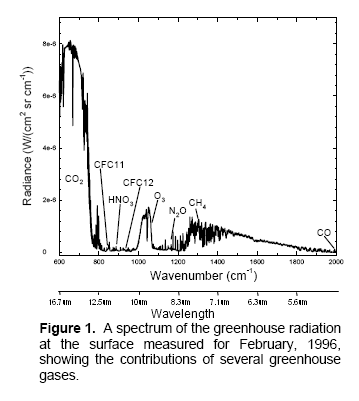
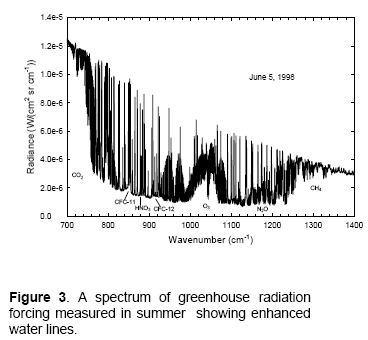
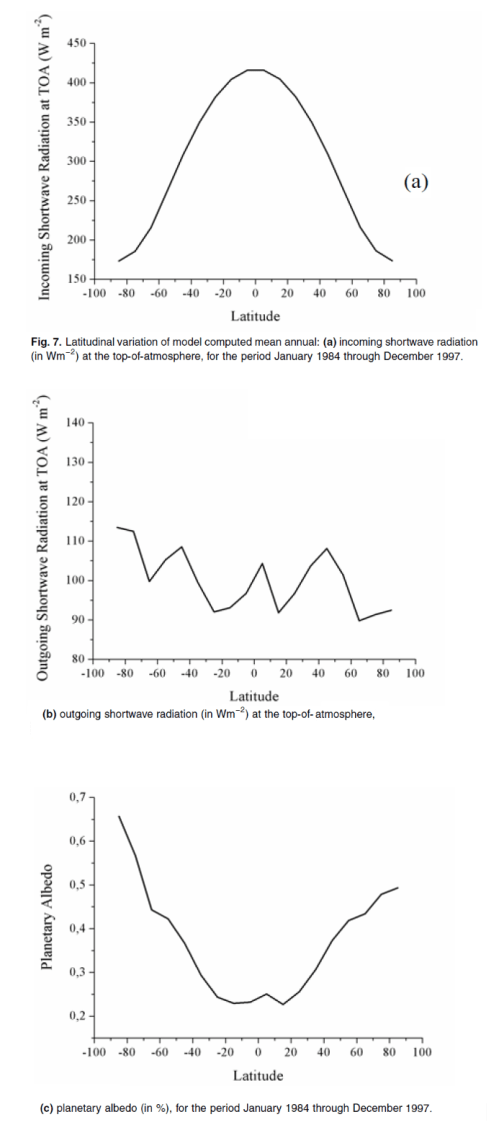
The surface of Venus is around 730K (457°C) – why is it so hot? Although there is still much to learn about Venus the basics have some simple explanations.
Energy Absorbed from the Sun
While earth is 150M km from the sun, Venus is only 108M km away, a ratio of 0.72.
The “solar constant” (as it has been historically called) at the earth is 1367 W/m², and, as energy intensity is proportional to the square of the distance away, or “r²”, the solar constant at Venus is 1367/(0.72²) = 2,636 W/m² – Venus is closer, so it receives more solar energy per m².
The earth has an average albedo of about 0.3, meaning that 30% of the solar radiation is reflected. This is by clouds, by aerosols in the atmosphere, and by the surface of the earth. (Watch out for “The Earth’s Energy Budget – Part Four – Albedo”). If Venus had the same albedo as the earth, the energy absorbed per m² of surface area would be, E = 2,636 * (1-0.3) / 4 = 461 W/m². [corrected – thanks to Bill Stoltzfus for pointing out my mistake]
For an explanation of why the value is divided by 4, see The Earth’s Energy Budget – Part One
This value equates to an “effective radiating temperature” of 300K (27°C). This is nothing like the surface of Venus. [corrected as well – thanks to Bill Stoltzfus for pointing out my mistake]
In any case, it turns out that Venus has a much higher albedo than the earth, with an albedo of 0.76 – meaning that 76% of the solar energy is reflected.
Redoing the calculation, E = 2,636 * (1-0.76) / 4 = 158 W/m² – which equates to an “effective radiating temperature” of 230K (-43°C). The same calculation for the earth gives 255K (-18°C) – see CO2 – An Insignificant Trace Gas? – Part One.
So in terms of a simple energy balance with the sun, Venus should be colder than the earth.
In the case of the earth, as laid out in the CO2 series, the reason the surface of the earth is so much warmer than predicted from simple energy balance is because various trace gases, including water vapor and CO2, absorb the upward radiation from the earth’s surface and reradiate it in all directions. As some of this is downward, the surface of the earth receives more energy than it would without these gases and so it is hotter. See CO2 – An Insignificant Trace Gas? Part Six – Visualization for some more insight into this.
So is Venus so much warmer at its surface because of the inappropriately-named “greenhouse” effect? Or is it for other reasons?
The Venusian Atmosphere
The atmosphere of Venus is quite unwelcoming for us earth-dwellers. The atmosphere is mostly CO2 (97%), with the balance made up mostly of nitrogen (N2), and trace amounts of water vapor and many other gases in minute quantities.
The mass of the Venusian atmosphere is around 100 times that of the earth, and consequently the pressure at the surface of Venus is much higher – at 92bar compared with 1bar for the earth.
Now some people say that the reason for the high temperature at the surface of Venus is because of the high atmospheric pressure and the depth of the atmosphere. For example, Steve Goddard on Wattsupwiththat and echoed by Lubos Motl. This explanation isn’t one that you can find in atmospheric physics text books.
A Quick Review of the Earth’s Surface
This is just to explain a few basics for some perspective.

From Trenberth and Kiehl (1997)
There’s much of interest in this diagram from Earth’s Annual Global Mean Energy Budget by Trenberth and Kiehl (1997) but we’ll focus on a few key elements for the purposes of this article.
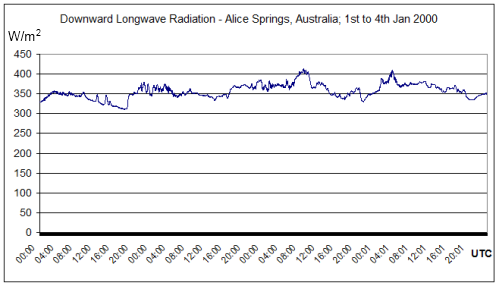
The surface of the earth receives an average of about 170 W/m² from solar energy (with an additional 70W/m² of solar radiation absorbed by the atmosphere). The earth’s surface also receives an average of 324 W/m² of radiation from the atmosphere. So in total the earth’s surface receives about 490 W/m² (annual global average).
Now the average radiation from the surface of the earth is 396 W/m² (or 390 W/m² in the diagram above, which is close enough for our purposes). Convection and conduction remove the balance of around 100W/m². If you take a look at Tropospheric Basics you can see more about the temperature profile in the troposphere (lower atmosphere) and why convection is a more effective re-distributor of heat within the troposphere.
The principal point is that the warming of the air from the surface radiation, conduction and convection causes the air to expand. Air that expands is less dense, and so this air rises, moving heat by convection.
The temperature profile, or lapse rate, from convection can be easily calculated, both for dry air and moist air. Dry air is just under 10°C/km, while moist air depends on the amount of water vapor, but can be as low as 4°C/km. (And the environmental lapse rate, or what we find in practice, is around 6.5°C/km).
So in the case of the earth’s surface, it would be radiating out 490W/m², but for the fact that conduction and convection remove some of this energy from the surface, and then convection redistributes this energy up into the atmosphere.
The Surface of Venus
Energy radiated from a surface is proportional to the 4th power of absolute temperature. This is known as the Stefan-Boltzmann law but visualizing the 4th power of something isn’t that easy. However, calculators are readily available and so if you punch the numbers in you will see that for a surface of T=730K with an emissivity close to a blackbody:
E = 16,100 W/m² – compare this with the surface of earth (288K, 15°C) of around 390 W/m²
This is over 40x the energy radiated from the surface of the earth – for a temperature only 2.5x greater. That’s the real world, very non-linear.. (And note that if the emissivity is not equal to 1, the energy radiated is simply the value above multiplied by the emissivity).
So if we think about the top of atmosphere of Venus, it is radiating round about 158 W/m². This balances the absorbed solar radiation. And yet the surface is radiating 16,100 W/m² – does the high pressure of the Venusian atmosphere explain it?
No
Think about it like this. For the surface of Venus to be radiating at 16,100 W/m² it has to be receiving this energy from somewhere. It receives a maximum of 158 W/m² from the sun (if all of the solar energy absorbed is absorbed in the surface and nothing in the atmosphere).
The explanation from others about a temperature gradient between the surface and the tropopause (top of the tropopause or lower atmosphere) only explains anything when the surface heats the atmosphere from below. In that case the atmosphere heats up, expands and rises – moving energy via bulk movements of air.
Can the atmosphere create heat from pressure and transmit this heat to the surface?
In the case of the earth’s surface, the extra radiation to the earth’s surface (caused by the “greenhouse” effect) heats the atmosphere from beneath and causes convection – with a lapse rate (or temperature profile) of between 4 – 10 °C/km. And convection moves some of this heat from the surface up into the atmosphere. In the case of Venus the argument that relegates the role of the “greenhouse” effect and promotes the role of atmospheric pressure doesn’t have a heat transfer mechanism.
Picture the starting condition where the surface is very cold. What heats it up?
There are three ways of moving heat – radiation, convection and conduction. Conduction in gases is extremely low and anyway the top of the atmosphere is around 230K – if the surface starts off colder what causes heat to flow to the surface to create such a huge emission of radiation?
Convection needs to work by warming a gas from below. Where is this mechanism if the surface is not already heated by the “greenhouse” effect?
And radiation has been ruled out (as the main mechanism) in these arguments from Steve Goddard and others.
How Can the Surface Get so Hot? An Over-Simplified Climate Model
Let’s take a look at the ignored radiation and the super “greenhouse” effect.
How can a surface get so hot from “back-radiation”? Isn’t that just as crazy an idea?
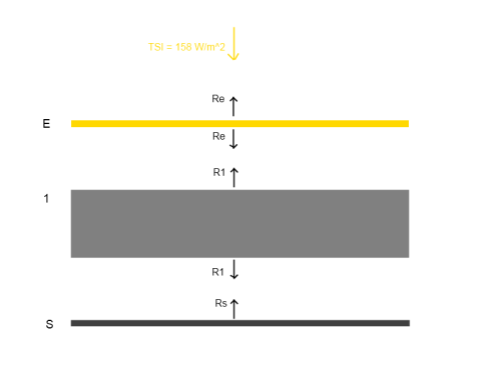
We will take a simple idea – as all models are at their start, just to demonstrate a point. There’s a little bit of maths, unfortunately, but possibly (if you haven’t seen this concept before), the concept might actually seem harder to grasp. Here’s a very simple (and not very realistic) model of a planetary surface and atmosphere (idea from Radiation and Climate from Vardavas and Taylor (2007)):
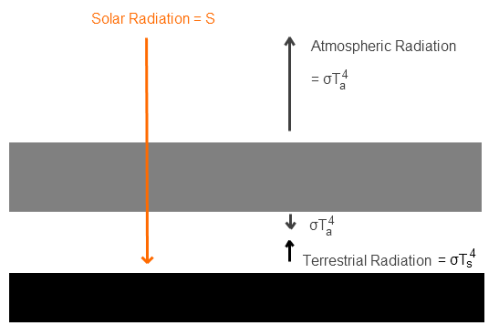
Simple climate model - atmosphere perfectly transparent to solar radiation, and totally opaque in the infra-red
The surface receives radiation from the sun, S. In the case of Venus this value would be (averaged across the surface), S = 158 W/m².
Now the surface is at Ts and radiates to the atmosphere, which heats it up. The atmosphere is perfectly transparent to solar radiation, but totally opaque in the infra-red and all at one temperature, Ta. Therefore, the atmosphere radiates σTa4 upwards and σTa4 downwards.
Note that “totally opaque” means that no surface radiation makes it through this layer of the atmosphere. In this scenario we can calculate the surface radiation. If you are new to this kind of model, it is easiest to follow the small amount of maths against the graphic. First, using the simple energy balance at top of atmosphere, the outgoing radiation at the top of atmosphere equals the absorbed solar radiation averaged over the surface of the earth:
σTa4 = S [equation 1] – this is the Stefan-Boltzmann law, where σ = 5.67 x 10-8
Second, the surface radiation balances the energy received at the surface – which is from the sun and the atmosphere:
S + σTa4 = σTs4 [equation 2]
Therefore, substituting [1] into [2], we get:
σTa4 + σTa4 = σTs4
2σTa4 = σTs4, or 2S = σTs4
and solving we find, Ts = (2S/σ)1/4
In the case of S = 158 W/m², Ts = 273K
Now effective temperature at the top of atmosphere is 230K, so an opaque atmosphere has increased the surface temperature significantly – but not to 730K. (Barton Paul Levenson has a model like this, commented on in CO2 – An Insignificant Trace Gas? Part Eight – Saturation)
Now with a very optically thick atmosphere, we simply add more and more layers to our model. The equations get slightly harder to solve, but each time we add a new totally opaque layer the temperature rises yet more.
For example, with 3 totally opaque layers the solution to a similar set of equations (with 4 equations and 4 unknowns) is:
Ts = (4S/σ)1/4, or Ts = 328K
It should be easy to see how the surface temperature gets extremely hot from radiation with many layers of opaque atmosphere (yet transparent to solar radiation).
So the Surface Temperature is Infinite, you Dummy!!
Well, if we can keep adding layers, and each one just increases the “back radiation” anyone can see that this can go on forever and the temperature will be infinite!
Obviously the model is wrong..
Not quite (well, if we could keep doing this, the model would be wrong). In the model above we have one totally opaque layer of atmosphere. But once we add multiple layers we are effectively dividing up the real atmosphere and saying that each layer is totally opaque. As we keep sub-dividing the atmosphere into more and more layers eventually they start to get optically thin and the radiation from the layer below will not be completely absorbed.
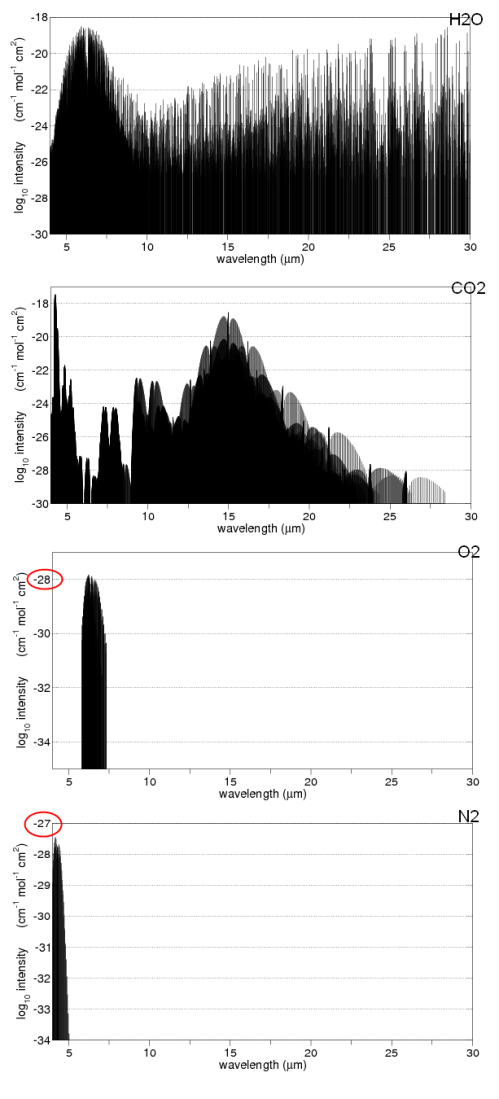
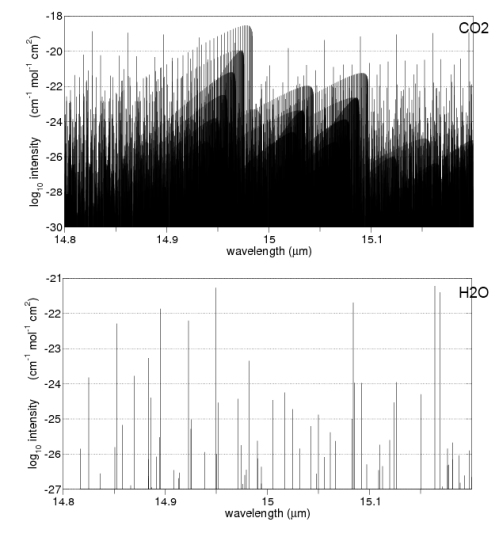
Radiation from CO2
All the above model does is demonstrate how the presence of significant radiatively absorbing gases can significantly increase the surface temperature. A 97% CO2 atmosphere is different from the model above for two reasons:
- CO2 doesn’t absorb at all terrestrial wavelengths so it isn’t a perfect absorber
- convection will moderate the surface temperature increase – once induced by radiation – as with the earth’s surface
So to calculate the effect of the CO2 atmosphere we have to solve the radiative transfer equations, which you can see in CO2 – An Insignificant Trace Gas? Part Three and Part Five (and the whole series).
These are fundamental equations of absorption and emission, but aren’t really solvable on a pocket calculator – despite so many people appearing to do just that in so many blogs. Note as well that CO2 spectral lines broaden with pressure so that CO2 (and water vapor) become a much more effective absorber in the lower Venusian atmosphere than the earth’s.
And we have to consider that once we have very high temperatures at the surface, convection will begin to move heat more effectively. This essentially moderates the effect of radiation.
But for those who believe that high Venusian atmospheric pressure and the ideal gas laws cause the high 730K surface temperature – they have to explain how the heat is transferred to the surface so that it can radiate at 16,100 W/m².
Real Solutions
One early approach to using real atmospheric physics on this problem was by James Pollack (reference below) in 1969 who showed that that plausible amounts of water vapor and the very high levels of CO2 could explain the high temperatures – using the radiative transfer equations and a convective model.
Bullock and Grinspoon (reference below) did this more recent calculation of the temperature profile in the Venusian atmosphere:
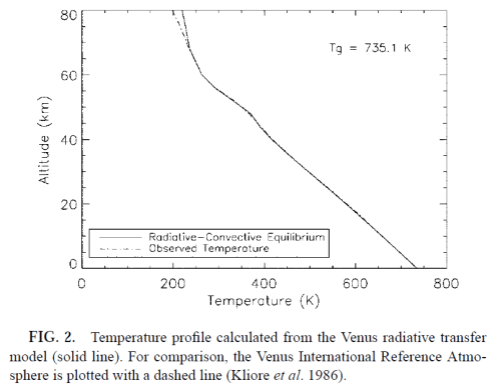
And they note a few possible reasons for the divergence above 70km. The model produces this spectrum of outgoing radiation:

The Planck function for an “effective radiating temperature” of 232K is shown. Note that the much higher levels of flux (in comparison to the 232K curve) demonstrate that at lower wavelengths (higher wavenumbers) the atmosphere is less opaque. This tends to limit further temperature rises, as the presence of any “window” regions allows a higher surface temperature to radiate out efficiently to the atmosphere.
A Mental Model
One mental model for people new to the inappropriately-named “greenhouse” effect is to think about the sun as an internal heat source, and CO2 as some kind of insulator.
Picture an ambient temperature of 20°C and a surface which has a constant internal heat source. As you add more and more insulation around this surface the temperature will keep rising – as heat is less able to flow away from the surface. For a given insulation there will be an equilibrium temperature reached that we can calculate, and it will be a function of the properties of the insulation.
Even though the temperature might reach 100°C or 200°C doesn’t mean that energy is “created” in this model – and this is probably clear to everyone.
Whether or not mental models “work” doesn’t change the realities of physics, but of course everyone wants to understand a subject conceptually.
Conclusion
Venus follows the same physical laws as the earth, so explaining the high surface temperature should be possible, even though many details of the atmosphere of Venus are hidden from us.
Some people who have attempted to explain the high Venusian surface temperature have used the ideas about the relationships between pressure and temperature in ideal gases without the strong “greenhouse” effect of a 97% CO2 atmosphere.
However, these ideas seem to lack a heat transfer mechanism whereby the surface of Venus can radiate at 16,100 W/m². This is the missing element in ideas which eliminate or relegate the role of CO2. In contrast, high surface temperatures in very strongly absorbing atmospheres can be explained using the radiative effects. A simple model can demonstrate very high temperatures, but a thorough calculation does require solution of the radiative transfer equations.
Update – New articles – Convection, Venus, Thought Experiments and Tall Rooms Full of Gas – A Discussion
Venusian Mysteries – Part Two
Reference
A Nongray CO2-H2O Greenhouse Model of Venus, James Pollack, Icarus (1969)
The Recent Evolution of Climate on Venus, Mark Bullock and David Grinspoon, Icarus (2001)
Read Full Post »