Do Trenberth and Kiehl understand the First Law of Thermodynamics?
Yes
But many people claim that they don’t after reviewing their well-known diagram from their 1997 paper, Earth’s Annual Global Mean Energy Budget:
The “problem” is – how can the absorbed solar energy be 235W/m2 when the radiation from the surface is 390W/m2? Where is this energy coming from?
Clearly they have created energy and don’t understand basic physics!
There have been many comments to that effect on this blog and, of course, on many other blogs.
Instead of pointing out that many of these values can be easily measured and checked, we will turn to a simple experiment which might help the many who believe the answer to the title is “No“.
The Thought Experiment
Picture, for the practical among you, building some kind of simple heat chamber in your garage. A wooden or a plastic box, with a light bulb in the center. You want to test whether some new gizmo really works at the high temperatures claimed. Or you want to find the melting point of gold.
The principle is simple – the thicker the material and the higher the energy from the bulb – the hotter it will get inside the heat chamber.
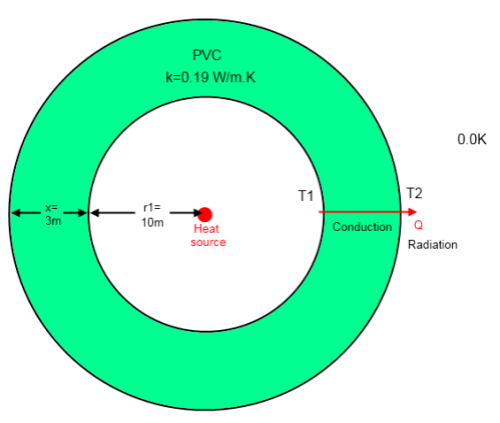
As a method of simplifying the calculations, my chamber will be spherical (because it makes the maths easier than when it is a box) and we will place it in the vastness of space and assume that the ambient temperature is 0.0K. Again, this is just to make the maths simpler to understand.
The inner radius of the sphere is 10m, and the thickness of the wall is 3m. (In a followup comment or post I will show how the values change with x).
The material used for this experiment is PVC which has a thermal conductivity of 0.19 W/m.K – I’ll explain a little more about this parameter in a moment. Probably down at such low temperatures the thermal conductivity won’t be this value but it doesn’t matter too much. We will also assume that the emissivity = 0.8.
You can see on the diagram that the outer surface temperature is T2, the temperature inside the sphere is T1 and the “ambient” is 0K. We don’t yet know what T1 or T2 is, we want to find that out.
In the center, we have our super-light-bulb, which radiates 30,000W. It is mysteriously powered, perhaps it is a nuclear device, or just electric with such a thin power cord we can’t detect it – we don’t really care.
Now – the first law of thermodynamics – energy cannot be created or destroyed. So for our thought experiment, the system receives 30,000W. The “system” is the entire PVC sphere, and everything it encompasses, right to the outer surface. No other source of energy can be detected.
Start Your Engines
Now that we have turned on the energy source the inside of the sphere will heat up. It has to keep heating up until the energy flow out of the sphere is balanced by the energy being added inside the sphere.
How does heat flow out from the center of the sphere?
- First, by conduction to the outer surface of the sphere
- Second, by radiation from the outer surface of the sphere to the vastness of space
Both of these processes are governed by very simple equations which are shown in the maths section at the end. Here, I will just attempt to explain conceptually how the processes work.
We start with consideration of the complete system and after equilibrium is reached the energy gained will be equal to the energy lost.
Energy gained, q = 30,000W = Energy lost
(Note that we are considering energy per second). For a rigid stationary body in the vastness of space, the only mechanism for losing energy is radiating it. All bodies radiate according to their temperature and a property called emissivity. Using the Stefan-Boltzmann law, we can calculate that:
Outer surface temperature, T2 = 133K
With this temperature, at an emissivity of 0.8, the whole sphere is radiating away 30,000W.
Time Out
So at this point, surely everyone is in agreement. We have calculated the steady-state temperature of the outer surface of the sphere as 133K. We can see that the mysterious energy source of 30,000W is balanced by the outgoing 30,000W radiated away from the outer surface.
The first law of thermodynamics is still intact and no one has to fight about anything..
Systems check?
But What’s the Story Inside?
We also want to calculate T1, the temperature of the inner surface. This is also very easy to calculate. The only mechanism for transferring heat from the inner surface (where the energy source is located) to the outer surface is by conduction.
The maths is below but effectively heat is transferred through a wall when there is a temperature differential between two surfaces. The higher the differential, the more heat. And the property of the material that affects this process is called the thermal conductivity. When this value is high – like for a metal – heat is transferred very effectively. When this value is low – like for a plastic – heat is transferred much less efficiently.
Once the system is generating 30,000W internally the inner wall temperature will keep rising until 30,000W can flow through the wall and be radiated away from the outer surface.
If we use the simple maths to calculate the temperature differential we find that it is 290K.
That is, to get 30,000W to flow through a hollow sphere with inner radius 10m and outer radius 13m and conductivity of 0.19 W/m.K you need a temperature differential of 290K.
Which means that the inner surface is 423K.
Everyone still ok?
What is the Radiation Emitted from the Inner Surface?
With an emissivity of 0.8 and a surface temperature of 423K, the inner surface will be radiating at 1,452 W/m2.
So the total radiation from the inner surface will be 1,824,900W.
What??? You have created energy!!!
Before bringing out the slogans, find out which step is wrong. If you can’t find an incorrect step then perhaps you should consider the possibility that this system is not violating the first law of thermodynamics.
Well, perhaps everyone is comfortable with the idea that with sufficient insulation you can raise the inner temperature of a box or sphere to a very high value – without having to build a power station.
In any case, the system is not creating energy. Inner surfaces are receiving high amounts of radiation while also emitting high amounts of radiation – they are in balance.
And of course, this has nothing whatever to do with the earth’s climate system so everyone can rest easy..
Update – Do Trenberth and Kiehl understand the First Law of Thermodynamics? – Part Two
And new article on the real basics – Heat Transfer Basics – Part Zero
Maths
The System
In equilibrium, the outer surface of the sphere has to radiate away all of the heat generated internally. This is the first law of thermodynamics.
The internal energy source, q = energy radiated from the outer surface
q = εσT24.4πr22 – the Stefan-Boltzmann equation for emitted energy per m2 x the surface area
where r2 = radius of the outer surface = 10 + 3 = 13
If q = 30,000W, r2 = 13m and ε = 0.8
T2 = q / (εσ.4πr22)1/4 and so T2 = 133K
If you recalculate back using the Stefan-Boltzmann law you will find that 133K with an emissivity of 0.8 radiates at 14.2 W/m2 (corrected-thanks to John N-G) and if you multiply that by the surface area of 4×3.14×132 = 2,124 m2 you find the emitted energy = 30,000W.
Conduction through the Sphere
The equation of heat conduction is very simple:
q = -kA . ΔT/Δx
This is for a planar wall. For a hollow sphere the equation is quite similar:
qr = -kA . dT/dr = – k (4πr2) . dT/dr and the important point is that qr is a constant, independent of r
After a small amount of maths we find that:
qr = 4πk . (T1 – T2) / (1/r1 – 1/r2)
So for the values of k = 0.19, r1 = 10, r2 = 13:
T1 – T2 = 290, therefore, T1 = 423K
Internal Radiation
Therefore, the radiated energy from the inner surface will be 1,452 W/m2 or a total of 1,824,900W (= εσT14.4πr12).



I understand that you’ve assumed the thickness of the thing to be 3m. I don’t quite understand why you designate it as x to be honest (the calculations are rather confusing this way around).
Good point, I drew the diagram first and then started the maths. At the end I was too lazy to change the drawing, but I have now updated the drawing.
And later I will look at how the values change with x.
x=3 has all the hallmarks of a variable that it turned out to be convenient to make fixed 🙂
“I don’t quite understand why you designate it as x to be honest (the calculations are rather confusing this way around).”
Because you can then see what happens when you make the chamber thicker.
But even so, at 3m thickness, you can see that there is more power inside than the light bulb produces.
You could calculate the thickness that makes the power emitted by the interior face of the PVC equal to 30,000W.
Perhaps the
q_r = 4πk . (T1 – T2) / (1/r1^2 – 1/r2^2) should be
q_r = 4πk . (T1 – T2) / (1/r1 – 1/r2)?
See note below and article now updated.
Only if you’re integrating wrt r.
But maybe you could give your workings, Joe.
q_r=4πr^2 . k . dT/dr
dT = [q_r/4πk)] . dr/r^2
T1 – T2 = [q_r/4πk)] . (1/r1 – 1/r2)
q_r = 4πk . (T1 – T2) / (1/r1 – 1/r2)
W = (W/m-K) . K / (1/m)
See, always helps when you put the workings out!
Joe Born:
Thanks, you are correct.
In fact, when I checked my calculations I appear to have used the right formula, but typed out the equation wrong. I will update the equation in the article.
Hi, in the maths section, where you have:
T2 = q / (εσ.4πr2^2)^1/4 and so T2 = 133K
I think should be:
T2 = (q / (εσ.4πr2^2))^1/4 and so T2 = 133K
Here is the plot of temperature (inner and outer) vs the wall thickness:
Note that the outer temperature declines slightly as the wall thickness increases. Why?
Because the same amount of heat is radiated from a larger surface area. That reduces the W/m^2, which implies a lower temperature.
It is a good example/analogy but I don’t see the back-radiation. I see a general increase from a point source at 30,000 W/m2 to 1,495 W/m2 at the inner surface to 17.7 W/m2 at the outer surface.
The 30,000 Watt point source should be comparable to the Sun which emits at 63,200,000 W/m2 at its surface but is reduced to 1,366 W/m2 at the Earth surface due to the inverse square law.
I especially don’t see the back-radiation from T2 to T1 which is supposed to simulate the Earth surface to the TOA I presume.
It might not be a good example, as well, since the thermal conductivity of the PVC at 0.18 W/m.K is considerably slower than the lower atmosphere which is on the order of 0.03 W/m.K
There is just a gradual increase in the radiation levels from T2 to T1 just like there is from the Earth surface to the troposphere. It is resistance to energy transfer versus a back-radiation.
Bill – The point source is 30,000 W, not 30,000 W/m2. At a 10m radius the irradiance from the point source is only 23.8 W/m2, not counting internal reflections from the PVC sphere’s inner walls due to incomplete absorption.
Just to nitpick a bit, you can’t really measure W/m^2 for a point source, as it is generally assumed to be dimensionless. The heat source is considered to be 30000 W. You could imagine the 30000W heatsource has a radius of 10m as well (the size of the heatsource doesn’t make much difference here).
And here is the total inner wall radiation (logarithmic plot) as a function of wall thickness.
Note that the value of total inner wall radiation when x=0 is – of course- the same as the total outer wall radiation.
Bill Illis:
Thankfully, it’s nothing like the climate system so the many people who know that current descriptions of the earth’s climate system violate the first law of thermodynamics can rest easy.
And in this nice safe environment of space I hope that many people will also learn a few basic principles.
Actually, just one basic principle.
The radiation from a surface inside a system can be many times the radiation leaving a system.
I’m hopeful that the many people who know this can’t happen show up and explain what is wrong with the model.
Or perhaps they are busy rewriting their previous work..
“If you recalculate back using the Stefan-Boltzmann law you will find that 133K with an emissivity of 0.8 radiates at 142 W/m2…”
I get 14.2 W/m2 (actually 14.1 W/m2 using the more precise value of 132.84K). The rest of the post seems fine.
Your point might be driven home more emphatically by noting that the inner surface absorbs only 23.8 W/m2 from the super-light-bulb, yet in the 3m-thick example it emits the afore-mentioned 1452 W/m2 (and also absorbs 1452 W/m2 of wall-emitted radiation).
Thanks, corrected the error.
You could have even more fun by considering where the 1,452 W/m2 of energy radiated by the internal walls goes. Doesn’t it land on other internal walls and raise their temperature? No, each square meter of internal wall is losing 1452 W by radiation and gaining (1452+23.9) W by radiation, 23.9 W being 30000 W from the source spread over 1257 m2 of inner surface area. This 23.9 W/m2 is on its way to the surface, where the increasing surface area with radius will reduce it to 14.2 W/m2. (The 142 W/m2 mentioned in SOD’s post appears to be missing a decimal point. There is also a misplaced parenthesis in the formula for T2.)
However, for our amusement, some of the 1452 W/m2 radiated by the inner walls is going to strike the power source in the center, stealing some of the energy needed to keep the inner walls at 423 degK and raising the temperature of the source. So we will have energy flowing from the cooler walls to the hotter source. The source will warm up, until it radiates 30,000 W plus whatever energy it intercepts from the walls, allowing the walls to remain at 423 degK despite the shadow cast by the source.
As you say, the thought experiment is a bit removed from K&T. What it demonstrates is what is intuitively known from observing the lagged hot water pipes in the home – the temperature of the insulation’s outside surface is considerably lower than inside. I would offer this further comment. The experiment is static in nature. However, the system would take time ‘s’ seconds to heat up to equilibrium with 423K and 133K on the inside and outside surfaces, respectively. During this time the mysterious power source would supply 30,000*s watt seconds of energy to the system and the cavity including the surface would achieve a uniform temperature of 423K. The Stephan-Boltzmann equation in its complete form takes ambient temperature into account which in this case is the same as at the surface and surface radiation is zero. Just guessing – if s = 60.8 seconds, the amount of energy supplied to the system would be about the number you attribute to inner surface radiation. Its units would be watt seconds, not watts as you have them. It would represent created energy, not by the system which would violate the 1st law, but externally and suppled to the system.
There is another strange feature in the Trenberth and Kiehl figure: 519 W/m2 of energy leaves the atmosphere as long wavelength radiation, but much more than half (350 W/m2) is directed back towards the surface. Should downward radiation emitted by GHGs and clouds be equal to upward (which is 195 W/m2)? Is this asymmetry (195 W/m^2) due to reflection of OLR by clouds?
If reflection is responsible, reflection of long wavelength radiation would involve more than twice as much energy as reflection of SWR from the sun by clouds. In that case, reflection by clouds would be the source of half of DLR, emission by GHGs would be the source of less than half of DLR, and emission by water in clouds would be the source of the rest.
John Millett:
The system has reached equilibrium.
The second part of this is confused.
The Stefan-Boltzmann equation is the integral of the planck function across all wavelengths and in all directions – thereby being the total energy radiated from a surface.
And so?
I calculated the equilibrium values using Watts – energy per second. If I calculated total energy over 60s I would need to use Joules. But I didn’t..
Is there something wrong with the equations that you would like to point out?
So a system heating up is violating the 1st law of thermodynamics?
In fact, when we consider any given system energy must be conserved.
So when we consider the system starting up from cold, the actual equation will be:
E(in) = E(out) + E(retained)
When the system reaches equilibrium, E(retained) = 0, and so E(in) = E(out) – this is the case above.
“In any case, the system is not creating energy. Inner surfaces are receiving high amounts of radiation while also emitting high amounts of radiation – they are in balance.”
This sounds like a welcome agreement between us:
“The Stephan-Boltzmann equation in its complete form takes ambient temperature into account which in this case is the same as at the surface and surface radiation is zero.”
Frank:
If you take a thin layer of the atmosphere it will radiate equally up and down – in fact it will radiate isotropically.
However, the atmosphere is not a thin layer.
The temperature, pressure and concentration of radiatively-absorbing gases changes with altitude.
The DLR we measure at the surface isn’t from the same average location as the OLR (outgoing longwave radiation) we measure by satellite.
Energy is being exchanged between different “layers” in the atmosphere via convection and radiation.
And OLR isn’t reflected by clouds. The amount of reflection of longwave in the atmosphere is insignificant.
Thanks for the help on the interactions between LWR and clouds. On further thought, I may understand the asymmetry in energy flow from the atmosphere. The average GHG emitter in the atmosphere has just as many is GHG absorbers between itself and space as between itself and the surface. It is equally likely to emit an upward photon as a downward photon as a downward photon. However, GHG’s closer to the surface are warmer – and therefore more likely to be in a excited vibration state – than those higher in the atmosphere.
How come a Point Source can receive any energy? Isn’t it dimensionless and thes evades any photons and phonos directed towards it?
“How come a Point Source can receive any energy?”
The photons have wavelength, therefore are not point sources.
There is also the factor of resonance which can increase the cross section of interaction.
It’s also a silly thing to say since changing this would merely complicate the maths to no avail.
Planar emission, for example, assumes an infinite plane. But such would require infinite power to generate.
But the error between a large (10^6 times bigger than the photon) and an infinite plane introduces negligible differences.
“As you say, the thought experiment is a bit removed from K&T. What it demonstrates is what is intuitively known from observing the lagged hot water pipes in the home – the temperature of the insulation’s outside surface is considerably lower than inside”
And the inside is hotter than the external pipe wall would be if you removed the lagging completely.
Where did this extra temperature come from???
I know.
T&K know.
SoD knows.
Do you?
“Its units would be watt seconds, not watts as you have them.”
Those would be “Joules”, rather than Watt seconds.
Watt=Joule/second
Watt*second=Joule*second/second = Joule
“The Stephan-Boltzmann equation in its complete form takes ambient temperature into account which in this case is the same as at the surface and surface radiation is zero.”
The surface of what? The surface of the PVC sphere is not zero, except right at the beginning. As soon as the thermal pulse goes through (at the speed of sound in the medium, so a millisecond or so) and reaches the outside, the radiation is not zero.
And radiative equiliobrium is what we’re talking about here, so the transients (time varying) elements are assumed static.
For energy balance, the differences are meaningless, since at balance overall change is zero, therefore the time varying part is zero.
This is quite common (and necessary in many cases) to solve the QM Schrodinger equation for real things (real things that work, like semiconductors in your CPU).
When you have found the equilibrium you can then manage to solve your equations for LTE where there’s a *slight* imbalance and see how the system reasserts itself (if indeed it does. if it doesn’t, you have catastrophe). But since you’re now only talking about DIFFERENCES, you can use differential equations and assume linearity (because differentials are infinitesimally small differences) which makes the equations tractable.
Would it be clearer to eliminate conduction by considering two thin opaque shells in a vacuum. Then you only need to do radiation.
I don’t know what you’re thinking of, but your comment put me in mind of this WUWT post by the estimable Willis Eschenbach: http://wattsupwiththat.com/2009/11/17/the-steel-greenhouse/#more-12889.
“Would it be clearer to eliminate conduction by considering two thin opaque shells in a vacuum.”
I wouldn’t have thought so because you’d need to work out the temperature of the opaque shells.
This hasn’t worked in Venus pt2 so I don’t think it would work here either.
Yet EVERYONE knows about conduction and how it works and that you lag hot water pipes and the water is hotter for the same boiler or you can use less boiler for the same heated water.
Basically we did the two shell calculation in our reply to Gerlich and Tscheuschner, (go to the link to the preprint) also the one shell, also the flat plat. Eli DID the damn experiment with a light bulb.
Any you know what. The clowns (and the Bunny uses the word advisedly) stuck their thumbs in their ears and manufactured denial. Sorry at that level you have to take it as deep, Goodwin class denial and nothing is going to shake them as SoD, Pielke Sr. and Spencer are finding out.
Poking about the rabbit warren and here’s a link to Roy Spencer who is having similar problems as SoD is having here:
http://www.drroyspencer.com/2010/07/yes-virginia-cooler-objects-can-make-warmer-objects-even-warmer-still/
And for RPSr:
1. By virtue of the second law of Thermodynamics, heat cannot be transferred from a colder to a warmer body, and
2. Since solar energy is the basic source of all energy on Earth, if we do not change the amount of solar energy absorbed, we cannot change the effective radiating temperature of the Earth.
Both of the above statements are certainly true, but as we will show, the so-called “greenhouse theory” does not violate either of these two statements.
Even those who love them loath their “defection” to science…
If Earth is in thermal equilibrium, in some time-average sense, then it’s just a game of conserving energy. One can consider the “3” systems in the TK97 figure:
1) The whole planet Earth system:
net flux (W/m^2) absorbed by Earth = net flux (W/m^2) emitted by Earth into space
2) The surface system:
Add up all the values in the arrows going directly into Earth’s surface and set equal to the sum of all the values of the arrows pointing out of the Earth’s surface.
3) The atmosphere system:
Again, add up all the arrows going directly into the atmosphere system (indicated by a gray horizontal band in the middle/right half of the diagram) and set equal to all the arrows directly pointing away from the atmosphere.
Of course, only 2 of these 3 sets of equalities are independent. Thus one may choose a variable (i.e., one of the flux quantities W/m^2) in any one of these 3 sets of equations, and insert it into either of the two equations
e.g., sticking the 2nd equation into the 3rd equation results in an equation describing the planetary system balance — equation 1.
sticking equation 1 into equation 3 results in an equation describing the surface balance — equation 2.
and sticking equation 1 into equation 2 results in an equation describing the atmosphere system balance.
Try it. With the figure in hand, set up the three sets of equations describing each of the three systems using the numbers given to you in the figure for each of the “3” systems. It should then make much more sense to you. Science of Doom has already got you started.
If this doesn’t work, then either one needs to brush up on their addition — or else the numbers are indicating a thermal imbalance in which the net flux absorbed by the Earth system is *not* equal to the net flux emitted by the Earth system. In this case, the surface and atmosphere systems will move to adjust until the Earth system balance is re-established. The TK97 figure assumes an Earth in thermal equilibrium.
And although it’s refreshing to see someone who normally eschews science when uncomfortable, there’s a little niggle from Roy Spencer’s explanation of backradiation here:
http://www.drroyspencer.com/2010/07/yes-virginia-cooler-objects-can-make-warmer-objects-even-warmer-still/
“Yes, thermal conduction involves energy flow in only one direction. But radiation flow involves energy flow in both directions.”
This isn’t really true.
Imagine how things would go with a hot conducting bar with a cold conducting bar brought into contact. The thermal loss from the hot bar is higher.
If you run with the same bar (reheat as necessary) with a *warm* bar put next to it, what is the thermal loss?
Well, what’s making the thermal loss less?
Now put a hot bar next to it. No loss? How?
In a solid, the thermal energy is in vibrations of a lattice that is the solid structure. The higher the temp, the more vibrant the activity.
When you bring another bar in contact that solid too will have vibration and that vibration will be at a speed that is a solid analogue of the kinetic free velocity of a gas.
Therefore putting a cold bar next to it, the vibrations at the joining edge of the hot bar hit the lattice of the cold bar rather more firmly than the cold bar hits back. Therefore a net transfer of vibration velocity. With a warm bar, the hot bar is still hitting harder, but there is more hitting back, so the transport of this vibrational energy is lower: more banging back.
And with an equally hot bar, there is as much smacking going one way as the other. No net transport of vibrational velocity.
The only way the second law can be understood on the micro scale is to allow each body do its thing independently.
And that means “backconduction” in solids.
Convection is a little different in that there’s some diffusion going on which would constitute “back convection”, it doesn’t go far unless the size of a convective cell is not much larger than the mean free path of the gas in that locality.
Where the MFP is ~1mm, reverse convection cannot go much more than a few mm. When the upward motion is in large fractions of m/s and the energy difference in Joules, the difference in “backconvection” hardly get a look in.
Such an effect is probably more effective on Mars and may be dominating in the Moon’s atmosphere, but that atmosphere is so tenuous, there’s no gas to have “temperature”.
While I think it would be better to drop the idea of backradiation rather than introduce ‘backconduction’, I think the analogy of the hotter vs colder body might be useful. What I mean is that a really really cold body is always warmer than vacuum in terms of emitted radiation (if it’s emitting ofcourse). As I believe it is rather intuitive to say that a warm body will lose energy faster to a colder body than a warm body (even if they are both colder than the losing body) we can conclude that any body will lose radiative energy faster to a body than it not being there (vacuum is really, really cold). Hence the presence of a cold body is warmer for the warm body than the non-presence of it.
You’re welcome to think that.
“As I believe it is rather intuitive to say that a warm body will lose energy faster to a colder body than a warm body”
It doesn’t mean it’s right. After all, it’s more intuitive to say the Sum moves around the Earth.
“any body will lose radiative energy faster to a body than it not being there”
Only because without a body being there, conduction is not possible.
But explain why it happens with different temperatures of body in contact, but the same conductivity.
Of course, there is another more extreme case to consider.
A perfectly lagged iron bar connected to an electrical power source. OK, the lagging will have to be made of unobtanium…
But if 100W goes in and 0W goes out, what happens to the temperature?
If 100J causes a 1C warming of the iron bar, then the bar will heat 1C in 1 second.
The next second, yet another 1C is added to that increased temperature (nothing gets out, remember!).
And so on.
After 1 hour, the iron bar is glowing hot (except all the energy is kept inside, so you can’t see the glow) at 3600C warmer than it was when you flicked the switch.
Yes, the lagging is unobtanium, but if such a device were made, why would the temperature NOT increase each second?
Yet still the lagging is no source of energy. It merely stops it passing one way.
Mark I believe you have described the conditions necessary to achieve the ‘big bang’; well done.
With cold and hot sources, shouldn’t the correct description be that the radiation from the cold[er] source decreases the rate of cooling of the hot source, not that the cold[er] source warms the hotter source?
With eli’s light bulb [can I have one eli, I’m sick of changing the so-called long-life non-flourescent ones?] and the other variations of continual heating should there not be a little black hole in the container to represent the [negative feedbacks] we are fortunate to have in the atmosphere?
If you are talking about SoD’s container then no – no black holes are needed. I’m quite sure this isn’t an earth model, but rather an illustration of how stuff can be more hot inside than outside. Which means adding weird stuff only makes it pointless (not that black holes would be a good way to represent negative feedbacks).
“I believe you have described the conditions necessary to achieve the ‘big bang’”
Do you? Well that’s unique of you.
Care to explain how you come to that conclusion?
“shouldn’t the correct description be that the radiation from the cold[er]…not that the cold[er] source warms the hotter source?”
No.
“and the other variations of continual heating should there not be a little black hole in the container to represent the”
No.
So far as I can tell, there are no black holes involved in the generation of electricity. Much of it produced by burning black rocks, but no black holes.
Hmm..
so middle aged adult males who call themselves ‘bunnies’ conclude that a global heat transfer system is seeking to minimize entropy?
that’s very interesting – quite the contrapositive of perpetual motion.
ah, well. it’s the age where Barney is in charge of Fannie.
This probably entirely offtopic, but I couldn’t resist (my apologies). Where are the bunnies, how are they minimizing entropy and who is Barney?
They were being a troll, Mait.
Eli’s blog persona is Rabbett.
I.e. “Rabbit” and probably an allusion to JB Haldane’s “Fossil Rabbit in the Precambrian” statement as to the falsifiability of evolution (in response to a creationist who wanted to show that evolution was just as unfalsifiable therefore creationism is just as good an answer).
And Nonsarky man is just being a troll.
Non sarky? No.
Man? Probably not that either.
Mark; you’re the one who invented unobtanium which, by the sounds of it, you are going to pour the total energy of the universe into the dohickey made of unobtainium:
“Yes, the lagging is unobtanium, but if such a device were made, why would the temperature NOT increase each second?
Yet still the lagging is no source of energy. It merely stops it passing one way.”
So, with the passing of time the energy goes in, stays there until eventually there is infinite energy inside and none outside; that sounds like a recipe for a big bang to me but perhaps you are being modest.
“which, by the sounds of it, you are going to pour the total energy of the universe ”
Nope, only 100W.
Do you think that the universe could not support 100W?
“So, with the passing of time the energy goes in, stays there until eventually there is infinite energy inside and none outside; ”
That would take infinite time, which would never happen.
I notice you can’t answer the question:
“but if such a device were made, why would the temperature NOT increase each second?”
All you’ve said is that the universe cannot give 100W and that after infinite time, you’d have a problem.
The first is obviously false.
The second pointless and no problem at all.
Mark,
I actually thought that I was agreeing with you in most part (with a different wording), but apparently I was wrong (the other explanation would be that you are in a strong disagreement with yourself).
Anyways, I’m fairly certain that in most cases the part about colder body cooling warmer bodies faster than warm bodies is in most cases true as well in addition to being intuitive (it’s one of those nicer cases where reality isn’t weird). I didn’t quite understand from what you wrote, whether you think it’s false or were just trying to make a point that intuitive things aren’t always true (to be honest I can’t see why it would be relevant to the discussion at hand for the latter).
I didn’t quite understand your last question to be honest (the explaining part). If possible it would be nice if you could elaborate on that.
“I didn’t quite understand your last question to be honest (the explaining part).”
Explain how you get a lower loss of energy when you have a warm bar touching a hot one than you do when you have a cold bar touching it.
The example is about conduction losses, not radiation ones. An earlier spot was talking about radiation.
This may be where we are crossing wires.
“Anyways, I’m fairly certain that in most cases the part about colder body cooling warmer bodies faster than warm bodies is in most cases true as well in addition to being intuitive”
Yes, so explain it at the molecular level.
I’ll take a stab at it 😉
You’re just really touching on the inception o thermal dynamics and its implications on heat engines, with the back ground state limiting the efficiency o an engine/ it cant exhaust at a lower state than the back ground state.
I think there is a difference though between variable conduction losses through varying entropy of its surroundings, and energy transferee through radiation. As you have stated, limiting conduction losses is the result of molecules not being able to transfer energy from a lower state to a higher state/ Or more energy across a boundary than its own equilibrium state, so the conductivity of the material will limit the energy loss vrs time across its boundary, as it can only transfer the differential between its state and its neighbors.
Where as with radiation, it is literally transferring energy across the boundaries of two radiating systems both ways. Rather than limiting the rate of loss.
On the molecular level… entropy, is the chaos/excitation of molecules. The way matter reacts to energy. So the molecules are vibrating, So a highly excited molecule collides with a less excited molecule, transferring energy, but it can only transfer the differential between its state and its less excited neighbor.
“but it can only transfer the differential between its state and its less excited neighbor.”
Errr, obviously, i was meaning half the differential.
Aye, that’d be the long word version.. !
But it shows that there is some of the exact same process going on “against the flow” that causes the flow in the first place.
Hence, like radiation, there’s an opposing flow in the other direction and the net flux is the difference between them.
Additionally, relating it to velocity (I would expect it to be that, but it could be momentum for solids, since you don’t have free particles and therefore momentum doesn’t *have* to be conserved by individual molecule in the solid) helps explain the differing heat capacities of solids.
If it is more massive a unit, slower movement and therefore less temperature increase for each joule added. If it’s more strongly bound, same thing.
If temperature was the simple expression of energy, then we wouldn’t have different heat capacities.
I’m still not sure I understand your question correctly, but on molecular level it can be explained by conservation of momentum as Mike wrote. If you bounce off slower moving molecules you will become slower hence your temperature will be lower than with bouncing with faster moving molecules (assuming mass is the same for both the fast and slow moving molecules in this example).
On macro level heat flux ought to be a product of conductivity and the negative temperature gradient, which pretty much should explain it as well. Bigger gradient for the colder object, hence higher heat flux.
This is a bit inaccurate actually. The temperature of a gas is more associated with kinetic energy than velocity. For monoatomic gases adding the same amount of energy for a gas with molecules with weight of 7 and 14 ends up with the same increase of temperature, althought the velocities of the gas with a weight of 7 will have increased a lot more than the velocities of the gas with weight of 14 (the heat capacities of monoatomic gases are the same).
“The temperature of a gas is more associated with kinetic energy than velocity”
Would this not be true of all matter? Molecular density would be what counts for thermal capacity i would have thought. But the kinetic energy of a heavier element would be what sets its T.
The state of elements is also set by its molecules entropy, a gas is a gas because of its T, same for solids and liquids. Thus a lighter element is a gas or liquid at lower T’s than heavier elements due its greater velocities for the same energy.
Well, ive always assumed this 🙂
“The temperature of a gas is more associated with kinetic energy than velocity.”
cf earlier:
“but on molecular level it can be explained by conservation of momentum as Mike wrote.”
same post.
Which is it, because momentum goes with velocity and energy goes with velocity squared.
Even though only 30,000W are input into the system – and therefore 30,000W leave the system once it reaches equilibrium – somehow the inner walls are radiating 1,824,900W.
And yet no one appears to have a problem with the example.
I’m not so surprised.
Many people are familiar with conduction of heat and lagging of pipes. Everyone can intuitively tell that the inner surface will get much hotter than the outer surface – that in fact it has to get much hotter.
And so it’s probably vaguely clear to everyone that therefore the radiation emitted by this surface must be much higher.
Hopefully anyone reading this article and thinking about it can see that, therefore, there is no creation of energy in the standard averaged energy balance diagram of the earth’s climate system.
Energy has to balance at the earth’s surface – and it does.
Energy has to balance for the whole climate system (at top of atmosphere) – and it does.
Semi-technical note: strictly speaking “energy balance” is a working assumption in the Trenberth and Kiehl 1997 paper because our current measurement capability is not good enough to produce global annual average numbers at TOA with uncertainties less than about 5W/m^2. (And the uncertainty is much greater at the surface).
However, the measured numbers at TOA are quite close. It is quite likely that in any given year there will be a difference between energy in and energy out – this will be reflected in a heating or cooling of the climate system.
Here is a thought experiment for you. There is no math required. Picture a hollow evacuated sphere with a surface absorbtivity of 0. Now picture a hot ball suspended in the center (actually position is irrelavent). The hot ball radiates but does not cool because all the radiation is reflected back to it. The hot ball must be reabsorbing its own radiated heat.
Your math is correct, however, the blackbody law states emitting bodies will emit energy based on their temperature because that is the sole (or major) energy loss mechanism framed in the law. It does not state that, coupled to other systems by conductive pathways, that they will continue to radiate in that fashion.
Could you explain the mechanism of energy transfer from the inner shell that allows energy to be absorbed from the point source and conducted away AND reradiated at the same time?
Further, you have created a hypothetical situation to answer a question which does not exist to anyone with a grasp of arithmetic and eyeballs:
At the top layer of the figure, there is
342 IN
107 and 235 OUT
Sum = ZERO
At the surface layer of the figure, there is
168 and 324 IN
24 and 74 and 390 OUT
Sum = ZERO
For both systems (the atmosphere+earth) or (earth) the total energy appears to be meritoriously conserved. What’s the question again?
Buffoon:
Yes it does.
To calculate temperatures and heat flows within a system you need to take into account conduction, convection and radiation.
Regardless of the heat transfer by conduction a body will still radiate according to the same law (which is the blackbody radiator modified by the emissivity of that surface = εσT^4).
I will assume that I stand corrected on my first point.
Could you explain the mechanism of energy transfer from the inner shell that allows any energy to be absorbed from the point source and conducted away AND reradiated at the same time?
I must admit I’m not sure I understand the question correctly, but I think you might be forgetting that the absorbed radiative energy at any part of the inner sphere is the energy radiated by the energy source incident on the observed part plus the energy radiated by the rest of the inner sphere incident on the observed part. A few integrals later you should find out that the energy radiated by the rest of the inner sphere nicely equals the energy radiated by the observed area according to the Stefan-Boltzmann law. This should leave us with the net radiative balance of the source, which will be happily conducted away towards the outer surface where it will escape into oblivion.
“Could you explain the mechanism of energy transfer from the inner shell that allows any energy to be absorbed from the point source and conducted away AND reradiated at the same time?”
Because there’s an energy source and some of that is being conducted away. If there was no conduction away then there would be a steadily increasing temperature and therefore steadily increasing radiation from the inner surface.
If 1% of the energy is conducted away then 30KW production inside the sphere becomes 3MW radiation from the inner surface.
All that means is that 300W is being conducted away and 3MW being radiated within the sphere. The radiation is unable to leave because the solid sphere is, well, solid.
“Further, you have created a hypothetical situation to answer a question which does not exist to anyone with a grasp of arithmetic and eyeballs: ”
You have created a hypothetical situation where nobody would ask that question.
People have.
Just have a look at the comments Roy Spencer has to field (link earlier).
“But the atmosphere is not isotropic at CO2 absorption wavelengths”
So, a CO2 molecule does not emit isotropically? I can only assume you are confusing direction and intensity; if the atmospheric emissions are not isotropic then the cold to warm conceit is reintroduced.
“How close to 200 do you want?” You are missing the mass effect in the real atmosphere where because of that mass the increments are crucial; rounding up is a big fudge. I don’t think I can take you seriously anymore.
“So, a CO2 molecule does not emit isotropically? ”
So you can’t read?
“But the atmosphere is not isotropic at CO2 absorption wavelengths”
Even when you quote it, you can’t read it, apparently.
Yes, a CO2 molecule does emit isotropically.
But the atmosphere is not isotropic. It’s thicker out to space and thinner into the earth.
“You are missing the mass effect in the real atmosphere where because of that mass the increments are crucial; ”
They are absolutely immaterial to the balance.
The earth has been warming under the sun for 4.5 billion years.
Plenty of time for the masses to heat up.
I notice that you haven’t tried to ascertain how quickly it heats up.
Well, Nassif tried and got 0.21C/second.
That would mean that to make up the 11C change CO2 is responsible for in toto is about 55 seconds.
But you don’t seem to understand science, do you.
Buffoon’s comment got me thinking; damn you Buffoon!
If we take SoD’s sphere and assume the light source puts out 100 units of radiation per unit of time and 1/2 of that 100 units is reradiated back into the inner sphere and 1/2 is conducted away then in the 2nd unit of time the source emits another 100 units which is combined with the 50 units from the 1st unit of time so that 150 units is available to be absorbed by the inner surface with 1/2 or 75 being reemitted and 75 being conducted away. This is, of course, a limiting sum geometric series:
Sn=a/1-r where a=1 and r=0.5; or 1/n^2 for n=1 to infinity is 2.
That is, the radiation available in the room will not double until infinity; the asymptotic aspect of this is, of course, what happens with additional CO2 in the atmosphere.
Cohenite,
I believe you have proven that an equilibrium will be reached as long as the characteristics of the system are not changed. The asymptote is that equilibrium point. That says nothing about what happens when you change the characteristics of the system.
What happens if you increase the amount reflected?
BTW, I don’t think you really understood the model that SoD used, or the real point he was making. There is no inner sphere or room.
ChrisG; you miss the point; an asymptote is a declining factor; the radiation in my hypothetical only doubles at infinity which is the nature of asymptotes. The purpose of my hypothetical was to show that with other constants and a continual radiation but increasing capture that there will not be runnaway or infinite build-up of energy/heat. As you say it says nothing about a complex system when the characteristics of the system change; Miskolczi has noted this scenario and you should read Christopher Game’s comments here:
However, you have successfully captured the idea that the level of energy between the source and the system emitter can be higher than what is finally emitted. In your example, this is by a factor of 2.
This still has nothing to do with changing the characteristics of the intervening layer, or the amount of CO2 in the atmosphere.
Except, in your example, capture was constant at 0.5.
Yes the constant was 0.5 but the amount of radiation was increasing at every stage; the scenario where both the amount of radiation and the proportion captured increases would mean a decrease in the rate of conduction away; why would that happen? It would only happen if the conducting material had a threshold whereby if the incoming radiation increased the rate of conduction decreased per unit above that threshold; there are permutations of this, but it should be remembered that the total of the amount conducted and reradiated cannot exceed the total of radiation available; so if one goes up the other can only inversely and proportionally go down, or go up at a lessor rate so that the total radiation is not exceeded.
The radiation was increasing on the inside because you started the re-radiating layer at 0 energy, obviously not an equilibrium state. It’s not surprising that an equilibrium will be reached when the rate of energy emitted outward through the re-radiating layer is equal to the rate of energy added to the system.
We’re getting off topic; you’ve already provided an alternate way of arriving at the same conclusion as SoD. At your asymptote, the re-radiating layer is putting out energy toward the inside at a greater rate (200 units energy/unit time) than energy is being added to the system (100 units/unit time).
However, increasing CO2 is analogous to increasing the rate of capture in your hypothetical model. Increasing the rate of capture is kind of the foundation of global warming. Using your own math, what happens if you increase the rate of capture, let’s say after 16 time units when the re-radiated energy inward is at 199.99847 units/time?
You can round that to 200 if you like.
For grins, let’s say the capture rate is increased to 0.51. You’ll see that the internal energy level increases to a new asymptotic value, and at this new level, the rate of incoming energy is again equal to the rate of outgoing.
In this scenario, the radiative energy directed inward approaches
100/(1-0.51) ~= 204.0816.
Let’s check outgoing.
204.0816 * (1-0.51) = 100
Yeap, outgoing is still equal to incoming, and the internal energy is higher than it was at the lower capture rate.
I think I understand the math and the implications of your model well enough.
I suppose you’ll want to talk about rates of attenuation at this point. It’s off topic and been covered before.
I choose 0.5 as the reemission amount because of the isotropic property of CO2. By increasing that to 0.51 you are distorting that and I believe mistaken is saying: “However, increasing CO2 is analogous to increasing the rate of capture in your hypothetical model.” In fact the 0.5 constant is equivalent to increasing CO2 and by increasing that you are making assumptions about feedbacks; which don’t exist in my hypothetical.
In addition, starting at 0 is equivalent to a pre-greenhouse atmosphere condition; I didn’t say anything about equibrium; my hypothetical does not have an equilibrium state; that’s due to the asymptotic nature of the radiation build-up; you keep rounding it up to 200 which, as I say, would only occur at infinity. You don’t work for the BoM by any chance?
“I choose 0.5 as the reemission amount because of the isotropic property of CO2. ”
But the atmosphere is not isotropic at CO2 absorption wavelengths.
“By increasing that to 0.51 you are distorting that and I believe mistaken is saying: “However, increasing CO2 is analogous to increasing the rate of capture in your hypothetical model.””
No, you’re distorting it by saying that “the atmosphere is optically thin and that this won’t change when you add CO2.” This is wrong.
“In addition, starting at 0 is equivalent to a pre-greenhouse atmosphere condition”
It’s also a non-equilibrium one when you add CO2 to the atmosphere.
“my hypothetical does not have an equilibrium state;”
That’s not the only thing your hypothesis lacks and these lacks are why it falls down when you try to apply the hypothesis to analysis.
“you keep rounding it up to 200 which, as I say, would only occur at infinity.”
How close to 200 do you want?
10 cycles and you have over 199.9.
10 feedback cycles takes a lot less than infinite time…
You don’t happen to want to waste people’s time with irrelevancies and diversions, do you?
Cohenite,
I don’t know if it will do any good, but let me explain where I think you are having some disconnects between concepts.
I suspect you have gotten confused regarding the nature of the radiation that CO2 captures and emits. Fifty percent of what it absorbs goes outward and 50% goes inwards. You are right that this does not change. The part I suspect you are missing is ‘of what it absorbs’; the rate at which it absorbs changes with how much of it there is. More CO2 means LW is captured at a higher rate.
Also, the sun has been shining (the light bulb has been on) and GHGs have existed for a very long time. Starting the system at energy level 0 is like the solar system before the sun started shining and there were GHGs on earth. The greenhouse effect, convection, solar irradiation, etc., all were in existence prior to the industrial revolution. All we are doing is changing the balancing point of all the pluses and minuses. The questions are really only where will the new equilibrium point be, how fast will the earth move in that direction, and what effects will that have on living conditions.
Buffoon
The mechanisms are independent.
The simplified form of the equation for conduction is
q= kA . ΔT/ Δx
which says that the heat flow per unit area is proportional to the temperature difference across the material.
The equation for radiation from a surface is dependent on T^4, where T is absolute temperature in K of that surface.
The equation for absorption of radiation is a property of the material which depends on wavelength (and sometimes, direction).
All of these equations need to be satisfied to solve the complete problem.
The reason the K+H diagram is so difficult to accept for some are the huge numbers for upwelling and downwelling radiation in comparison to the incoming solar. It just doesn’t look right. You previously said that these numbers were observed however you didn’t seem to realize that only the net radiation was observed, while the upwelling was calculated using Stefan-Boltzmann and the temperature of a black dome on the instrument and the downwelling is calculated from the difference between the two. Now its important to note that the net radiation alone would be enough for the diagram, and that’s what the NASA diagram uses. Putting it that way also happens to bring it home that we are not really talking about heating from the atmosphere but reduced cooling of the Earth.
Clearly it isn-t possible to separate out downwelling radiation via a separate instrument so the numbers calculated and derived are not able to be independently verified. So is the calculation actually correct? I’d submit that it probably isn’t. Now you might say in your usual haughty fashion I’d be disbelieving the Stefan-Boltzmann equation but no I don’t. The problem is in ignoring convection. Imagine that the heat the instrument experienced rising from the earth is mostly transported there by convection rather than radiation. Additionally some conduction through the instrument would occur from that convection. Then any calculation that assumes only radiation heat transfer will greatly overestimate the numbers.
That’s my deep suspicion from having done such heat transfer calcs that absolutely had to be verifiable rather than taken on faith. Basically the experience of anyone who has had to do this to compare models to real life, is that radiative heat transfer only dominates when the source is at a very high temperature. For low temperature objects, the secondary convection arising from the radiation is dominant and the direct radiation heating itself is minor. You can test this yourself with a radiator next Winter. Touch it and it’s hot thanks to direct conduction. Hold your fingers a cm out and you’ll feel only the rising air currents but no direct radiation. Now go close to a wood fire with a source temperature much higher and you’ll really feel the radiative heat hitting you. The Earth temperature is rather lower than even a radiator so likely there is very little direct radiative heating on the instrument.
Anyway that’s my tuppence worth. But, as I said, the disbelief and argument about numbers would be null and void if they used only the net value which is 66. Everyone can agree that’s sensible. An enhanced greenhouse would just show a reduced net value. What therefore is the point of showing back-radiation? It doesn-t even make any sense that there is no radiation upwards that equally matches the back-radiation, as the theory requires.
None of this is denying AGW, or speculating about the 2nd law which probably doesn’t even apply to the atmosphere anyway. It’s perfectly possible to dislike the back radiation concept just because the numbers for it look just plain wrong. Since they aren’t actually observed numbers as you wrongly stated previously, they are calculated, there is no need for so much mockery to those who merely notice the numbers look wrong.
JamesG
What do you believe is the value of thermal radiation from a 15’C typical earth surface?
“It just doesn’t look right.”
You know what a round Earth just doesn’t feel right to me. I just cannot fathom how those upside down Australians manage to hold on.
Doesn’t feel right. Therefore it cannot be.
“Clearly it isn-t possible to separate out downwelling radiation via a separate instrument”
Why not?
The sun radiates with practically no power below 4um and downwelling atmospheric radiation practically no power above 4um.
A simple bandpass filter for either would do.
“That’s my deep suspicion from having done such heat transfer calcs that absolutely had to be verifiable rather than taken on faith.”
You do seem to have taken on faith the supposition I quoted earlier, however.
“What therefore is the point of showing back-radiation? It doesn-t even make any sense that there is no radiation upwards that equally matches the back-radiation, as the theory requires.”
It doesn’t match, and the theory requires it not to.
You seem awfully confused.
“The problem is in ignoring convection.”
The problem of ignoring convection is one of your invention: you’ve ignored the convection (Thermal) vector.
It’s 24W/m^2.
JamesG:
Here’s the setup of EBEX and the measurements of upward and downward shortwave and longwave radiation:
From The Energy Balance Experiment EBEX-2000. Part III:
Behaviour and quality of the radiation measurements, Kohsiek (2007).
The location was a cotton field of 800m × 1600m at coordinates 36◦06’ N, 119◦56’W, approximately 20 km south-south-west of the town of Lemoore CA, USA.
I’m still fascinated to find out your take on the upward longwave radiation from a typical 15’C surface.
[…] Further reading – Do Trenberth and Kiehl understand the First Law of Thermodynamics? […]
A question concerning the Trenbeth and Kiel picture:
Would the picture be incorrect, if we deleted the downward 324 W/m^2 arrow (backradiation) and changed the 390 upward value to 66 W/m^2, of which 40 W/m^2 is lost directly to space and 26 W/m^2 is lost to the atmosphere from the surface?
Mait:
Yes, because it would no longer be the real world.
I’m not quite sure how it wouldn’t be the real world any more. I just replaced two way numbers with a net number. I’m guessing you have good reasons why you think it’s a bad way to represent energy balances (without the separate downward/upward fluxes), but it would be appreciated if you explained why.
PS. I already mentioned this before somewhere, but please do not quote only a part of a sentence, otherwise you will missrepresent what is being said.
Because you’d have an earth that should be 185K rather than the 290K it is.
If it were hotter than 185K, then the upward radiation would be higher, and that means your arrow of 66W/m^2 would have to be changed from reality.
So, either the earth’s 100C too cold or it isn’t radiating anywhere near enough.
Either is not the real world.
“but please do not quote only a part of a sentence, otherwise you will missrepresent what is being said.”
No, you’ve complained about it before. But if the quotation remains within context, then there’s no misrepresentation.
It is telling that this second time you complain, you have still not managed to display what misrepresentation happened.
This would be because there isn’t one.
Or, more succinctly, the real world has radiation going both ways, so a single arrow would be not what’s happening in the real world.
If you changed the arrow to be 66W/m^2 then you’d have a temperature of 185K.
Your problem then would be “how does the earth register a different temperature?”.
Why the big hate-on for back radiation?
That there is a NET flux of 66 shows that the second law is not being disobeyed: heat flow is from higher to lower temperatures.
If you wanted to calculate the temperature from a net radiation balance you’d be right. But I don’t quite understand why you’d do that.
On the quoting part – in my opinion there is a profund difference between saying there is no downwards radiation and saying that it can be presented as a reduction (in terms of total energy loss) of upwards radiation (or in other words net balance). I’m pretty sure there are good reasons why this isn’t a good idea, however, to say it’s just wrong seems a bit confusing to me. The numbers should still all add up in the Trenberth and Kiehl picture.
So how would you explain the temperature of the earth???
Mait:
The idea behind the energy balance diagram is to understand all of the surface fluxes and all of the TOA fluxes.
If you replace the up and down flux with a net flux you have removed useful information from the diagram.
Anyone who wants to do further calculation has now lost this information.
The net flux won’t explain the surface temperature or the atmospheric temperature.
To calculate absorption and therefore atmosphere heating you need to consider the absorption of both up and down fluxes through the atmosphere. Not net flux.
In fact, net flux is mostly a useless number in radiative transfer.
It’s a little different in conduction where the heat flow is proportional to the temperature difference, and not dependent on the absolute temperature. And the heat flow in each direction doesn’t need to be separately considered to calculate the net.
The only reason I can see for removing “back radiation” and “surface radiation” from the diagram and replacing with net flux is make people who don’t understand the subject a little happier.
I would say that I’ve just hidden some of the information a bit rather than removed it. It’s quite easy to find back radiation value from the net if the temperature and surface albedo are assumed (which they are anyway on the picture I believe).
I would argue that neither does back radiation to be honest. As there are more methods of heat transfer besides radiation, I would say that temperature explains the back radiation not the other way around.
To be honest I didn’t think the purpose of the picture was to calculate temperatures somewhere, but rather roughly illustrate how energy moves in the atmosphere. However I don’t quite see any particularly big difference between using net radiation and source radiation and using source radiation and back radiation.
Shouldn’t radiative heat transfer be proportional to temperature difference as well as a bigger temperature difference indicates bigger difference between radiation output of different parts of the object thus the colder parts heat up faster the colder they are (as they are losing less energy through emission as they are colder) and hotter parts cool down faster for the same reason.
I’ve always kind of assumed the purpose of this blog (at least in some part) was to help make people who don’t understand the subject understand it a little better. I just tried to present a bit of a different view on the same subject, which might be somewhat easier to digest for some people. It should be more – is a ping pong ball falling on earth or earth falling on ping pong ball – kind of a question I would say.
Is there a slight transposition error in the opening paragraph? The figure shows the absorbed solar energy is 168W/m2, however the problem definition reads:
“how can the absorbed solar energy be 235W/m2 when the radiation from the surface is 390W/m2? ”
I realize it does not affect the proof itself, but it confused me for a second.
BTW, as a layperson I think that this site and skepticalscience provide wonderfully complementary resources. Thank you!
There’s 67 being absorbed by the atmosphere directly from the sun, Eilleen.
Add to 168 and you get 235.
It’s a little hard to see because there’s a concentration on what’s going to the ground, so easy to miss.
Aha! Thanks much!
A fascinating model.
I can NOT identify where it is wrong BUT somewhere it is.
Perhaps the equations fail to be valid as you have defined the parameters of the materials? What happens at the barrier between the empty sphere & the inner wall?
My understanding of radiative heat rransfer is that if 30KW comes in & 30KW goes out, then at ALL points in between 30KW is also going out. The temp profile must be a continuously decreasing curve as you go out. (like the Earth)
Regardless, a few thoughts re global warming for you to ponder:
1. The half up half down problem:
The traditional diagram (235 in, halfout to space & half returned to the ground, is an instantaneous initial condition – definitely NON equilibrium. This is NOT reality.
Reality is equilibrium (or always rapidly approaching)since the 235 number varies in reality from 0 at night to 2×235 at noon.
At equilibrium the intial solar input in is 235, it bounces at ground is absorbeb by GHGs, half (235/2=117.5) goes to space & half down where it is again absorbed & reemitted along with the continuous input of the solar insolation in. (Th diagram ignores that the half down must go somewhere (ie back out again)
ON the 2nd bounce at the ground you get (235 + 117.5)=352.5/2 to space, same to ground,
On the 3rd ground bounce it is 235+352.5/2=235+176.25=411.25 …
Which as you all pointed out is asymptotic to double the intitial 235 in.
ie at equilibrium, which takes about 8-10 bounces or absorbtions,(I’m a practical engineer, not a theorist!)energy out= energy in.
Eli once told me that each absorbtion takes a few nanoseconds, so equilibrium takes at most 100 nanoseconds – ie FAST
2. The K-T diagram is not real. It is an annual average construct. It assumes that the “world begins at the TOA and ends at the ground surface”. It is missing a term at the bottom that represents the energy provided by the Earth temperature (your 30KW lightbulb). At night when the solar insolation in equals zero, but the ground still radiates IR energy out, the GHE still continues albeit at a slightly reduced rate. The question then is where does this energy come from, since solar insolation is actually zero? AND where is it counted in the K-T diagram? The ground energy is real- conventional wisdom is that it comes from radiaoactive decay. I claim it comes from the forces of gravity impacting the Earth- eg the moon causes tides which cause friction which generates heat. OR The Sun gravity causes Earth torque which causes friction which rotates the inner core faster than the outer core faster than the mantle faster than the winds- hence friction & heat. (comments please?)
3. The K-T diagram is missing time dependance. We all know that the Earth’s orbit is eccentric- hence closer & further away at times, hence the 235 number varies with time. NOW when the Earth is further away, what happens to the Earths gravitic potential energy? Doesn’t it increase as we get further up the (Suns) gravity well? Where does the energy come from? Does it get taken out of the Earths kinetic energy (ie temperature)? hence we get colder further from the sun?? (along with less energy coming from solar insolation at the greater distance?) So if the total energy in varies based on distance, AND the total energy in from gravity and PE is MUCH MUCH greater than the solar insolation, then could it be that a proper K-T diagram would make the values presented so trivial that they do Not matter? That in reality the GHE is on the order of trivial,(but still real- or maybe it is not real & just a model construcr made up to explain real temp differences) and the Earth’s temperature is dictated by the varying forces of gravity & PE? See http://www.scribd.com/doc/27343303/Gravity-Causes-Climate-Change or search for John Dodds at http://www.scribd.com for more info. & explanation
4. When looking at the GHE look at the concept that it is NOT that more GHGs means more warming per Arrhenius & IPCC, but rather it is that the amount of incoming photons is limited & limits the amount of the GHE thus leaving EXCESS GHGs in the air, so adding more just adds more GHGs & does NOT add warming.
Love the tone & exchanges of this site
Thanks- John
“I can NOT identify where it is wrong BUT somewhere it is.”
Ah, try opening your mind to the possibility it IS right and that eminent scientists who have studied this for years have it right and you have it wrong.
“My understanding of radiative heat rransfer is that if 30KW comes in & 30KW goes out”
That’s what’s happening.
TOA: In 342. Out 342.
“then at ALL points in between 30KW is also going out.”
Why?
“Eli once told me that each absorbtion takes a few nanoseconds, so equilibrium takes at most 100 nanoseconds – ie FAST”
Eli didn’t tell you that. Go and find out how long it takes to emit 15um from CO2. And how long between collisions is it at STP?
Compare the two.
A CO2 molecule doesn’t have time to radiate before some other bugger comes along and bumps it.
“2. The K-T diagram is not real. ”
No, it’s real.
“3. The K-T diagram is missing time dependance.”
Well, yes. It’s an indication of the energy balance. This is the time-independent part. Changes over time take things out of balance, therefore putting time dependency in makes it not an energy balance diagram.
“but rather it is that the amount of incoming photons is limited & limits the amount of the GHE”
Nope, the number of photons only matters until the first absorption.
And any such paucity is irrelevant in any case: there are a lot of photons in 1W.
“thus leaving EXCESS GHGs in the air, so adding more just adds more GHGs & does NOT add warming.”
This doesn’t follow on.
It ads to the effect because TOA gets higher and TOA radiation with the same profile is reduced. Therefore there’s an imbalance in the TOA fluxes and the atmosphere heats up more.
If the atmosphere heats up more, then ground is heated by the atmosphere more.
Until equilibrium is reached at a higher temperature.
Don’t seem to love the science, do you.
John Dodds:
Nice to see someone make the claim. It’s this strong belief that drives the reaction to the Trenberth & Kiehl diagram.
And by the way, that diagram is simply the latest in a long line (100 years) of papers to attempt to calculate the average values for different aspects of how energy moves through the climate system.
Suppose – crazy thought I know – but suppose you are wrong. Just entertain the thought for a few minutes.
How would you demonstrate it?
You would be wrong if:
– a) the temperature at the inner surface was greater than the temperature at the outer surface
AND
– b) the Stefan-Boltzmann equation was true
Or looking at it another way, you have 2 ways to be right:
– 1. The temperature at the outer surface is the same as the temperature at the inner surface
OR
– 2. Radiation doesn’t follow the Stefan-Boltzmann equation
Which one do you want to pick?
Otherwise it’s clear that the inner surface radiation is much higher than the outer surface radiation.
The only way that the inner surface radiation can match the outer surface radiation is to be at the same temperature. But everyone can see that it will be hotter.
The first law of thermodynamics simply states that energy cannot be created or destroyed in a system.
No energy is created or destroyed in this system. Therefore, this system does not violate the 1st law of thermodynamics.
John Dodds:
Just to pick one point out of your comment:
Averages are never “real”. They are averages. So long as you point out it’s an average there’s nothing wrong with that. The usefulness of an average can be questioned of course..
It includes the energy from the earth’s surface temperature – see below.
The sun heats up the atmosphere. The atmosphere can only radiate (lose) heat at a certain rate compared with the stored heat. This is why it doesn’t drop to absolute zero.
In fact, the explanation is simple. You can measure the temperature of the atmosphere at night. It isn’t absolute zero. Therefore it will still emit radiation.
So it doesn’t matter than solar insolation is zero.
“where is it counted in the K-T diagram” – it is in the average. The average “backradiation” globally annually averaged is 324 W/m^2. The global annual average includes the night-time.
No, convectional wisdom is that it comes from the retained heat in the surface of the earth. The earth cools down slowly. During the night certain surfaces might drop 10-20’C, the ocean surface might drop a degree or two, likewise with the atmosphere.
Mait:
Perhaps I could have been clearer.
Whereas conducted heat is linearly proportional to temperature difference, radiated heat is proportional to T1^4 – T2^4.
I’m not quite sure I understand your point, but probably the fourth power makes the calculations a bit more nasty. However you should still be able to calculate transfered heat from T1 and the temperature difference. From what I understand that is actually the same for conducted heat (that you need to know T1 and temperature difference), as conductivity is usually temperature dependendent.
Or use energy fluxes.
Then it’s a linear problem.
And to do that you need … wait for it … BACK RADIATION!
🙂
Response to SoD
Could you explain for your readers the difference between a scalar quantity and a time dependant vector quantity?
If you sum the values across the K-T diagram at any level you get zero, ie equilbrium at any particular instance of time.
However if you try to add the 350 outgoing flux to the 324 backscattered flux you get a positive value of 674. Just one problem: the outgoing flux happens a few nanoseconds BEFORE the backscattering. so they are not at the same time and can NOT be added. In fact the two values are probably using the same photon of energy which gets absorbed and a few nanoseconds later backscattered.so by adding the two you just created energy and violated conservation of energy. If they cancel vector wise then you are left with the equilibrium 235 in equals 235out
To paraphrase you “THINK ABOUT IT!”
The diagram is supposed to be equilibrium- ie in=out. 235 in =235 out at every point. at a particular instance of time.
In your model, I can calculate the temp at 1m, “T2-” or 1micron less that 10m and at T1+ or one micron outside the 13m sphere, ALL using the Steffan-Boltzmann Law just by a simple ratio of the 30KW divided by the area. Its a steadily declining temp profile. just like the Earth. What the EarthPVC sphere does is delay the transmission UNTIL it climbs back to equilibrium (which is what we are showing in the diagram)rapidly( given that an absorbtion and return to the air by collision (~95%) & by reemission (~5%) is within microseconds- or multiple absorbtions each of several nanoseconds). Given that the transmission of the photons is a vector, then adding the multiple backscatters needed to get back to equilibrium (which is what your T1 calculation did) is NOT mathematically nor physically valid. (the backscatters happen sequentially multiple nanosecords after each other for a specific photon going out.)
The SBL by definition occurs at every point on the way out. I can always create a “system” that is the 10m inside the PVC sphere. or the sphere itself or half teh sphere etc & at each surface energy in MUST equal energy out (ie the sum =zero- try it on the K-T diagram at the ground or the t1 & t2 points, and at every point in between.. SBL MUST conserve energy altho as you said the density decreases because the area gets bigger.
Mathematically you can NOT warm the interior by backscatter and add the energy without violating conservation of energy.
The K-T diagram itself confirms that the only way to increase the energy & add warming is to add more energy from somewhere outside the system.
“The diagram is supposed to be equilibrium- ie in=out. 235 in =235 out at every point. at a particular instance of time.”
And this is a problem *how*?
“Mathematically you can NOT warm the interior by backscatter and add the energy without violating conservation of energy.”
Yes you can, very easily.
Otherwise explain how radiative insulation (the silvered inside of a vaccum flask) works.
“The K-T diagram itself confirms that the only way to increase the energy & add warming is to add more energy from somewhere outside the system.”
Yes, like the sun.
This is not a problem for us for probably another 8 billion years.
And you need both energy from outside the system and an impediment to that energy leaving the system.
Which is what the GHG effect is all about and why we’re getting global warming from anthropogenic production of CO2.
Mark , you missed my point, or I failed to explain enuf.
In order to warm you have to add MORE energy than what is already coming in. If 235 is coming in & going out, (& it basicly has NOT changed since the 1960s) and causing the existing GHE 33C warming effect, then in order to increase the temperature you MUST add more energy. ie from the sun (or some other source – eg gravity) Just adding CO2 will not do it. CO2 can’t create energy -see more below in response to SoD)
As for the thermos flask, it does NOT add energy, it slows down the loss to maintain the warmth IF it added energy it would boil away the contents. Same applies to pipe insulation. By adding insulation, you reduce losses over time & it so the continued addition of gas or electric heating allows you to maintian the same temp or heat it up if you add more electricity from outside, & hence it works better.
When you drive to work, it takes time and in that time an interval of linear distance is traversed.
Therefore you have an average speed.
This is a real phenomenon predicated on the simple maths that the average speed can be calculated by taking the total time and dividing by the total distance traveled.
That you never went at that speed for any significant time is irrelevant to the supposition of whether your average speed can be calculated.
They even have speed cameras based on that phenomenon.
Why the hate-on for it here?
John Dodds:
No. The values are not equilibrium at any particular instance of time. The values are the global annual average for each component.
And as the paper explains, equilibrium has been assumed because we can’t measure accurately enough to confirm equilibrium.
I don’t understand what you are claiming or pointing out.
These values are global annual averages.
Let’s put that to one side to try and understand what you are claiming.
“..the outgoing flux happens a few nanoseconds BEFORE the backscattering..
This kind of comment is quite confusing. Outgoing flux from the surface is happening continuously, as is incoming radiation from the atmosphere (DLR).
We aren’t following individual photons here. You won’t find any useful cause and effect from individual photons.
..so by adding the two you just created energy and violated conservation of energy..
I’m not adding the two.
Energy into the surface equals energy out of the surface. That is conservation of energy.
And then onto my model. I don’t really understand any of your comments. You finish up with:
Do you think the temperature of the inside of the sphere is higher than the outside? This is an important question to answer.
I used the equation for heat conduction and Stefan-Boltzmann’s law, both of which are fundamental thermodynamics.
I don’t understand your comments about “warming the interior by backscatter”.
1. The outer surface of the sphere will radiate energy out to space, and while the sphere is radiating less energy than it is receiving it will heat up. Eventually a temperature will be reached where energy in = energy out.
This defines the equilibrium outer temperature.
2. The equation governing the temperature difference between inner and outer surfaces is simply application of the 1st law of thermodynamics with the material property of the sphere. If 30,000W flows from the inner surface to the outer surface then the temperature difference is easily calculated.
Heat can’t flow through this wall until there is a temperature differential. The temperature differential will keep increasing until enough heat flows out to balance the heat added to the system.
3. The equation governing the radiation from the inner surface is simply the Stefan-Boltzmann equation, using the temperature already calculated.
Go step by step through the maths and explain where the error is, e.g., which equation is incorrectly used or which assumption is not valid.
First I really appreciate you guys responding scientifically so please continue.
I shall attempt to explain my position. using the K-T diagram & basic global warming theory & if I can to relate it to the sphere model. I would like to avoid analogies , greenhouses, water pipes and thermos flasks etc since they usually add extraneous physics that doesn’t apply
My bottom line is that adding more GHGs to a system without adding more energy or photons will not result in more warming. You just get more excess GHGs in the air and the GHE temp stays constant. (unless you also add energy)
So you are left with 30KW or 235W/m^2 at r=0, at r=just under 10m or 10-, at r=10m+ and at r=13m- and r=13m+ This results in a steadily declining temp profile as the surface areas increase. So yes it is warmer inside than further out. BUT it is not warmer due to back radiation or the GHE., or added insulation. It can not get any warmer than the only source of energy, the 30KW. Your model calculation of temp at T1 being way more than 30KW going out is just not possible.- you did not add any energy. OR you violated the first law of thermo.(somehow – not sure exactly how yet- but I think that the heat transfer equation is scalar, not vector, & so it implicitly added back radiation and outgoing together multiple times)
Basically my interpretation of the SBL is that you can define any “system” you want ie the sphere up to 10m-, or the PVC hollow sphere from 10 to 13m, or the entire thing, and you must be able to get a continuous E in =E out. at ALL points at equilibrium. If not then how can the SBL calculate 30KW out at r=0 and at r=10m- (ie one micron inside the 10m line), but your greatly enlarged number at 10m (just a micron away), at the edge of the hollow sphere. You just can’t get such a differential & have it be real. (I do like the model- it simplified everything down. makes it easier to discuss.)
I think the confusion comes about because of the various diagrams like K-T, and like the simplified halfup- half down type diagram. They mix and match numbers under differing circumstances.
Let me explain: -the classic half up half down model has 235 coming in to bounce at the ground to get 235 going out, which then hits the GHG layer which is infinitesimally far away – ie an assumed flat plate, r at the GHG level equals r at the ground = r at the space boundary (to eliminate the enlarging surface area phenomenon- which is fine)
BUT since half goes up & half goes down, only 235/2 escapes to space. Obviously this is NOT equilibrium.
If you do an equilibrium condition you get 235 in , & 235 out to space and 235 NET out at every point in between. The way you get this is by the transitory state of coming to equilibrium, On the first ground bounce 235 is converted from incoming solar insolation, of 235W/M^2) to 235 (also 235W/M^2)of IR, this then hits the GHG layer where 235/2 bounces down (as back radiation) & 235/2 goes out to space. The half down bounces for the 2nd time at the ground where it is absorbed & reemitted ALONG WITH the continuous 235 from Solar insolation for 235+ 235/2 going up (NOTE that the diagrams always fail to address what happens to the back radiation but it has to hit the ground and add to the 235 continuously coming in. You can’t have the solar insolation be absorbed but the back radiation NOT be absorbed)
At the 2nd GHG absorption half of this which is now 235+117.5 or 352.5/2 or 176+ goes down instead of the original 117.5 half down.
This continues for 8-10 bounces (about a microsecond) at ground or GHG absorptions, until the half up and half down equals 235- ie equilibrium. Bottom line the half up half down diagram is NOT equilibrium. .It is an instantaneous depiction of the first absorption process that actually takes a few nanoseconds. It is NOT real- we are never at a such a situation as depicted- but it sure explains what is going on. At equilibrium the NET of the time dependant vector energy is 235 up at all points. You do not add the 235 up and the backscatter down unless you apply the up and down signs to get the net number. At the theoretical point between the ground and the GHG layer, the net result is 235 out going plus the equilibrium 235 of to be reflected to space back radiation, minus the final 235 of the last back radiation for a total of 235 NET It is my gut feel, (can’t prove it) that what your T1 calculation using heat transfer equ. did to get the very high T1 number (& to implicitly try to show that the GHE can result in warming without adding energy), was to add all the outgoing plus back radiations without properly subtracting the back radiation pieces. I think it added the the multiple back radiation values required to get to equilib without using negative values since these are (time dependant) vectors not scalar numbers.- this may be way off base, but I cant understand HOW you can get such a high temp when the ttransfer rate at t1- (ie 1 micron inside the the hollow central sphere at r=10- must equal 30KW/area & the temp at 1 micron inside the PVC shell must be so much higher when they HAVE TO be about equal. My answer- the equation while valid for heat transfer is not valid for calculating the temperature ????) I would believe the SBL equation (nothing happens from r=0 to r=10-) before I would raise the temp & apply the heat transfer equ backwards. MAYBE???? My bottom line conclusion is that you cant get a higher temp or increased radiation without adding more energy – ie adding CO2 alone can NOT cause extra warming. & I think your model just proved it.
NOW lets look at the K-T diagram: , which is more complex but also shows more energy pathways & tries to balance them out. (a very useful picture- even if not instantaneously real either) As you pointed out the numbers are annual averaged- they are not real. They are trying to show what happens at equilibrium, ie 235 in and out. If you sum the numbers horizontally at any level like the ground, space or GHG level you get a net change =zero. ie equilibrium. at which time it is in equals out and at every point in between (you can’t create energy!) It also means that there is no heat transfer from the ground/air “System” to the “system” of the rest of the earth.(underground/underwater) but we know very well that it gets warmer in coal and gold mines, and the earth temp at the liquid/solid core boundary is about 5700K or so (source: wiki) AND it varies year over year. So how did it get so hot if we are always at equilibrium and no energy crosses the ground barrier- at least according to K-T??
I also have a problem with the 235 number, or actually the 342 which is 235 plus reflected solar of 107. I can’t get to 235 or 342 from the solar constant of 1366W/M^2!! Even when applying the standard conversion of the Earth diameter area to the half a sphere area that receives the incoming energy. There is missing energy (the subject of a 2010 Trenberth paper) My theory is that the missing energy is not actually missing, it is in the form of outgoing forces of gravity and magnetic field energy that is not measured by the satelites.
I also have a problem with reality. Supposedly the moon (& sun’s ) gravity causes ocean tides.from which we can extract tidal power. SO if it is there doesn’t it also contribute to the ground or ocean temperature? ie the K-T diagram (& the climate models) at the base has ignored this gravity source of energy. Then there is the sun’s and planets gravity. Supposedly the forces of gravity result in maintaining the Earth’s rotation. and in so doing result in Earth torque, which spins the solid core faster than the liquid core causing friction heat, (think ice skater with arms out or in) AND spinning the mantle causing friction and spinning the oceans causing friction and spinning the winds (along with solar insolation heating) causing friction. So with all these heat sources, at all times and especially for the half the time when there is no solar insolation, Wouldn’t these gravity forces and energy, which are way larger than solar insolation, result in an outflow of energy that causes the Earth to radiate at night at a lower temp than during the day when the Solar insolation adds more energy? While the solar insolation & ground heat is providing the 33C of GHE and the direct to space 40 W/m^2 is adding some, wouldn’t these gravity sources provide the remaining 255 of Earth 289K of temperature? AND since these gravity sources vary over time (Earth eccentricity)- especially relative to Jupiter and Venus) couldn’t they be responsible for the time variations in the temperature EVEN when the Solar insolation is relatively constant (since the 60s) and providing a relatively constant GHE of 33C (except for long term gravity caused variations)?, AND thus account for the very real change in the Earth temp (global warming) from the 70s thru 1998? Basically if the insolation is constant and so the GHE is relatively constant there MUST be another source of variable incoming energy & its missing in the K-T diagram.
For further explanation see my paper at http://www.scribd.com http://www.scribd.com/doc/27343303/Gravity-Causes-Climate-Change.
The paper shows how the Earth’s 60 year cooling /warming cycle (…from back a few thousand years to 1880-1940-1998-2058…)corresponds to the Jupiter/Saturn 60 year resonance orbit which causes Earth eccentricity. It also shows how Jupiter itself will cause ~12year temperature sub-cycles (eg1998 to 2010) that result in temperature variations of ~12 year orbital duration (similar to sunspots & solar insolation variations) as Earth approaches Jupiter at their closest point of maximum enegy transfer. It also identifies how Jupiter’s eccentricity also causes Venus’s eccentricity and they combine their closest point to Earth to get temp peaks in 1998 and 2010. (Oct 2010 will be the next peak- 2010 WILL absolutely be the warmest year on record (since 1850) & CO2 has nothing to do with it.) ALSO Jupiter’s eccentricity has ~900+ year cycles that result in roman warming (- 1 to 2 degrees warmer than now according to GISP2 ice core data), middle ages cooling, Viking/Medieval warming, little ice age cooling and current warming.. Jupiter also has an orbit that goes above & below the Sun-Earth ecliptic plane that results in a north -south variation in the incoming energy which coincides with the N-S movement of El Nino/La Nina, explaining that phenomenon. Finally Jupiter has a long term eccentricity of about 50K(needs to be pinned down??) years, that corresponds to Ice age cycles. and since the Milankovitch theory is lacking sufficient solar insolation energy variation to explain the loss of energy in ice ages (see WIKI Milankovitch problems/footnotes), then the addition of gravity induced energy variations solves that problem. Finally increasing CO2 can NOT explain cooling, unless you also explain the variation/reduction in incoming photon/gravity energy.
Please. I would welcome any comments! (on or off line). If you think I’m nuts please explain why scientifically, or ask me to just go away.. Note that if my “Wobble Theory of Climate Change” (170 additional pages also at scribd.com) is correct then we will continue to get cooler and warmer for thousands of years but you will have to change your name from ScienceofDoom to something more cyclical as you continue to work on the myriad of new science that the theory opens up. eg a new K-T diagram. or confirmation of all the numbers I identified , or a new method of predicting droughts monsoons and hurricanes and El Nino changes since we now have a method of identifying and predicting the energy sources vs time. Sorry for the length!
John.
John Dodds
You’re an engineer right…. so this should be obvious, now the properties of what the energy is passing through matters… replace the thought experiment sphere with two spheres, one made of aluminum, one of steel. Now if you input the same amount of energy into each sphere, will they only reach the same T?
Obviously not, aluminum(damn American spelling) is vastly more conductive than steel, the energy will pass through it to the outer shell vastly faster than through steel… so, if the same energy is being input into the spheres, what happens to the extra energy that is input into the steel sphere over the longer time it takes for the energy to reach the outer shell? There will be more energy in the steel sphere…
With the atmosphere, N2 and O2 are largely transparent to terrestrial radiation, so with out the GHG’s the radiation would pass through it without interacting with it. This would mean there is a greater differential in T’s between the surface and atmosphere… So as well as the radiative losses, there will be greater conductive losses from the surface. Im sure you would agree this would mean a cooler surface?
So with the addition of the GHG’s, terrestrial radiation, is absorbed in the atmosphere, this excites them, and they pass off the energy into what ever they bump into(N2 & O2 mostly) So now we have an atmosphere with a smaller differential in T’s with the surface than we would other wise, yes?
So right off, this would obviously mean less conductive losses, even if we ignore radiation… But the same basic principle applies for radiation as for conduction. So the atmosphere is at a higher T, the excited N2 & O2 collides with GHG’s exiting them so they pass off radiation, in all directions. Some will be in the direction of the earth, so this will offset radiative losses from the ground up…
But this is NOT new energy, created energy… this is just slowing its migration to space. The same as steel will hold more heat than aluminum.
Mike, I’m 7th Generation Australian living in the US. I understand Aluminium.
Yes different temps, Since both sphere/shells have 30KW coming in AT EQUILIBRIUM they will both have 30KW going out. and hence by SoD’s SBL equation, the same outer temperature UNLESS the emissivity of 0.8 is different (which it would be) which would result in different surface temperatures in reality but the same total energy in =out. (we can NOT create energy!) They wll get to equilibrium at different rates due to the transmission heat transfer differences and hence different 0.8 emissivity numbers. So at equilibrium there WILL be more energy in the steel sphere, true.. The inner surfaces temps of the shells still can’t differ because the temp is dictated by the SBL radiative heat transfer from the point source to the inner surface.
BUT if the steel shell gets hotter on the inside edge by retaining heat (slower transfer) it will then conduct more energy faster and with the lower emissivity to vacuum end up hotter at the surface. in order to get the same 30KW out.
Para 3, Not sure I understand the “conductive losses at the surface” Our spheres, actually shells, are either steel to vacuum, aluminum to vacuum, or outer edge of atmosphere in a two component double shell, (earth simulator) to vacuum In all cases the (modified with emissivity) SBL applies since ALL energy ( what comes in is going out) is radiated (only) into the vacuum, but at different temps due to the different emissivities. At equilibrium, you can’t lose/retain or gain any transmitted energy along the way in the material or else it will get hotter (colder) and hence radiate faster (slower) and cool (warm) back down (up) to equilibrium. I visualize the SBL as being Mother Natures automatic equilibrium enforcer. It WILL be at equilibrium , in =out, or the SBL will force it back there.
If as you describe the atmosphere is totally transparent (no GHE but can use conduction? & also direct radiation!) Then the rate of heat transmission changes (faster- since a photon that would have been GHE absorbed will now go straight to space with less delay (- conduction is probably changed minimally) in getting to the equilibrium surface temp (which in the no GHE case will differ (lower or cooler ground surface as you said- , not sure about the differential Ts since the TOA temp will also change with the change in no GHE.), and yes the with GHE case does result in higher temperature (but without adding energy after reaching equilibrium conditions) But since the emissivity changes with no GHE, at equilibrium there must be 30KW going out at the lower temp. (or else it will continuously get hotter & radiate more energy out faster to get back to equilibium.)
I think we are consistent in the results. (except the change in conduction) No energy is created or different from equilibrium at any point, but you can get different surface temps and temp profiles for different materials.
My key point (not addressed in your response unless I misunderstood) is that yes you get temp and rate changes during transitions to equilibrium, (happens every morning and night) or with different materials BUT on Earth at equilibrium, even if man adds more GHGs you do NOT get more absorptions or changes in that GHE warming because without adding photons from outside you can NOT get more GHE warming. All the photons are already in use causing the equilibrium and maximized GHE, because there are excess GHGs in the air. and no excess photons. The number of photons, not the number of GHGs dictates the GHE warming and the temperature. I think SoD’s model actually proves my case that you can not create energy, and by adding (creating) energy photons (ie warming) whenever you add more GHGs you actually prove that the IPCC model violates the first law. If more GHGs causes more warming why doesn’t the water in the ocean vaporize immediately? Arrhenius and IPCC are wrong. with their “more GHGs causes more warming”
Ok john, 1st i shall clarify a point.
” Not sure I understand the “conductive losses at the surface” Our spheres, actually shells,”
Taken in context of the paragraph, i wasnt talking about the spheres, but surface interactions of a planet with an atmosphere. But maybe i was just confusing the issue by bringing conduction into it…
But i shall just get this straight, you acknowledge that the properties of a material, will effect its energy carrying capacity? And you understand how the GHE increases the T of the surface of the planet, by increasing the energy contained in the system?
But you believe the amount of energy contained in a system cant change? Or that energy contained in a system doesn’t effect it, passed a certain point?(i suppose to some extent youre right… but the earth is being radiated by a star… and we certainly arnt at equilibrium with its surface)
There are a few contradictions in your logic
So i have a aluminum sphere, we input energy into it, till it reaches equilibrium, then we encase the aluminum sphere with a steel sphere, now by your own reasoning(and mine) this will mean more energy is contained in the system… but from your reasoning, this would mean that this extra energy dosnt effect the system? It cant just disappear.
Energy in will still always equal energy out, but the amount of energy contained in a system matters.
[…] 5, 2010 by scienceofdoom In Part One we looked at a thought experiment designed to make it easier to understand a basic principle […]
John Dodds:
Let me explain.
The Stefan-Boltzmann law tells you the thermal radiation from a surface. And it is proportional to T^4 (where T is absolute temperature).
Within the PVC sphere the only net heat will be due to conduction. Conduction only occurs when there is a temperature differential.
Radiation occurs when there is a temperature above absolute zero and an emissivity >0.
But radiation and conduction are two quite different mechanisms for transferring heat.
Follow the equations. They are correct. And see Part Two.
By the way, the key is that in the case of the sphere, the net radiation at the inner surface is only 30,000 W.
SoD
That ebex experiment was new to me. Of course i was referring to all the other radiation measurement instruments that purport to observe things that are actually achieved by calculation. I cannot get my hands on tech details of ebex but it seems they are measuring heat fluxes rather than radiation. That being the case they’d be measuring convection and ascribing radiation to it. If they are measuring radiation rather than assuming it then I withdraw my objection though I still find the numbers strange and I still see no need to split out back radiation from the net value in the first place.
I don’t see the relevance of your question. I do know that you cannot separate out radiation and convection in 1D calcs and hope to get the right answer. You need an iterative 3D radiative-convective model for that because there are just too many interactions and potentially false assumptions. So forgive me if I am a bit dismissive of these simplified hand calcs that never have to be checked against the real world.
Mark
Try not getting confused yourself before you accuse others of it. The equal up and down radiation refers to the atmosphere radiating in all directions. The T+K diagram does not show that and as far as I can see this is because it would not be possible to make the numbers then match up. Hence the cartoon is misleading from the outset. As I said though there is no need for separating out up from down radiation because nothing depends on it. You can happily use the net value. Hence you ask why the hate, but I’d ask why the love for back radiation? On reading one of Craig Bohrens books I noticed he said some physicists prefer to talk about net and others prefer to talk about back radiation. Nobody needs to be castigated for not accepting one definition over another. IOW There is no need for a love-in with back radiation.
Lastly why would I not believe that generations of scientists have ignored convection in favour of radiation in their atmospheric calcs, when that is exactly what they did with a real greenhouse from which the name greenhouse effect comes? Secondly the textbooks have wrongly ascribed aerofoil lift to the Bernoulli effect for nearly as long. Yes generations of scientists making the same assumtions and making few backup experiments can indeed be wrong en masse. As I keep repeating though, the net effect is the important one. Nasa believes it too.
For the smart guy who understands gravity and why Australians don’t fall off, now try to explain where is all the missing anti matter and just what is dark energy and dark matter anyway? Its easy to be a smart ass but really clever people tend to like to admit to the huge amount we still have to discover before mentioning the remainder that they think they know. Even more clever people have studied scince history and found that the majority have very often been incorrect and always defend the point of view they have spend 30 teaching to others. The single biggest mistake we make is to assume x is not important therefore we can concentrate on y. One day you might learn that.
JamesG
“where is all the missing anti matter”
Not co existing with matter! which explains why we dont really deal with it… and the universe is a big place.
and just what is dark energy and dark matter anyway?
Well the problem with dark matter(and thus dark energy) is that it dosnt interact with matter… the only way thus far found to measure it is through gravitational lensing, its interaction with gravity, the way it warps time and space, and this warps effect on light.
But what we are dealing with here, is measurable/quantifiable daily reality. Have you ever wondered why a piece of iron that is heated up around the 1200k mark glows red? I can see with my eyes the short wave radiation from the sun, and feel its effect on my skin. And how exactly is a satellite going to measure “heat” flux?
Not all is known about the universe, but that dosnt make what can be tested false by default.
[…] of Doom has a nice simple, fully detailed calculation to set people straight on the part or radiative physics they are bound and determined to be […]
Buffoon was right. If you actually had 1.8 plus million Watts radiated off the inner surface, you could use it to power anything you like, without affecting the 30,000 emitted by the source (and thus by the outside surface of the sphere). So you could not only tap the original 30,000, outside the sphere, you could tap that 1.8 million inside it. (Prove that it can’t be done, if you don’t believe that.) If you used that 1.8 million to run another 60 such spherical generators, you could use those to run another 3600, and those to run another–well, you should see that you are fooling with utterly fantastic and entirely unreal amounts of energy, all from that original 30,000 watts. So the mistake is obviously that you don’t know what you are talking about, while Buffoon had it right: The inner surface of that sphere cannot emit as a blackbody, equally obviously because it is in thermal contact with the outside world. I don’t expect you to accept this, but that is the real truth. Your entire exposition is ignorant fantasy. And “climate science” a la Trenberth et al is ignorant fantasy.
To tap into the internal energy, and use it, requires that you are moving that energy from a higher concentration to a lower, so it will have a very finite response before you have reduced the energy down to the equillibrium of the incoming energy/ and out going… you cant get more out o it than 30kw.
Harry Dale Huffman:
Perhaps you can find a thermodynamics or heat transfer textbook which says “the surface cannot emit as a blackbody because it is in thermal contact with the outside world“.
Or perhaps not.
“I don’t like the answer so the explanation is wrong” is not science.
Regardless of the amount of heat being conducted the emission of thermal radiation from a surface follows the same formula.
And just to be pedantic, the example I gave here isn’t “blackbody” radiation. This example has an emissivity of 0.8. A blackbody has an emissivity of 1.0.
The important heat transfer concept is that regardless of whether a surface is conducting 1W/m^2 or 100000W/m^2 it still radiates according to the Stefan-Boltzmann equation (radiation is proportional to the 4th power of temperature of the surface).
Claiming this is “ignorant fantasy” isn’t science.
If we move to the climate system.. satellites measure outgoing longwave radiation from the earth’s climate system of 239 W/m^2 (averaged globally annually), while measurement of the surface reveals that it does follow the Stefan-Boltzmann equation and is globally annually around 396 W/m^2. Check out The Amazing Case of “Back Radiation” – Part Three under the sub-heading “If All the “Back Radiation” Was Reflected..” for one of the measurements.
And back to the hollow sphere in question – the surface radiation can’t be “tapped off” as an energy source. The reason can be easily found by applying basic heat transfer equations.
Well, to prove the example wrong needs a demonstration of which equation is incorrectly applied, or where the maths is wrong.
All you have so far done is explain that you “don’t like it”.
So, please, identify the mathematical flaw.
Some possible places to start –
— Prove that the temperature of the inner surface is incorrectly calculated.
– Prove that the Stefan-Boltzmann equation is wrong
Ignorant fantasy? Or a result that you just don’t like?
You’re adding 1.8 million plus joules per second to the inside of that sphere. You’re going to vaporize your source, or that sphere is going to explode. [moderated out the insult]
Harry Dale Huffman:
Or perhaps you haven’t understood the basics.
Each square meter of inner surface is receiving 1452W from radiation, and also radiating 1452W.
Each square meter of inner surface is receiving 23.9W from the energy source and is also conducting 23.9W through to the outer surface where it gets radiated away.
So how can this inner surface reach a vaporization temperature? We have found the equilibrium, so therefore the system is nicely in balance.
Equilibrium is found when enough temperature difference exists between the inner and outer surface to conduct away the heat from the energy source. Conduction requires a temperature differential to drive heat.
At this temperature difference each part of the inner surface becomes quite hot and is therefore radiating substantially. But its “extra energy” is simply the radiation it receives from the rest of the inner surface.
So instead of insulting people and embarrassing yourself why not try to understand the subject?
1. Do you think the inner surface is at the same temperature as the outer surface?
2. If so, how is any heat conducted through the sphere?
3. If the inner surface is, therefore, at a higher temperature how much radiation does each square meter radiate and how much radiation does each square meter receive?
Okay. In the real world, your hollow sphere would come to thermodynamic equilibrium, the inner surface would have the same temperature as the outer, and it would be in equilibrium with the radiation from the source, and is THEN emitting as a blackbody, just like the outer surface, a total 30,000 watts, which is cancelled by the 30,000 from the source.
The 390 W/m^2 being emitted from the Earth’s surface in the Trenberth diagram is higher than the average total power provided by the Sun, and is unphysical. The whole world of science is incompetent. Believe it or not.
And you’re still [moderated out the insult] for letting this go on so long, and even when the thought occurs to you that the inner and outer temperatures of the sphere must come to the same temperature, you throw away that gleam of commn sense. Well done for thinking of it, F for throwing it away.
I suggest–strongly, and as a professional scientist–that you shut it down for a while, get in touch with everyone linking to this web site as something to follow for good science, and consider very seriously what I said about the general level of competence in science.
Harry Dale Huffman:
If the inner surface is at the same temperature as the outer surface then no heat will be conducted from the inner surface to the outer surface.
Conducted heat requires a temperature differential.
For a planar wall, the simplest case to understand:
q = -kA . ΔT/Δx
ΔT is the temperature difference, Δx is the thickness of the wall, and q is the heat flux. If ΔT = 0, then q=0.
This means that the energy source of 30,000W has no way of leaving the inner surface – because the only heat transfer mechanism to reach the outer surface is conduction.
As a result, in the scenario you paint, there would be an accumulation of energy but no change in temperature. This would be a violation of the first law of thermodynamics.
It doesn’t solve the problem. If the inner surface emits “like a blackbody” (see note) and emits 30,000W – where is it radiating?
This 30,000W is being radiated from the inner surface. This doesn’t transfer it to the outer surface, it simply moves it from one area of the inner surface to another.
Please explain how energy is moved from the inner surface to the outer surface if there is no temperature differential. What is the mechanism:
– conduction?
– convection?
– radiation?
– magic?
And if energy is not moved from the inner surface to the outer surface why doesn’t the inner surface temperature increase?
Note – “radiating like a blackbody” is not something that surfaces do when they have an emissivity of 0.8. Many surfaces do not radiate like a blackbody, and the example in this article is stipulated to radiate with an emissivity of 0.8. Probably the concept intended is to radiate according to the Stefan-Boltzmann equation, which is blackbody radiation x emissivity.
Harry Dale Huffman:
Suggest all you like “professional scientist”.
If you can prove that heat flows through a material when the inner and outer surfaces are at the same temperature you will be on that coveted plane to Stockholm to pick up your Nobel prize. Everyone will know your name.
Pick up a thermodynamics text book and look at the “Conductivity” section. You will find that the equation of conducted heat requires a temperature differential:
q = -kA . ΔT/Δx
See the comment above for more explanation, written before I pulled your last comment out of the moderation queue. It picks up childish insults.
Pick up 50 books on thermodynamics and you will find they all say the same thing.
So if you are a “professional scientist” you should easily be able to explain the questions posed above.
[…] This post “follows” on from Heat Transfer Basics and Non-Radiative Atmospheres and Do Trenberth and Kiehl understand the First Law of Thermodynamics? and many other posts that cover some […]
New post added – Heat Transfer Basics – Part Zero
My email has been screwed up so I didn’t see this until today . Over on
http://hockeyschtick.blogspot.com/2010/09/shattering-greenhouse-effect.html
you said :
ScienceofDoom has left a new comment on the post “Shattering the
Greenhouse Effect”:
I’m not sure what the relevance of your example is . The issue is the
temperature mean and profile for externally heated planets . I contend
that the mean temperature for an externally heated sphere will come to
the temperature corresponding to the total energy calculated acording
to Stefan-Boltzmann&Kirchhoff impinging on its surface .
I contend the only way for Venus to exhibit a surface temperature more
than twice that of a gray sphere in its orbit is to have an internal
source of heat . Perhaps you are agreeing with me ?
Your comment here jogged my memory that myself and a few others explained the same point a few times – so if you don’t agree with it no point going over it again.
Absorptivity/emissivity is a function of wavelength. Solar radiation is centered around 0.5um, while terrestrial radiation is centered around 10um – with little overlap.
This means that a planet can have an absorptance for solar radiation that is totally different for its emittance for terrestrial radiation. This is the case with earth.
The effective result is that solar radiation warms the earth from the planet’s surface (because the atmosphere is mostly transparent to solar radiation), while the planet cools to space from high up in its atmosphere (because the atmosphere is opaque to terrestrial radiation).
This is similar to the example in this article.
You still did not address in what way your example with an internal heat source is relevant to the externally heated balls .
I just posted this on another blog :
Ah , peer review !
What is absurd is that even PhDs in “climate science” either have never learned this most basic physics , or think it is optional . The Stefan-Boltzmann law says energy density is proportional to the 4th power of temperature . Virtually all our energy comes from the sun . It subtends about 5.41e-6 of the total sky . The most commonly cited value I’ve seen for its temperature is about 5778 kelvin . Thus
( 5.41e-6 * 5778 ^ 4 ) ^ % 4 |->| 278.7
which is about 9 or 10 centigrade below the 288 kelvin commonly cited as our observed mean temperature , up perhaps 0.6c since before the steam engine , essentially nothing since the 1930s .
As I learned reading boy’s science books in the 1950s , and was essentially Kirchhoff’s ( and Stewart’s ) insight 150 years ago , this computation applies to any gray ( flat spectrum ) ball , however dark or light . What’s absurd and an indictment of our government education is that anybody graduates from highschool physics without understanding this fact .
The commonly claimed 255 kelvin computation is in fact a little beyond what is possible for even the most extreme spectrum which displays our approximately 0.7 absorptivity with respect to the Sun’s spectrum .
If you disagree with this extremely classical computation , experimentally falsify it .
I DARN well know how to calculate the temperature for any multi-colored ( multi-spectral ) ball heated by sources of any spectra .
It does NOT come close to explaining Venus .
Bob Armstrong:
I can’t even understand what you are writing.
If your point is something different from the earlier discussion, then have another go.
Energy absorbed by the earth’s climate system (surface and atmosphere) = 239 W/m^2 (globally annually averaged).
This is because the albedo is approximately 30%, ie reflected radiation is 30%.
We measure this reflected solar radiation by satellite.
We measure incident solar radiation by satellite.
There is no confusion about these numbers.
We can relate 239W/m^2 to an “effective radiating temperature” of 255K. (This is the blackbody which would produce 239W/m^2).
See The Earth’s Energy Budget – Part One .
It’s the most straight forward computation of the energy impinging on a point or ball in our orbit ; size doesn’t matter . Since the computation is temperature to temperature , the Stefan-Boltzmann constant drops out . Multiplying the total energy from the sun
( 5.41e-6 * 5778 ^ 4 ) |->| 6.03e9
by the SB constant gives 341.9 W/M^2 which correspond to the temperature I calculated of 278.7 .
This is the temperature for any gray ball , NOT just a black ball because absorptivity/emissivity is constant across the spectrum and thus falls out .
Your absorptivity value of 0.7 is essentially the measured correlation of of the earth’s spectrum with that of the sun . The claim , which gets sloppy is that the spectrum of the earth without an atmosphere is so (infra)red that the correlation with all of the sky other than the sun is approximately 1 . Even with the most extreme “anti-greenhouse spectrum that can’t be true because the emissivity is measured to be on average 0.7 over the solar spectrum .
The essential point is that the unbiased hypothesis is that of a flat spectrum gray ball . From that , given the observed absorptivity/emissivity spectrum of a planet , a precise equilibrium temperature can be calculated . It appears the moon is slightly “red” because its mean temperature is measured to be a few degrees below the gray temperature .
I hope this clarifies things .
Now I’ve got to get out and vote for Freedom candidates and issues .
Clarifies?
The earth’s climate has incident radiation of 342 W/m^2, but absorbs only 239 W/m^2. We know this because we measure the reflected portion.
The top of atmosphere radiation measured from the climate system is also 239 W/m^2.
Which is unsurprising. Energy in = energy out, within measurement uncertainty.
However, from your many previous comments I don’t think you understand these simple points.
A GRAY ball of 0.7 absorptivity absorbs 239 W/m^2 .
It also emits 239 W/m^2 .
Thus its albedo makes NO difference .
It is only the assumption of an extreme “anti-greenhouse” spectrum which comes close to producing the asserted “cold earth” , as Marty Hertzberg puts it . That is an unwarrented BIASED assumption , and a mathematical confounding of mean absorptivity/emissivity with spectrum .
Given any particular spectrum , the equilibrium temperature is a rather straight forward calculation .
It’s the first law of thermodynamics that means the energy in = energy out.
The emissivity & absorptivity of the earth’s climate system for longwave radiation is NOT 0.7.
There is no requirement for the absorptivity of a body at 0.5um to be the same as the absorptivity of the body at 10um.
Snow and ice for example are highly reflective for shortwave (solar) radiation, and yet are highly absorbing for longwave (terrestrial) radiation.
This is very basic and has been explained to you a few times already. It’s in all the thermodynamics textbooks that cover radiation.
Ok then , calculate the ACTUAL equilibrium temperature for the spectrum of snow . Prove that “snowball earth” would be stable given the current temperature of the sun .
In the several years I’ve been diverted from productive activity by this endless debate , I have seen no forward progress from the simple parroting of the most crude and extreme calculation .
I have not , in fact , seen evidence that the “climate science” community actually knows how to calculate actual values .
Surely some student interested in the field has the time , which I do not , to actually find and confirm the relevant broad band spectra , and present actual equilibrium temperatures for substances of interest .
Bob Armstrong:
Your question does not make sense.
There is no such thing as “the equilibrium temperature for the spectrum of snow“.
By that statement there is no equilibrium temperature for any object .
It just reveals what I’ve been pressing all along , tho you parrot the case of an object which due to its spectrum absorbs only 0.7 of the radiation of the sun’s spectrum , yet ( impossibly ) radiates 1.0 of the heat it absorbs , you don’t know how to compute the actual temperature for any actual spectra .
This is why I keep pounding at the pathetic state of the understanding of this very most basic physics .
If one doesn’t know how to compute the temperature of a radiantly heated uniformly colored ball , how can one say anything quantitative about more complex problems ?
Bob Armstrong:
There will usually be an equilibrium temperature for an object in a particular situation.
That wasn’t your question before.
It is very easy to calculate the equilibrium temperature of a planet heated by the sun.
Ein = I.(1-a)/4 [eq 1], where I is incident radiation, a is albedo (reflectivity)
Eout = εσT^4 [eq 2], where ε is emissivity, σ=5.67×10^-8 and T is absolute temperature.
Ein = Eout [eq 3]
Therefore, T= (I.(1-a)/(4εσ))^0.25
Solving for the conditions of the earth, a=0.3, ε=1, therefore T=255K.
The factor of 4 in [eq 1] is to allow comparison of incident radiation from a small angle (“captured” by a disc of area pi.r^2) with radiation emitted from all around a sphere (with area of 4.pi.r^2).
And ε ≠ (1-a)
The reason why is that “a” and ε are both functions of wavelength.
In the equation above ε is the value around 10um (the peak wavelength of radiation from a 290K body), and “a” is the value around 0.5um (the peak wavelength of radiation from a 5780K body.
If the solution to the equation meant that the equilibrium temperature was much higher, then obviously we would need to know ε at the relevant range of wavelengths.
And please, read this and study it before sounding off in your usual manner about what an idiot everyone else is.
You seem unable to even take in a simple graph of absorptivity from a textbook.
1. Absorptivity is a function of wavelength and direction
2. Emissivity is a function of wavelength and direction
3. Absorptivity = emissivity from Kirchhoff’s law – at the same wavelength and direction
4. Solar radiation is emitted in a totally different range of wavelengths than terrestrial radiation
Read these 4 points. Try and understand them.
If you don’t understand them, don’t assume that the rest of the world is talking nonsense. There is an alternative explanation.
Actually , the computation of the equilibrium temperature of any arbitrarily irradiated , arbitrarily “colored” sphere is what the algorithms on my website address .
You keep repeating the same well understood facts , but you clearly don’t know how to go from the spectra of radiation sources impinging on a sphere , and its pattern of colors ( spectra ) , to its equilibrium temperature .
Bottom line , you do not know how to calculate the temperature of a radiantly heated colored ball .
I do , and am working to make the executable algorithms more and more understandable on my website .
Since you’ve dropped a number of comments at my site referencing this post, I thought I would return the favor with a new post titled
“Why ‘Science of Doom’ doesn’t understand the 1st Law of Thermodynamics”
http://hockeyschtick.blogspot.com/2010/11/why-science-of-doom-doesnt-understand.html
Firstly, the system described in this “thought experiment” doesn’t violate the Second Law. Energy input to the system = energy output from the system. The second law says nothing about consequent internal heat transfer/flows in the system at all. The high internal temperature of the sphere results from all the 30kW/s radiation which heated the sphere before it attained equilibrium. At equilibrium, the 30kW/s merely maintains the external radiation.
Secondly, there isn’t a FLOW of 1,824.9kW/m² from the inner surface inwards, it’s a FLUX – each sq.m of the surface is opposed by the sq.m opposite, which is radiating the same. The fluxes have opposite direction, therefore sign, and cancel out. There’s no net flow towards the centre, from the initial phase where the interior of the sphere is quite cold, to the equilibrium state when it’s hot. There’s nothing fundamentally wrong with this post’s conclusions about this thought experiment.
Lastly however, does this validate Kiehl and Trenberth’s “energy budget” diagram? Only if they’re correct in assuming that GHGs don’t radiate isotropically – in every direction. If their 1997 figure of 324 W/m² DOWNWARD is correct, then this must be mirrored by an equal UPWARD amount. This would mean the GHGs are radiating a total of 648 W/m². SB with e=1 (which they incorrectly assume) gives a temperature of 72.5°C. A little warm IMHO.
Note also, the diagram shows no albedo or emissivity effect for absorption of LWIR from GHGs at the surface, nor an emissivity <1 for radiation from the surface. I was glad to see this article use a nominal emissivity of 0.8 for the sphere. There's plenty of evidence for supposing that the earth's emissivity is similar. It most certainly can't be a perfect black body with emissivity 1. If that were so, radiation physicists wouldn't need to struggle to create reliable black body reference sources for their experiments – all they'd need would be a handful of dirt or a bucket of sea water.
After pointing out the flawed heat transfer analysis in the fascinating article – Why ‘Science of Doom’ Doesn’t Understand the 1st Law of Thermodynamics
I asked:
I should have suggested a new title for the article as well, but that would have been cruel.
I wondered what the response was to my comment, but the article has disappeared.
Let’s hope it is temporary.
No doubt a reworked article with correct heat transfer calculations is in the making.
The old article is back, but it still has the same flaws.
The comments from the writer indicate they have realized that they don’t know how to do the calculation.
To All:
Please read the comments beginning here
http://hockeyschtick.blogspot.com/2010/11/why-science-of-doom-doesnt-understand.html?showComment=1289569099824#c6931012645424475282
SOD’s use of the conduction equation was done wrong.
He used 30,000 watts for the value qr when qr is actually the watts tranfered by conduction (2390 watts).
He then came up with a temperature difference of 290K when it is actually 23.1 K and back calculated the watts at the source to be 1.85 MEGA-WATTS!
No doubt SoD will now rework his article with correct heat transfer calculations
On the Hockey Schtick we have a system with 30,000W input and 24,000 output. If the system is at equilibrium then 30,00W MUST be transmitted and radiated at the outer surface. What happened to the 6,000W that’s missing? Arguing vectors can’t explain a violation of the conservation of energy law.
Arguing also that a 30kW heater can’t heat the interior of a surrounding 20m sphere to more than 135.47 K won’t cut it – it implies that it would be impossible to heat a large house or small hall with a 30kW heater.
I hesitate to say it, but the following is a masterpiece of confused logic and maths:
24,000 Watts = 19,200 watts absorbed by outer sphere and re-radiated + 2390 watts transferred by conduction to the outer sphere surface and radiated + 2410 watts stored in the PVC that is also radiated.
It seems that 19,200 watts magically appears at the outer surface – it can only get there by conduction from the inner surface. That conduction is however stated to be 2390 watts, and apparently doesn’t include the 2410 watts “stored” which somehow (more magic) instantly appears at the outer surface.
The miscalculated 24000 watts is either conducted to the outer surface or it’s not. If only 2390 watts is conducted then that’s all that will appear at the outer surface (somewhat less per sq.m of course, because of the larger outer surface area). If only 2390 reaches the outer surface then the system can’t be at equilibrium, and the calculation of the surface temperatures is wrong.
As I said before, I can’t see any obvious defect in the heated sphere setup outlined here, though I disagree with the implied vindication of K&T.
In my opinion – as an engineer – this kind of articles are just showing that both K+T and the authors of this article have no idea neither of simple thermodynamics nor of basic physics.
The example you are showing, of a sphere (Earth) whose inner core is radiating 30.000 W to the surface is ridicolous.
Please, ask yourself this simple question:
Why inner core of Earth reaches temperatures up to 5000-6000 ° C, while at surface we have just temperatures ranging from the lowest – 89.2 °C (Vostock, Antartide, 1983) up to the highest + 58 °C (Libya desert, 1922) , and averaging to 15°-16° C?
And why inner core of Sun reaches temperatures up to MILLIONS °C, while at Sun’s surface we have “just” 5600° C?
Because of PRESSURE, PRESSURE, (and again) PRESSURE!
Forget all these Stephan-Boltzmann + equations of heat conductions, you completely missed the point.
Heat from inner core of Hearth cannot flow to surface, simply because of THERMAL GRADIENT which prevent this could happen.
Whenever you have atmospheric gases + Earth layers (from Earth surface to inner core) having different thermal gradients, you simply have to look at this general equation of gases:
PV = RT
i.e.: P is pressure, V is volume, R is universal constant 8.31 Joule/°K and T is Temperature.
And so, you can write down also PV = T but this means that whenever pressures rise at the same volumes, then temperatures rise too!
So, heat cannot flow from inner core to outsider cores, just because of HIGH PRESSURES, that’s all,and of course outsider cores at lower temperatures cannot raise the temperatures of inner cores (and that’s 2nd thermodynamics: lower temperatures cannot raise higher temperatures)
And of course atmospheric gases at lower temperatures and at lower pressures cannot raise the temperatures of Earth surface (where you can find higher temperatures, and higher pressures).
Even if you to Termosphere, which is highest point of terrestrial atmosphere (between 80 – 200 km. up) you theoretically can find very high temperatures, because of huge kinetic motion of molecules, but in reality it is VERY COLD, because of very low PRESSURE and DENSITY of air.
And of course K+T diagram is ridicolous, because first of all you don’t have 390 W/m2 radiation at surface while having just 168 W/m2 solar irradiance (no sperimental evidence of 390 W/m2 at surface!), but above all you cannot have 390 W/m2 from surface less 324 “backradiated” (budget is just 66 W/m2) while having at the same time 235 W/m2 outgoing to space.
390 – 324 cannot give 235 outgoing radiation to space.
The point is that K+T diagram puts 168 W/m2 solar radiation in surface less 78 W/m2 evaporation/convection, less 24 W/m2 conduction, then there are just 66 W/m2 at surface, OK?
But now they magically add up a fake 324 W/m2 going both out and in as “backradiation”, just to show that there’s a bogus greenhouse effect, and of course 324 out – 324 in gives always 0, the system “equilibrates”.
But system equilibrates even whenever you put 1000 + 66 W/m2 going out less 1000 W/m2 backradiated, or if you put 10 + 66 W/m2 going out less 10 backradiated, as Heinz Thieme pointed out, you can have infinite figures like x – x and the result is always 0.
Therefore, forget the inner core of Earth and 30.000 W: K+T DON’T know thermodynamics!
Ah, by the way…
There is no need to “fight” against the K+T diagram, because – looking carefully – it does not prove ANY greenhouse effect at all!
Even allowing that we have 390 W/m2 surface radiation outgoing to the atmosphere, and 324 W/m2 “backradiated” by gases…. and so?
If you have 390 out and 324 in, through a surface, then you have just 66 W/m2 budget, because according to heat flux equations, http://en.wikipedia.org/wiki/Heat_flux
two heat fluxes passing through a surface, and coming from opposite directions have to be SUBTRACTED, and not added up.
Therefore, if the budget at surface is just 66 W/m2 out, this means that there’s no increase of temperatures on surface by “greenhouse gases”, but just a “slowing down” of the rate of cooling.
But, as IPCC maintains that greenhouse effect means that gases INCREASE the temperatures at surface, sorry but this cannot happen, even according that diagram.
As I said above, the most important things are thermal GRADIENTS and PRESSURES of gases.
But you don’t ’have a greenhouse effect even whenever you have a thermal inversion, as at Poles, or in polluted urban areas, or after volcanos’ eruptions.
In those case you have surface temperatures LOWER than the one of atmosphere up to 2-3 km., and so gases do not succeed either to increase temperatures at surface, as greenhouse theory pretend.
Two centuries ago, after the great volcanos’ eruptions in Indonesia, Earth surface temperatures dropped, and there was no summer in 1816, although atmospheric temperatures were higher.
Where is greenhouse effect on Earth?
Greenhouse theory is just a bogus theory, no experimental evidence can prove it!
Engineer:
Read The Amazing Case of “Back Radiation” – you will find that the back-radiation is a measured value all around the globe.
You don’t understand radiation. If the atmosphere did not interact with the radiation then you would expect the top of atmosphere upward radiation to be the same as the surface upward radiation.
The atmosphere does interact and you need to solve the radiative transfer equations to calculate the change in radiation.
Read Theory and Experiment – Atmospheric Radiation.
On why higher pressure doesn’t necessarily cause higher temperatures, read Convection, Venus, Thought Experiments and Tall Rooms Full of Gas – A Discussion.
That should be enough for now. Feel free to post your questions about those articles in the comments there.
[…] 14, 2010 by scienceofdoom When I wrote Do Trenberth and Kiehl understand the First Law of Thermodynamics? I imagined that (almost) no one would have a problem with the model created. Instead, I thought […]
Sorry, but your “answer” doesn’t answer nothing.
I didn’t say that there isn’t backradiation, clearly there is!
But first of all it is not true that there is an “average” figure for 324 W/m2, and it doesn’t make sense to calculate an “average” figure, from a physical standpoint.
Moreover, what means this:
“If the atmosphere did not interact with the radiation then you would expect the top of atmosphere upward radiation to be the same as the surface upward radiation.
The atmosphere does interact and you need to solve the radiative transfer equations to calculate the change in radiation.”
This means nothing, gases cannot send downward 100% radiation, because gases do transmit radiation in every direction, so you should get at most 50% down and 50% up, and not 100% up and 100% down, as ridicolously wrote Gavin Schmidt, forgetting that irradiance law does not allow an inert gas to reflect, transmit, absorb more than 100% of received radiation.
Moreover, it is not true that higher pressure do not cause higher temperatures, and regarding Venus, you disregarded both the 92 bars at surface, but above all the fact that – as prof. Nasif Nahle rightly wrote – Venus HAS NO MAGNETIC FIELD (while Earth has a strong magnetic field) and therefore solar winds and plasma heated particles are not deflected by a magnetic field and they do heat Venus surface.
Finally, regarding the K+B “model” it is enough to say that even NASA does not use anymore that model, and they do not consider at all backradiation, in their model of radiative exchanges.
Moreover…
It is not true, as you wrote above, that without atmosphere IR radiation from surface would 100% escape to space.
Without atmosphere, and without clouds and albedo effect, there would be more solar irradiance at surface, http://www.climate4you.com/ClimateAndClouds.htm#Cloud%20cover%20effects%20on%20climate and Earth surface, and soils would keep solar radiation more than Stefan-Boltzmann predictions, therefore Earth would heat more!
As Alan Siddons + Gehrlich correctly demonstrated, Moon (that has no atmosphere at all!) is warmer for 40°C than predictions from Boltzmann constant, because Boltzmann is right just for flat 2D surfaces, but not for 3D rotating planets, and it is uselles to calculate how much heat is retained by heated soils.
NASA knew that as early as the beginning of sixties, it is well known, they did not use Boltzmann + Kirchhoff + Wien to calculate Moon temperatures for Apollo’s Moon landings at the end of sixties.
But above all remember what Von Neumann once said: “Give me a model, and I can make an elephant dance!”
K+ T model is wrong and useless to depict real heat exchanges in Earth atmosphere, mainly because Kirchoff law + Stephan Boltzmann (on which those fluxes are being calculated) are theoretical equations that can ONLY describe RADIATIVE emissions of heated bodies.
But, as heat echanges on Earth + planets and stars are mainly CONDUCTIVE (i.e. changes of kinetic energy of a body) + CONVECTIVE (i.e. heat transmission from heated fluids movements) heat exchanges, a very rough equation like W/m^2 = σ T^4 fall short of exactly calculating the amount of heat which is stored by planets surfaces, and therefore results are wrong and misleading, and so are “models” like K+T.
It is easy to prove it sperimentally.
According to “blackbody” predictions, Earth surface should be 33°-34° C colder, then the simplistic hypothesis is that the difference is due to the presence of atmosphere which is “warming” the surface as a greenhouse effect.
But please, read hereinafter real data collected by NASA + satellites:
Click to access Rethinking_the_greenhouse_effect.pdf
Venus receives 2164 W/m2 solar constant irradiance, but in the range between 0.1 up to 1 atmospheric bars, Venus temperature raises for 100° C
Earth receives 1368 W/m2 solar irradiance, though its temperature raises for 68° C
Jupiter receives just 50.5 W/m2 solar irradiance, but its temperature raises for a big 53° C
Saturn gets just 14.9 W/m2 from Sun, but temperature raises for a huge 50°C
Uranus gets a small 3.71 W/m2 from Sun, yet its temperature raises for a great 23°C
Neptun receives a very small 1.51 W/m2 solar irradiance, tough its temperature raises for an enormous 17° C!
What data above mean?
Simply that they clearly contradict the Boltzmann “blackbody” calculation, Jupiter heats more than 150% than Boltzmann preditions!
ALL planets and satellites like Moon show true temperatures which are very different from “blackbody” radiative hypothesis.
On the contrary, true data show that gravitation, atmospheric pressure, convective heat transmission, are the main factors to explain heat trasmission, while radiative forcing and Boltzmann equations are clearly rough and misleading for this purpose.
We are about 10c warmer that a gray ball in our orbit . If you’re an engineer , show me the actual calculated temperature for a ball with our measured spectrum .
I think you’re talking at cross purposes, and neither side fully understanding the other because the different paradigms not fully appreciated. I’ve been following some of the discussions here and although not a scientist or mathematician can see where the problem is because recently I cracked it for myself in a discussion elsewhere, and the arguments from the applied scientists here have reminded me of this. I think it was John Millet who said something like ‘you’re missing out a step, there is a medium’.
How I discovered the sticking point was in discussing the AGWScience concept of ‘Carbon Dioxide accumulates in the atmosphere and stays up hundreds and even thousands of years’, the amount varies and arguments about how long between those two not important, it’s the concept that it ‘stays in the atmosphere for extremely long periods of time and accumulates’ which is topic.
After several frustrating discussions where I was being told that ‘carbon dioxide is well-mixed in the atmosphere and doesn’t separate out’ in reply to my saying the concept can’t be true because carbon dioxide is heavier than air, I finally decided to concentrate on finding out why I was being told this, and so, why I wasn’t being understood.
In the standard science I was taught, gases had weight and separated out unless work was done to change this. In this final discussion I eventually managed to show from lots of examples that carbon dioxide did indeed flow to the ground and in large amounts and would pool, as it does in breweries, volcanic eruptions, Cameroon lake a recent grave problem and so on, and gases in general would separate out as well known in mining, where not to know this aspect of gas’s properties can be detrimental to one’s health.
This point was conceded to me. However, I was arguing with a PhD in physics AGW who taught his subject, and he would not relinquish the idea that carbon dioxide is well-mixed in the atmosphere and can’t then separate out.
I suggested a scenario to make sure we fully understood what the other was saying. Take a room, Carbon Dioxide is introduced in a large enough quantity for it pool on the floor. Nothing changes in the conditions of the room from what it was which allowed the carbon dioxide to pool, i.e. no work was done, no window opened, no fan switched on.
I said it would stay where it was. He said it would diffuse into the atmosphere and become thoroughly mixed and it would take work to unmix it.
I said his idea was impossible because carbon dioxide was heavier than air, 1.5 times heavier, and couldn’t on it own volition rise back into the atmosphere. He said that molecules travel at great speeds knocking into each other and so thoroughly mix up together. He pointed me to gas laws to prove that this was the behaviour of gas molecules so I was wrong. I began researching gas laws.
And discovered that these referred to ideal gases, not real. The ideal gas is an imaginary gas, it doesn’t actually exist. It is useful in some calculations just as the imaginary average is useful. To find how a real gas acted in the real world, stages would have to be gone through from the initial gas law to arrive at an approximation for a real gas. The ideal gas laws do not describe real gases, in the real world. They do not have volume, not subject to gravity and so on, to use them to describe how CO2 would act in the real world is actually non-sensical, but because of AGWScience this is firmly believed to be true.
Recently I saw a research paper from AGWScience which set out to prove that methane separated out at the ceiling of a mine would thoroughly mix back into the atmosphere of the mine. It didn’t happen. The AGWScientist concluded that there must have been an undiscovered source of methane entering the test mine which was causing the methane to continue pooling at the ceiling. The reality in RealScience is that methane separates out because it is lighter than air and so will form a layer at the ceiling of a mine. Miners know this well, they used to, perhaps still do in countries where mining is not well equipped, cover themselves in wet towels and enter new mines with a lit candle on a long pole to set fire to any methane there before mining could begin.
But as with the above example, the AGWScientist I was arguing with couldn’t let go of his erroneous view, he proposed that the CO2 wasn’t actually sinking on its own, because of this idea that gases are well mixed, so it was actually sinking in a ‘package’ of air in which it was the predominant constituent. It is so much simpler to accept standard, well known, observed, tried and tested, real science, than propose answers when to explain an imaginary hypothesis when observation fails to confirm it.
Air is a medium. Our atmosphere is not a vacuum. Our atmosphere is like an ocean of air above us and is subject to gravity and pressure and the molecules comprising it have volume, react with each other – they do not travel at huge speeds in empty space bouncing off each other. Real gas molecules move in a real medium, air, not empty space.
The ideal gas is not real, it does not describe a real gas molecule in the real world because it does not behave as a real gas.
Carbon dioxide is 1.5 times heavier than air, it sinks through air, displacing it. The key word here is displacing. A description from RealScience. That’s how weather works. That’s how water vapour which is lighter than air rises bringing heat away from the earth’s surface, forming clouds.
A good way to appreciate how air is a medium is to think of how sound waves travel, as does water in the sea, the water doesn’t move, energy moves. Sound doesn’t move through empty space to reach your ear, nor does it move in a packet of air to reach your ear; the air hardly moves. Neither does carbon dioxide move in a mixture of molecules to pool on the ground bringing this mixture down with it..
And so the problem here with the laws of thermodynamics. These are well known and used by practical scientists in all kinds of disciplines, their work would be nonsense if things didn’t happen according to these laws; tried, tested, observed, utilised. If you can’t work out why there is this extra heat don’t rewrite the laws as your first step. When something is a law it is not a hypothesis, nor even a theory, try fitting the parameters into the laws first. If these laws didn’t work as they are actually written then we would know by now, the majority of our modern applications wouldn’t exist.
Good luck, I’m interested to know why the first plate heats up.
Here’s a good description of how sound travels in the medium air. http://www.mediacollege.com/audio/01/sound-waves.html
Engineer:
When you write statements like (November 15, 2010 at 4:10 pm):
It demonstrates that you haven’t taken even a short amount of time to try and understand the subject.
Take a look at the K&T diagram. They include sensible and latent heat (convection). Conduction is only important at the surface of the earth as part of the convective heat exchange as the atmosphere has an extremely low conductance.
Read their paper.
Myrrh,
Do these rooms where CO2 pools have wind or convection currents? Wind can actually do work and mix gases. Air samplings at various altitudes have confirmed this. We see the same sort of thing with the creation of clouds. The condensation nuclei for the water droplets are very tiny pieces of dust, salt, etc. These are even more weighty than CO2, but they get up there.
Of course, you already know this answer if you actually read answers to your previous questions.
No, the set up of conditions for CO2 pooling in the room was that there were no windows open or fan on, no movement of air. So I said that without a window being opened or fan put on the CO2 would stay pooled, he said it would diffuse into the atmosphere without any work being done, such as no window opened or fan put on.
I read up quite a lot in investigating this, it’s surprising how something remembered from early school days takes on a new lease of life as it’s explored further! An interesting aspect here, in my previous discussions about the CO2, was that other examples of its properties were also dismissed and slightly tweaked as with the weight to conform to AGWScience, tweaked enough to make it seem plausible. I was given examples using dry ice to show that it diffused into the atmosphere at room temperature, that CO2 is invisible had to be ignored, (it took a while to discover they were mistaking water vapour for CO2), and I was given a long explanation of how it was toxic to the body, regardless that it has long been designated as non-toxic in RealScience!
It sometimes seemed to me that there had to be ‘a someone’ originally who knew CO2 properties really well to be able to create examples of experiments and explanations to deliberately confuse in the first place. There was one such on the BBC a while ago.
Supposedly a group of believers and one of sceptics were taken through an experiment where a flask of air and a flask of CO2 were heated, it ‘proved’ they said that CO2 gets much hotter and more quickly than air and to oohs and aahs the ‘sceptics’ were convinced there must be something in AGW after all. That there was no explanation of heat capacity, and so the real differences between the two which they were observing wasn’t forthcoming.
There was enough ‘theatre’ around this that it wouldn’t have registered to the ‘average’ person who might vaguely have remembered something from school days or if younger never been taught, how experiments should be conducted. I don’t know how many like me were watching who didn’t trust the example and so made an effort to explore further. That’s how I learned about heat capacity of gases.
If they had also timed how long it had taken each sample to cool they would have been able to explain this in real science terms and so wouldn’t be taken in by such AGW explanations as ‘CO2 traps heat forming a blanket’, because they would have seen that CO2 also loses heat very quickly while the flask of air which took longer to heat would also have taken longer to cool down – because it must have had consituents with a higher heat capacity, such as water vapour, which take longer to heat up and so to cool down. And I’ve just realised that is was knowing this that has helped me think through something recently, that water vapour in the atmosphere takes heat away from the earth as it rises to form clouds and precipitate out as rain, and that the earth would be a lot hotter if that wasn’t the case, like deserts.
I was originally obsessed with this nonscience because of learning in childhood science books what is essentially the result of Ritchie’s 1833 demonstration that albedo does not effect the temperature of a gray body .
The understanding of experimentally confirmed macroscopic physics has declined sadly since my school days .
For new readers, Bob Armstrong keeps popping up saying how downhill science has gone since 1833.
Various people have tried to explain the absolute basics to Bob, including me in – this explanation.
Pointless to explain it again to Bob. Just a note for new readers.
Until I see someone actually calculate the the equilibrium temperature of a radiantly heated ball with a given absorption/emission spectrum , I will maintain my assertion that the general level of understanding of the essential basic physics of this field has regressed since the advent of the label “climate science” .
I suggest anyone who wants an introduction , with actual computations , to the problem browse my website .
I actually only found that Ritchie’s experiment , http://cosy.com/Science/Ritchie%27sKirchhoffExperiment.html , predated Stewart and Kirchhoff’s theories by 30 years . I had assumed it had been done in confirmation of those ideas in the early 20th century .
[…] the original article – Do Trenberth and Kiehl understand the First Law of Thermodynamics? – I used a simple heat conduction problem to demonstrate that temperatures can be much higher […]
q = -kA . ΔT/Δx
qr = 4πk . (T1 – T2) / (1/r1 – 1/r2)
What did you do with the minus sign in the rest of the maths?
The minus sign is because heat flow is in the opposite direction to the temperature gradient.
It’s not really necessary so long as we remember the direction of heat flow.
Unfortunately your calculations are totally incorrect:
a) The area of the outside sphere is 2122.64m2 giving an energy of 14.14W/m2.
b) The area of the inner sphere is 1256m2 giving an energy of 23.86W/m2.
The temperature of the outer surface is 10k not 133K
There is absolutely no back-radiation or GE apparent.
bananabender:
Your calculation in a) is correct.
Before pointing out your flaw in b) you have made a big mistake in this statement:
A surface with an emissivity of 0.8 radiating 14.1 W/m2 has a temperature, T2 determined by this simple equation:
E = εσT24
So, 14.1 = 0.8 x 5.67 x 10-8 x T24
Therefore T2 = (14.1 / (0.8 * 5.67 x 10-8)1/4
T2 = 133K.
Please review and comment and then I will explain what is wrong with b).
You have also made numerous basic arithmetical errors:
1) The area of the internal sphere is actually 1256m2.
The radiative flux density is 23.86W/m2
2) The area of the outer sphere is actually 2122.64m2
The radiative flux density is 14.14W/m2
3) You said:
“where r2 = radius of the outer surface = 10 + 3 = 13”
In fact r2 = 13*13 = 169
Considering that you can’t even calculate the area of a sphere correctly I think you should give up trying to disprove the Second Law of Thermodynamics.
bananabender:
You haven’t actually shown what they are.
If you read the article you will find that I have stated the outer surface area = 2124 W/m2 and I have agreed with you that the radiative flux density = 14.1 W/m2 from the outer surface.
Actually it is 14.13 W/m2 but I don’t think we are arguing about rounding errors and number of significant figures we are working to.
If you do want to be picky:
Outer surface area = 2123.7166 m2.
Flux from outer surface = 14.126178 W/m2.
The reason you have the wrong value is you have used π = 3.14, but actually π = 3.14159265….
You should be clear that if you use pi to only 3 significant figures you will not get an answer for area which is accurate to 6 significant figures.
And so – back to my comment of March 21, 2011 at 8:35 pm – redo your calculation of temperature with this flux value and you will see that my original number is correct.
Unless you have a different formula for emission of thermal radiation from a surface?
Or you have a different approach to calculating the 4th root of a number?
Why don’t you explain which exact bit is wrong in my formula or arithmetic?
Before you get too carried away with how clever you are, take a look at the maths section.
r2 = outer radius. This is not r2.
A radius is measured in meters, or dimensions of length in other units.
So r2 = 10+3=13 meters.
And the area of the outer sphere = 4πr22.
And if you calculate the area accurately you will find it really is =
2123.7166 m2. Or we can just write it as 2124 m2 for now.
By the way this article is not about the Second Law of Thermodynamics, it is about the First Law of Thermodynamics – and Heat Transfer Basics.
Why are you using the Stefan-Boltzmann Law?
A black body is an idealised cavity radiator of zero thickness surrounded by a perfect vacuum.
Your model is really a radiator surrounded by a thick insulator.
This is an engineering problem requiring a heat transfer solution not a radiative physics problem.
bananabender:
You are now happy with my arithmetic? You have now realized that the mistakes are your own?
Ok.
So, onto the emission of thermal radiation.
It appears you haven’t read any heat transfer textbooks. Don’t let that hold you back from telling people how it’s done.
The correct equation for emission of thermal radiation from a planar surface is:
E = εσT4
where E = rate of energy emitted per meter squared
ε = emissivity, a material dependent property which is usually a function of wavelength
σ = 5.67 x 10-8
T = temperature of the surface
I will paste in a few textbook samples by way of illustration, no doubt in vain. Why would textbooks on heat transfer be more valuable than the blogs you have come to believe in?
bananabender:
Some textbook examples as promised.
From Siegel & Howell, Thermal Radiation Heat Transfer (1968):
From Incropera & DeWitt, Fundamentals of Heat & Mass Transfer (2007):
From Lienhard & Lienhard, A Heat Transfer Textbook (2008),
Chapter 1:
Chapter 10:
You can cut and paste all the textbooks you want but it doesn’t make you correct.
Your understanding of your own model is totally, utterly and unequivocally wrong!
Your model is nothing but a giant thermos with a heater.
Everyone knows a normal thermos can’t heat the coffee.
However if you put a heater inside the thermos the coffee gets hotter. Why? Because the input of heat exceeds the output. However it is the heater that heats the coffee not the outside of the thermos.
You falsely assumed that the outside of the sphere radiates energy towards the inside. This is totally impossible because the only heat source is the internal radiator. Heat transfer is unidirectional from the inside surface of the sphere to the outside along a thermal gradient. The external surface only radiates to space which has zero temperature
I will do some thought experiments to show why you are wrong:
a) Imagine a 30KW heat source surrounded by a perfect vacuum (a radio-transparent perfect insulator):
radiant flux at 10m = 23W/m2. The temperature is absolute zero.
radiant flux at 13m = 14W/m2 . The temperature is still absolute zero.
Why? Because a vacuum is devoid of mass and energy. The radiation simply pases through without heating.
b) Imagine a 30KW heat source surrounded by a perfect radio-opaque insulator(eg a mirrored thermos flask) :
radiant flux at 10m = 23W/m2. The temperature continues to increase (towards infinity).
radiant flux at 13m = 0W/m2 . The temperature is absolute zero.
Why? Because all the energy is trapped inside the inner sphere.
c) Imagine a 30KW heat source surrounded by a radio-transparent perfect conductor.
radiant flux at 10m = 23W/m2.
radiant flux at 13m = 14W/m2 .
The temperature on the inner and outer surfaces of the sphere are identical.
Why? Because the entire sphere reaches perfect thermal equilibrium due to 100% energy transfer.
d) Imagine a 30KW energy source surrounded by a radio-opaque perfect conductor.
radiant flux at 10m = 23W/m2.
radiant flux at 13m = 0W/m2 .
Temperature on the inner and outer surfaces of the sphere are identical due to conduction.
bananabender:
First you claimed – March 21, 2011 at 1:38 pm – that I had incorrectly calculated the outer surface temperature, T2, of the outer surface.
You said – without proof or even citing a formula – that the temperature, T2 was 10K not 133K.
I provided the calculation and so second – March 22, 2011 at 4:44 am – you switched to claiming arithmetic errors on my part.
However, you used pi = 3.14000 and claimed that I couldn’t do arithmetic accurately.
I got the correct answer because I used the correct value of pi.
You don’t dispute this and so third – March 23, 2011 at 1:46 am – you made the bizarre claim:
I explained, with reference to heat transfer textbooks, that the Stefan-Boltzmann law is not about black bodies. The Stefan-Boltzmann law is about emission of thermal radiation from all bodies. And the last 100+ years of experimental heat transfer confirms this.
So now, fourth, you switch again and start making all kinds of inaccurate claims. For example:
No. I assume that the outside of the sphere radiates energy towards the outside only.
Given that you are not able to admit your mistakes earlier – or alternatively, you are not able to defend your original calculations or claims – clearly you aren’t interested in finding out the difference between true and false.
Instead of ranting you should be interested to find that you are plain wrong about the Stefan-Boltzmann law.
You should be interested to find out that the Stefan-Boltzmann law is not just about the ideal case of blackbodies.
You should be interested to find out that heat transfer textbooks use it as the only equation for emission of thermal radiation.
But instead the truth makes you angry.
Another point: a Black Body has zero thickness!
You incorrectly assume that there are two separate Black Bodies on the inner and outer surfaces of the sphere. In fact there is a SINGLE Black Body spherical film of zero thickness located at the exact Centre of Mass of the sphere. In this case the radiative flux and temperature are identical on both the inner and outer surfaces of the film.
However your model as depicted is simply that of a heat source contained in an insulator – literally a light bulb in a box. This is a thermal conductivity problem rather than a radiative physics problem. The correct method for calculating that is to use the heat equation – not the Stefan-Boltzmann Equation.
BTW I don’t get my information from blogs.
However (unlike you) I know that:
a) you can’t use the Stefan-Boltzmann Equation for Black Body radiation to solve a conductivity heat transfer problem.
b) energy cannot flow from a cool body to warm body without a heat pump.
and
c) the atmosphere is heated by a molecular-kinetic process as correctly described by the Ideal Gas Law pV=nRT rather than a totally mythical “Greenhouse Effect”.
You should try reading the article instead of making stuff up.
The transfer of heat from the inner surface to the outer surface is by conduction.
It flows from hotter to colder and uses the equation:
qr = 4πk . (T1 – T2) / (1/r1 – 1/r2)
You can see this if you actually read the article.
The transfer of heat from the outer surface to space is by radiation.
What other mechanism do you think will transfer energy into a vacuum?
It uses the Stefan-Boltzmann formula which is the only equation which describes emission of thermal radiation and is not an equation only for blackbodies.
So if you want to discuss a subject instead of ranting – explain which formula you will use for emission of thermal radiation from the outer surface of the sphere.
Or admit that my formula is the correct one.
Most of what you have claimed so far shows you haven’t even read the article.
Most of what you have claimed so far shows you can’t admit to making mistakes. Is my arithmetic wrong for the surface area of the sphere – or, is yours wrong?
For a radius of 13m, is the surface area of a sphere = 2124 m2 or is it as you claim = 2122.64 m2. You were the one who wanted to make an issue out of it.
It is pretty simple stuff and you are wrong.
A temperature can be assigned to a point in a vacuum simply by adding up all the energy impinging on it from all directions . That will give the temperature of any gray , flat spectrum , ball at that point . SB applies to any gray object , not just black , because for any shade isotropic gray body absorptivity/emissivity is constant across the spectrum .
As I posted way back , the issue with the example is its relevance . The issue is the temperature of spheres radiantly heated from the outside , not the temperature distribution of internally heated spheres .
Dear dear ScienceOfDoom!
I applaud your astonishing patience in dealing with the world’s fools. You do seem to bring them out of the woodwork! Your heat transfer model is brilliant, a wonderfully counterintuitive way of leading fools into making absurd statements. It a very clever example, and I congratulate you for designing it. The mathematics are clear and unequivocal, yet so many have trouble understanding the implications for climate and weather science.
I wonder if part of the problem people have understanding the Trenberth and Kiehl (1997) diagram is that only the ground radiates like a black body. The back radiation from the atmosphere radiates only at frequencies/wavelengths that are part of the absorption spectrum of the radiating gases. This is a consequence of the Schwarzschild equation. Thus, the spectrum of the “downward” longwave radiation is quite different from the spectrum of the “upward” radiation; the spectrum of the “downward” radiation is the same as the absorption spectrum of the greenhouse gases, i.e., H2O and CO2.
I have, by the way, taught both thermodynamics and quantum mechanics.
Cheers!
Peter Offenhartz:
Thanks.
I’m certain that if I had presented the PVC hollow sphere with no reference to Kiehl & Trenberth, and not calculated the inner surface radiation I wouldn’t have had a single argument from anyone.
Of course a hollow sphere heated up from inside will have a higher internal temperature than external. Everyone knows that.
Of course the radiation away from the outer surface must match the energy being supplied inside. Almost everyone knows that.
No argument from anyone (probably).
Show the radiation from the inner surface and many people’s conceptual problems trump the obvious heat transfer concepts.
Still, I didn’t expect to get so many people leaping in with their patented heat transfer ideas.
The author has created a hypothetical perpetual motion machine consisting of a light bulb in an styrofoam box that is magically heated by back-radiation.
To achieve this amazing result he has incorrectly merged emissivity, radio-transparency and thermal conductivity into a single “radiation” entity to produce his apparent “back-radiation”.
Like Arrhenius he has created a hypothetical PVC aether with magical properties that defy the rules of electromagnetism to obtain his answer.
By definition a black body is a unidirectional (outwards) cavity radiator of zero thickness. If we assume that the inner surface of the sphere is a real black body then the radiation is only inwards towards the radiator. So the PVC is not heated at all and remains at absolute zero.
The radiative flux at the outer surface = 0W/m2.
Therefore no inward energy flow is possible from the PVC sphere even if the radiator is turned off.
From real world experience we know that cooling of an insulated box will progressively occur from the radiator to the outer surface. Therefore the temperature must always be: T.radiator >T.inner sphere >T.outer sphere.
bananabender:
You are imagining the perpetual motion machine – and this is a problem in your head.
Not in the model.
If the source of energy is turned off, the PVC sphere will cool back down to 0K (actually to 2.7K, the background temperature of the universe).
A body with an emissivity of 0.8 and radiating 14.2 W/m2 will necessarily have a temperature of 133K.
This is taught in all heat transfer textbooks (as already explained – and shown from scanned textbooks).
The fact that you don’t like it and so make random false or irrelevant claims:
a) the area of the sphere is wrong (false)
b) a blackbody is not a real object (irrelevant)
c) it’s a perpetual motion machine (false)
– just demonstrates to all the other readers that you don’t have any idea what is wrong with this model – you just don’t like it.
Which is not a scientific argument.
And many before you have made similarly random claims.
So please, explain what the surface temperature of the outer sphere is, for a body with an emissivity of 0.8 radiating 14.2 W/m2 – with reference to a formula from a heat transfer textbook.
The fact that you can’t do this simply demonstrates that you don’t like the model but don’t actually know what is wrong with it.
In fact, I think that you have realized the correct temperature is 133K but don’t want to admit it.
And so demonstrating that you don’t want to discuss science.
————
If I now take your most recent comments you claim that the outward flux from the sphere (into space) is zero?
Or you think I claim it? (Hard to tell as you have made so many different claims)
Previously you stated that the outward flux into space was 14.2 W/m2 (actually you claimed, incorrectly, that it was 14.1 W/m2).
Now it’s zero?
And yet to explain how you derived your outer surface temperature.
So why not explain, with reference to heat transfer textbooks, what the outer surface temperature, T2 is?
It should be simple for you.
bananabender:
In fact you have previously made 2 testable claims.
I am going to ignore all your new random rubbish until you clear up these 2 simple points.
1. What is the surface temperature of a body with an emissivity of 0.8 and a flux of 14.2 W/m2 – or 14.1 W/m2 ? With a reference for your temperature calculation.
2. What is the surface area of a sphere with a radius of 13m. Is it 2123.7166 m2 as I claimed.
Or is it 2122.64 m2 as you claimed (on March 21, 2011 at 1:38 pm) and made a point of.
Readers with a calculator can check no 2 very easily. Area = 4 x 3.141593 x r2.
Reader who want to check no 1 can do so very easily by referring to the formula provided:
E = εσT24
So, 14.1 = 0.8 x 5.67 x 10-8 x T24
Therefore T2 = (14.1 / (0.8 * 5.67 x 10-8)1/4
T2 = 133K.
So, bananabender, let’s see if you can decide to join a scientific debate and defend your original claims.
“Moving on” is not an option.
The answer is correct but the question is wrong.
A calculated temperature of 133k for the outer surface is incorrect because have used a method that is only applicable for a two-dimensional surface.
The 30KW of energy is emitted from the entire volume of the PVC not just the surface
You need to use equations applicable to a solid-state such as the Debye Model or the Einstein Solid Equations.
http://en.wikipedia.org/wiki/Einstein_solid
http://en.wikipedia.org/wiki/Debye_model
An internally heated sphere emits radiation at all levels not just at the surface. Because the sphere has a variable internal heat distribution it will emit radiation at a continuum of wavelengths according to a Gaussian Distribution. It doesn’t emit 30KW of monochromatic light corresponding to a single particular temperature at the surface.
An observer outside the sphere equipped with a wide-spectrum radiometer would measure the mean temperature of the sphere not the surface temperature.
A real world experiment using thermometers would probably show that calculated values are way out anyway. If anyone has a few million bucks to waste they can try this experiment at home.
Joules:
I don’t think you understand the relevance of an Einstein solid to this problem.
Solids do not radiate just from “the surface”, but the radiation from more than a few microns below the surface is absorbed by those last few microns.
The calculation to demonstrate this is simply the Beer-Lambert law of absorption.
You also say:
You imply that I am claiming that the surface emits monochromatic radiation.
I have not claimed that at all.
The surface will emit radiation according to Planck’s law, modified by the emissivity of 0.8 which is assumed here (for the purposes of the thought experiment) as the material property of the PVC. This is what it will look like:
Well, if you want to prove that the entire sphere – “all levels” – radiates to space please produce your calculation of the transmittance of the sphere to radiation through 1mm of PVC and through 1um of PVC.
When you have completed this calculation you will understand why all heat transfer textbooks show the emission of thermal radiation from solid surfaces as a dependency on the surface temperature and the emissivity of the surface – and not with a dependency on the temperature profile below the surface.
E = εσT4 (the real formula)
See if you can find a textbook with a different formula, like this one perhaps:
E = εσ ∫f(T(x)).dx (what the formula would look like if Joules was correct)
Here is an extract from Fundamentals of Heat and Mass Transfer, by Incropera & DeWitt (2007, 6th ed):
I have completely recalculated this problem from first principles.
Note that their are very small discrepancies due to rounding errors.
The temperature of the inner sphere is only ~152K
The radiative flux is only ~24Wm2 on the inner surface.
There is no radiative forcing.
All calculations have been double-checked by back-calculation to obtain the original 30KW of energy input.
The energy at both the inner and outer surfaces is 30KW. Energy has been fully conserved.
Part 1. Calculating Inner sphere temperature:
radius=10m
area = 4*pi*r2
=4*3.14*10*10
=1256m2
radiative flux = 30,000/1256
= 23.9W/2
temperature of the inner sphere surface:
P=30,000W
emissivity=0.8
area = 1256m2
Stefan-Boltzmann Constant = 5.67×10^-8
Therefore:
30000 = 0.8 *1256*5.67*10^-8*T^4
30,000/0.8 +1256*5.67*10^-8*T^4
37500/7.12*10^5 =T^4
T^4=5.27*^10^5
T=151.7K
The temperature of the inner sphere is ~151.7K
I back checked this by feeding the numbers into the following online calculator. A temperature of 151.7K means an energy input/output of 30KW.
http://www.endmemo.com/physics/radenergy.php
Part 2. Outer sphere radiant flux:
energy at inner surface = 23.9W/m2
conductivity = 0.2W.m^-1*k
thickness = 3m
energy at outer surface = 0.2*23.9*3
= 14.31W/m2
Surface area of outer sphere = ~2120m2
14.31W/m2*2120m2 = 30375W (due to rounding errors)
A triple check:
Radiant flux of inner surface = 23.9W/m2
Inner surface area = 1256m2
Total radiant flux = 23.9*/*1256
= 29390W (rounding error)
radiant flux at outer surface = 0.2*23.9*3
= 14.31W/m2
Surface area of outer sphere = 2120m2
14.31W/m2*2120m2
= 30375W (due to rounding errors)
29390 =30375 = energy balance (with 3% rounding errors)
Joules:
There is a small problem with your calculation.
First, let me confirm that you believe the outer surface temperature, T2 = 133K ?
This is calculated the same way as your method for the inner surface temperature.
I should add that this is the correct way, as I have explained to various people, most recently, bananabender.
Here is the problem:
How is 30,000 W conducted through the PVC sphere?
Heat is not conducted by magic. Heat is conducted due to a temperature difference.
From the article (and from heat transfer textbooks):
qr = 4πk . (T1 – T2) / (1/r1 – 1/r2)
So this means, with k = 0.19, r1 = 10, r2 = 13,
qr = 1,935 W
So – either the equation of heat conduction is wrong OR some mysterious new heat transfer mechanism exists OR – the real answer..
The real answer, the inevitable answer, the temperature of the inner surface keeps rising until 30,000 W can flow by conduction to the outer surface.
Otherwise the First Law of Thermodynamics will be violated.
Once you have grasped this essential point you see that everyone claiming the inner surface is NOT 290K above the outer surface is confused about the first law of thermodynamics.
Or confused about the equation of heat conduction. From Fundamentals of Heat and Mass Transfer, by Incropera & DeWitt (2007, 6th ed):
How about a real world experiment.
Place a heater in the middle of a small room.
Wait an hour.
Place your hand on the wall.
The wall is cool – not hot.
How about answering the question.
Is the equation of heat conduction wrong?
Or does the temperature difference between the inner and outer surface have to be 290K to conduct 30,000W through the PVC?
I suggest you:
a)discuss this problem with a mechanical or chemical engineer who has practical experience.
or
b) makes some observations rather than just quoting textbooks.
In the real world (as opposed to theoretical physics textbooks):
– there are no black bodies – only very crude approximations such as light bulb filaments. However a light bulb surface is not a black body.
– black body calculations are totally worthless for large three-dimensional bodies. The Moon is 60K hotter at night than predicted by theory due to stored heat in the upper surface rocks.
– thermally conductive solid bodies (except rods and thin plates) have very uniform internal heat distribution (<<1K/m temperature difference) except at interfaces. A car engine block is uniformly at ~90C after a few minutes running except for ~1mm of metal in direct contact with the combustion gases.
– thermal equilibrium is sometimes approached but is never achieved.
– in practice heat transfer across large objects is infinitely slow. eg Applying a blowtorch to a brick barely heats the other side more than a few K even after a week.
<b<What really happens in your example (as opposed to the theory) ?
1) The heater heats the inner surface.
2) Heat immediately diffuses to the outer surface which begins to radiate.
3) The outer surface reaches a temperature of ~2K at which point radiant heat loss is faster than the heat transfer rate. This is because the entire 7,000 tonnes of PVC acts as a heat sink.
4) The entire sphere after a few weeks develops a uniform temperature of 2-3K.
When you put a light bulb inside a styrofoam box the inside surface temperature stabilises within a fraction of a second. The surfaces always remain much cooler than the surface of the light bulb. This can be confirmed by touching the inside of the box.
As expected, your random claims continue.
What does a blackbody have to do with this? The emissivity of this surface is 0.8. That means, by definition, it’s not a blackbody.
What is the point of your engine example? Metals have a very high thermal heat conductivity, so therefore, a very low temperature difference can cause conduction of a large amount of heat. The exact same principle means that as thermal conductivity reduces, the temperature difference required to conduct the same amount of heat increases.
Reaching thermal equilibrium in this example – PVC – takes a very long time. You can see in Part Two that it takes many years.
People who do practical heat transfer use these equations. Or use 3d thermal modeling programs that use these equations.
That’s why heat transfer textbooks contain these equations. Because they are correct.
The fact that you don’t like textbook equations being used is very revealing.
So, once again, what is the temperature differential between the inner and outer surface that will cause 30,000W to be conducted?
The answer is right in front of you. You just don’t like it.
Are you claiming the textbooks are wrong? No. Because that would be embarrassing. Are you claiming the answer can be calculated differently? No. Because that would be even more embarrassing.
Don’t answer the question. Don’t do the calculation. Divert attention.
Thereby proving the point.
There is nothing wrong with the equations.
The problem is that you are incorrectly applying these equations to “prove” a nonsensical idea.
You are applying equations intended for a hypothetical two-dimensional static model to a real world three-dimensional dynamic model.
To use an old computing phrase “garbage in – garbage out”.
What does a blackbody have to do with this? The emissivity of this surface is 0.8. That means, by definition, it’s not a blackbody.
It’s a grey body. Stop engaging in semantics.
Your calculations are for a sphere that only emits radiation at the inner and outer surfaces and has a substantial temperature gradient exists across the sphere.
This is totally wrong as:
a) A large solid body simultaneously emits radiation from all regions of the body not just the surfaces. This also creates a relatively uniform internal heat distribution.
b) No real system can ever be in thermal equilibrium.
What is the point of your engine example? Metals have a very high thermal heat conductivity, so therefore, a very low temperature difference can cause conduction of a large amount of heat.
Not in the real world. Heat exchangers use the thinnest walls (often <1mm) possible even though they are made of highly conductive metals. This is because the heat transfer rate is negligible otherwise.
Concrete has very poor conductivity. However the heat distribution within the walls of a large building is uniform except at the interfaces with the atmosphere. The only real case where heat distribution is highly non-uniform is when a huge temperature gradient exists across an effective insulator eg applying a blowtorch to the back of an asbestos tile.
Reaching thermal equilibrium in this example – PVC – takes a very long time. You can see in Part Two that it takes many years.
It doesn’t take “many years” to reach thermal equilibrium it takes an infinite amount of time (except in a theoretically perfect conductor).
In the real world each extra K of heating takes T^4 times as long the previous K of heating due to radiative heat losses. In the case of sphere the radiation occurs simultaneously from all regions of the sphere above absolute zero.
eg Heat transfer rate = 1K/sec .
Temperature = 99K
It takes (100)^4 = 1*10^8 seconds or 1,157 days to add only 1K
Therefore to reach a temperature of n degrees K it takes:
= 1+ (2)^4 + (3)^4….(n)^4 seconds.
In practice the time is infinite because each of the the last micro-degrees takes billions of years
Unless the the energy input per unit mass is extremely high there is no possibility of equilibrium ever being approached.
Only very small masses with extremely high energy inputs such as light bulb filaments (~10KW/g) or burning gases can ever even approach the theoretical temperature.
In your example:
Energy input = 30KW = 30,000W = 30,000J/s.
energy input at inner sphere surface = 23.9W/m2 = 23.9J/s
calculated volume of the sphere = 5003m3
mass of the sphere:
PVC density = 1,400m3
= 7,004,200kg
The specific heat capacity of PVC
=0.9Kj(Kg.K)
How long to heat the sphere (assuming no radiative losses)?
dQ = m c dt
where
dQ = heat supplied (kJ)
m = mass (kg)
c = Specific Heat Capacity (kJ/kg.K)
dt = temperature change (oK)
Expressing Specific Heat Capacity using (1)
c = dQ / m dt
therefore:
0.9 = 30KJ/7,004,200. dt
= 47,368s/K
= 2.1*10^-5K/s
To reach 133K =73 days
Actual rate of heating:
The sphere radiates energy proportional to the 4th power of the temperature above absolute zero. So the rate of heating is inversely proportional to the 4th power of temperature.
If the initial rate of thermal transfer is 2.1e-5K/s
dT=1/t^4
where: time = t
temperature =T
t=0, dT =0
t=1, dT=1/(2.1e-5) = 2.1.e-5K/s
t=2, dT=1/(2.1e-5)^4 = 1.99e-19K/s
It takes more than the entire age of the universe just to reach 2k.
If you don’t believe me put your hand on the outside of plastic electric kettle and turn it on. The outside temperature will barely increase despite the walls only being 3-4mm thick and a 1KW/kg energy input for several minutes.
Yet your model has an energy input of only:
(assuming PVC density = 1,400kg/m3)
30,000W/7,004,200kg
= 4.28*10^-3W/kg
Your model has 30x the energy input of the kettle. 7 million times the mass and walls 1000x as thick.
It will never warm up.
People who do practical heat transfer use these equations. Or use 3d thermal modeling programs that use these equations.
These equations are useful approximations for designing objects like heat exchangers or tube boilers which are essentially two-dimensional surfaces. They are only accurate to about 5% even for these situations.
Engineers also rely very heavily on practical experience and measured results rather than just using calculations.
NASA actually measured the surface temperature of the Moon in the mid 1960s rather than relying on theoretical calculations. This is because the engineers refused to risk the lives of astronauts based on dubious theoretical physics. In this case the measured temperature discrepancy was up to 60K.
That’s why heat transfer textbooks contain these equations. Because they are correct.
The equations are grossly simplistic descriptions of reality and are only correct for certain hypothetical situations.
They cannot be applied universally.
You have mis-applied the equations to obtain a ludicrous conclusion.
So let’s take it one step at a time. You claim that I have misapplied all these equations.
That is your claim. So far without any proof.
1. What is the radiation from the sphere to space?
You claim that radiation takes place from all depths within the sphere without any proof.
I asked you to calculate the transmissivity of PVC for 1mm and 1um.
Please provide these numbers to demonstrate you have any idea what you are talking about. (If you calculate them you will find exactly why the textbook cited earlier explains that radiation from a surface takes place from the top micron).
OR – Alternatively, provide an equation for the emission of thermal radiation from the outer surface of this sphere.
While waiting for you to produce your evidence for the emission of thermal radiation from within a solid PVC sphere, I thought I should comment on this statement of yours:
From April 4, 2011 at 4:46 am:
and then following my comment:
What does a blackbody have to do with this? The emissivity of this surface is 0.8. That means, by definition, it’s not a blackbody.
You said on April 4, 2011 at 10:46 am:
First of all, these equations don’t just appear in “theoretical physics textbooks”.
They appear in all practical heat transfer textbooks. These are the textbooks used to teach engineers how to calculate heat transfer.
And the reason is because these are the measured, proven results over the last 130+ years.
Every solid opaque body radiates according to the Stefan-Boltzmann equation, which is not an equation for “theoretical bodies”. It is an equation for practical bodies.
I don’t know where you have learnt these ideas, but you haven’t learnt your ideas from practical engineering textbooks. Or from experimental results.
I also would be fascinated to find the source of your claim that the moon doesn’t radiate according to the Stefan-Boltzmann law. It is another wild blog claim, last seen in a non-published paper with a picture of a purple balloon as its best feature.
Well, people can write anything on the web.
So please if you are interested in science, produce some evidence.
If the Stefan-Boltzmann equation is wrong and inapplicable to the emission of thermal radiation from a body, demonstrate it with:
– textbooks on heat transfer (you will be out of luck there), OR
– properly recorded experimental evidence to demonstrate that all the heat transfer textbooks are wrong, OR
– theoretical considerations demonstrating how this equation must be flawed
The heat exchange equations totally ignore quantum surface effects to simplify calculations. That is why “impossible” values occur at interfaces. The calculations simply assume T1-T2 and are not concerned with how the energy is transferred across a junction.
The only reason your model gets the wacky feedback results is because of interface effects. However if you think you can get 60x as much energy out of a system as you put in you should try and patent it.
A probabilistic distribution of heat occurs at any point within a system according to quantum mechanical-statistical principles. That is why heated bodies radiate over a range of temperatures rather than emitting monochromatic light.
Newtonian mechanics are completely invalid at the atomic level. So are heat exchange equations.
I worked as a scientist in the food and beverage processing industry for many years so i have plenty of practical experience with heat exchangers, cookers, refrigeration and many other aspects of heat exchange.
You assume that engineers spend huge amounts of time using the infallible thermal transfer equations to design an optimum system.
Nothing of the sort happens. Engineers typically work with completely standardised components such as pumps, heat exchangers and pipes. They don’t design custom hardware to solve every problem. They tend to solve problems based on years of practical experience and some estimates. They then join the components together and test them. If the desired result doesn’t occur they add or subtract a few heat exchanger plates or swap the pump for a bigger model.
Car makers test drive prototype vehicles in hot conditions. If the engine overheats they simply make the grille slightly bigger or install a different radiator.
Electronics makers just use a bigger heat sink and fan if the device runs a bit warm.
Joules:
Your problem is conceptual.
You claimed the equations I cited were right but misapplied. No proof.
Now that they are invalid. No proof.
The equations are close? The equations are totally wrong? No one has any idea how to calculate heat conduction through a hollow PVC sphere? It’s unknown?
Practical problems of heat transfer have strange shapes and materials of variable and sometimes poorly known properties. Everything here is spelt out for you.
You don’t have a scientific argument, you are trying to find a way to change the result because you believe it to be wrong.
Take a look back. 200 comments, mostly of people with all different ideas about why this is wrong but not a single piece of proof. That should be a clue.
Part Three of this series might help you with your conceptual problem.
Joules:
This is your conceptual problem. Do I claim this? I explain this confusion on your part in Part Three.
Basically if you try to do anything with this energy “bouncing around” then you will cool down the inside of the PVC sphere very quickly.
You can only get out the energy you have put in.
In any given second 1m2 receives lots of energy radiated from the rest of the inner surface and radiates it back out. Why is this creating energy?
This is equilibrium (note 1). The inner surface has stopped heating up and 23.8 W/m2 is conducted through to the outer surface. The rest is just absorbed and re-radiated.
The energy for this re-radiation has come from the energy stored during the heating phase – which happened because until the inner surface temperature gets high enough, 30,000W cannot all be conducted through the sphere.
Think about it.
If you lag a hot water pipe it increases the inner temperatures.
Note 1 – And yes, I know that equilibrium is never really reached, you can see it in the graphs in Part Two. However, it is a useful concept and we can speak about getting to 99% of the equilibrium temperature in a certain time, or 99.9%. I can write caveats around everything if necessary – but it stops onlookers learning, and prevents commenters who can’t find proof for their important claims being able to sound like they have said something important and relevant to the discussion at hand.
Hello Bananabender
Brilliant commentary – explanation on the thermos thing.
Also Joules.
Good stuff.
It seems I like this blogg for all the wrong reasons.
There are scientists commenting.
Science of Doom
I have briefly flicked through comments here and it seems you always want to apply a “partial” or “incomplete” heat balance so that you can highlight effects that are important to you.
Every real world system must account for all heat or energy involved not just “important” bits.
Perhaps you have taken as true the many “authoritative” outlines of the CO2 AGW theory. Mainstream science does not acknowledge this theory which is confined to”Climate Science” magic.
Part reading of comments by Bananabender and Joules show they are well educated in this area of science which can be quite difficult to follow without years of study.
DanR:
Perhaps you have a scientific point to make rather than your opinion?
bananabender has provided a claim about the emission of thermal radiation from the outer surface of this sphere and provided no justification for it.
Perhaps you can help that commenter out.
Or are you applauding because bananabender just made it up?
Joules claims – without justification – that the PVC sphere emits from radiation from within the sphere.
Perhaps can you help out Joules with proof of that claim.
Or are you applauding because Joules just made it up?
You say:
So I look forward to your careful explanation of where my analysis fails to account for any heat. After all, I have applied the first law of thermodynamics for the equilibrium condition.
And doesn’t it seem strange to you that all the different commenters claiming the PVC sphere analysis is wrong have incompatible reasons?
And no one likes textbooks anymore?
What is this – some secret society of “heat transfer gurus” who don’t accept textbooks, don’t agree with each other, and can’t even produce equations for their claims when asked?
Doesn’t seem slightly odd to you?
The “PVC sphere analysis” is just diverting attention away from the problem.. which is “Does Man Made CO2 induce Global Warming?”.
You don’t need equations to show the basic CO2 hot belt in the sky thing is wrong.
If the CO2 hot belt was present at the altitude proposed there is one big physical drawback.
Any earth bound radiation would not get very far through the atmosphere and certainly would not reach the surface.
At the most, if there was an effect, the heated air receiving this “dangerous” energy from the CO2 would rise, pushing the CO2 out of the way and creating a partial vacuum lower down with attendant winds which would create evaporative cooling of the surface.
Other stumbling blocks exist but there is no real point talking about them here.
As others have said the basic model is wrong and text books wont help you if you have not learnt to use them properly.
Climate Science is VooDoo.
DanR:
So your claim is that the PVC sphere heat transfer calculations are wrong?
Or your claim is that the PVC sphere heat transfer calculations are right?
Be specific. I don’t know why you are bringing up CO2 – it is irrelevant to this discussion.
Easy to claim isn’t it? But if you understood the subject yourself you would be able to explain how to use the textbooks.
I realize that you can’t.
Otherwise you – like the many before you – would have explained which equation is wrong.
By now someone would have explained the correct method of calculating heat transfer through this sphere. And with reference to an equation from a textbook.
And, DanR, I don’t expect to convince you. But, along with the many before you who claim this model is wrong, you serve a valuable purpose.
I can of course “you would be able to explain how to use the textbooks.”
You have to go to University and study thermodynamics for about four years before its starts to become clear.
If you haven’t done this it shows up to those who have.
You can’t hide that.
Well then, instead of pretending you understand the subject why not demonstrate it?
1. What is the correct equation for emission of thermal radiation for a surface of emissivity=0.8?
2. If a surface of area = 2,124m2 radiates 30,000W and has emissivity = 0.8, what will be its surface temperature
3. What is the equation for heat conduction through a hollow sphere?
4. And what is the temperature difference required to conduct 30,000W through a hollow sphere with inner diameter 10m and outer diameter 13m?
Yet you don’t know the answers to questions 1-4 do you?
Come back when you have found out.
You still don’t get it do you Science of Doom.
Before you can use any equations you MUST put together a sketch of all processes and heat flows in the system under study.
Then you try to quantify as many knowns as possible.
It is a long involved process to make a road map of the chain of events in a problem.
If you can’t do this then equations are useless as the IPCC Trenberth, Mann etc have demonstrated so well.
As explained in the article:
There is no other mechanism for transferring heat through the PVC than conduction.
And there is no other mechanism for transferring heat from the PVC sphere to space than radiation.
It’s not complicated and figuring it out isn’t a “long involved process”.
Unless you have discovered some new physics that is unknown to all of the people who have written textbooks on heat transfer.
You keep claiming, claiming, claiming.
Your claims and your opinions are not interesting – although they are entertaining.
Come back when you have evidence.
OK.
I see the problem now that you have given this detail:
“Now that we have turned on the energy source the inside of the sphere will heat up. It has to keep heating up until the energy flow out of the sphere is balanced by the energy being added inside the sphere.”
To make this system replicate that of Earth, once the temperature of the system has been brought to equilibrium we must turn off the heat source that is equivalent to the internal heat of earth. Just leave an amount of heat equiv to solar input.
Does that seem logical?
It’s in the article. So you mean you didn’t read it?
Reading people’s articles, books and papers is a good way to understand what they actually think and why they think it. I recommend this approach.
So please, take some time to read the article.
Then – using your amazing knowledge of heat transfer – or talking with the many experts you know – explain what is wrong with it. With reference to science – via textbooks, papers or your own experiments. Not with reference to your opinion.
Or confirm that the calculations are correct.
Not liking what it might or might not confirm about the earth’s climate system is not an appropriate method of evaluating the correct result of this heat transfer thought experiment.
Obviously if you can deduct that I hadn’t read the bloggs apart from the first few lines then perhaps you have the makings of a scientist.
Now try to work out how the Climate Community has been able to hold this joke together for so long.
It will fold.
Many scientists are now getting very unhappy about the lise supporting AGW and the IPCC.
I am coming late to the party, and someone might already have said this, but it seems that you have described an energy storage system, much like a capacitor where some of the energy leaks out of the outside surface …
There will be a period of time where the inside surface heats up to its equilibrium temperature, because more energy is being input than lost, but after that, with the same energy being lost through the outside surface as is being gained from inside, that 423C inner surface temperature remains the same.
Of course, every square meter of the inner surface is radiating 1,452 W/m^2 (or whatever the number is), but that is fine because it is receiving the same amount from the rest of the inner surface so that has no net effect on the temperature of the inner surface.
If the material of the insulator were better, and the material could withstand very high temperatures, it would seem that there is nothing preventing the temperature of the inner surface reaching 5000C, 10,000C, …
Thank you for this interesting example.
If I have got anything wrong, I would appreciate corrections.
Your review is correct. And you are right on the mark with the point that increasing the insulation will increase the equilibrium temperature.
I might have made the same point earlier, but if I had done all of the calculations with no comment on the radiation from the inner surfaces I am certain that there would have been none of these howls of protest.
That is, heating a body from inside will obviously lead to a rise in temperature until such time as the heat conducted through the body is equal to the heat source inside.
I’ve contacted Trenberth directly about this graph to get some clarification.
[Moderator’s Note – rest of comment snipped as already promised – continual restatements of your confusion add nothing to this blog and are very annoying for everyone else.
Further restatements will just get the whole comment deleted without comment.]
[…] the Palaver? – Kiehl and Trenberth 1997 Do Trenberth and Kiehl understand the First Law of Thermodynamics? Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Six – […]
If a stream is flowing up to a mound dam of loose dirt, it will start to pool up at the base dam. It will build and build up into a reservoir until the pressure of the reservoir water overpowers the dam (let’s say, in one spot, where it breaks down and flows through). Let’s say the reservoir does not continue growing at that point, meaning the same amount of water flowing into the reservoir is now the same as flowing out through the breach in the dam. There is then a dynamic equilibrium where water flows into the reservoir and out of the reservoir at the same rate, thus keeping the reservoir the same size (volume).
Though there is a lot more water in the reservoir than in one-second’s flow either into or out of it, it is evident that extra water has not been created here, and that no conservation laws have been broken. You could not draw more water of the reservoir than is being put into it, because any siphoning off will affect the flow out of it.
I tried discussing the idea of applying the second law of thermodynamics to greenhouse theory on the Hockey Schtick site, but was cut off after not just accepting additional restatements of the same point as basic physics (including the idea that the second law applies to parts of systems as well as the whole). it is very easy to come out ahead in a discussion where one gets to moderate the responses as well as make them.
Boy, I made a hash of that first post, and was not a great typist in the second. “Let’s say the reservoir does not continue growing at that point, meaning the same amount of water flowing into the reservoir is now the same as flowing out through the breach in the dam” should end as “the same amount of water flowing into the reservoir is flowing out through the breach.”
Oh, dear. 6/10 I am afraid.
It was all going so well until you calculated a radiant heat output from the inner surface.
“What is the Radiation Emitted from the Inner Surface?”
Zero. Nothing. Nil.
The inner surface isn’t radiating anything. It is absorbing. Approx 23.87W/m2.
A.
Andrew,
The inner surface emits radiation and absorbs radiation. In steady state these values are equal.
If you claim a surface (above 0K) with non zero emissivity does not emit radiation then you either:
– have no idea what is taught in all heat radiation textbooks, or
– have a new discovery for the world
Heat radiation textbooks give a formula for emission of thermal radiation from a surface. This value = emissivity x absolute temperature to the 4th power x Stefan Boltzmann constant
It is always the same formula.
Either produce your textbooks or explain how your new discovery overturns the last 150 years of physics.
My textbook says …
P=eσA(T1^4 – T2^4)
There is no radiant heat output from the wall. It is absorbing heat.
Andrew,
Heat and energy are not the same thing. Using the definition from classical thermodynamics of heat, which is that heat is the amount of energy that must be transferred from a hotter object to a colder object to make the temperatures of the two objects equal, all heat is energy, but not all energy is heat. Heat flow would then be the rate of heat transfer.
That definition doesn’t require that the colder object must not radiate. All real objects have a finite emissivity so all real objects above absolute zero produce EM radiation at all times. Your textbook equation is derived by subtracting the Stefan-Boltzmann equation, which is the integral of the Planck equation over all wavelengths assuming constant emissivity, of one object from the S-B equation from the S-B equation for the other. Your equation is valid for infinite parallel planes where view factor is not an issue and both planes have the same emissivity. The total power per unit area radiated by each plane at T1 and T2 is then:
P1 = εσT1^4
P2 = εσT2^4
The net energy transport or heat flow is then:
P1-P2 = εσ(T1^4-T2^4)
Hi DP,
Well we seem to mainly be in agreement (apart from I need to be a bit more accurate in usage of terms heat/energy). I’ve stuck in an A so we’re measure Watts, I guess without the A we’re measuring W/m2.
The difference of opinion seems to be around whether you can sensibly split the forumula into its components and get something that has any real world meaning.
I am not sure you can.
To take a slightly facetious example, you could just as easily write:
P1 – P2 = εσT1^4 + 1000000000 -εσT2^4 – 1000000000
That is still true, but you cannot say that one surface is radiating εσT1^4 + 1000000000 and also absorbing εσT2^4 + 1000000000 … it just isn’t something that is meaningful in the real world.
A.
Andrew,
For people like yourself unfamiliar with heat transfer basics known for 100 years I provided extracts from 6 heat transfer undergraduate textbooks in Amazing Things we Find in Textbooks – The Real Second Law of Thermodynamics.
Here is one example:
Read the words.
Here is another example:
Read the words.
Hi DP,
Mostly true. However stating that bodies radiate and absorb separately is a bit of a guess/unproven assumption. Isn’t it?
And I am not sure it is supported by observation. All we can ever observe is a net transfer.
It is entirely valid to say that in the example above, when we are looking at energy transfer, the wall not radiating any energy; it is absorbing.
That is real and observable.
I do not think you would be able to construct a test in the real world that would disprove this assertion.
Can you? Has it been done?
Radiation has a strange habit of behaving in odd ways; sometimes as a particle and sometimes as a wave. It also has a tendency to interfere with itself as demostrated in various experiments.
In the scenario above, who knows. Perhaps there develops a null standing wave where light ‘waves’ cancel each other out?
As far as I am aware, you cannot see/observe separate transmission and absorption energy flows. Assuming or guessing that they exist is not the same as them actually existing.
So the separate elements of the formula above could be considered more like ‘forces’ than an energy transfer. One is pushing one way, and one the other but the only energy that is is transfered is in the imbalance.
I probably have missed something, but that is my understanding of the current state of our knowledge.
A.
Andrew,
So you like textbooks until they contradict your assertions.
October 22, 2014 at 8:45 am:
November 27, 2014:
And now, after being presented with undergraduate heat transfer textbooks that state the sum of knowledge telling you that all surfaces radiate you say:
Of course you could overturn standard physics, but in the interests of focusing on climate science.. from the Etiquette of this blog:
“Basic Science is Accepted – This blog accepts the standard field of physics as proven.. This is aimed to reduce the continual stream of unscientific rubbish that gets placed here as comments.“
Andrew
No it’s not unproven – it’s unproven only in the sense that formally science never proves anything correct, it only proves something wrong and something else very accurately in agreement with observable reality.
Quantum Electrodynamics (QED) of Feynman and other scientists is one of the most accurately verified theories of physics. according to QED radiation and absorption are essentially independent processes. They are not totally uncoupled, but the coupling enters only as a very small correction to the approximation based on considering the processes as independent.
This is a proven fact as firmly as any of the most certain results of physics.
Hi PP,
Anyway, I am an engineer and not a physicist.
Energy transfer is a macro-thermodynamic and observable phenomenon.
SoD: I do not think I am misrepresenting known science in any way.
Indeed Feynman’s theories are well evidenced. And back-up what I am trying to say.
They are even more based on the fact that the real-world is based on the net effect. While individual elements of the computations are (generally) not coupled with each other, and can be calculated independently, they do not exist independently in the real world.
Is is absoutely clear from the QED approach that the probablilities of individual events combine together (various sums and products) to create an overall probablistic reality which we then observe.
The indivual elements, even those that are uncoupled (mathematically independent) have no independent existence in reality.
Possibilities combine to create an overall probability and that is the reality we see.
A.
SoD: I quite like your thought example. I think it gets right to the heart of the issue.
However, to answer your questions:
– I have a reasonably good idea what is taught in many (can’t claim all) radiation text books.
– I don’t have a new discovery for the world.
Just trying to stick to what we do already know and have discovered.
A.
Andrew,
I provided extracts from 6 textbooks in the linked article, and to save having to click through I provided 2 extracts in my comments.
What do they say?
They say that each surface emits radiation.
So you don’t have any idea what is taught in radiation textbooks.
Please scan a heat transfer textbook which backs up your claim. You can post the scan here, just use html img tags for an image you have uploaded to a url.
Or email the scan to me at scienceofdoom – you know what goes here – gmail.com.
Anyone want to place a bet on whether Andrew Gordon cites a person who shall remain nameless, but whose initials are C and J to justify his assertion?
Bet you £100 I don’t.
Shall I send you my bank details do you can tranfer the cash?
Andrew,
I’m assuming here that you are, in fact, a real person and not a sock puppet for someone that has been previously banned here.
The hypothesis that objects with a temperature above absolute zero only emit EM radiation when there is a target that is at a lower temperature and at a rate proportional to the difference of the fourth power of the temperatures of the emitter and receiver requires previously unknown physics. If the hypothesis were true, then there must be some physical mechanism to transmit information to every excited state on the surface of the object to allow or forbid emission. Standard physics requires no such mechanism.
In order to show that there must be some such new mechanism, you would have to demonstrate that the new hypothesis explains measurements better than the standard. Note that’s better than, not the same. Good luck with that.
Hi DWP,
Hmm. I can see that point of view. When you introduce a distance that takes TIME to cross, you can see that somehow radiating elements would need ‘pre-cognition’ about the eventual target to decide how much energy to send over!
Which is clearly ridiculous. At least I assume so. It is reasonable to assume that things would have to happen independently first and then interact later. At the very least in the intial start-up states before we reach a (dynamic) equilibrium state.
EMR (light) is a funny thing though. Indeed everyhting gets stranger the harder you look at it. Sometimes the things that seem reasonable are proven false when looked at in more detail.
QED tells us that “possible” events (energy transfers and so on) are probablities that combine to create an eventual reality. So a surface has a probability/potential to radiate but until we combine that with the all the other probabilities in the system we do not have a valid description. No?
At first when not interacting with any other system, it very likely does actually radiate as per one-side of the equation. When is starts to interact with something else … who knows?
I haven’t yet ready about how time comes in to this. Maybe it takes time for probablities to propagate/interact? Yeah, probably does. I suspect some speed of light limitation here.
Nevertheless, we can only ever observe/measure a net energy transfer (I think). I do not have any new hypothesis that explains anything differently to current … all current maths/physics etc. is accepted.
I am only pointing out that while the distinct/individual maths terms may be correct … they do not necessarlily have meaning in the real world and assuming they do is taking an unnecessary step.
In the real world only the net energy transfer is measurable/relevant/real. Therefore in the example given I still maintain that you can say that there is no energy radiated from the inner surfaces.
We can measure a net heat transfer – we can only imply the individual terms.
Maybe it is an irrelevant distiction, but I think it is important.
A.
And, yes, I am a real person. Not been banned (yet), but I am just starting … 🙂
Hi DwP & SoD,
Is it possible to PROVE that real radiation is occuring as separate flows, and not just as a potential irradiance which we can only see in the net effect?
Well (and here is where I might have to take back what I said above) I think there might be.
Radiation absorption and emisison creates a radiation pressure, because photons have momentum. Not much, but some. And the emitted photons and absorbed photons would both be generating outward pressure on the inner surface.
In the example you give, if there is this a large multiplication of radiation being absorbed and emitted as suggested then the radiation pressure inside the sphere would be much bigger than just that that which would be generated by the heater alone. Yes?
And this should at least in theory be measurable? Then I would concede that the separate terms are not merely mathematical.
A.
Andrew,
The radiation pressure of solar radiation has been observed and its influence on spacecrafts calculated using standard theory of radiation. Solar sails are based mainly on radiation pressure. They emphasize reflection, but absorption and emission must be taken into account to determine correctly the force.
http://en.wikipedia.org/wiki/Radiation_pressure
http://en.wikipedia.org/wiki/IKAROS
Hi PP,
Yes, radiation pressure is real and we know its effect on spacecraft and the concept of a solar sail.
The question above is about exactly what that radiation pressure would be in the ‘thought experiment’ above.
Would the radiation pressure on the inner surface be just from the 30Kw net radiation (input power) absorbed.
Or would it be much higher? Like more than 100 times higher! From the 1.8Gw emitted plus the 1.8Gw absorbed + 30Kw absorbed.
A.
The formula for calculating the radiation pressure can be found from Wikipedia. For the emission it is
Pe = (εσ/c)T4
If the intensity of the radiation that meets the surface were equal to the blackbody radiation at the temperature of the surface, absorption would add an equal contribution and reflection so much that the total would be equal to that obtained bu replacing the emissivity ε by the value 2. Thus measurement of the radiation pressure allows for the determination of a combination of emissivity and the level of radiation that meets the surface.
σ is the Stefan-Boltzmann constant and c speed of light. When the value 2 is used and the temperature is 288 K, the radiation pressure is 0.84 µPa.
Hi PP and SoD.
Sorry been away for a bit …
Anyway, yes you can calculate radiation pressure as per below. The formula you quote is probably correct (I haven’t looked at it too closely as it is not fundamental to the argument).
However, my question is can you measure it?
If there is genuinely 1.8Gw of radiation whizzing backward and forward in the above case, then you should be able to measure an increased radiation pressure. Not theory, conjecture or formulae. But an actual measurement.
If it is there, then you can show that the separate elements of radiation occur as more than just mathematical constructs of the calculation of net heat transfer.
It is possible, however, that a sort of EM standing wave is created where there is no real photon transfer (they’d cancel each other out) and the only real photon transfer occurs NET. This would be aligned to the SQED approach I outlined above, rather than a discrete particle approach.
In that case, there wouldn’t be any real individual radiation component, only the net effect and the radiation pressure would be commensurate with the 30kW net power transfer.
And actually I don’t know which outcome would occur, until tested. Do you?
I couldn’t even guess.
How astounded were the people who did the single-photon inteferometry experiments that a photon could interfere with itself!!! You don’t know until you try.
Anyone know if this type of experiment has actually been done???
This is, as far as I know, the only way to resolve this issue. All the other pyrgeometer style of pseudo-scientific nonsense doesn’t actually measure anything it is representing to do until this issue is resolved!
If someone can show me that there is a radiation pressure effect in a similar example to the above of the incoming plus outgoing radiation and the two do not cancel each other out, then I would happily concede that my earlier statement that:
“However stating that bodies radiate and absorb separately is a bit of a guess/unproven assumption. Isn’t it?
And I am not sure it is supported by observation. All we can ever observe is a net transfer.”
.. is entirely and completely wrong!
If you can show BY EXPERIMENT IN THE REAL WORLD that we can observe more than just a net transfer, then everyone must rationally concede that the two occur independently.
Otherwise and until such an experiment is conducted, it remains pure and unproven conjecture … and remains a mathematical and non-physical artifact. You cannot state with certainty that it exists in the real world.
Regards,
Andrew
And this isn’t a:
Standard physics works. Until you can show an example where it doesn’t, there is no reason to complicate calculations.
That’s not the way science works. It’s up to you to show by experiment that the separate elements of radiation don’t exist and that non-existence results in measurable consequences. I don’t really care what you believe if you can’t back it up with data rather than hand waving.
interferometry (sigh – no edit function!)
@DWP
I really, really hope you re-read what you have written ….
Science is not faith. You have to prove the separate flows exist in the real world. There is currently no proof that they do, as far as I am aware.
You can prove that the net effect occurs. That is real. We are okay with that. That is science. That the individual components have any physical reality is unproven conjecture. For now.
Until you actually prove that these things exist separately (and I’ve even outlined a possible way of providing an experimental proof) then you are the one who is not following scientific method.
A.
No, you don’t. You obviously aren’t a working scientist if you think that.
You do not prove a hypothesis is correct, you can only disprove it. Proof that a theorem is correct is for math. And even then you run into Gödel’s incompleteness theorem. Separate flows is the simpler hypothesis. It does not require the extra physics that a unidirectional flow requires. It works. Until you can show an example where it doesn’t or come up with a simpler hypothesis that works as well or better, then, by Occam’s Razor, separate flows is the preferred hypothesis.
Andrew,
By the way, I’m curious why you think you’re well enough educated to instruct us on how science and particularly the physics of emission and absorption of radiation actually works.
I have a B.S in Chemistry (1965) from the California Institute of Technology and a Ph.D. in Analytical Chemistry from the University of Texas at Austin. Further name dropping, Richard Feynman was the lecturer for my freshman and sophomore Physics classes at Caltech. I presume you’ve heard of the Feynman Lectures on Physics. That’s where they came from.
Of course that doesn’t guarantee that I’m right and you’re wrong, but it represents a large hurdle for you.
Andrew,
As you have presented it, the whole question of existence of two separate flows is not well defined. We are discussing phenomena that cannot be described properly without Quantum Mechanism and that means that the concepts have to be defined carefully, before questions like that can be asked.
We know what the overall physical situation is. That has been verified empirically well enough. We know also that the simplest way of explaining that physical situation is in terms of separate and largely independent emission and absorption. This is a fact in the present way of looking at the physics and that’s good enough to every working physicist as long as some very small deviations from the simplest model are not studied.
There’s nothing to prove in the above. In the present way of looking at the physics the answer is a fact that need not be proved any further.
There may be other ways of formulating the physical theories. in some other formulation the same words may refer to different concepts. With the different concepts another statement may be true, but at the present there’s no need to worry about that. Any new formulation of the theory must be equivalent with the present formulation or alternatively it must explain all physical phenomena at least as well as the present formulation does. Good luck for any attempt to create such an alternative formulation.
There’s only one physical reality, but it may be possible to describe that reality in many quite different ways. Some questions of philosophy of science (or metaphysics) may get involved.
@SoD … you have constructed an excellent site, and your patience for trying to educate the willfully uneducable borders on Sisyphean.
I have one small nit to pick however .. I think you often use the term “thermal equilibrium” when you actually mean “thermal steady state” … according to the 0th law of thermodynamics, thermal equilibrium only applies to the case where heat transfer between two (or more) bodies in contact has ceased because they have reached the same temperature (that actually provides the definition of temperature in thermodynamics). In your example (as well as the K & T) diagram, what is actually being calculated is the steady state condition, where the fluxes into and out of the system have achieved balance. It’s not the same thing, because one or more of the phenomena (e.g. conduction or convection) are driven by thermal gradients, which wouldn’t exist in a true condition of thermal equilibrium. I think this is more than a minor semantic point, because the whole point of AGW is that the Earth is experiencing a net heat gain due to a relatively small imbalance of the incoming and outgoing fluxes, arising from the increased greenhouse effect due to man-made emission of GHG’s.
DarkLordKelvin,
DeWitt Payne has also picked me up on this point and I generally agree. Although I often use less technical language than I would prefer as a means to communicate.
It’s a fine balance.
In this case I think my own internal terminology was incorrect anyway.
@Andrew Gordon .. I am baffled by your inability to understand (or accept) that any object with non-zero emissivity and a temperature above 0 K MUST emit radiation. I am sure you agree that if you put such an object in deep space, with a background temperature that is as close to 0K as you can get, then it would emit radiation. What do you think changes when you put that body in a different environment? It will still emit radiation at a rate consistent with its surface temperature, in accordance with the S-B equation.
Have you ever used an IR thermometer? They are great .. you can just point them at any reasonably non-reflective, non-transparent surface, and they’ll read the temperature. They do this without having any thermal contact with the object, and the measured temperature doesn’t change as you move the thermometer closer to or further from the object (within reason). How do you think those work, if not by measuring the thermal emission from the surface of the object being measured? Note that if you point them at an object that is *colder* than the ambient temperature (i.e. is experiencing a net absorption of radiant heat from the room), they still read the temperature accurately. By the logic you seem to espouse in your posts, such “colder than ambient” objects should not register at all on an IR thermometer, because they are “net absorbers” or radiant energy.
DLK,
The simple, inexpensive IR thermometer works the same way as a pyrgeometer, but is less precise and has a much smaller field of view. It measures the heat flow either into or out of the surface of a block covered by a lens to restrict the field of view in the case of an IR thermometer or by a hemisphere designed to pass IR radiation in a wavelength range of ~5-50μm in the case of a pyrgeometer, by measuring the temperature difference with a thermopile across a thin film of known insulating properties attached to the exposed surface of the block.
Heat flows in if the area being measured has a higher effective temperature than the block and out if it is lower. The heat flow is converted to effective temperature using the S-B equation, the emissivity of the exposed surface of the insulating film and the measured temperature of the block.
If you look around the internet, though, you can find lots of hand waving claiming that pyrgeometers, and thus by extension, IR thermometers don’t actually measure radiative flux or temperature or something. It’s hard to say because what passes for logic in these screeds is impenetrable. But the conclusion is always that radiation only flows one way, from hot to cold.
I completely agree with your nitpick,by the way. It is not thermal equilibrium, which has a very specific meaning in thermodynamics. OTOH, in chemistry one refers to chemical equilibrium, which is when the rate of the forward and backward reactions are equal. A is being converted to B at the same rate as B is being converted to A so the bulk concentrations of A and B are constant. So in that sense, an energy balance at the TOA could still be considered an equilibrium state.
DeWitt … I am a chemist, so I appreciate your comparison. However, I think I disagree that chemical equilibrium is a good analogy for “energy balance at TOA”. A system in chemical equilibrium is one were (as you correctly stated), the concentrations of reactant and product have stopped changing *because* the forward and reverse reaction rates are equal. Analogously, a system in thermal equilibrium is one where the temperature has stopped changing *because* heat exchange between the various parts of the system has stopped (i.e the temperature distribution is isotropic). The TOA situation is better described as steady state, because heat is still being exchanged with sources and sinks. Similarly, a chemical system that happened to have (temporarily) constant concentrations of reactant and product due to the respective rates of addition of reactant and removal of product being in balance, would also be described as “steady state”.
Also, it’s very possible that the person you addressed can’t answer because he is one of the few people who have earned a ban here for violating blog etiquette, specifically that Basic Science is Accepted. At least someone whose last name starts with G has been banned “who thought radiation in opposite directions cancelled out “because they are vectors”.
The Earth as whole is in thermal equilibrium when the net radiative imbalance at TOA is zero.
The Earth is never in thermodynamic equilibrium as long as it’s heated by some source to a higher temperature than that of cosmic background radiation.
@Pekka .. I suppose that is not an unreasonable operational definition of thermal equilibrium, but it is also not the “correct” one used in thermodynamics, which as I mentioned is co-defined with temperature in the 0th law. The Earth is in thermal *balance*, or at a thermal *steady state*, but since it is still exchanging heat with sources and sinks, it is not at “thermal equilibrium”. Furthermore, “the Earth” is not even in thermal equilibrium with itself .. in order for that to be true, it would have to have an isotropic temperature distribution, which it clearly does not.
Just in case it wasn’t clear from what I wrote, I should have said, the Earth *would be* in thermal balance (etc.) … if the net radiative imbalance at TOA were zero.
DLK,
We can discuss equilibrium of a specified whole with outside systems without going into the internal structure of the whole. A body as whole can be in thermal equilibrium with it’s surrounding while not internally in thermodynamic equilibrium.
When it’s clear from the connection what kind of equilibrium is meant, and when other expressions are for some reason not natural, it’s acceptable to use thermal equilibrium to mean that the single balance that’s being discussed is in equilibrium.
Whenever confusion can be created, it’s certainly best to avoid using the word equilibrium loosely.
[…] Do Trenberth and Kiehl understand the First Law of Thermodynamics? & Part Two & Part Three – The Creation of Energy? […]
“Do Trenberth and Kiehl understand the First Law of Thermodynamics?”
They do, but they apparently don’t understand that there is more than one way that a joule can pass from the atmosphere to the surface. The diagram would have us believe that the only way a joule can pass from the atmosphere to the surface is via EM radiation. Surely to space that is the only way, but not to the surface.
Of all the potential flaws in this diagram, it’s quite surprising no one notices this one when it’s a glaring flaw. Unless you believe that the only way a joule can pass out of the atmosphere is by EM radiation, there is no way the numbers presented in the diagram can be correct.
What rubbish. The energy calculated from the inner surface does not exist. If it did then a thermos flask would result in the contents getting hotter.
AT,
If you put a heat source inside a thermos flask it heats up. If you increase the insulation it heats up more.
Why do people lag pipes? It increases the temperature of the water inside the pipes by reducing the heat loss to the outside.
Plumbers and sellers of lagging for pipes are just con artists.
Of course, why should you apply any thought or analysis to the problem..
“Internal Radiation
Therefore, the radiated energy from the inner surface will be 1,452 W/m2 or a total of 1,824,900W (= εσT14.4πr12).”
You have used the T1 temperature of 423 K back calculated from the conduction formula Q = U * A * dT in the S-B equation.
This calculation is illegal.
The heat transfer mechanism at surface 1 is 99.9% conduction. If you use emissivity of 0.0145 for surface 1 it all works fine. By applying the S-B equation incorrectly you have effectively and inappropriately doubled the heat in the system.
The warm earth surface moves heat into the atmosphere primarily by conductive/convective/latent processes. Inserting 15 C or 288 K into S-B to get 390 W/m^2 upwelling is absolutely
WRONG!!!!!!!!
http://writerbeat.com/?search=schroeder&category=all&followers=all
Nicholas,
Perhaps you can quote from a heat transfer textbook or some kind of new amazing paper that has revolutionized heat transfer.
The equation for emission of thermal radiation is well known – and as given in the article – it depends on temperature and on emissivity. Emissivity is a material property.
The equation for conduction of heat is well known – and as given in the article.
You have defined two new laws, unknown in heat transfer textbooks:
1. “heat transfer at surface 1 is 99.9% conduction” – invented
2. [not sure how to sum it up in words, but something like] “emissivity changes to suit the requirements and is not a material property” – invented
I await your textbook extracts with interest.
With 30,000 W radiating from surface 2 out into space per S-B & ε = 0.8 GB equation result is T2 of 132.8 K.
Check.
Using Q = U * A * dT and the 3m thick PVC conductivity of 0.19 W/m-K T1 = 413 K. (U = k/x & averaged areas 1 & 2. SoD got 423 K, used 43% of ave? Temp diff = 290 or 280 C.)
Check
Insert T1 of 423 K in S-B 0.8 GB equation to get new current radiative flux from inner surface 1.
“Internal Radiation
Therefore, the radiated energy from the inner surface will be 1,452 W/m2 or a total of 1,824,900W (= εσT14.4πr12).” (413 K = 1,319)
Hold on. You can’t do that! Over 60 times the 30 kW input? Yeah, that’s energy out of nowhere!! All 100% of the 30,000 W have been transferred from surface 1 to surface 2 by conduction, there are ZERO W left for radiation from inner surface 1.
BTW PVC is opaque so that’s twice as much zero radiation.
Radiation from surface 1 is simply NOT possible no matter the surface temperature or S-B theory!!
Now 100% radiation heat transfer between surfaces 1 & 2 requires a vacuum between surface 1 and surface 2, i.e. no molecules, no conduction, convection, latent, etc. for a S-B 0.8 GB T1 of 151.5 K, a difference of 18.6 C. Energy moves, i.e heat flows, from high to low with little dT when there’s no insulative crap in the way.
Any media placed between the two surfaces that impedes conduction will increase the surface T1 temperature. This is what warms the earth, not down/“back” radiation. Placing any media between the surfaces, e.g. air, CO2, water, clouds, lucite, glass, wood, concrete, etc. will all have their unique combination of conduction, convection and radiation and inner surface 1 T1 consequences.
K-T diagram Trenberth et al 2011jcli24 Figure 10
Method A balance, mixed:
Incoming:
240 ASR at 100 km = 80 atmos + 160 surface
Outgoing:
17 convec + 80 latent + 63 LWIR = 160 surface + 80 atmos = 240 OLR at 100 km
Incoming = outgoing. That’s it, all balanced, nothing left for more radiation, certainly not up/down/back of 333.
Method B balance, 100% radiation:
15 C, 288 K, S-B BB = 390 W/m^2 with emissivity of 0.615 upwards = 240 W/m^2 OLR
You can use Method A OR Method B, you may NOT use BOTH!!!!!
And neither method A or B makes the surface warm/er.
You’re forgetting that radiation emitted from the inner wall is also absorbed by the inner wall. The area of the source is negligible. Even if it weren’t it wouldn’t matter. The emissivity of the inner wall also doesn’t matter as long it’s opaque. Any radiation not absorbed by the wall will be reflected until it is absorbed. That’s why a sealed box with a tiny hole is a very good approximation of a blackbody with the temperature of the walls even if the walls of the box have an emissivity much less than 1. Energy is not being created, so the First Law hasn’t been violated.
Nicholas
The result was calculated from applying conservation of energy.
Lots of confused people before you have used the same argument from incredulity along with trying to redefine basic physics (you can see their comments above). All in inconsistent and/or incoherent ways.
In Do Trenberth and Kiehl understand the First Law of Thermodynamics? Part Three – The Creation of Energy? I summarize the main themes of incredulous commenters.
You will find your theme there as well.
If you believe that emissivity is not a constant material property, or that the equation for emission of thermal radiation is not the Stefan-Boltzmann equation then you should get to work with your paper.
It will be a ground-breaker. Overturning 100 years of heat transfer will create a stir in the physics world.
Usually when I try to pin down confused commenters on whether or not they accept what is in all physics textbooks and please provide the correct equation I can never quite get an answer. For obvious reasons.
In another article I provided 6 extracts from textbooks on 2-way radiation and one of our most admired and entertaining commenters of the time (his time has now passed) said “you have cherry picked” (I had collected every book on the university library shelves on heat transfer and scanned the relevant pages). In an irrelevant but highly entertaining footnote to the episode, he then provided his own favored textbook which also gave the same equation.
Nicolaus wrote: “Hold on. You can’t do that! Over 60 times the 30 kW input? Yeah, that’s energy out of nowhere!!”
Your statement is incorrect, but the way this problem is worded makes it hard to see where the energy came from. This post specified the existence of an equilibrium and correctly deduced some logical consequences based on that equilibrium: a 423 K inner surface that is radiating AND 1,824,900 W and absorbing 1,824,900+30,000 W.
To understand where this “extra energy” comes from, let’s imagine starting this experiment with everything at 0 K. As 30,000 Joules per second flows into the inner sphere from the heat source, the temperature gradient across the 3 m of PVC (insulation) isn’t steep enough for any of that energy to be conducted to the outer surface of the sphere. When more energy flow into than out of an object, the law of conservation of energy demands that the difference becomes “internal energy” or a higher temperature. So – for a long time – a lot of energy will be stored as heat in the PVC, particularly near the inner surface. Eventually the PVC will reach 423K, which produces a temperature gradient steep enough for 30,000 W to be conducted to an outer surface at 123K. The energy that has been stored as the temperature has warmed is what allows the inner surface to emit 1,824,900 W (AND absorb 1,824,900+30,000 W AND conduct 30,000 W). Since the sphere is so well insulated, some of the heat that has been stored up becomes thermal vibrations and some is turned into a lot of photons that are continuously emitted and absorbed.
When thing are well insulated, they be very hot and still lose heat slowly.
The Earth is a classic example of an object that has a very weak power source and lots of insulation. The Earth is heated by the slow decay of some long-lived radioactive isotopes, including U-238. That provides enough power for 0.1 W/m2 of heat to escape through the crust to the surface. If we moved the Earth to interstellar space, its surface temperature might drop to about 36 K, the temperature of a black body that emits 0.1 W/m2. Fortunately, we are near the sun. Nevertheless, the center of the Earth is approximately 5700 K (roughly the temperature of the surface of the sun), because heat escapes only slowly through the crust (and mantle).
https://en.wikipedia.org/wiki/Earth#Heat
Above you might say: “Hold on. A 5700 K blackbody emits 60,000,000 W/m2 and radioactive decay provides only 0.1 W/m2. That is energy out of nowhere!” Nevertheless, the Earth is mostly molten rock above 1000 K with an INSULATING thin crust floating on top. Occasionally, that molten rock reaches the surface as lava, so there is no doubt the Earth is very hot inside.
SOD didn’t ask us to calculate anything about how fast the sphere would warm up – that requires taking into account heat capacity and heat flow for millions of time steps and locations in the sphere using the principles described above. Equilibrium is much easier to calculate, but it is easy to get wrong answers if you rely on intuition instead of unless you focus of the laws of physics, especially conservation of energy.
GWID,
Clouds reflect incident sunlight, about 20% of the total incident solar energy is reflected back into space by clouds. An additional 10% is reflected back to space by the surface.
There is essentially no vertical conduction by convection above the tropopause. Even in the troposphere, most convective energy transfer is in the lower part. Energy is lost to space only by radiation and the radiation emitted to space is a lot less then the total of radiative and convective emission from the surface. That sounds exactly like reduced conduction to me.
GWID wrote: “I can´t see either reflection or reduced conduction by the atmosphere.”
It is quite easy to literally see reflection by the atmosphere. Go outside in the daytime on a clear day. Look up at the sky. It is blue.
Since you state as a dictate “Don´t claim things you haven´t read up on” I assume that you know why the sky is blue and that “reflection” includes both specular reflection and scattering.
Above, Nicolaus discusses Methods A and B for describing heat flux on our planet.
Method B is obviously wrong. There is nothing on our planet at 288 K with an emissivity of 0.61. The surface and atmosphere emit a total of 240 W/m2 to space, but their temperature ranges from 200 to 310 K. The S-B eqn can’t define the emissivity of a heterogeneous, partially transparent system with a range of temperature.
In Method A, you specify that latent heat (of evaporation) carries 80 W/m2 from the surface to the atmosphere. I presume that you know that evaporation ceases when relative humidity is 100%. With 100% humidity, the most energetic water molecules don’t stop escaping through the surface of water to become water vapor. That process goes on at a rate that depends on temperature, not the relative humidity above the water. However, when relative humidity is 100%, just as many water molecules are entering the surface of the water as leaving it (equilibrium). Relative humidity over oceans is 80%. So the real situation is that about 400 W/m2 of latent heat leaves the surface and about 320 W/m2 of latent heat enters the surface. When water vapor condenses high above the surface, 80 W/m2 of latent heat becomes higher temperature.
Why the fuss about a two-way flux of latent heat? Because there is a two-way flux of radiation that some people ALSO ignore: an average of 390 W/m2 of upward radiation leaves the surface for the atmosphere and 333 W/m2 leaves the atmosphere for the surface. The net result of this process is 67 W/m2 upward. That is what the correct laws for radiation transfer (the Schwarzschild eqn) predict and what we measure. Planck’s Law (and the derived S-B eqn) apply only to radiation that is in equilibrium with the medium it is traveling through. (Planck didn’t take into account real molecules that absorb and emit differently at different wavelengths.) The Earth doesn’t emit like a black or gray body – the intensity vs wavelength plot is jagged, not smooth. Unfortunately, the Schwarzschild eqn is difficult to derive and use (requiring numerical integration). So college physics typically presents only situations that can addressed using the S-B eqn or Planck’s Law. Our atmosphere is not one of those situations, but no one told me that when I was in school.
The website below allows you to use MODTRAN (a program that solves the Schwarzschild eqn) to calculate radiation transfer on our planet. It is not a climate model; just the program that makes calculates radiation fluxes through our atmosphere similar to the way AOGCMs do. Note the absence of a blackbody shape to the intensity vs wavelength plots.
http://climatemodels.uchicago.edu/modtran/
Working on a similar thought experiment and wanted input on my solution.
A sphere radius r1 is internally heated such that its surface will radiate 100W/m2.
Outside the shell is 0K vacuum.
The sphere is surrounded by a theoretical black body shell with no thickness. It radius is r2. Solving for everything I’m getting that the emission out of the system:
100*(r1/r2)^2
Which means the surface must receive:
200*(r1/r2)^2
Tracing that amount back to the sphere means the sphere must be emitting:
200W/m^2
It seems intuitive that the sphere temperature would warm because of the shell, I don’t doubt that. The part that I was a bit shocked by was that the steady state temperatures will always be double, regardless of the radius of each. In my mind, if the ratio of radius were close to 1 it would have led to a higher temp than if the ratio was closer to 0. Limits of the function would have been at 1 a doubling of sphere output and at 0 the same sphere output as if there wasn’t a shell.
But alas, that’s not what I got when I actually walked through the exercise. So is my math wrong, or my intuition? I’m guessing the latter.
Brad,
Emitted power is doubled, not temperature, but only for values of r2/r1 close to 1. The temperature of the shell will decrease as its area increases. As r2→∞, Tshell→0.
DeWitt,
I see now I misspoke in my explanation. Meant to say emission, not temperature. Let me rephrase.
My intuition says the sphere would warm and thus emit more. However, by my calculation the introduction of the shell means that the steady state emission of the sphere is 470, regardless of ratio of the radii. I didn’t expect the emission of the sphere to be 470, regardless of the individual radii. My intuition was that the upper limit (ratio approaching 1) would have been for an emission of 470, while limit as ratio approached 0 would have been for a surface emission of 235. Again, my math says differently than my intuition though which is why I’m asking.
Thanks.
Brad,
DeWitt is right. The temperature of the outside sphere must approach zero as r2 becomes infinite… the inside sphere does not “know” anything about the outside sphere except it’s temperature, and it’s temperature falls to zero at infinite radius. The total emission out of the system of the two spheres is 100 watts/M^2 multiplied by the area of the inner sphere, no matter the size of the outer sphere.
So, when r1/r2 approaches 1 the emission from the inner sphere approaches twice (200 watts/m^2), but half of that is replaced by back radiation from the inside surface of the outer sphere, for a net radiative loss of 100 Watts/M^2 from the inner sphere. When r1/r2 approaches zero, the temperature of the inner sphere approaches the temperature of the same inner sphere when surrounded by empty space at zero degrees. You can use the Stefan Boltzman equation to calculate the actual temperatures of the spheres for any combination of radii.
Steve and DeWitt,
I’m getting numbers mixed up between posting here and working on a write up. My apologies, I’m making this more confusing. My focus isn’t on the temperatures but rather the emission of the various objects.
So the inner sphere is powered such that it emits 100W/m2. It has a radius r1. Power into the system is then 4pi*r1^2*100
Power out of the system must equal this in steady state. Obviously this happens on the outside surface of the shell, radius r2, so:
e(out) *4pi*r2^2= 4pi*r1^2*100
e(out) = (r1/r2)^2*100
This means total emission of the shell is double the above:
e(shell) = (r1/r2)^2*200
So the inner sphere must emit enough radiation to balance (r1/r2)^2*200 at radius r2 which looks like:
e(inner sphere) = (r1/r2)^2*200/(r1/r2)^2 = 200
The introduction of the shell means the sphere will be emitting 200W/m2, regardless of the associated radii. Does this look and sound correct?
Brad,
DeWitt,
From your example:
“That means that the shell is emitting 25W/m² and has a temperature of 144.9K. So we plug that into the heat transfer equation
100 = 0.0000000567(T1^4 -144.9^4)
T1 = (100/0.0000000567 + 144.9^4)^0.25
T1 = 216.68K for an emission of 125W/m² not 200 W/m²
I agree that emission on the outside of the sphere must equal 25w/m^2 for the given radii ratio of .5. But that would mean the total emission of the shell is 50, correct. Solving that I got a shell temp of 172 and surface temp of 227.
Cancel that last question. I see now that is really the initial point of my misunderstandings.
Brad,
Not all the radiation from the outer sphere will strike the inner sphere. The emission from the inner surface will have a cosine distribution, but you are implicitly assuming that it will all be emitted radially toward the center. For a large outer sphere, only a small fraction of the inner surface emission will be absorbed by the inner sphere, most will miss it and be adsorbed by a different part of the outer sphere. Since the total emitted from the inner surface is fixed, the fraction that will return to the inner sphere will decrease as the outer sphere gets larger. As others have said, when the outer sphere is infinite, it will have no effect on the inner sphere.
Mike,
I don’t see in my equations above where I made any implications about the radiation received by the sphere from the inside of the shell.
I set the emission from outside of the shell equal to peer into the system.
I then determine the total emission of the shell, inside and out, which is double the amount from above.
Since the inner sphere is the only source of power for the shell, I calculated how much radiation it would have to emit to match the radiation the outer shell must radiate for steady state. When I did this I found the inner sphere emission to be 200, regardless of radius.
I agree that the shell temp will drop as it gets larger. My intuition told me the emission (and subsequently temperature) of the inner sphere would vary as well with radius. By my math though, I’ve found it to be constant. A doubling of emission regardless of radius of the shell.
I’m by no means trying to say I’m right. I’ve shown my equations so if I’m incorrect please slow me where in my equations I went wrong. My intuition had been wrong in the past and I’ve made plenty of mathematical errors. I’ve rechecked the math and tried to rethink it from different angles and come to the same conclusion each time.
Brad,
It’s always good to learn some basics.
Here is an extract from A Heat Transfer Textbook from MIT that you can freely download. Worth reading through the relevant few chapters on radiative heat transfer, then most questions disappear:
Click to enlarge
Click to enlarge
So you can see that the view factor is the key. As the radius of the outer sphere increases it is mostly just radiating to itself. Try the maths out for r2 = 1.01r1, r2 = 2r1, r2 = 100r1 and you will see how it works out.
Thank you all for the responses. SoD, will look into the info from MIT.
Looks like my intuition was correct but my math and reasoning failed me this time.
Brad,
Consider the logical constraints. The total power emitted from the outer sphere can’t possibly be more than the net power emitted by the inner sphere (100 watts/M^2). so the problem is really very simple: calculate the area of the outer sphere relative to the inner sphere, and use the Stefan Boltzman equation to determine the outer sphere temp. Then calculate the temperatue of the inner sphere with stefan boltzman assuming “space” is at the temp of the outer sphere instead of zero. You will always get the right answer.
Steve,
I think it is slowly coming together. Reading on view factor will probably help reveal the flaw in my logic. I’m still a bit stuck on the fact that for DeWitt example of ratio of radii to be .5
This means the shell will emit 50w/m2 (25 out each side). In my mind it only has a single source of power, the inner sphere. So the inner sphere has to supply power for the shells emission.
I understand the concept that the majority of the emission on the inside of the shell doesn’t reach the inner sphere. But energy emitted from one spot on the inside of the shell to a different spot of the shell doesn’t increase the power into the shell, it is a 0, no gain nor loss.
So that’s where my equation came from. The outside of the sphere being the only source of power for the shell, since transmission of radiation from shell to shell is amounts to no change.
Brad,
I haven’t been through your example in detail, but maybe a minor point – defining your boundary conditions clearly is also a critical item.
You said earlier (maybe you are working on another assumption now):
This is different from the boundary condition that the inner sphere has a constant power source of x. That might be what you meant to write.
You have effectively defined a constant temperature which means that the internal source may be changing its output as a result of the outer shell. And it also means that regardless of external circumstances the sphere will be at a constant temperature – which makes it very easy to work out the temperature of the sphere.
If you meant to define a constant power source then you would say that the power source = 100A – where A = surface area (correction a few seconds later, no ε needed).
SoD,
I took Brad to mean an internal constant power source equal to 100 watts/M^2 on the inner sphere’s surface. (eg. plutonium (very) slowly degrading and generating heat)
Seems to me you are making this far more complicated than it really is.
Brad,
That’s the point. If there is no gain and no loss, then there is no need for excess power to supply that fraction of the emission. It’s supplied by the rest of the sphere The view factor for the shell to the sphere is 0.25. That’s the only power that needs to be supplied by the sphere. So emission from the sphere is 100 + 25 W/m².
Consider if the sphere were actually hollow and the heat source was evenly distributed in the surface of the spherical shell. No energy will be lost by emission from the interior surface of the shell because energy out equals energy in. So the power required for an emission to empty space at a temperature of 0K of 100 W/m² isn’t 200 W/m², it’s 100 W/m².
Steve F.,
It’s not the setup that’s complicated, it’s the mistake that Brad is making by ignoring the view factor. That makes the emission from the surface of the sphere, and thus it’s temperature, constant regardless of the radius of the shell, which is wrong.
I was just going for clarification. It’s easy to get confused with heat transfer. Easy to write down the wrong equation because you misunderstood a boundary condition. I’m not trying to complicate it at all.
DeWitt,
The pieces finally just fell into place in my head. I’m understanding now where I was incorrect in my reasoning.
Brad: I think you may be confusing yourself by not clearly distinguishing between power and power flux (W/m2). We can apply conservation of energy to the former, but not the latter. For clarity, I’ll use r for r1 and R for r2, and T and t for T1 and T2. The outer sphere has the lower temperature.
and power flux (W/m2). We can apply conservation of energy to the former, but not the latter. For clarity, I’ll use r for r1 and R for r2, and T and t for T1 and T2. The outer sphere has the lower temperature.
Brad: “A sphere radius r1 is internally heated such that its surface will radiate 100W/m2.”
Instead try: A sphere radius r with a power source that emits P Watts and therefore a flux of P/(4*Pi*r^2) W/m2 from its surface. P is conserved, power flux is not.
Brad: “The sphere is surrounded by a theoretical black body shell with no thickness. Its radius is R.”
If the system has reached a steady-state (and no components are changing temperature and thereby consuming power), then the total power leaving the outer shell is still P and the power flux is P/(4*Pi*R^2).
The ratio of the fluxes leaving the outer and inner spheres is (r/R)^2. This is the inverse square law.
If the outer sphere is a perfect blackbody then it must emit (both inward and outward) and therefore have a temperature t given by:
P/(4*Pi*R^2) = ot^4
——————-
The calculation gets much more complicated when you ask about the temperature of the inner sphere. Some of the radiation emitted inward by the outer sphere is absorbed by the inner sphere and added to its internal power P. The rest of the inward radiation is absorbed by the inner side of the outer sphere. I won’t do the trigonometry of these “viewing angles” (including applying Lambert’s cosine law when a flux isn’t perpendicular to the absorbing surface). Let’s just say some fraction (f) is absorbed by the inner sphere. So the FLUX being absorbed by the inner sphere is:
f*P/(4*Pi*R^2)
and the POWER being absorbed (and added to the internal power P) is flux times area:
f*P/(4*Pi*R^2) * 4*Pi*r^2 = f*P(r^2/R^2)
making the total power available to the inner sphere:
P * (1 + f*(r^2/R^2))
From this we can calculate the temperature the inner sphere must have (if it is a blackbody or graybody). In other words, the presence of an outer shell raises the temperature of the inner sphere – driving those who naively apply the 2LoT ABSOLUTELY NUTS. However, this is just insulation – reducing heat loss – but the 2LoT fanatics believe the concept of insulation only applies to heat loss by convection, not radiation. Nevertheless, the net flux of energy – heat – still flows from warmer to colder.
————————————
According to this website, the view factor f = r^2/R^2, making the total power available to the inner sphere:
P * (1 + (r/R)^4)
Click to access Radiation%20View%20factors.pdf
————————————
You might wonder about the inner wall of the outer sphere. It is emitting P/(4*Pi*R^2) and absorbing only (1-f)*P/(4*Pi*R^2) from itself. It also absorbs P from the central power source, but that is passing through the outer sphere and then radiating away to space. Why isn’t its temperature falling? That is because the inner sphere is hotter than it would be without the outer sphere and the extra radiation it emits keeps the outer sphere at a constant temperature. (:))
Thanks Frank.
I can see that sticking to power as the basis rather than flux, could save me from making errors. In this case however, that wasn’t my issue.
The first step or two of my equations were correct. That is that the outer surface of the shell must emit P (or as I was writing it in terms of flux, 100W/m^2 * (r/R)^2, which is properly accounting for inverse square law). The other part I had write was the amount of flux, or power the entire shell was emitting, inside and out.
Where I went wrong, and the pieces just finally came together, is that i considered the only source of power for the shell to be the sphere. 3 or 4 people said the same thing but in my mind it simply wasn’t making sense why emission from shell wall to shell wall could conceivably count as power into the shell. To me this sounded as if the shell were creating power. I could see and understand DeWitt’s equation but simply wasn’t grasping why the shell to shell emission want some violation of conservation of energy.
Then I realized, it isn’t about the shell adding power to the shell, it is about emission that is occurring that isn’t removing energy from the system. It would be as if i you and I were in a boat that is taking on water. We are each using a bucket to scoop water and dump it. However, you are dumping yours overboard and I’m just dumping mine into the boat. I haven’t added water to the boat that wasn’t already there, but I haven’t removed water from the system either. You are using your effort to emit water from the boat while I’m making the same effort but I’m not emitting the water from the boat.
Brad wrote: “my mind it simply wasn’t making sense why emission from shell wall to shell wall could conceivably count as power into the shell.”
My comment was held up in moderation and so missed the back and forth.
If you go back to the problem posed by this post, the 3 m of rubber provides a lot of insulation. The outside temperature is 133 K and the inside 423 K. That is roughly a 100-fold difference in T^4. So the internal transfer of energy from one location on the inner surface of the sphere to another is very large – but no heat transfer is involved since the net flux is zero (by symmetry).
SOD cleverly didn’t give his internal heat source a definite size and therefore didn’t need to get bogged down in details of viewing angles, the “shadow” cast by the central power source, nor energy returning to the central power source (:)) It is an elegant teaching example as written, but one that is much more challenging (and therefore less clear) if the central power source has a defined size. The smaller the power source gets, the hotter it must be to radiate 30,000 W.
Many commenters above were outraged about how much power was traveling inside the sphere. Where did it come from? If you imagine starting the system up from 0 K, negligible energy is escaping through the rubber sphere for a long time. Insulation keeps that energy inside – it builds up because it can’t escape until there is a large temperature gradient. Power * time is energy – the energy that is going nowhere when being radiated from spot on the inner surface to another.
I had this realization today:
“The Stefan–Boltzmann law describes the power radiated from a black body in terms of its temperature.” …emphasis added
I’ve read this plenty of times and have failed to fully grasp the …from a body….. I’ve been thinking of it as emission away from a surface.
In the green plate effect on Eli’s site the plates had to have a temperature equivalent to double the emission from a single side. That’s obviously a different case since the emission away from a plane can’t be absorbed by the same object. I realize everyone that has responded understands this very clearly, just sharing in case it is a stumbling block for someone else that passes through the blog.
What? No! They have a temperature equivalent to the emission of one side. Not double.
Brad,
It’s the surface area that doubles when you consider both sides, not the radiant flux density in W/m². If only one side is illuminated, then the input flux density has to be twice the emission flux density at steady state.
Ah, yes. The temperature would just be equivalent to the emission from one side.
As you said DeWitt, the incoming flux would be double the emitted flux in the green plate experiment. So my incorrect thinking was that the flux arriving to the inside of the shell needed to support emission from both sides. My reasoning was that I was interpreting sb to be emission from a surface, regardless the direction the surface is facing. Sb though is for emission from a body. Incoming flux to the shell wouldn’t need to be double, as it was on the green plate experiment, since only the emission on the outside shell is “from the body”.
I think I need to stick to reading through that MIT textbook for now.
Brad: One surface vs two can be tricky. Imagine cutting a 2x2x2 cube into eight 1x1x1 cubes. The surface area doubles and so does the total emission. Where does the extra power come from? The temperature is still the same.
The extra power comes from twice as much internal energy being converted to radiation – its temperature falls twice as fast …. but only in intrastellar space. Everywhere else, there is a local temperature, local thermal radiation field and twice as much surface area to absorb it. So the temperature may not fall. Twice as much power could be exchanged by radiation, but not necessarily any “heat”.
Now consider reassembling the eight cubes into one big one. Then consider the difference between a solid sphere (with one surface) and a hollow sphere with two). In all cases, you need to consider both outgoing and incoming radiation at each surface.
This works well until you slice objects so thin they are semi-transparent and no longer behave like blackbodies. This happens when you consider layers of atmosphere. “Optically thick” layers by definition emit blackbody radiation, but our atmosphere isn’t optically thick at all wavelengths and eventually becomes optically thin at all wavelengths if you go high enough. “Optically thin” layers modify the radiation passing through them. Those modifications add up. That is why we use the Schwarzschild eqn for radiative transfer through the atmosphere, but the S-B equation elsewhere. The S-B equation is only appropriate for radiation in equilibrium (via repeated absorption and emission) with the medium it is passing through. In classical experiments, a black cavity with a pinhole was used to create such an equilibrium. When radiation is not in equilibrium with its surroundings (semi-transparency, for example), you need the Schwarzschild equation.
Your model is flawed: pure PVC will melt at 423K. You need some poisonous additives, otherwise there will be some ugly mess on your garage floor… 😉
Apparently the concept of a thought experiment is beyond you.
Great ppost