In Part Four we took a first look at overlapping gases. pH2O’s absorption band was changed to overlap pCO2’s absorption band. And remember that pH2O has a much higher concentration in the lower atmosphere.
For those who haven’t followed the series so far, these are fictional molecules with only a passing resemblance to the real molecules H2O and CO2. The massive complexity of real spectral absorption and emission makes it difficult for people to appreciate the key points of radiative transfer in the atmosphere.
And of course, many people don’t want to “just accept” the results of a hugely complex computer model..
The simple model results revealed some interesting points:
- With overlapping bands, increases in pCO2 still led to a reduction in TOA flux.
- With increasing pCO2, DLR (back radiation) remains constant and yet TOA flux reduces.
It’s important to understand these results, because it’s very common to see an implicit belief that the TOA results are some kind of “mirror image” of the surface downward results. They aren’t even though of course they are related.
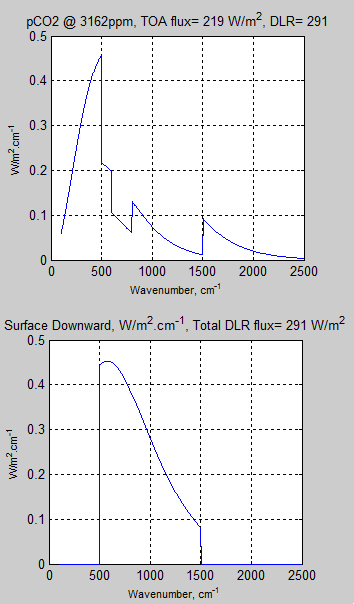
For the results shown in Figure 7 of Part Four, here is the last TOA spectrum and below, the corresponding DLR spectrum:
Figure 1
The balance of energy at TOA is what determines whether the planet warms or cools. Therefore, the spectral values at the surface are not the most important for determining which gases make the most contribution to the inappropriately-named “greenhouse” effect.
And the total value of back radiation at the surface is not what determines the long term surface temperature – because it is possible to reduce the TOA flux without increasing the surface downward flux. (Note 1)
Hopefully, this simple model demonstrates those points clearly.
Just for reference I have added this model version, v0.4.0 to the notes.
Stratospheric Temperatures and “Saturation”
The model results shown in Figure 7 of Part Four show that the TOA flux continues to reduce as the pCO2 concentration increases.
There is an important point here for the ever popular theme of “saturation”.
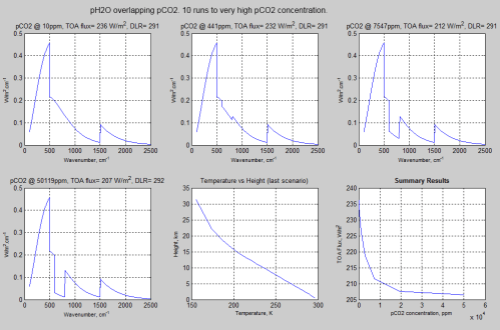
Let’s take a look at that model again, this time up to very high concentrations of pCO2:
Figure 2 – Click for a larger image
Notice that even as the pCO2 concentration has reached 50,000ppm the TOA flux is still reducing for increasing pCO2.
Also notice the temperature profile (5th graph in figure 2) – it’s important.
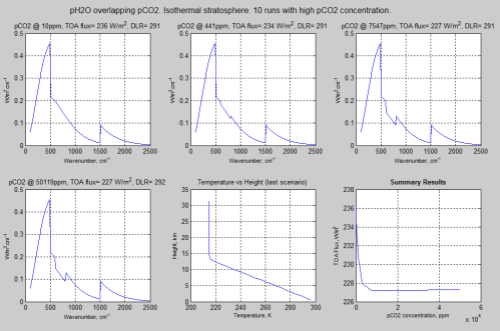
Now here is a similar model run with a slightly different constraint:
Figure 3 – Click for a larger image
These results show that “saturation” is reached much sooner. Notice the temperature profile.
The intensity of radiation is dependent on the temperature of the atmosphere from where the radiation takes place.
So if we have an atmosphere that keeps reducing in temperature as we go higher, then no matter how much the concentration of a “greenhouse” gas increases, the ever-higher radiation will be from a colder temperature – and therefore, will keep reducing in intensity.
Of course, eventually the atmosphere thins out to the point where even this effect disappears.
But hopefully the basic physics behind that idea is clear.
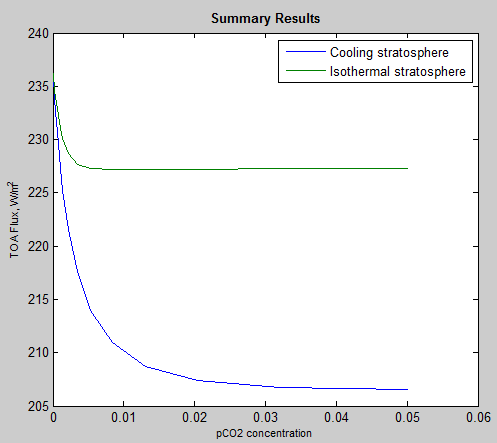
This is why in Figure 3 where the stratospheric temperature is held constant and isothermal (all at the same temperature), the changes in TOA flux level off much sooner. No matter where in the stratosphere the atmosphere radiates from it will be at the same temperature. (See the section “Why the Lapse Rate Matters” in Part Four which is covering a very similar point).
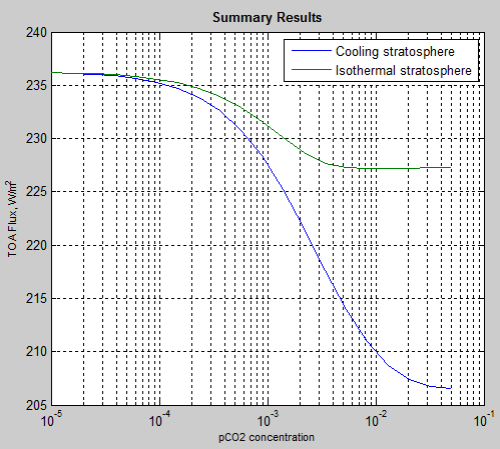
Here is the comparison, of 20 different pCO2 concentrations, where the stratosphere was held at 215K (isothermal) and where the stratospheric temperature was allowed to change according to the radiative heating/cooling:
Log plot
Linear plot
The temperature profile of the atmosphere does affect the “saturation” or not question by “greenhouse” gases.
Note that this model is still very simplistic – both of the gases have a fixed absorption within a band and zero outside. Real gases are much more complex and these complexities are very significant in the “saturation” question.
Conclusion
This article is more of a summary and consolidation so far, than any new ideas.
The next article, before covering line width issues, will cover some of the basic maths (and an explanation of the maths) behind how radiation moves through the atmosphere. At least that’s the intent at the moment.
Other articles:
Part One – a bit of a re-introduction to the subject
Part Two – introducing a simple model, with molecules pH2O and pCO2 to demonstrate some basic effects in the atmosphere. This part – absorption only
Part Three – the simple model extended to emission and absorption, showing what a difference an emitting atmosphere makes. Also very easy to see that the “IPCC logarithmic graph” is not at odds with the Beer-Lambert law.
Part Four – the effect of changing lapse rates (atmospheric temperature profile) and of overlapping the pH2O and pCO2 bands. Why surface radiation is not a mirror image of top of atmosphere radiation.
Part Six – The Equations – the equations of radiative transfer including the plane parallel assumption and it’s nothing to do with blackbodies
Part Seven – changing the shape of the pCO2 band to see how it affects “saturation” – the wings of the band pick up the slack, in a manner of speaking
And Also –
Theory and Experiment – Atmospheric Radiation – real values of total flux and spectra compared with the theory.
Notes
Note 1: Reducing the TOA flux = less heat leaves the planet = the planet warms; all other things being equal. More about this idea in The Earth’s Energy Budget – Part Three.
In an immediate sense the back radiation is one of the mechanisms by which the surface is at the temperature it is.
Think of the TOA flux as determining the long term temperature of the surface, and the back radiation as determining the current temperature of the surface.
And for the many who think that this means I am saying convection is unimportant, no I am not. I am explaining one effect on the surface temperature. The essence of understanding a complex subject is to be able to understand the separate effects, and then how they fit together.
Note 2: The Matlab code, v0.4.0:
The code is easiest seen by downloading the word doc, but here it is for reference:
======= v0.4.0 ======================
% RTE = Radiative transfer equations in atmosphere
% Objective – allow progressively more complex applications
% to help people see what happens in practice in the atmosphere
% v0.2 allow iterations of one (or more) parameter to find the TOA flux vs
% changed parameter
% v0.3 add emissivity = absorptivity ; as a function of wavelength. Also
% means that downward and upward radiation must be solved, plus iterations
% to allow temperature to change to find stable solution. Use convective
% adjustment to the lapse rate
% v0.3.1 changes the method of defining the atmosphere layers for radiation
% calculations, to have roughly constant mass for each layer
% v0.3.2 tries changing lapse rates and tropopause heights
% v0.3.3 revises element boundaries as various problems found in testing of
% v0.3.2
% v0.4.0 – introducing overlap of absorption bands
clear % empty all the variables, so previous runs can have no effect
disp(‘ ‘);
disp([‘—- New Run —- ‘ datestr(now) ‘ —-‘]);
disp(‘ ‘);
% SI units used unless otherwise stated
% ============= Define standard atmosphere against height ================
% first a “high resolution” atmosphere
% zr = height, pr = pressure, Tr = temperature, rhor = density
Ts=300; % define surface temperature
ps=1.013e5; % define surface pressure
% nmv=2.079e25; % nmv x rho = total number of molecules per m^3, not yet
% used
maxzr=50e3; % height of atmosphere
numzr=5001; % number of points used to define real atmosphere
zr=linspace(0,maxzr,numzr); % height vector from sea level to maxzr
[pr Tr rhor ztropo] = define_atmos_0_2(zr,Ts,ps); % function to determine (or lookup) p, T & rho
% Create “coarser resolution” atmosphere – this reduces computation
% requirements for absorption & emission of radiation
% z, p,Tinit,rho; subset of values used for RTE calcs
numz=30; % number of boundaries to consider (number of layers = numz-1)
minp=3e3; % top of atmosphere to consider in pressure (Pa)
% want to divide the atmosphere into approximately equal pressure changes
dp=(pr(1)-minp)/(numz); % finds the pressure change for each height change
zi=zeros(1,numz); % zi = lookup vector to “select” heights, pressures etc
for i=1:numz % locate each value
zi(i)=find(pr<=(pr(1)-i*dp), 1); % gets the location in the vector where
% pressure is that value
end
% now create the vectors of coarser resolution atmosphere
% z(1) = surface; z(numz) = TOA
% T, p, rho all need to be in the midpoint between the boundaries
% T(1) is the temperature between z(1) and z(2), etc.
z=zr(zi); % height
pb=pr(zi); % pressure at boundaries
Tb=Tr(zi); % starting temperature at boundaries
rhob=rhor(zi); % density at boundaries
% now calculate density, pressure and temperature within each layer
for i=1:numz-1
dz(i)=z(i+1)-z(i); % precalculate thickness of each layer
Tinit(i)=(Tb(i+1)+Tb(i))/2; % temperature in midpoint of boundary
p(i)=(pb(i+1)+pb(i))/2; % pressure in midpoint of boundary
rho(i)=(rhob(i+1)+rhob(i))/2; % density in midpoint of boundary
end
% ============ Set various values =========================
lapse=6.5e-3; % environmental lapse rate in K/m ** note potential conflict with temp profile already determined
% currently = max lapse rate for convective adjustment, but not used to
% define initial temperature profile
ems=0.98; % emissivity of surface
cp=1000; % specific heat capacity of atmosphere, J/K.kg
convadj=true; % === SET TO true === for convective adjustment to lapse rate = lapse
emission=true; % ==== SET TO true ==== for the atmosphere to emit radiation
tstep=3600*12; % fixed timestep of 1hr
nt=1000; % number of timesteps
% work in wavenumber, cm^-1
dv=5;
v=100:dv:2500; % wavenumber (=50um – 4um)
numv=length(v);
rads=ems.*planckmv(v,Ts); % surface emissive spectral power vs wavenumber, v
disp([‘Tstep= ‘ num2str(tstep/3600) ‘ hrs , No of steps= ‘ num2str(nt) ‘, numz= ‘ …
num2str(numz) ‘, minp= ‘ num2str(minp) ‘ Pa, Lapse= ‘ num2str(lapse*1e3) ‘ K/km’]);
% ============== Introducing the molecules ==============================
% need % mixing in the atmosphere vs height, % capture cross section per
% number per frequency, pressure & temperature broadening function
nummol=2; % number of radiatively-active gases
mz=ones(nummol,numz-1); % initialize mixing ratios of the gases
% specific concentrations
% pH2O = pretend H2O
emax=17e-3; % max mixing ratio (surface) of 17g/kg
mz(1,:)=(ztropo-z(1:numz-1)).*emax./ztropo; % straight line reduction from surface to tropopause
mz(1,(mz(1,:)<0))=5e-6; % replace negative values with 5ppm, ie, for heights above tropopause
% pCO2 = pretend CO2
mz(2,:)=3162e-6; % a fixed mixing ratio for pCO2
% absorption coefficients
k1=0.3; % arbitrary pick – in m2/kg while we use rho
k2=0.3; % likewise
a=zeros(nummol,length(v)); % initialize absorption coefficients
a(1,(v>=500 & v<=1500))=k1; % wavelength dependent absorption
a(2,(v>=600 & v<=800))=k2; % ” ”
% ========== Scenario loop to change key parameter =======================
% for which we want to see the effect
%
nres=10; % number of results to calculate ******
flux=zeros(1,nres); % TOA flux for each change in parameter
fluxd=zeros(1,nres); % DLR for each change in parameter, not really used yet
par=zeros(1,nres); % parameter we will alter
% this section has to be changed depending on the parameter being changed
% now = pCO2 conc.
par=logspace(-5,-2.5,nres); % values vary from 10^-5 (10ppm) to 10^-2.5 (3200ppm)
% par=1; % kept for when only one value needed
% ================== Define plots required =======================
% last plot = summary but only if nres>1, ie if more than one scenario
% plot before (or last) = temperature profile, if plottemp=true
% plot before then = surface downward radiation
plottemp=false; % === SET TO true === if plot temperature profile at the end
plotdown=true; % ====SET TO true ==== if downward surface radiation required
if nres==1 % if only one scenario
plotix=1; % only one scenario graph to plot
nplot=plottemp+plotdown+1; % number of plots depends on what options chosen
else % if more than one scenario, user needs to put values below for graphs to plot
plotix=[1 round(nres/2) 8 nres]; % graphs to plot – “user” selectable
nplot=length(plotix)+plottemp+plotdown+1; % plot the “plotix” graphs plus the summary
% plus the temperature profile plus downward radiation, if required
end
% work out the location of subplots
if nplot==1
subr=1;subc=1; % 1 row, 1 column
elseif nplot==2
subr=1;subc=2; % 1 row, 2 columns
elseif nplot==3 || nplot==4
subr=2;subc=2; % 2 rows, 2 columns
elseif nplot==5 || nplot==6
subr=2;subc=3; % 2 rows, 3 columns
else
subr=3;subc=3; % 3 rows, 3 columns
end
for n=1:nres % each complete run with a new parameter to try
% — the line below has to change depending on parameter chosen
% to find what the stability problem is we need to store all of the
% values of T, to check the maths when it goes unstable
mz(2,:)=par(n); % this is for CO2 changes
% lapse=par(n); % this is for lapse rate changes each run
disp([‘Run = ‘ num2str(n)]);
T=zeros(nt,numz-1); % define array to store T for each level and time step
T(1,:)=Tinit; % load temperature profile for start of scenario
% remove??? T(:,1)=repmat(Ts,nt,1); % set surface temperature as constant for each time step
% First pre-calculate the transmissivity and absorptivity of each layer
% for each wavenumber. This doesn’t change now that depth of each
% layer, number of each absorber and absorption characteristics are
% fixed.
% n = scenario, i = layer, j = wavenumber, k = absorber
trans=zeros(numz-1,numv); abso=zeros(numz-1,numv); % pre-allocate space
for i=1:numz-1 % each layer
for j=1:numv % each wavenumber interval
trans=1; % initialize the amount of transmission within the wavenumber interval
for k=1:nummol % each absorbing molecule
% for each absorber: exp(-density x mixing ratio x
% absorption coefficient x thickness of layer)
trans=trans*exp(-rho(i)*mz(k,i)*a(k,j)*dz(i)); % calculate transmission, = 1- absorption
end
tran(i,j)=trans; % transmissivity = 0 – 1
abso(i,j)=(1-trans)*emission; % absorptivity = emissivity = 1-transmissivity
% if emission=false, absorptivity=emissivity=0
end
end
% === Main loops to calculate TOA spectrum & flux =====
% now (v3) considering emission as well, have to find temperature stability
% first, we cycle around to confirm equilibrium temperature is reached
% second, we work through each layer
% third, through each wavenumber
% fourth, through each absorbing molecule
% currently calculating surface radiation absorption up to TOA AND
% downward radiation from TOA (at TOA = 0)
for h=2:nt % main iterations to achieve equilibrium
radu=zeros(numz,numv); % initialize upward intensity at each boundary and wavenumber
radd=zeros(numz,numv); % initialize downward intensity at each boundary and wavenumber
radu(1,:)=rads; % upward surface radiation vs wavenumber
radd(end,:)=zeros(1,numv); % downward radiation at TOA vs wavenumber
% units of radu, radd are W/m^2.cm^-1, i.e., flux per wavenumber
% h = timestep, i = layer, j = wavenumber
% Upward (have to do upward, then downward)
Eabs=zeros(numz-1); % zero the absorbed energy before we start
for i=1:numz-1 % each layer
for j=1:numv % each wavenumber interval
% first calculate how much of each monochromatic ray is
% transmitted to the next layer
radu(i+1,j)=radu(i,j)*tran(i,j);
% second, add emission at this wavelength:
% planck function at T(i) x emissivity (=absorptivity)
% this function is spectral emissive power (pi x intensity)
radu(i+1,j)=radu(i+1,j)+abso(i,j)*3.7418e-8.*v(j)^3/(exp(v(j)*1.4388/T(h-1,i))-1);
% Change in energy = dI(v) * dv (per second)
% accumulate through each wavenumber
% if the upwards radiation entering the layer is more than
% the upwards radiation leaving the layer, then a heating
Eabs(i)=Eabs(i)+(radu(i,j)-radu(i+1,j))*dv;
end % each wavenumber interval
end % each layer
% Downwards (have to do upward, then downward)
for i=numz-1:-1:1 % each layer from the top down
for j=1:numv % each wavenumber interval
% first, calculate how much of each monochromatic ray is
% transmitted to the next layer, note that the TOA value
% is set to zero at the start
radd(i,j)=radd(i+1,j)*tran(i,j); % attentuation..
% second, calculate how much is emitted at this wavelength,
radd(i,j)=radd(i,j)+abso(i,j)*3.7418e-8.*v(j)^3/(exp(v(j)*1.4388/T(h-1,i))-1); % addition..
% accumulate energy change per second
Eabs(i)=Eabs(i)+(radd(i+1,j)-radd(i,j))*dv;
end % each wavenumber interval
dT=Eabs(i)*tstep/(cp*rho(i)*dz(i)); % change in temperature = dQ/heat capacity
T(h,i)=T(h-1,i)+dT; % calculate new temperature
if T(h,i)>500 % Finite Element analysis problem
disp([‘Terminated at n= ‘ num2str(n) ‘, h= ‘ num2str(h) ‘, z(i)= ‘ num2str(z(i)) ‘, i = ‘ num2str(i)]);
disp([‘time = ‘ num2str(h*tstep/3600) ‘ hrs; = ‘ num2str(h*tstep/3600/24) ‘ days’]);
disp(datestr(now));
return
end
% need a step to see how close to an equilibrium we are getting
% not yet implemented
end % each layer
% now need convective adjustment
if convadj==true % if convective adjustment chosen..
for i=2:numz-1 % go through each layer
if (T(h,i-1)-T(h,i))/dz(i)>lapse % too cold, convection will readjust
T(h,i)=T(h,i-1)-(dz(i)*lapse); % adjust temperature
end
end
end
end % iterations to find equilibrium temperature
flux(n)=sum(radu(end,:))*dv; % calculate the TOA flux
fluxd(n)=sum(radd(1,:))*dv; % calculate the DLR total
% === Plotting specific results =======
% Decide if and where to plot
ploc=find(plotix==n); % is this one of the results we want to plot?
if not(isempty(ploc)) % then plot. “Ploc” is the location within all the plots
subplot(subr,subc,ploc),plot(v,radu(end,:)) % plot wavenumber against TOA emissive power
xlabel(‘Wavenumber, cm^-^1′,’FontSize’,8)
ylabel(‘W/m^2.cm^-^1′,’FontSize’,8)
title([‘pCO2 @ ‘ num2str(round(par(n)*1e6)) ‘ppm, TOA flux= ‘ num2str(round(flux(n)))…
‘ W/m^2, DLR= ‘ num2str(round(fluxd(n)))])
% —
%subplot(subr,subc,ploc),plot(T(end,:),z(2:numz)/1000)
%title([‘Lapse Rate ‘ num2str(par(n)*1000) ‘ K/km, Total TOA flux= ‘ num2str(round(flux(n))) ‘ W/m^2’])
%xlabel(‘Temperature, K’,’FontSize’,8)
%ylabel(‘Height, km’,’FontSize’,8)
grid on
end
end % end of each run with changed parameter to see TOA effect
if plotdown==1 % plot downward surface radiation, if requested
plotloc=nplot-plottemp-(nres>1); % get subplot location
subplot(subr,subc,plotloc),plot(v,radd(2,:)) % plot wavenumber against downward emissive power
title([‘Surface Downward, W/m^2.cm^-^1, Total DLR flux= ‘ num2str(round(fluxd(n))) ‘ W/m^2’])
xlabel(‘Wavenumber, cm^-^1′,’FontSize’,8)
ylabel(‘W/m^2.cm^-^1′,’FontSize’,8)
grid on
end
if plottemp==1 % plot temperature profile vs height, if requested
plotloc=nplot-(nres>1); % get subplot location
subplot(subr,subc,plotloc),plot(T(end,:),z(2:numz)/1000)
title(‘Temperature vs Height (last scenario)’)
xlabel(‘Temperature, K’,’FontSize’,8)
ylabel(‘Height, km’,’FontSize’,8)
grid on
end
if nres>1 % produce summary plot – TOA flux vs changed parameter
subplot(subr,subc,nplot),plot(par*1e6,flux)
title(‘Summary Results’,’FontWeight’,’Bold’)
ylabel(‘TOA Flux, W/m^2′,’FontSize’,8)
xlabel(‘pCO2 concentration, ppm’,’FontSize’,8) % ==== change label for different scenarios =========
grid on
end
disp([‘—- Complete End —- ‘ datestr(now) ‘ —-‘]);


[…] « Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Three Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Five […]
I’m not fully across your model, but I have a minor question which may have already been answered.
Do you account for density changes as well? I expect that at any given level of atmosphere at any given temperature, there will be a preferential energy flow in the direction of lower density. Simply because the mean free photon path will be longer in that direction.
Is this assumption correct?
If so, is it accounted for in your model?
Jerry:
Yes, you can see it in the pressure graph shown in Figure 2 of Part Two:
But for completeness here is a plot of density vs height:
And with density shown as a log plot:
Hello Science of Doom.
Thanks for the response, but I think you missed the point of the question, which was does a density gradient favour photo radiation flow from the more dense to less dense part? If so, do you account for that in your model?
It is nice to note that “science of doom” uses consequently the quotation marks around the word “greenhouse” when describing the response of the atmosphere to the thermal radiation from the surface. This is because the underlying physics is not about “the glass houses for cultivation of vegetables” but about “the roasting of a chicken in the microwave oven”.
Thus, we have a chicken (here the atmosphere) and the source of microwave radiation (here the thermal radiation from the surface). The differences are that the source of radiation covers a wide range of wavelength, the heater is places inside “the chicken” and the heat is allowed to escape from the system. The latter indicates that we will always expect the appearing of the temperature gradient independently if the “chicken” absorbs radiation or not. Besides, the chicken meat does not allow the convection process, which makes the chicken to be less useful as a model example.
But let’s stop talking about the roasting of a chicken since this will scare people to death.
So back to the real issue. The surface of the Earth has some temperature and warms the surrounding air by convection, conduction and radiation. The addition of the absorbing molecules increases the efficiency of the absorbing process. As a consequence, this leads to the additional raise of the temperature of the air and of the heater itself. The efficiency of the molecules to affect the temperature of the system by radiation can be found by means of, for example, a transient experiment when studying how the temperature of the transient heater is changing as a function of time due to the different concentrations of the molecules in the air. Such an experiment has nothing to do with the temperature lapse across the air and can be performed inside the greenhouse if one lacks the access to a more suitable place for carrying out the experiment.
The presence of the lapse makes, however, both the theory and the computations more complex, especially since the change of the lapse influences also the efficiency of convection. However, intuitively one would expect that the impact of the absorbing molecules on the temperature of the system will be stronger when the flow of heat is prohibited out of the system (the system is kept at a constant temperature) than when we allow a part of heat to leave the system through the outer surface. The results presented by “science of doom” confirm this, if I have understood the figures correctly.
I would also like to emphasize what all the other participants are mentioning in their posts, namely that “science of doom” is doing an excellent job.
The impact of the atmosphere on the climate of the Earth might be more clearly understood from studying the functioning of the electric bulb lamp.
Energy, delivered to the bulb is loosed to the surrounding by the thermal emission. But, notably, the air is evacuated from the bulb in order to achieve the higher temperature of the radiation element. With the air present around the heating element, the temperature of the heating element would be lower. This is due to the fact that the element is now cooling not only by the radiation losses but also by the heat conduction and convection through the air. The doubling of CO2 in the air within the bulb will not change the situation much.
The effect is thus opposite to what the climate science is expecting from the adding of the atmosphere to the radiative system.
But it should be remembered that in the case of the bulb we are talking about very large temperature gradient. The heat convection through the air is in such a case much stronger than the radiation from the air back to the heating element.
If we lower the power input into the heating element, the temperature of the heater will decrease, and the efficiency of the convectional heat losses will decrease, too. This will also decrease the radiation out from the heater and thus the efficiency of heating the air through the absorption of radiation. This drop of efficiency might be compensated by the increase of the absorbing molecules in the air. This will not affect the convection but will increase the amount of radiation that is absorbed by the air.
Assume that we succeed in absorbing all the radiation that leaves the heater. A part of this absorbed radiation will still manage to leave the system through radiation while the rest will be radiated back to the heater. This will certainly slow down the cooling of the air and the heater, but the temperature of the heater will still go down until it has stabilized at the new, now slightly higher level than previous one before enriching the air by the absorbing molecules. But even so, this new temperature will be lower than that before letting air to surround the heater.
Well, this is the problem that makes me uneasy about the explanation given by the climate science, namely, that the variation of the temperature of the Earth is explained by the variation of the absorbing gases in the atmosphere, solely. The changes of the composition of the air might affect the general trend of the temperature changes observed from the diagram of the temperature variations on the million years scale, but the absorbing gases seems not to be the main cause for these general temperature trends. I must therefore agree with DeWitt Payne, mentioning that the dominating factor causing the temperature trends on the Earth might be prescribed to the drifts of the continents and the changes of the energy flow from the inside of the Earth to the surface (which might result in volcanoes and collisions between the continental plates).
SOD: The results are getting more and more elegant.
One caveat: On the real earth, the stratosphere is warmer than the tropopause because oxygen and ozone absorb solar UV at this altitude. Increasing CO2 causes the stratosphere to cool because it is absorbing photons in the CO2 band from locations that are mostly cooler. This phenomena produces small spikes in the TOA spectrum when the spectrum is calculated well above the tropopause. Your model may be incapable of simulating this phenomena and the stratospheric cooling/tropopause warming “fingerprint” of GHG forcing.
However, I am confused about some aspects of your model, which may have changed. In Part II, you showed a temperature profile with a fixed lapse rate in the troposphere ending at about 12 km, a tropopause from 12-17 km, and a rising temperature in the stratosphere. I initially thought this temperature profile with altitude was unchanging. I also assumed that a similar unchanging profile was used by MODTRAN and HITRAN (but changing in the IPCC’s definition of forcing). Now it appears that the temperature profile above the troposphere or tropopause is being allowed to reach radiative equilibrium in your model while the troposphere has a fixed lapse rate. If you want to model the complications introduced by a warm stratosphere, you need to introduce a heat source at these altitudes OR, second-best, have a preset temperature profile with altitude. The model also needs the TOA (which was at 17 km in one case) to be well above the tropopause.
Frank:
The model is still simple with no solar heating. Effectively the model has a totally transparent atmosphere (to solar radiation). And of course, with no solar heating of ozone the stratosphere won’t increase in temperature. This might be something to be added in due course.
The temperature profile shown in Figure 2 of Part Two is the “starting” profile.
Usually the model then develops its own temperature based on the effect of radiation absorbed vs emitted, with a “convective lapse rate” adjustment.
So if the temperature is too cold as height increases, then the temperature is adjusted to the lapse rate.
The lapse rate is a parameter that can be varied, as shown in Part Four.
In the case of Stratospheric Temperatures and “Saturation”, I then added the ability in the model to fix the temperature of the stratosphere at one value. This helps to see how “saturation” can occur – at least in the case of an absorption band with very simple characteristics.
The model runs since Part Four have had the TOA lifted, usually above 30km. It is the parameter called “minp” in the code – which is in pressure, in v4.0 it is 3000Pa, or 3% of the surface pressure.
SOD: If you are looking for more things to try with your model, I’d love to see the pCO2 band broken up into 9 or 19 sub-bands of 20 or 10 cm-1 and one central band (680-720 or 690-710) twice as wide as the others. If the central band has an absorption coefficient of 0.8 or 1.6 and dropping two-fold each step from the center, the average absorption will remain about the current 0.3. With 32-fold or 512-fold changes in absorption, as CO2 increases we may see the center of the band saturate, while some section of the “wings” continues to grow with changes of a few W/m2 at the TOA. With modest changes in CO2 (2-3 fold), you might need to superimpose plots to see the difference or plot the differences. (I won’t be surprised to find that you are already several steps ahead of these suggestions.)
SOD’s model allows atmospheric temperature to reach radiative equilibrium above the top end of the fixed lapse rate. Does anyone know what happens with MODTRAN or HITRAN? Do these use a fixed temperature vs altitude profile (a “standard atmosphere” representing some sort of global average) or a “radiatively equilibrating” atmosphere or can you select from a variety of options? The IPCC defines forcing after radiative equilibration in the stratosphere, so this capability exists somewhere.
I have mentioned previously about ”the roasting of a chicken in the microwave oven” and “bulb lamp”. These two examples relate mainly to the rubric “Understanding …..” rather than to the actual problem of the temperature gradient across the atmosphere. So let me finish quickly my digression by being more precise on how these examples might contribute to the Understanding.
The example with roasting of a chicken shows that the effects of absorption of radiation can be explained, and even studied, in a quite simple manner. The simplicity of it is that a part of radiation from the heater is used for the raising of the temperature of the chicken instead of being emitted “unused” out to the space as it would be in the case if the chicken had lacked the absorbing molecules. This is as simple as that, so the climate scientists must stop using the word greenhouses, which is only misleading in this connection, and makes people to treat these scientists as being completely ignorants. The experiments by use of the microwave oven (not necessary with a chicken) might help a) to study the “saturation” effects, b) to check both the different theories and results of computational calculations concerning the formation and maintaining of the lapse across the object of study and c) to investigate the “TOA” effects.
The example with the bulb lamp points on the risks for misunderstanding the real processes when presenting explanations with convection and radiation being separated from each other. The evacuation of the air from the around of the filament reduces to zero (more or less) the contribution from the convection without changing the “back radiation” from the surrounding, here coming from the glass housing. This is pointing on the importance of convection and on its role in counteracting the influence of the downward radiation on the temperature of the source. On the experimental level, one is talking about the line source experiments, which are a well-known technique for studying the thermal properties of gases and liquids. Such a method might be used for the determination of the properties of air with or without different absorbing agents in the air. The experimental creates also the lapse across the material under the study which might be studied by means of the optical interferometric technique.
SOD, I’m having trouble with the DLR being independent of pCO2 concentration. I can see that Part 4 started with a dependence which was lost in the last simulation run, yet I can’t see what changed in the model to make it so.
David Reeve:
Problems in understanding are critical to making progress – it’s a serious point. At least, I have generally found this to be the case for myself.
Mathematically the answer is correct. Nothing has changed in the underlying physics of the model.
What you seek is conceptual understanding of how this can happen.
Here is my best attempt at a conceptual model for you. Picture a lot of pH2O in the lower troposphere. And picture a lot of pCO2 in the upper troposphere.
The pCO2 radiates into the pH2O which absorbs it all – and IN TURN – radiates down to the ground.
The pCO2 radiates up to the sky, which doesn’t absorb anything, so this is the radiation to space, or TOA flux, or “cooling to space”.
The pH2O in the lower troposphere radiates down to the ground ACCORDING to its temperature.
The temperature of pH2O is determined (primarily) by the surface temperature (and the radiative/convective linkage between the surface and the lower troposphere).
So pCO2 – in the SHORT TERM – cannot affect the downward radiation of the pH2O, because the temperature of the surface is the main determinant of the pH2O temperature.
But pCO2 – in the long term – CAN affect the surface temperature because less outgoing radiation (less cooling to space) means the climate heats up.
Exactly how that is “mediated” to the surface depends on many factors, but ultimately the surface will keep warming until the TOA flux balances the absorbed solar radiation (all other things being equal).
So long term (all other things being equal) an increase in pCO2 will cause more DLR from the pH2O. But it won’t show up in the short term, it is all “blocked” by the pH2O.
Jerry:
Thanks for the response, but I think you missed the point of the question, which was does a density gradient favour photo radiation flow from the more dense to less dense part? If so, do you account for that in your model?
Yes.
The absorption and emission is dependent on density.
There is a term in the code, called “trans”:
trans=trans*exp(-rho(i)*mz(k,i)*a(k,j)*dz(i)); % calculate transmission, = 1- absorption
This is the calculated transmittance, dependent on density for that layer x concentration of that gas in air.
So the equations automatically take care of effect of the density changes.
SOD, thanks for the explanation. I now can see that what has changed in the model is the fact the pH20 absorption band overlaps the pCO2 band.
When the bands are separate, the two components independently contribute to DLR from the lower (and presumably different, ie optical range will differ with wavelength) layers of the troposphere, regardless of the fact that pH20 is at a far higher concentration than pCO2. Thus changes in pCO2 must impact the DLR.
However, when the absorption band of pH20 overlaps that of pCO2, the high pH2O concentration means pCO2 will have minimal impact on the signal. I can see this is not a simple relationship and is safer to model than it is to work out on the back of an envelope.
David Reeve:
Glad my comment was some help.
Frank,
HITRAN is a database of molecular absorption lines, not a program to calculate atmospheric emission and absorption. Talking about a temperature profile in HITRAN has no meaning. SpectralCalc is a line by line program that uses the HITRAN database. That program does use different temperature profiles as does MODTRAN, which is a band model rather than line by line.. The standard profiles are tropical, mid-latitude summer and winter, sub-Arctic summer and winter and US 1976 standard atmosphere (SpectralCalc also has a Martian atmosphere). In MODTRAN when you change the surface temperature offset, it changes the temperature in the troposphere for altitudes from 0-11 km only by the magnitude of the surface temperature offset. The temperature (and pressure, concentration, etc.) profiles in MODTRAN can be accessed by selecting the save text output for later retrieval option and then clicking on the link at the bottom of the right hand pane after results have been calculated.
SpectralCalc
http://www.spectralcalc.com/info/about.php
MODTRAN
http://geoflop.uchicago.edu/forecast/docs/Projects/modtran.orig.html
Thanks DeWitt. SOD’s model makes me wonder what I’ve been looking at. There are a variety of possible temperature vs altitude profiles. Do these profiles adjust to radiative equilibrium above the tropopause (as required for calculating radiative forcing according to the IPCC’s definition) or are we seeing a fixed profile until the TOA.
The profiles are fixed above the tropopause (actually 12 km and up for MODTRAN as implemented on the web) so forcings calculated using MODTRAN or SpectralCalc do not correspond to the IPCC definition.
The lapse rate doesn’t change either. You don’t get the upper tropical troposphere warming faster than the surface when you change the surface temperature using the surface offset. You need a radiative/convective model for that. Except no such thing actually exists. Convection is handled by a kludge in GCM’s. The entire 100 km2 cell is convectively re-arranged whenever some stability parameter is exceeded.
Some argue (Gerald Browning for example in several long threads at Climate Audit) that even weather forecasting models don’t properly model convection. The needed resolution may be on the order of 1 mm. While weather models are much finer than GCM’s they’re still orders of magnitude away from that. Then there’s the problem that as you go to finer resolution, the stability of the solution gets worse exponentially. Spatio-temporal chaos is really messy.
The density of the molecules is essential for the greenhouse effect. The amount of the molecules determines the pressure increase. Therefore the following diagram:
http://www.bilder-hochladen.net/files/h9qc-b-jpg.html
[img]http://www.bilder-hochladen.net/files/h9qc-b-jpg.html[/img]
I want to leave a general comment. Firstly, thank you for all this work. It has been a continuing inspiration and resource for me.
I do believe there are two important aspects of radiative transfer this series has not addressed.
The first is that there is a disconnect between the surface and the TOA in all saturated GHG outgoing bands. This concept follows simply from either modtran or hitran transmission to the tropopause, which in the case of CO2 is zero in the saturated bands.
You can try to parse from the available information exactly how far OLR can get in these saturated bands. It seems like maybe 5 meters for the WN 667.4 fundamental CO2 band, somewhat more in the negative and positive P and R rotational bands on either side. You can find estimates ranging from 10’s to hundreds of meters for the composite saturated bands.
It doesn’t matter if the answer is a meter or 7 kilometers. If transmission to the tropopause is zero, there is a gap.
The reason this is important is that satellites (including CERES) looking down do not see energy communicated from the surface in these bands. They see CO2 bands reenergized indirectly by solar energy collected by stratospheric ozone.
The second is that the tiered structure of CO2 resonance is not addressed.
Properties of that strange linear molecule dictate that every transition after 667.4 is a “hot band” incremental and dependent on the fundamental. There is a second order of three far weaker transitions on which all further yet far weaker transitions are incremental.
To be sure, there are weird “crossover” resonances that somehow avoid the principal quanta. The troposphere is basically a mess of “self” and otherwise broadening, collisional induced emission, coriolis affected rotations, and doubtless other imponderables.
Yet zero is zero. Both the fundamental and second order transitions are squarely in the saturated bands. That leaves us with the nearly trivial further incremental transmissions to work with in the wings.
Your matlab work shows striking concentration of “wing” energy at less than WN 500. That makes sense, but the important question is where on the approximately logarithmic Beer diminution does all this leave us?
The models doubtless set an initial condition of 1 and let the logarithm run…
There is no disconnect or gap. There is continuous absorption and emission over the whole column. The way to look at it is to use thin enough slices that the absorptivity, even in the center of the band, is small in each slice. It turns out, however, that isn’t necessary for radiative transfer calculations. Thick slices give you a reasonable approximation.
You’re also neglecting rotational transitions in your diagram. The tens of thousands of transitions from that are the origin of the band structure.
You are saying this is wrong and there actually is transmission to the tropopause in the saturated bands?
This and many other modtran and hitran findings very clearly show zero transmission.
gymnosperm: SOD’s graph shows the fraction of LWR emitted by the surface that reaches space at every wavelength. However, this is only a small part of the big picture – only about 15% of the LWR reaching space is emitted by the surface. The atmosphere from 0-1 km above the surface also emits LWR (both up and down). Since the GHGs in the atmosphere don’t emit like a blackbody, that flux is concentrated in a fewer bands. SInce the temperature drops about 6.5 K/km in the atmosphere, slightly less total energy (about 4%) is emitted than would be if the air were at surface temperature. Still, essentially all of the center of the CO2 band fails to reach space, but transmission on the sides of the CO2 band does increase.
Now consider what the atmosphere between 1 and 2 km above the surface emits. The same pattern continues: more radiation is transmitted on the side of the bands and the atmosphere is colder. It is also less dense: fewer GHGs to emit and the lines begin to narrow slightly (less collision and Doppler broadening).
Continue this process for every kilometer layer of the atmosphere. You are above most of the water vapor within a few kilometers. You are above half of the CO2 at about 5 km. It stops getting colder near the tropopause (10-17 km) and then begins to warm. By 20 km, all but the strongest line in the CO2 band have become somewhat transparent. Somewhere SOD has published a plot showing the W/m2 reaching space from photons emitted by each layer of the atmosphere. The source of photons reaching space is spread over the whole troposphere, with the average source altitude being around 5 km.
Then remember that 60% of the sky is cloudy and LWR is emitted from cloud surfaces as well as the atmosphere and the ground surface.
So there is a gap in surface emission that reaches space for the CO2 band, but the planet as a whole emits a lot of energy through the CO2 band, with essentially all of it coming from the atmosphere, not the surface. And since the atmosphere is colder than the surface, less energy is emitted through the CO2 band than nearby wavelengths. However, emission from this band is NOT zero.
The changes absorption and emission produce in upward LWR on the way to space are difficult to fully understand without the assistance of software. The link below allows one to use the MODTRAN software package for calculating upward LWR at any altitude (the “look down” altitude) from any atmosphere you want to define. If you really want to understand what happens, start with the simplest system: No GHGs except 4 ppm of CO2. Look down from 1 km, to see what a tiny bit of CO2 does in the first 1 km above the surface – almost nothing since 1 km of atmosphere with 4 ppm of CO2 is effectively transparent; meaning you see only blackbody radiation from the surface of the planet. Now look down from 10 km and 70 km (the highest, called the Top of the Atmosphere or TOA, above which no significant change in outgoing OLR occurs). When you understand 4 ppm of CO2, try 40 and then 400 and then 4000 ppm. When you understand CO2, then look at the other GHGs.
http://climatemodels.uchicago.edu/modtran/
I believe your Schwarzschild approach ignores the fact that there is no radiation above a few hundred meters in the saturated CO2 bands to pass on to higher layers.
The P, Q, and R channels of the fundamental bending mode of CO2 centered at WN 667.4 and extending from 630 to 715 basically defines the MODTRAN zero transmittance to the tropopause at 280 ppm preindustrial CO2.

Satellites, high flying aircraft, and balloons looking down from above 20 km see CO2 radiance with flat spots sitting on Plank temperatures that correspond to the lower stratosphere. These flat spots correspond to the saturated fundamental spectra of CO2.
I believe a fundamental error is the assumption that instruments from above 20 km are seeing radiation communicated from the surface. I believe what they are seeing is CO2 radiation kinetically re energized indirectly by ozone.
The existence of this substantial gap in CO2 radiance from a few hundred meters to the lower stratosphere renders “area below the curve” approaches meaningless.
This interpretation is supported by observational evidence.

The above from Mlynczak (2014) at Nasa is measurements taken from a plane. It clearly shows the “gap” @ WN 667.
Interestingly there is a lot going on in the H2Ov spectra on the 500 range at a surprisingly high altitude.

This one also shows the gap and upper tropospheric water hotspot. Surprisingly the CO2 radiance shown here is concentrated at a far higher altitude (basically the stratopause) and Planck temperature than the Nimbus sensors were seeing, and far above the ozone layer.
I have often wondered why the stratosphere keeps warming long after the ozone is gone. CO2 has only a few tiny bands in the incoming solar spectra so it can’t be doing it.
Something else is indirectly lighting up CO2 up there
Gymnosperm wrote: “I believe your Schwarzschild approach ignores the fact that there is no radiation above a few hundred meters in the saturated CO2 bands to pass on to higher layers.”
Frank replies: There is plenty of observational data showing that the Schwarzschild equation correctly predicts the LWR flux at all wavelengths traveling upward and downward through the atmosphere.
Gymnosperm wrote: “I believe a fundamental error is the assumption that instruments from above 20 km are seeing radiation communicated from the surface. I believe what they are seeing is CO2 radiation kinetically re energized indirectly by ozone.”
Frank replies: Of course, instruments at 20 km are not seeing 650-700nm photons emitted by the surface. Those photons have all been absorbed. The instruments are seeing photons emitted from CO2 molecules between 15 km and 20 km in altitude, possibly mostly between 19 km and 20 km. Your phrase “communicated from the surface” suggests you, like many others, misunderstand how most CO2 molecules (and other GHGs) are excited in the troposphere and lower stratosphere. These altitude are in local thermodynamic equilibrium (LTE), which means that the average CO2 is excited and relaxed by collisions much faster than it is excited by absorbing a photon and relaxed by emitting a photon. The number of CO2 molecules in an excited state does not depend on how much “radiation is communicated from the surface” – it depends only on the local temperature (the amount of energy in molecular collisions). LTE greatly simplifies the problem of calculating radiative transfer through the atmosphere – you only need to know the local temperature and composition of the atmosphere to know how many photons it will emit and what fraction of the entering photons it will absorb.
The local temperature in the troposphere is the result of heat transfer by both radiation and convection – with the latter maintaining an average drop in temperature of 6.5 K per km of altitude. In the stratosphere, convection is not important and temperature is due to radiation transfer alone. That includes both LWR absorbed by CO2 and O3 and H2O, but also the SWR absorbed by O3. So CO2 in the stratosphere is “energized indirectly by ozone” and the mechanism of energy transfer is collisions – thermal energy/temperature.
The real gap you see in the plot of Net Flux vs Altitude and wavelength is due to the temperature of the molecules that emitted the photons being measured.
The 650-700 nm photons emitted by CO2 don’t travel very far before they are absorbed until you reach high altitudes where the atmosphere is less dense. It is very cold (200-240K) at those altitudes, so few photons are emitted.
Note: You “Net Flux” is OLR minus DLR, further complicating the analysis.
Your last graph shows heating and cooling rates in different colors, a subject discussed several times by SOD. The results are from radiative transfer CALCULATIONS using the Schwarzschild eqn. The dotted line is the tropopause. Above the tropopause, the stratosphere is in radiative equilibrium and the temperature isn’t changing. So all of the radiative cooling at the wavelengths emitted by CO2 and O3, is being balance by absorption of UV by ozone at wavelengths not shown on this graph. CO2 and O3 both emit and absorb LWR, but the energy from absorption of UV makes it warm enough for these GHGs to emit much more LWR than they absorb. Below the dotted line, there is also more radiative cooling. That is due to some absorption of SWR in the troposphere, but mostly due to absorption of SWR by the surface followed by convection to the upper troposphere, which emits more LWR than it absorbs.
You obvious didn’t spend much time exploring with MODTRAN as I suggested. Before you can say that the Schwarzschild equation fails to predict how radiations fluxes change as they pass through the atmosphere, you need to know what they actually DO predict (and then understand why). THEN show us how the Schwarzschild equation fails.
Frank,
Minor nitpick. Oxygen also absorbs in the solar UV. If it didn’t, there wouldn’t be any ozone.
Thanks, DeWItt.
Just reviewing and noticed that ozone radiance is also concentrated @ 45 km. Either this is an anomaly or the standard ozone profile is wrong.
gymnosperm,
You should carefully read Frank’s replies to your comments and work to understand them. Then you won’t make so many mistakes.
Ozone radiance depends on two things: ozone concentration and temperature. In the stratosphere, maximum ozone concentration is at about 25 km and the maximum temperature is at about 50-55 km. So the maximum emission from ozone should be somewhere between those two altitudes.
Just not seeing my “mistakes”. My point regarding ozone and temperature was that maximum concentration does not coincide with maximum temperature. You have restated this well known feature of the standard conception of the stratosphere.
I provided an observation which showed strong CO2 radiance centered at ~45 km and seemingly going strong where it is cut off ~60 km. As you know, above 55 km or so the lapse rate goes negative again. Clough 1995 graphic also showed a weaker ozone radiance centered somewhat lower ~43 km and contained within the stratosphere.
As you have pointed out, ozone concentration is thought to fall off sharply at 25 km.
I read Frank’s comments carefully, and I see another waiting, but so far I have not learned anything new.
What I would like to learn is 1. why ozone is radiating so strongly near the top of the positive stratospheric lapse rate when its concentration is so low there? 2. Why the Nimbus IRIS instruments do not see radiance at appropriate Planck temperatures for these altitudes? 3. What is lighting up CO2 at the top of the stratosphere into the mesosphere when even ozone radiance has flagged?
gymnosperm,
You wrote: “Just not seeing my “mistakes”. ”
Frank has patiently pointed out and explained a number of them. There is no point in my repeating what he has done so well.
You wrote: “My point regarding ozone and temperature was that maximum concentration does not coincide with maximum temperature.”
There is no reason why they should coincide.
You wrote: “ozone concentration is thought to fall off sharply at 25 km.”
Actually, it drops off gradually above 25 km.
You wrote: “What I would like to learn is 1. why ozone is radiating so strongly near the top of the positive stratospheric lapse rate when its concentration is so low there? ”
A big factor is temperature. There is also at least some absorption of the emissions from lower altitude.
You wrote: “2. Why the Nimbus IRIS instruments do not see radiance at appropriate Planck temperatures for these altitudes?”
Who says they don’t?
“3. What is lighting up CO2 at the top of the stratosphere into the mesosphere when even ozone radiance has flagged?”
The energy comes from thermal excitation, which depends on temperature. The concentration of CO2 drops more slowly than O3 since CO2 has a nearly constant mixing ratio, so the concentration is proportional to air density. The O3 mixing ratio decreases with altitude in the upper stratosphere.
Gymnosperm wrote: “What I would like to learn is 1. why ozone is radiating so strongly near the top of the positive stratospheric lapse rate when its concentration is so low there?”
Look carefully at the units on the scale in the bottom Figure you provided: degK/day-cm-1. Radiance is W/m2. This is a cooling rate (per cm-1). When only a small mass is involved, it doesn’t need to radiate away much energy to cool rapidly. However, it is NOT cooling rapidly – it is gaining as much energy absorbing SWR (UV) as it is losing as LWR. In fact, it absorbs so much UV that is it roughly 50 degK warmer than it is at the tropopause (where there is little UV left to absorb).
The authors of this Figure are using a program like MODTRAN to perform the radiative transfer calculations that produced the Figure you posted. Why are you unwilling to use it to inform yourself? Then you might be reminded that O3 is a greenhouse gas that reduces planetary emission of LWR by about 5 W/m2 despite its radiating so strongly from the middle of the stratosphere. Do you understand why this happens and how it appears on your graph of cooling rates?
This implementation of MODTRAN has a graphs showing the O3 concentration and temperature vs altitude used by the program. Conduct some experiments with it and come back with questions.
To clarify, MODTRAN is integrating the Schwarzschild eqn from the surface to the look down altitude as the intensity of upward radiation (I) of a particular wavelength (lambda) is changed (dI) by passing through a layer of atmosphere of thickness, dz, and the density of the GHG is n, its absorption cross-section is o and its temperature is T.
dI = emission – absorption
dI = n*o*B(lambda,T)*dz – n*o*I*dz
When emission and absorption are in equilibrium, dI is zero and I = B(lambda,T), blackbody intensity. The blackbody curves on the MODTRAN output show you the intensity the radiation expected where absorption and equilibrium at a particular temperature.
I’m so mad I was looking at a MODTRAN graphic yesterday morning and failed to save it. Can’t find it now, but what it showed was pasically your prescription to scroll down through MT CO2 at different concentrations. What it showed was unsurprising except that the curves flatlined at the angle of the Planck/blackbody function through what I am calling the gap.

My best efforts to describe the lost file likely having fallen short, I submit below a graphic to clarify my position:
The base graphic is an effort to describe “cooling”. As you well know, radiation is a double edged sword causing largely kinetic warming when it is blocked by interfering absorbers, and cooling when quanta are free to escape to space.
The units in the base graphic are 10^-3 degree K/C wavenumbers.
The colors are fairly intuitive except for the hypercenters going coolish again.
Negative numbers are gray scale and represent warming.
Having done this little exercise, I understand a bit more why Nimbus saw radiation in the bands of my own graphic. Ozone was warming there.
Why?
I get the integration of the double edged functions.
What defines the transition?
You can also look up with MODTRAN. In that case, the program is integrating from the top of the atmosphere, defined as 100 km, down to the look up altitude.
Gymnosperm: Now that you are actually working with MODTRAN, I presume you have noticed that:
1) If there are no GHGs, OLR is simply the blackbody radiation emitted by the surface of the planet. (Surface temperature varies with your choice of atmosphere, tropical, US Standard etc).
2) As the concentration of CO2 is increased from a few ppm, less and less OLR reaches space up to a certain point – near 220 K on the blackbody emission scale. The center of the CO2 band reaches this limit at about 40 ppm and is still touching it at 400 ppm. At 4000 ppm the center of the band rises but the wings are still touching. Obviously we are not dealing with simple absorption of the blackbody radiation emitted by the surface of the earth, based on the difference between 4 ppm and 40 ppm, all of the surface emission is absorbed in less than one kilometer. By the time we get to 400 ppm, in the center of the CO2 band we are seeing photons emitted by CO2 in the atmosphere. The intensity of that radiation varies because those CO2 molecules have different temperature. If you look at the temperature vs altitude plot (which varies depending on whether you choose the US Standard Atmosphere – what I’m looking at now – or a Tropical Atmosphere), there is a broad minimum in temperature at 217 K from 11 to 21 km. When the center of the CO2 band has a blackbody intensity of about 220 K, most of the photons reaching space were emitted from this broad tropopause. Most of the photons emitted from below were absorbed and there isn’t enough CO2 to change the number of photons arising from near the tropopause by emission or absorption.
3) If you crank CO2 up to 4,000 ppm or 40,000 ppm, there is enough CO2 above the tropopause to change the intensity of radiation rising from below. Eventually the intensity of the center of the CO2 band rises to 260 K, which is about the average temperature between 40 and 60 km.
If you look at the Schwarzschild eqn that is interacting with our atmosphere to produce this complex behavior, you will see that when the radiation passing through a layer of atmosphere has blackbody intensity, it isn’t changed (dI = 0, absorption equals emission). If it has greater than BB intensity (for the local temperature), dI is negative (more absorption than emission) and the opposite is true when the incoming radiation has less than BB intensity for the local temperature. The change is always towards BB intensity and the rate at which BB intensity is approached depends on the density of the absorbing gas and the strength of its interaction with radiation (its absorption cross-section at a particular wavelength).
For your final exam, try to explain the radiative HEATING and cooling produced by ozone in the stratosphere in your Figure.
Frank,
The problem with using MODTRAN at high CO2 is that the temperature profile above 13km or so never changes. High levels of CO2 would, in the real world, rapidly cool the stratosphere and decrease emission from the center of the CO2 band to space. The safest thing to do is look down from the tropopause, defined as the altitude just before the temperature starts to increase, and ignore the stratosphere. To a good approximation, radiation to space from above the tropopause would not increase after the stratosphere was allowed to equilibrate. MODTRAN does not allow this to happen.
DeWitt: All radiative transfer calculation start with a defined atmospheric composition and temperature profile. This is their fundamental limitation. The calculations can tell us how a change in GHGs will change radiation – but not how temperature and composition (humidity, O3) will change. In the troposphere, temperature is controlled by both radiation and convection, a phenomena RT can’t describe. So some RT calculations stop after simply calculating the change in OLR and DLR. The stratosphere is in radiative equilibrium and its change in temperature in response to increasing GHGs and changing radiation can be calculated by repeated approximation. SOD’s RT program did so. MODTRAN probably does not.
The point of my EXERCISE with 4,000-40,000 ppm was to demonstrate how the B(lambda,T) term in Schwarzschild equation interacts with the temperature profile and the GHG concentration to change OLR. Hopefully, the software is using the temperature profile displayed in the graph, which doesn’t change with changes in CO2. It demonstrates that the GHE is fundamentally linked with the temperature gradient in the atmosphere – and not an inmate property of GHG’s. Since temperature usually drops with altitude where most photons escaping to space are emitted, there is usually GHE in most atmospheres. However, it has recently been recognized that the negligible lapse rate in Antarctica results in no GHE over most of the continent. Those who have been trying to detect warming in the bulk of Antarctica (to verify the existence of GLOBAL warming) may be chasing something that isn’t there (except fame).
As usual, you are correct: it would be a big mistake to think MODTRAN models the changes in both OLR and temperature (and humidity and probably O3) that would accompany 4000-40,000 ppm CO2. However, once the stratosphere has equilibrated, I think it is OLR-DLR at the tropopause and TOA are the same, not OLR alone.
I am confused about both OLR and radiative forcing (RF = OLR-DLR) not changing between the tropopause and the TOA. MODTRAN at 400 ppm CO2 certainly shows negligible change in OLR, but it also shows 9 W/m2 of DLR (at the tropical tropopause). Is UV absorption by the stratosphere the source of this 9 W/m2. The alphabet soup of names for radiative forcing (iRF, FI, Fa) adds to my confusion.
Mostly it’s UV absorption, but there is some absorption of LW OLR from the troposphere that contributes to the total energy absorbed in the stratosphere and thus to the total emission up and down. If you set the stratospheric ozone scale to zero, OLR at 70km for the tropical atmosphere increases about 5W/m². I should look at the Petty book, but as I remember, the cooling curve is slightly positive at the tropopause and negative everywhere else because of LW IR absorption.
Unless I’m reading Hansen’s paper, I forget which is what on forcing too.
” By the time we get to 400 ppm, in the center of the CO2 band we are seeing photons emitted by CO2 in the atmosphere.”
That is not what I see. I see flatline across the fundamental bending gap with no articulation whatsoever at 667.4.

Where do you think the light is coming from? It is coming from the surface. If it is extinguished at pick a number, a hundred meters, a kilometer, there is no more left. It is dissipated by kinetic interactions. Done gone.
CO2 does not just love to swim, it loves to dance. It grabs every photon in the fundamental bending strike zone and will not let it go until it has danceathoned it to death.
Light cannot spontaneously generate at higher levels of the atmosphere. CO2 can be kinetically kinetically lit up but it cannot do so against the tropospheric lapse rate. It does not get lit up again until ozone helps it out at the stratopause. Ozone kinetically energises CO2 both directly and likely more importantly indirectly through the following resonance with Nitrogen:
In case you are uncomfortable with the cooling units in the prior graphic, I submit the following :

I trust your suggestion that my prior image was model based. I don’t know everything , and neither do you.
This, from the best of my information was a learjet outfitted by NASA with a spectrophotometer intended to deal with the intense tropospheric doppler/rayleigh/coriolis/pressure broadening noise.
[A bit of conjecture here] the circular descending paths over a coastal area suggest Langley.
This is absolutely an observational dataset.

Here is the original image for context.
What do you see? I see a gap, a gap partially filled by incursion just below the tropopause by doppler/rayleigh/coriolis/broadening effects of water. Not water vapor, which as you have correctly pointed out is largely absent at this altitude; liquid water in the form of tropical thunderclouds, and ice, in the form of cirrus. Liquid water and ice have remarkably similar spectral properties which differ significantly from vapor.

Now, I really like your final exam question. I have been struggling with this. Why would CO2 resonance be focused in a narrow range in the NIMBUS data and ozone free to roam, pretty much tracking the atmospheric window?

You tell me. I am just a naked ape. so are you. Let’s both continue to learn.
Frank wrote: “” By the time we get to 400 ppm, in the center of the CO2 band we are seeing photons emitted by CO2 in the atmosphere.”
Gymnosperm: “That is not what I see. I see flatline across the fundamental bending gap with no articulation whatsoever at 667.4. Where do you think the light is coming from? It is coming from the surface. If it is extinguished at pick a number, a hundred meters, a kilometer, there is no more left. It is dissipated by kinetic interactions.”
Frank replies: If 4 ppm CO2 causes a significant loss of transmission and 40 ppm lets only 1/3 of the surface radiation through, how can any radiation between 650 and 700 reach space through 400, 4,000 and 40,000 ppm of CO2. MODTRAN shows more radiation reaching space at some CO2 wavelengths with more CO2. Obviously a negligible fraction of the photons emitted by the surface at these wavelengths reach space at 400 pmm. Thus my conclusion that the photons reaching space must be emitted by the atmosphere.
Emission of photons obviously requires getting some CO2 molecules into an excited state. Many people mistakenly think that CO2 molecules are only excited by absorbing a photon (or at least the majority are excited by absorbing a photon). This is completely wrong. In the troposphere and stratosphere, the vast majority of excited CO2 molecules are produced by collisions that create a Boltzmann distribution of vibrational and rotational energy states. The Planck function in the Schwarzschild eqn and Planck’s Law contains the temperature dependence of the fraction of CO2 molecules found in an excited state.
Gymnosperm fantasizes: “CO2 … grabs every photon in the fundamental bending strike zone and will not let it go until it has danceathoned it to death.
Light cannot spontaneously generate at higher levels of the atmosphere.
Returning to reality, Frank says: The average vibrationally excited state of CO2 lasts for a few microseconds before emitting a photon. However, the kinetic theory of gases shows that the time period between collisions is about 1 nanosecond at 1 atm.
http://hyperphysics.phy-astr.gsu.edu/hbase/kinetic/frecol.html#c1
Collisions both excite and relax CO2 molecules much faster than photons do in the lower atmosphere. That is why we say the troposphere and stratosphere (up to about 100 km) are in local thermodynamic equilibrium. The emission term in the Schwarzschild equation depends only on temperature, not the local radiation field. This equation only applies when such local thermodynamic equilibrium exists.
The emission of thermal (blackbody) radiation by solids and liquids is widely accepted and understood, but many people fail to recognize that the same fundamental physics applies to gases. There are two differences: 1) The heterogeneous environment and constant collisions in solids and liquids often broadens the emission to a broad band or continuum. 2) Solids and liquids are so dense that absorption and emission are usually in equilibrium. We expect them to emit radiation of blackbody intensity, although we apply an emissivity fudge factor to the real world. The absorption and emission of photons frequently have not come into equilibrium (which depends on temperature) in the atmosphere. The radiation passing through the atmosphere in many places does not have the intensity expected for blackbody radiation at the local temperature. Thus the surface of the planet emits like a simple blackbody, but the radiation reaching space is very different.
Frank,
Actually, the decay time for a free vibrationally excited CO2 molecule (bending mode) is on the order of 2 seconds, not microseconds. Also, only a small fraction of collisions are inelastic and cause a change in energy so the average time between inelastic collisions is much longer than 1nsec, a few μs is more like it. But emission intensity still only depends on the average number of molecules in the excited state at any time and the inverse of the decay time. The number in the excited state in a given volume, in turn, only depends on the energy level and degeneracy (=2 for CO2) of the excited state, the number density of CO2 molecules and the temperature. Since the energy level and degeneracy and decay time are constants, intensity is a function of temperature and number density only.
Collisional excitatation and deexcitation being several orders of magnitude higher than absorption and emission of photons is required for the Boltzmann distribution to apply and other things like Kirchoff’s Law to be valid. It’s also called Local Thermodynamic Equilibrium or LTE.
DeWItt: Thanks for the correction on the lifetime of excited CO2. I thought the value I cited came from you, but obviously my memory failed me. I did a quick search to confirm what I remembered before commenting above, but failed to find anything useful. Are you aware of any good sources?
For CO2 at the surface and tropopause, I’d really like to have in one place:
1) Fraction in excited vibrational state – from hv and Boltzmann distribution*
2) Collision rate – from hyperphysics.
3) Relaxation rate. “non-elastic collisions”
4) Excitation rate. (2,3,4 linked)
5) Einstein B12 and A21 coefficients
6) Absorption cross-section(s) (linked to 5)
The only source I have that ties all of this material together is a bit too comprehensive and dense (not to mention obscure):
http://www.hindawi.com/journals/ijas/2013/503727/
Frank,
In one place is tough. The Einstein A21 is in the HITRAN database. spectralcalc.com lets you do a few searches for free. Here’s the data in HITRAN format for CO2 at 667.66 cm-1:
21 667.661347 2.981e-19 1.542e+00.0740.1020 106.12970.710.000000 0 1 1 01 0 0 0 01 Q 16e 465550 6 2 1 1 1 0* 33.0 33.0
Translating, That’s molecule #2, isotopologue #1 , transition frequency 667.661347 cm-1, line intensity 2.981E-19, Einstein A21 1.547s-1. The rest are data used to actually calculate a spectrum see here: https://www.cfa.harvard.edu/hitran/formats.html
I remembered the A21 being greater than 1, but I forgot that was per second. So the 1/e decay time is 0.65 seconds, so I was off by a factor of 2. The A21 and B21 coefficients are related. You know one, you can calculate the other. Similarly B12 can be calculated from B21. But stimulated emission is not a significant factor in the atmosphere for CO2.
An on line calculator and reference that would do everything you want would be nice, but I’m not holding my breath.
Further indication from MODTRAN that the layer by layer radiative transfer concept is incorrect. Here we look both up and down from various altitudes. The “background run” (red) in al cases is looking down.

The clear result of this balloon ride is that CO2 emissivity below a kilometer is essentially zero. CO2 photons are not being handed up to higher levels in the atmosphere.
gymnosperm,
Your misinterpretation of the data is hilarious. I want more.
I would give you a serious reply, but I don’t see the point.
Frank,
“Emission of photons obviously requires getting some CO2 molecules into an excited state. Many people mistakenly think that CO2 molecules are only excited by absorbing a photon (or at least the majority are excited by absorbing a photon). This is completely wrong. In the troposphere and stratosphere, the vast majority of excited CO2 molecules are produced by collisions that create a Boltzmann distribution of vibrational and rotational energy states. The Planck function in the Schwarzschild eqn and Planck’s Law contains the temperature dependence of the fraction of CO2 molecules found in an excited state.”
Then why is the emission/re-emission only narrow band and not broadband? The emission, except if from the water or ice in clouds, is claimed/established to be narrow band originating only from GHG molecules, yet it is also thought to be established that the atmosphere emits based on its temperature in the same way a dense body emits broad band Planck emission from its outer surface.
A thermometer in the atmosphere measuring temperature cannot distinguish between a flux of incident photons and a locally present kinetic flux of molecules in motion. The measured temperature in a given spot will always be the combination of the two, even in the lower troposphere where the molecules are the densest.
It does seem that the atmospheric science community is arbitrarily using the notion of LTE as it applies in a liquid or solid to that of a gas, and a very thin radiating gas as is the Earth’s atmosphere.
I understand the mainstream model of atmospheric radiation only works and gets the right answer when Kirkchoff’s law is applied to each wavelength independently, for which I also understand there is no actual inherent requirement for this. Kirchoff’s law can be fully satisfied without the incident flux of photons absorbed being collisionally transferred to the N2 and O2 in the atmosphere, provided the translational energy of the massive particles, i.e. the N2 and O2, are in LTE with each other and the IR emission is equal to IR absorption.
Or at least I’ve yet to see a fully credible and logical refutation of this.
Of course, a collision of non-GHG molecule with a GHG molecule can trigger a photon emission from the collided GHG molecule, and seems to be the reason why the mainstream model works. But what is the actual mechanism by which an absorbed photon, whose energy is stored as internal vibration energy, can transfer this energy to the N2 and O2? This seems to just be arbitrarily assumed to happen in the same way it does in liquid or solid.
That should have said:
“……provided the translational energy of the massive particles, i.e. the N2, O2 [b]and GHGs[/b], are in LTE with each other and the IR emission is equal to IR absorption.”
RW,
Go away. If we told you all the mistakes you made in your above post, you still wouldn’t believe us. Maybe Frank will have the patience, but I don’t.
How about one simple question answered, and that is what is the actual mechanism by which an absorbed photon by a GHG molecule, whose energy is stored as internal vibration energy, transfers this energy to the N2 and O2 when it collides into them? No doubt when a photon is absorbed by a liquid or a solid this is what happens, but how is it established mechanistically to occur in a thin, internally radiating gas of only narrow band emission?
I mean it would be one thing if he gas was dense enough to emit broad band emission, but I understand it’s not (or least the mainstream view is the emission is only narrow band).
RW,
I am sympathetic to DeWitt’s response to your earlier comment, but since you ask a fairly specific question here, I will try to answer it.
You wrote: “what is the actual mechanism by which an absorbed photon by a GHG molecule, whose energy is stored as internal vibration energy, transfers this energy to the N2 and O2 when it collides into them?”
It is stored as vibrational and rotational energy. Both are transferred to N2 and O2 by collisions. The rotational energy is transferred easily but the vibrational transfer is slow (meaning that any given collision is very unlikely to transfer the energy) because a quantum of vibrational energy is typically large compared to thermal energy. These processes have been extensively studied in great detail by physical chemists for many decades. Atmospheric scientists merely draw upon that existing knowledge.
You wrote: :No doubt when a photon is absorbed by a liquid or a solid this is what happens”
There is no real difference between a condensed phase and a gas, except that energy transfers are much slower in a gas.
You wrote: “but how is it established mechanistically to occur in a thin, internally radiating gas of only narrow band emission?”
I don’t know what you mean by “internally radiating” or “narrow band” or what you think they have to do with the original question.
Mike,
“It is stored as vibrational and rotational energy. Both are transferred to N2 and O2 by collisions. The rotational energy is transferred easily but the vibrational transfer is slow (meaning that any given collision is very unlikely to transfer the energy) because a quantum of vibrational energy is typically large compared to thermal energy. These processes have been extensively studied in great detail by physical chemists for many decades. Atmospheric scientists merely draw upon that existing knowledge.”
How is it established that the absorbed photonic energy is stored as rotational energy? I understand that rotational energy would be fairly easily transferred via collisions, even in a thin gas. I understood the absorbed IR energy was mostly stored as internal vibration energy and not via rotational.
“You wrote: :No doubt when a photon is absorbed by a liquid or a solid this is what happens”
There is no real difference between a condensed phase and a gas, except that energy transfers are much slower in a gas.”
This seems to be the conventionally accepted, albeit somewhat loose and arbitrary wisdom.
“You wrote: “but how is it established mechanistically to occur in a thin, internally radiating gas of only narrow band emission?”
I don’t know what you mean by “internally radiating” or “narrow band” or what you think they have to do with the original question.
By internally radiating, I mean photons are being emitted all within the gas, i.e. there is no effective radiating surface or surfaces within the gas, and by narrow band I mean not broad band Planck emission. The atmosphere does not emit broad band Planck emission (unless from the water or ice in clouds), but only narrow band from GHG molecules. The N2 and O2, which make up the bulk of the atmosphere don’t actually emit IR (or least not any significant amount).
This is what is bizarre and doesn’t seem quite right with the idea that the absorbed photonic energy is transferred via collisions and the atmosphere subsequently radiates based on its temperature in effectively the same way a dense body, i.e. a liquid or a solid, emits broadband Planck emission from its outer surface (based on its kinetic energy within). It seems if this was what was happening the emission from the atmosphere would (or should) be broad band Plank emission and not narrow band emission.
LTE arises in a liquid or solid because all incident energy, vibrational, rotational, photonic, kinetic, is equalized by collisions — and the body radiates based on its temperature (and emissivity), but is this also a requirement for LTE in a thin radiating gas that only emits narrow band?
https://en.wikipedia.org/wiki/Thermodynamic_equilibrium
““It is important to note that this local equilibrium may apply only to a certain subset of particles in the system. For example, LTE is usually applied only to massive particles. In a radiating gas, the photons being emitted and absorbed by the gas need not be in thermodynamic equilibrium with each other or with the massive particles of the gas in order for LTE to exist. In some cases, it is not considered necessary for free electrons to be in equilibrium with the much more massive atoms or molecules for LTE to exist.”
This at least suggests otherwise. As long as the massive particles, i.e. the GHGs and non-GHGs have fully equalized their linear kinetic energy via collisions with each other, the photons moving through the GHGs need not be transferred to the non-GHGs — for LTE to exist and Kirchoff’s law to be satisfied.
RW asks: “Then why is the emission/re-emission only narrow band and not broadband? The emission, except if from the water or ice in clouds, is claimed/established to be narrow band originating only from GHG molecules, yet it is also thought to be established that the atmosphere emits based on its temperature in the same way a dense body emits broad band Planck emission from its outer surface.”
You haven’t specified what you mean by narrow and broadband. I suspect that broadband means “blackbody intensity” or “blackbody spectrum” to you – the predictions of Planck’s Law. The first assumption in the standard derivation of Planck’s Law is that radiation is in EQUILIBRIUM with surrounding “quantized oscillators”. So “blackbody radiation” (which you appear to be calling broadband) is the radiation that is exists where absorption and emission have come into EQUILIBRIUM with the material photons are passing through. Originally, heated black cavities with a pinhole to sample the radiation inside were used to create such an equilibrium.
Radiation traveling through solids and liquids usually interacts strongly enough that absorption and emission have come into equilibrium by the time the radiation leaves the surface of the material. (Some is reflected at the surface, creating emissivity.) The absorption cross-section of any material is a measure of the strength of the interaction between radiation and that material – how fast (or short a distance) it comes into equilibrium. The heterogeneous intramolecular interactions between vibrating and moving molecules in solids and liquids often broaden energy levels into a continuum so that the radiation leaving the material has blackbody intensity at all wavelengths.
Gases are less dense and the energy levels of individual molecules are often not perturbed by the behavior of neighboring molecules. Thus gases emit as a series of lines whose width is controlled by temperature (Doppler broadening) and pressure (broadening caused by energy levels being perturbed by nearby molecules). This may be what you call “narrow band” emission.
Radiation traveling through the atmosphere often does not come into equilibrium with the atmosphere. The blackbody radiation by the surface of the earth emitted at wavelengths of the “atmospheric” window doesn’t interact strongly with gases in the atmosphere (small cross-section), so it passes through without its intensity changing appreciably. At the strongest absorption line for CO2, the average photon emitted by the surface travels only 1 m before it is absorbed and replaced by a photon emitted by a photon emitted by a CO2. Equilibrium exists at this wavelength. At 10 kilometers, the temperature is about 220 K and radiation with blackbody intensity (in equilibrium with its surroundings) is less intense than near the surface. At a high enough altitude, the density of CO2 drops enough so that absorption and emission are no longer in equilibrium with the surroundings.
Blackbody radiation is not fundamental – it is a shortcut that applies only in some situations: If radiation has come into equilibrium with its surroundings, it will have the intensity given by Planck’s Law. If not, you need to take into account the details of how radiation interacts with and is changed by its surroundings. Fundamentally, the rate of emission and absorption depends on Einstein coefficients, which can be used to calculate an absorption cross-section when molecules are in local thermodynamic equilibrium. In that case, the Schwarzschild equation can be used to calculate how radiation changes as it passes through molecules that absorb and emit radiation. That equation predicts that emission depends on temperature, the density of the gas and its absorption cross-section, but not the intensity of the radiation passing through the gas.
FWIW, according to Petty’s book (p 126), Kirckhoff’s Law applies only when local thermodynamic equilibrium exists. Apparently it doesn’t apply to situations where LTE doesn’t exist – lasers, LED’s, fluorescent light build, our atmosphere above about 100 km.
RW,
” I understood the absorbed IR energy was mostly stored as internal vibration energy and not via rotational.”
I suppose I was being needlessly picky. When IR is absorbed, there are changes in both vibrational and rotational energy. But the change in vibrational energy is much larger.
“This seems to be the conventionally accepted, albeit somewhat loose and arbitrary wisdom.”
Nothing loose or arbitrary about it. Well established by both experiment and theory.
“By internally radiating, I mean photons are being emitted all within the gas, i.e. there is no effective radiating surface”
I think that everything is what you call “internally radiating”. An “effective radiating surface” is just an approximation, useful when the penetration depth of radiation is small compared to some length of interest. Much more likely to be useful for solids or liquids, but not due to some fundamental difference.
“by narrow band I mean not broad band Planck emission. The atmosphere does not emit broad band Planck emission”
Sound like maybe by “broad band” you mean blackbody radiation? Sounds to me like a distinction without a difference here.
” It seems if this was what was happening the emission from the atmosphere would (or should) be broad band Plank emission and not narrow band emission.”
You seem to be assuming some fundamental connection between density and emission spectrum where there is none. Gases absorb at some wavelengths and not at others. So do liquids and solids. Water is opaque in much of the IR, but transparent in the visible. Germanium is just the opposite.
“LTE arises in a liquid or solid because all incident energy, vibrational, rotational, photonic, kinetic, is equalized by collisions — and the body radiates based on its temperature (and emissivity), but is this also a requirement for LTE in a thin radiating gas that only emits narrow band?”
Yes.
“It is important to note that this local equilibrium may apply only to a certain subset of particles in the system.”
Right. So the gas may be in LTE, while at the same time the radiation field is not in LTE. Maybe not realizing this is the source of your confusion?
“As long as the massive particles, i.e. the GHGs and non-GHGs have fully equalized their linear kinetic energy via collisions with each other, the photons moving through the GHGs need not be transferred to the non-GHGs — for LTE to exist and Kirchoff’s law to be satisfied.”
If the gases are in LTE, then any energy absorbed must be transferred. But the photons may not be in LTE; at some wavelengths where the gas is transparent they might be representative of a temperature other than that of the gas, since those photons are not interacting with the gas.
Mike,
“I suppose I was being needlessly picky. When IR is absorbed, there are changes in both vibrational and rotational energy. But the change in vibrational energy is much larger.”
How much larger and how is it mechanistically established that when a ‘charged’ GHG molecule collides with a non-GHG molecule (or another GHG molecule) that its internal vibration energy is transferred into the linear kinetic energy of the non-GHG molecule?
This is what has to happen if the absorbed photonic energy stored as internal vibration energy is transferred to the other gases in the atmosphere, i.e. mostly O2 and N2, right? Do we at least agree on this?
“Nothing loose or arbitrary about it. Well established by both experiment and theory.”
OK, what experiment specifically?
“Sound like maybe by “broad band” you mean blackbody radiation? Sounds to me like a distinction without a difference here.”
How is it a distinction without a difference? My point is the atmosphere doesn’t emit a Planck spectrum of energy, and in fact the N2 and O2 which make up the bulk of the constituents of the atmosphere don’t even emit radiation at all. I don’t understand why you don’t understand the difference between broad band emission and narrow band emission.
When a GHG molecules emit photons for any reason, they emit single wavelengths only, where as on the other hand when a dense body like a liquid or solid emits radiation the emission is multi-wavelength, i.e. it emits a broad band spectrum of wavelengths according to Planck’s law.
““LTE arises in a liquid or solid because all incident energy, vibrational, rotational, photonic, kinetic, is equalized by collisions — and the body radiates based on its temperature (and emissivity), but is this also a requirement for LTE in a thin radiating gas that only emits narrow band?”
Yes.”
Why? The scenario is if the absorbed photonic energy stored as internal vibration doesn’t have that energy transferred via collisions with non-GHG molecules (and other GHG molecules), increasing their linear kinetic energy, i.e. their thermal temperature, then what will eventually happen is the GHG molecule will keep accumulating absorbed photonic energy until the energy state gets high enough that the absorption of a photon will trigger or ‘excite’ an emission of another photon from that GHG molecule a short time after the photon is absorbed; and this will become the dominant way re-emission is triggered in the atmosphere. Meaning the absorbed photonic energy is primarily moving through the atmosphere, i.e. through the GHGs, without this energy being shared with the other constituents of the atmosphere.
This was proposed by someone here as an alternative to the mainstream model of atmospheric radiation. It was claimed this does not constitute non-LTE emission, because the massive particles are in LTE with each other, i.e. their linear kinetic energy is equalized among each other by collisions (with each other).
“If the gases are in LTE, then any energy absorbed must be transferred.”
Not according to that wiki definition quoted for the case of radiating gas. It seems the concept of LTE is not universally defined or established.
“But the photons may not be in LTE; at some wavelengths where the gas is transparent they might be representative of a temperature other than that of the gas, since those photons are not interacting with the gas.”
I’m only talking about absorbed photonic energy at some discrete layer where the condition of LTE exists — I’m not including photonic flux transmitted through such a discrete a layer.
RW,
I do not have the time or patience to try to unpack all of this. So just a couple of points that may be at the center of your confusion.
You wrote: “I don’t understand why you don’t understand the difference between broad band emission and narrow band emission.”
You appear to have in your mind some idea of what those terms mean. That idea does not seem to connect to the understanding of physicists and chemists. Not being able to read your mind, I can do no more than guess at what you mean.
“When a GHG molecules emit photons for any reason, they emit single wavelengths only, where as on the other hand when a dense body like a liquid or solid emits radiation the emission is multi-wavelength, i.e. it emits a broad band spectrum of wavelengths according to Planck’s law.”
No, photons are emitted one at a time and a particular photon has a single wavelength. That is true for both gases and condensed phases. The collection of all photons emitted will have a range of wavelengths, according to the properties of the substance. Again, true for both gases and condensed phases. The spectrum of emitted light might be close to blackbody over some ranges of wavelengths and very different over other ranges. Again, true for both gases and condensed phases.
Look at the CO2 spectra at: https://scienceofdoom.com/2009/11/28/co2-an-insignificant-trace-gas-part-one/ (the last two figures before “Measurements in the Lab”). The CO2 band centered at about 670/cm has a width of about +/- 100/cm. These are “wavenumbers” and energy is proportional to wavenumber. So the energy of the photons absorbed or emitted can range from about 15% less than the vibrational energy to about 15% greater. That is due to changes in rotational energy. The fundamental laws involved are no different for a gas than for a liquid or solid.
For a liquid, the bands are broader since the vibrational motions of the individual molecules interact, creating transitions that are not present in isolated molecules. But that is a difference in the detailed properties, not a difference in fundamental behavior.
Mike,
“No, photons are emitted one at a time and a particular photon has a single wavelength.”
Yes, of course. The point is a singular stream of photons of the same wavelength absorbed by a liquid or solid are not re-emitted as a single wavelength, but are re-emitted as a broadband Planck spectrum of energy.
The atmosphere does not absorb specific wavelengths and re-emit, i.e. convert them upon re-emission, into broad band Planck emission.
Mike,
As I said above, you’re wasting your time. RW doesn’t have a clue about molecular spectroscopy. That would be OK if he were interested in learning, but he’s not. He has his fixed ideas and won’t change them.
DeWitt – I agree.
RW – Since you obviously have no interest in learning anything, I will not respond further.
Mike and RW: According to the equipartition theorem, energy is distributed “equally” among all possible modes: translation in three directions, rotation about three axes, stretching and bending vibrations. QM modified this principle by quantizing rotational and vibrational states and populating them according to the Boltzmann distribution. The lowest vibrational states require too much energy at terrestrial temperature to gain an equal share of the energy. For example, there is a roughly 10:1 ratio of ground:1st excited vibrational states for CO2, while many rotational states are nearly equally populated. So there is just as much energy in each of three translational states (0.5*k_B*T) as in rotation about any axis, but less in vibration. (Rotation about the long axis of linear molecules like CO2 and diatomic molecules doesn’t count.) When gases get hot enough so that many vibrational states are occupied, they also get their “equal” share of the available energy (1*k_B*T per mode in this case). In the sun, electronic excited states are also populated. In the kinetic theory of gases, this “equipartition” among populated modes roughly explains the heat capacity of gases. When more modes are available to share the energy, the more energy that must be added to raise the temperature of the gas.
https://en.wikipedia.org/wiki/Equipartition_theorem
https://en.wikipedia.org/wiki/Heat_capacity#The_simple_case_of_the_monatomic_gas
Frank,
You are correct as to the equilibrium partitioning of energy. But the absorbed IR initially goes mainly into vibrations, then gets partitioned among all modes as a result of collisions.
RW wrote: The atmosphere does not absorb specific wavelengths and re-emit, i.e. convert them upon re-emission, into broad band Planck emission.
This is completely wrong. You have seen the absorption AND EMISSION spectra of various GHGs in many places. This is how these gases behave in the atmosphere.
No molecule or combination of molecules EMITS “blackbody radiation” – with the spectrum/intensity predicted by Planck’s Law. Blackbody radiation is what you get when emission AND ABSORPTION produce when they have come into EQUILIBRIUM with the medium radiation is traveling through. The change in radiation at a given wavelength (dI) as it passes an incremental distance (dz) through a GHG (or any other material in LTE) is given by the Schwarzschild eqn.
dI = emission – absorption
dI = n*o*B(lambda,T)*dz – n*o*I*dz = n*o*[B(lambda,T)-I]*dz
where n is the density of the GHG, o is the absorption cross-section for the GHG at that wavelength, B(lambda,T) is the Planck function and I is the intensity of radiation at that wavelength entering the dz increment of distance. When just as much radiation is absorbed as emitted by that incremental distance, dI = 0 and I = B(lambda,T). In other words, the radiation has blackbody intensity for that temperature. If I is greater than B(lambda,T), there will be more absorption than emission and the intensity of the radiation will decrease. (This is what causes the GHE.) If I is less than B(lambda,T), there will be more emission than absorption and the intensity of the radiation will decrease. The RATE of intensity change (with distance traveled) is proportional to the density of the GHG and the absorption cross-section (a measure of how strongly radiation of a given wavelength interacts with the GHG).
To see this more clearly, go to the MODTRAN calculator at:
http://climatemodels.uchicago.edu/modtran/
Tell MODTRAN to look up (towards empty space) from 70 km. For simplicity, make all of the GHGs zero except 400 ppm CO2. (I’ve also chosen the US Standard Atmosphere with no clouds or rain.) Looking up from 70 km, you see no radiation – there aren’t enough GHGs above this altitude to emit a significant amount of downward radiation. (above, n is effectively zero). You are seeing empty “black” space. Now look up from 30 km. There is enough CO2 above this altitude for some downward radiation emitted by CO2 to reach this altitude. It does not have a blackbody spectrum!!!. Now look up from 20 and 10 km. The intensity on the sides of the band increases, but the middle stays the same (appropriate for blackbody intensity for 220 K). The photons in the middle of the band emitted above 30 km are being absorbed and replaced by photons emitted between 10 and 30 km where the temperature averages about 220 degK. The outside of the band increases in intensity because o (cross-section) is smaller and the rate of change with distance traveled (from absorption and emission) is SLOWER. You can increase the CO2 concentration to 4000 ppm and see that more CO2 does not result in more radiation arriving in the CENTER of the band, only the edges. Absorption and emission are in equilibrium at this altitude in the center of the band. As you look up from lower and lower in the atmosphere you are seeing the net result of absorption and emission by denser CO2 that is warmer and warmer and therefore it has a higher blackbody intensity across most of the band. However, there is still negligible intensity at wavelengths outside 550-750 cm-1.
You can try the same thing with all atmospheric GHGs and see that you still don’t have a blackbody spectrum. If you compare to tropical atmosphere (with very high humidity and aggregates of water vapor molecules with new absorption bands – the “water vapor continuum”), you are closer to blackbody, but there are still wavelengths where the DLR does not have blackbody intensity – where emission and absorption haven’t come into equilibrium.
(You can also look down to see how OLR changes with altitude, but that is complicated since the radiation emitted by the surface does have a blackbody spectrum.)
When discussing LTE, you talk about massive particles (actually molecules) being in LTE and photons not always being in LTE. Where the density of a GHG is high (n) AND it interacts strongly with photons of a particular wavelength (o, the absorption cross-section is high), you can think of the photons at THAT WAVELENGTH as being in LTE – they are absorbed and emitted often enough to be in equilibrium. Photons at visible wavelengths are never in LTE with the atmosphere because the atmosphere is not hot enough for collisions to produce the excited electronic states that emit at visible wavelengths. For solids and liquids, such an equilibrium is usually present at all thermal infrared wavelengths and thus blackbody radiation leaves their surfaces (after some reflection at the surface). However, if you look at a thin enough slice of any solid material or a “thin film”, it will be semi-transparent and no longer emit like a blackbody. In that case you will need to integrate the Schwarzschild eqn., just like we do for the atmosphere.
Frank,
I think I understand most of what you’re trying to convey here. I understand the so-called Schwarzschild eqn. integrates the emission and absorption from the discrete layers to arrive at the net change in absorption, like from 2xCO2 (which is about 3.7 W/m^2, resulting in effectively an instantaneous -3.7 W/m^2 deficit at the TOA). This of course includes both up and down emission from each discrete layer from its layer through to the surface and from its layer through to the TOA.
What I’m discussing is independent of this. Even if what I’m putting forth is correct, the Schwarzchild eqn. and calculating atmospheric radiative transfer still holds just the same.
RW: The concept of LTE is clearly defined by wikipedia. It says that LTE includes all molecules – but not photons. Photons may or may not be in thermodynamic equilibrium with the molecules they are passing through. Absorption and emission of photons by gases in LTE don’t perturb LTE because collisions re-distribute the energy gained or lost among neighboring molecules faster than any other process occurs.
https://en.wikipedia.org/wiki/Thermodynamic_equilibrium#Local_and_global_equilibrium
LTE is important because it guarantees that we can correctly calculate the fraction of molecules found in an excited state using the Boltzmann distribution and the local temperature. This fraction is the first step in the derivation of Planck’s Law. When LTE doesn’t exist, Planck’s Law doesn’t apply and the Planck function B(lambda,T) in the Schwarzschild eqn won’t correctly predict emission. Fortunately, our atmosphere is in LTE at the altitudes that are relevant to climate (those that modulate heat flux into and out of the atmosphere near the surface.
Frank,
“The concept of LTE is clearly defined by wikipedia. It says that LTE includes all molecules – but not photons.”
But this is not the way LTE is used and applied in the mainstream view of atmospheric radiation. It includes and stipulates the GHG absorbed photonic energy is transferred, i.e. is distributed/shared equally via collisions, along with all other modes of energy, to all the other molecules (GHG and non-GHG).
Absorption and emission of photons by gases in LTE don’t perturb LTE because collisions re-distribute the energy gained or lost among neighboring molecules faster than any other process occurs.”
This is precisely what the wiki definition says is not a requirement for LTE in a radiating gas. The photons/electrons are moving through the GHGs need not be shared, i.e. have their energy transferred to the other gas molecules for LTE to still exist. That what is meant by ‘free electrons’. That is, the absorbed photons are moving through the GHG molecules and being re-emitted by the GHGs without their energy being shared with the other molecules, i.e. the absorbed energy is not thermalized. It says the only requirement is the massive particles, i.e. GHGs and non-GHGs, be in LTE with each other, i.e. have their kinetic energy equalized by collisions.
RW: You are confusing two things: a) molecules that are in local thermodynamic equilibrium and b) the possibility that photons MAY be in thermodynamic equilibrium with the gas they are passing through. (If you want to call this thermodynamic equilibrium a “local thermodynamic equilibrium”, in our atmosphere “local” would cover tens of meters of distance or longer. Temperature changes 0.65 K on the average over 100 m of vertical distance.)
For a) = molecules in LTE, equilibration of energy among all populated kinetic, rotational and vibrational states in a group of rapidly colliding molecules must be faster than any process (including absorption or emission) that brings energy into or out of the group. That means that the group has a defined temperature (proportional to the mean kinetic energy). That temperature plus the Boltzmann distribution determines how many molecules will be in excited states capable of emitting photons. It also tells us what states are available to absorb a photon. Molecules do not remain in LTE when photons are being absorbed faster than the resulting excited states can be collisional relaxed.
For b), when photons of a particular wavelength are in thermodynamic equilibrium with an (isothermal) layer of gas they are passing through (which is in LTE), the mean free path of the photons must be much shorter than the thickness of the layer. That ensures that absorption rate and emission rate will come into equilibrium as radiation passes through the layer. That means that radiation at that wavelength will have the intensity predicted by Planck’s Law.
There is no such thing as an “absorbed photon”. A molecule in an excited (or higher energy) state is created whenever a photon is absorbed by a molecule in a ground (or lower energy) state. The photon does not exist after it has been absorbed. The excited state behaves the same way whether it was created by a collision (the dominant mechanism when LTE exists) or by absorbing a photon.
Re-emission is negligible where the atmosphere is in LTE. Remember LTE means that the fraction of molecules in an excited state depends only on the local temperature, not how many photons are passing through creating excited states than can ‘re-emit” a photon. After an excited state is created by absorption, that molecule will be collisionally relaxed and excited “hundreds” of times before that molecule emits a photon. 99% of the time the term “re-emission” is used in a climate blog, the author is confused.
Kinetic energies are not equalized by collisions. A Boltzmann distribution of kinetic energy is always present at LTE.
Frank,
“For a) = molecules in LTE, equilibration of energy among all populated kinetic, rotational and vibrational states in a group of rapidly colliding molecules must be faster than any process (including absorption or emission) that brings energy into or out of the group. That means that the group has a defined temperature (proportional to the mean kinetic energy). That temperature plus the Boltzmann distribution determines how many molecules will be in excited states capable of emitting photons. It also tells us what states are available to absorb a photon”
Again, not according to the cited wiki definition in the case of a radiating gas.
“For b), when photons of a particular wavelength are in thermodynamic equilibrium with an (isothermal) layer of gas they are passing through (which is in LTE), the mean free path of the photons must be much shorter than the thickness of the layer. That ensures that absorption rate and emission rate will come into equilibrium as radiation passes through the layer.”
OK, but the requirement that rate of emission be equal to the rate of absorption doesn’t necessarily require that the absorbed photonic energy is shared by collisions with all the other molecules. This is whole point I’m getting at, and is — right of wrong — what the wiki definition is saying. In a radiating gas, LTE doesn’t require the absorbed photonic energy to be transferred and shared by collisions for LTE to still exist (and for Kirchoff’s law to be satisfied).
You guys are unbelievable. I’m not even claiming the mainstream model is necessarily wrong. The truth is I don’t know. You guys are treating science more like religious belief than science. Even if what I’ve proposed is correct, it’s hardly a complete re-write of the theory of atmospheric radiation — but more only a fine tuning of already established theory that may reveal some finer nuances of how radiation behaves and affects things as it travels through the atmosphere.
First of all, why is the difference between narrow band and broadband emission not understood in this context? This seems truly bizarre to me.
Let me put it as simply as possible. If a liquid or solid or is receiving monochromatic radiant energy from some source, it doesn’t re-emit monochromatic radiation, but instead re-emits a broadband Planck spectrum of radiation. I understand this can also happen in gas if the conditions are right, meaning there is no inherent requirement that a gas cannot emit broad band Planck emission, i.e. convert the absorption of narrow band radiation into broad band emission.
The point is the Earth’s atmosphere doesn’t do this. Narrow band absorbed is re-emitted as narrow band — not broad band. The bulk of the constituents of the atmosphere, i.e. N2 and O2, don’t even emit radiation (or least very, very little)
The mainstream view of the application of LTE is that all energy modes, including photonic, are equalized by collisions and the atmosphere radiates based on its temperature (though again, strangely, the re-emission is narrow band only) . Meaning the mainstream view considers the mechanism described here:
“It is important to note that this local equilibrium may apply only to a certain subset of particles in the system. For example, LTE is usually applied only to massive particles. In a radiating gas, the photons being emitted and absorbed by the gas need not be in thermodynamic equilibrium with each other or with the massive particles of the gas in order for LTE to exist. In some cases, it is not considered necessary for free electrons to be in equilibrium with the much more massive atoms or molecules for LTE to exist.”
as a means of initiating emission to constitute non-LTE emission, i.e. emission occurring where the gas is not in LTE. I know this, because this is what Grant Petty says. I have his book and asked him several questions. Another participant here claimed that the GHG molecules getting energized high enough where the absorption of a photon by a GHG molecule ‘excites’ the emission of another photon was not non-LTE emission.
A central tenant of the mainstream view of atmospheric radiation is the notion of LTE.
There is none so blind as he who will not see.
RW
The emission from a specific atom in an exited state follow quantum laws, according to Bohr. That is the explanation that we can`t get the broadband emission from atmospheric gases. No stuff has a blackbody emission. Blackbody is an aggregate of different atoms, I think. Even iron has its own spectrum. It is the jump of electrons from one orbit to another orbit that create the photon energy at distinct levels, as narrow bands for each type of atom. That makes it possible to identify different kind of matter in the universe.
RW wrote: “You guys are unbelievable. I’m not even claiming the mainstream model is necessarily wrong. The truth is I don’t know. You guys are treating science more like religious belief than science.”
In science, a good theory is capable of predicting what we observe. If two theories make different predictions about what will be observed when an experiment is run, one of those theories MUST BE WRONG.
You keep presenting ideas that differ from “mainstream theories – ideas that make different predictions. Either you are wrong, or the mainstream theory is wrong. The mainstream theory has survived a century of experimental tests. You are almost certainly wrong, because your ideas almost certainly will make predictions that don’t agree with some experiment. This is the science method, not religion.
Sometimes there are two different ideas or explanations that seem to make exactly the same predictions about some phenomena. “Wave/particle duality”, for example. However, Feynman’s book QED or online lectures will tell you that light is a massless particle that obeys a very strange theory (quantum electrodynamics) that produces wave-like phenomena such as interference. This lecture shows why we now believe in this theory.
You appear to want to believe that re-emission of absorbed photons is important in the lower atmosphere. Laboratory measurements show that the excited (vibrational) state of CO2 lasts about 1 second before it emits a photon. Laboratory experiments show that the same excited state is relaxed by collisions at atmospheric pressure in less than 1 microsecond. (Collisions occur about once a nanosecond, but not all collisions result in relaxation.) Continuing to believe that re-emission of absorbed photons is important in the lower atmosphere under these circumstance resembles religion.
“RW wrote: “You guys are unbelievable. I’m not even claiming the mainstream model is necessarily wrong. The truth is I don’t know. You guys are treating science more like religious belief than science.”
In science, a good theory is capable of predicting what we observe. If two theories make different predictions about what will be observed when an experiment is run, one of those theories MUST BE WRONG.”
Of course, and BTW I was not referring to you but to Dewitt and Mike who seemed to be basically just decreeing various things more as if they were religious doctrine rather than science.
“You keep presenting ideas that differ from “mainstream theories – ideas that make different predictions. Either you are wrong, or the mainstream theory is wrong. The mainstream theory has survived a century of experimental tests. You are almost certainly wrong, because your ideas almost certainly will make predictions that don’t agree with some experiment. This is the science method, not religion.”
It has never been claimed that the mainstream model doesn’t work, i.e. get the correct final answer and correctly predict observable spectrum (it does); it was instead only claimed that it wasn’t an accurate model of the more micro processes actually occurring on a quantum level; and that a more accurate model of these processes reveals some nuances about how radiation behaves and affects things in the atmosphere, such as its ability to act to ultimately further elevate the surface temperature in particular (but that itself is separate issue).
“You appear to want to believe that re-emission of absorbed photons is important in the lower atmosphere.”
Not necessarily. I’m only considering the possibility, because the physical mechanism(s) that supposedly preclude this are fuzzy when one asks for the specific quantum mechanism by which the absorbed photonic energy, which is stored primarily as internal vibration energy, is transferred to the other gas molecules in the same way it is in liquid or solid.
“Laboratory measurements show that the excited (vibrational) state of CO2 lasts about 1 second before it emits a photon. Laboratory experiments show that the same excited state is relaxed by collisions at atmospheric pressure in less than 1 microsecond. (Collisions occur about once a nanosecond, but not all collisions result in relaxation.), Continuing to believe that re-emission of absorbed photons is important in the lower atmosphere under these circumstance resembles religion.”
Assuming the absorbed photonic energy is shared, i.e. actually transferred by collisions to all the other molecules, then yes of course re-emission being primarily triggered by the absorption of photons doesn’t make sense and would not be occurring. 100% agree with this.
Can you at least agree that if the absorbed photonic energy is mostly not shared or transferred via collisions, that the GHG molecule will eventually (and very, very quickly) reach a high enough energy state where the absorption of a photon will trigger the emission of another photon and this would become the dominant way photon emission is triggered in the atmosphere? I think the point at which a molecule does this is called its ionization potential or ionization energy level.
Sorry, that 5th paragraph of mine was not supposed to be in italics. No way to edit.
RW wrote: “Can you at least agree that if the absorbed photonic energy is mostly not shared or transferred via collisions”
I think that we can all agree that if the laws of physics were different than they are, then things would behave differently than they do. So what?
“that the GHG molecule will eventually (and very, very quickly) reach a high enough energy state where the absorption of a photon will trigger the emission of another photon and this would become the dominant way photon emission is triggered in the atmosphere? I think the point at which a molecule does this is called its ionization potential or ionization energy level.”
Molecules are not being ionized in the atmosphere below something like 100 km, so obviously your reasoning has gone wrong somewhere. If the energy were not thermalized by collisions, then it would be emitted as radiation; Frank gives the lifetime as 1 second, which sounds about right to me for the 675/cm band.
At equilibrium about 8% of molecules will be in that doubly degenerate excited vibrational state, so if emission = absorption, a molecule would absorb a photon about once every 12 seconds. It would take roughly 150 photons to reach the ionization potential, so that would take about 30 minutes. An eternity for a molecule.
Mike,
He’s sucking you in again. Resistance is not useless or futile in this case.
That phrase, by the way, goes back at least to the old Doctor Who. It was quite popular with the Daleks and the Cybermen. The Vogon guard in Hitchhikers Guide to the Galaxy used it as well as Borg Picard in ST:TNG.
Mike,
“Molecules are not being ionized in the atmosphere below something like 100 km, so obviously your reasoning has gone wrong somewhere. If the energy were not thermalized by collisions, then it would be emitted as radiation; Frank gives the lifetime as 1 second, which sounds about right to me for the 675/cm band.”
For what, spontaneous emission? I’m not sure what you mean here. Can you further elaborate on these numbers?
“At equilibrium about 8% of molecules will be in that doubly degenerate excited vibrational state, so if emission = absorption, a molecule would absorb a photon about once every 12 seconds. It would take roughly 150 photons to reach the ionization potential, so that would take about 30 minutes. An eternity for a molecule.”
Again, you seem to be assuming the absorbed photonic energy, which is stored as internal vibration energy, will be transferred via collisions. The point is if it’s not, or there is no real mechanism for it to occur, i.e. be transferred to the other molecules, in the same way it does in a liquid or solid, its ionization potential will be reached super quickly, especially in heavily saturated bands. How long does it take for a CO2 molecule to absorb 150 15u photons?
RW: If you want to understand how translational, rotational and vibrational energy are exchanged by collisions, watch the molecular dynamics simulation here:
http://physics.weber.edu/schroeder/md/InteractiveMD.html
Chose the “Diatomic” Preset. Slow down the action by reducing the time step (or steps per frame). You can gradually shrink the box (compress the gas) to see a closer view. The faster and slower button increase and decrease the temperature.
Note that this is a 2-dimensional, not 3-dimensional, simulation. It involves classical mechanics (balls connected by a spring and experiencing a van der Waals force following Newtons Laws of motion), so the rotational and vibrational energies are NOT quantized. However, it illustrates clearly how translational, rotational, and vibrational energy are exchanged during collisions. If you turn up the temperature (I’m looking at 15 units), the vibrations are easier to see and you can see molecules with lots of vibrational energy “relaxed” by a collision with another molecule. The collisional relaxation of rapidly spinning/rotating molecules is more realistic because those energy levels are closely spaced, dozens of energy levels are populated, and quantum effects are less important. If vibrations were quantized, you would see most molecules in the slowest vibrating state and only a few vibrating twice as fast.
When you turn down the temperature, the molecules will aggregate into a liquid droplet and then a crystalline solid. There are many other things you can try with this teaching tool. However, it doesn’t include photons.
There are roughly a billion collisions at surface temperature and pressure per second. CO2 takes one second on the average for an excited state to emit a photon (from its Einstein A21 coefficient). However, there are 10^19 CO2 molecules/L and about 10% (10^18) are in the first excited vibrational state (that emits at about 15 um) at any one time. Perhaps 1 in 10^8 (10^10) manages to emit before it is relaxed by a collision. This is what we mean by LTE – energy is locally re-distributed through all possible modes by collisions much faster than energy enters or leaves the local: The number of vibrationally excited CO2 molecules depends only on the local temperature.
I think the key question that needs to be answered here is why does (or would) the absorption of monochromatic radiation in a liquid or solid get re-radiated as a broad band spectrum of radiation and in the atmosphere it does not — it’s re-radiated as monochromatic or narrow band. If the same fundamental physics (equal distribution by collisions of all energy modes) is said and claimed to occur in the gas as it does in a liquid or solid (in a state of LTE), what accounts for this difference?
No doubt in liquid or solid, the absorbed photonic energy is thermalized, i.e. shared and distributed equally by collisions. But if the same thing is occurring in the gas as claimed, what specific physics accounts for the difference?
BTW, I had thought for a long time that the mainstream view was that the absorbed photonic energy was thermalized by collisions, manifesting LTE, and re-radiated as broad band emission, but was surprised to learn from Grant Petty that the mainstream view was that it was re-radiated as narrow band.
Now mind you, if it were re-radiated as broad band, then it makes logical sense that the absorbed photonic energy is being thermalized in mechanistically the same fundamental way as it is in liquid or solid, but it’s not.
Something must account for this difference.
“Now mind you, if it were re-radiated as broad band, then it makes logical sense that the absorbed photonic energy is being thermalized in mechanistically the same fundamental way as it is in liquid or solid, but it’s not.”
BTW, this is clearly what happens when IR wavelengths are absorbed and re-radiated by the condensed water (or ice) that make up clouds. That is, the absorbed photonic energy of any one wavelength or combination of wavelengths is re-radiated as a broad band Planck spectrum based on the temperature of the H2O.
This is not what happens in the gases that make up all but the cloud portions of the atmosphere, which is nearly the whole of the entire atmosphere.
RW,
“I think the key question that needs to be answered here is why does (or would) the absorption of monochromatic radiation in a liquid or solid get re-radiated as a broad band spectrum of radiation and in the atmosphere it does not — it’s re-radiated as monochromatic or narrow band.”
No, the key question is why you think there is something different between the two cases. The processes in a gas are fundamentally the same as in a liquid or solid.
When a substance absorbs a photon, it goes from one quantum state to a different state that is higher in energy by an amount equal to the energy of the photon. One necessary condition for that to happen, is that there must be quantum states with the right difference in energy. So liquid water, for example, absorbs in the infrared but not in the visible.When a substance emits a photon, it changes from a higher energy quantum state to a lower energy state. It can only emit at the same wavelengths it absorbs and then only if the higher quantum state is occupied as a result of thermal energy. So liquid water has thermal emission in the IR, but not the visible.
There is no fundamental meaning to the terms “broad band” and “narrow band”, they are merely convenient labels. Some things have bands of available quantum states that cover a wider energy range than others. But the processes involved are unaffected by that. The thermal emission is spread out over whatever energies are allowed by the state of the substance.
But I think that Frank and I have both explained that already, so I am probably wasting my time by writing this.
“The thermal emission is spread out over whatever energies are allowed by the state of the substance.”
should be
“The thermal emission is spread out over whatever energies are allowed by the quantum states of the substance.”
Mike,
“No, the key question is why you think there is something different between the two cases. The processes in a gas are fundamentally the same as in a liquid or solid.”
Let’s take this one step at a time, because I don’t understand how or why you don’t understand the difference.
As an example, lets say we have a device that can emit a stream of IR photons of only one wavelength and we point the device toward a container with liquid water in it (in a state of thermal equilibrium) so the stream of photons is absorbed by the liquid water, causing an energy imbalance. That is, the water is receiving more (net) energy flux than its radiating away, causing the water to warm and radiate more. Is the additional radiation of the emitted by the water (from the warming of the water) all re-radiated in the same wavelength as the single wavelength emitting device? Or is it re-radiated as a broad band spectrum based on the increased temperature of the water according to Planck’s law?
“As an example, lets say we have a device that can emit a stream of IR photons of only one wavelength and we point the device toward a container with liquid water in it … is it re-radiated as a broad band spectrum based on the increased temperature of the water according to Planck’s law?”
It is re-radiated as a broad band spectrum, but not in accord with Planck’s law since it is not a black body. There will be a range of wavelengths for which the emission is pretty close to that of a black body, reduced by some emissivity less than unity.
Now replace the water with CO2 gas. As long as the photons are within an absorption band of CO2, the answer is exactly the same. But the range of emitted wavelengths will be narrower.
Mike,
“It is re-radiated as a broad band spectrum, but not in accord with Planck’s law since it is not a black body. There will be a range of wavelengths for which the emission is pretty close to that of a black body, reduced by some emissivity less than unity.”
OK, fair enough. I was using the rough approximation of black body, but you get the point.
So then in this case do you understand and/or agree that what is occurring is a process of narrow band absorption being converted into broad band (Planck) emission?
BTW, I’m not ignoring your second paragraph. I just want to take this one step at a time.
Mike M.
Your fundamental problem with RW is that he insists on using the term ‘re-radiate’ as if it meant something. There is no significant emission from a molecule that has just absorbed a photon, i.e. re-radiation. There is, in practice, absorption and there is emission. They are separate events. I guarantee you won’t convince him to change his mind on this.
Dewitt,
Yes, I agree the notion of ‘re-radiation’ is a misnomer and not what really happens. So I agree that in reality there is only absorption and emission occurring as separate events.
Sorry if I caused any confusion as a result of using the term ‘re-radiation’, but it’s still a moot point relative to what I’m getting at.
In the case of the liquid water above, what matters is the end result of the conversion between the absorbed electro magnetic (photonic) energy into the kinetic energy of molecules in motion, and then back into the electro magnetic state via radiant emission. This can only be achieved by broad band Planck emission. There is no mechanism by which a solid or liquid can absorb a singular wavelength stream of photons, covert that energy into the kinetic energy of molecules in motion, and subsequently convert that energy back into the same single wavelength stream of emission.
BTW, relative to broad band and narrow band emission, there is no difference relative to joules of energy. One joule of narrow band emission is equivalent to one joule of broad band emission. The only difference is how many photons it takes for the same number of joules.
RW: “So then in this case do you understand and/or agree that what is occurring is a process of narrow band absorption being converted into broad band (Planck) emission?”
Tentatively. I reserve the right to withdraw that if it turns out that you plan to shift the apparent meaning of the idiosyncratic terms that you have not carefully defined.
RW wrote: “There is no mechanism by which a solid or liquid can absorb a singular wavelength stream of photons, covert that energy into the kinetic energy of molecules in motion, and subsequently convert that energy back into the same single wavelength stream of emission.”
There is no mechanism to do that in gases, either.
Mike,
“Tentatively. I reserve the right to withdraw that if it turns out that you plan to shift the apparent meaning of the idiosyncratic terms that you have not carefully defined.”
What is not clear or has been ambiguously defined? I feel I’ve been quite clear, and avoiding any ambiguity was whole point of taking this step by step.
At any rate, I’ll proceed assuming you’re answer is yes.
Do you agree that in the case of GHGs absorbing IR energy in the atmosphere, the same process of narrow band absorption being converted into broad band emission, as it occurs in the example of the liquid water above, does not occur in the atmosphere? That is, no conversion of narrow band absorption into broad band emission occurs?
“RW wrote: “There is no mechanism by which a solid or liquid can absorb a singular wavelength stream of photons, covert that energy into the kinetic energy of molecules in motion, and subsequently convert that energy back into the same single wavelength stream of emission.”
There is no mechanism to do that in gases, either.”
Let me clarify this. There is no mechanism by which a solid or liquid can absorb an additional singular wavelength stream of photons, convert that energy into an increase in the kinetic energy of molecules in motion, and subsequently convert that energy back into an equal additional increase in emission of the same specific single wavelength initially absorbed.
I’m using the term ‘additionally’ only because we can’t start a absolute zero and in all likelihood any liquid (or solid) is already absorbing some radiation of multiple wavelengths.
RW,
You wrote: “Do you agree that in the case of GHGs absorbing IR energy in the atmosphere, the same process of narrow band absorption being converted into broad band emission, as it occurs in the example of the liquid water above, does not occur in the atmosphere? That is, no conversion of narrow band absorption into broad band emission occurs?”
I most certainly do not agree, since that is flat wrong. I have already said that several times. I think that Frank and DeWitt have also said that, although I am not going to search the thread to check. The problem is that you refuse to give a clear definition of what you mean by “narrow band”; perhaps because you refuse to think about what you mean. Frank ( or was it DeWitt?) carefully addressed that point, but you pay no attention.
Mike M. wrote: “I am probably wasting my time by writing this.”
Confirmed.
Mike,
“I most certainly do not agree, since that is flat wrong.”
Well not according to Grant Petty, at least as I understood him. As best as I recall, he agreed and stated the mainstream view is narrow band absorption is ‘re-emitted’ as narrow band only, i.e. there is no conversion of narrow band absorption into broad band emission (except if absorbed by the water or ice in clouds). I remember because I was quite surprised to hear it at the time. I understand the observed wavelength proportions passing out the TOA can’t be reconciled unless the ‘re-emission’ is narrow band only (because there is so much or too much energy passing into space in the saturated absorption bands, i.e. saturated absorption bands from the surface ‘looking up’ through the whole of the atmosphere).
BTW, I recall Petty having no difficulty at all with the difference between narrow band and broad band emission, so I don’t know what’s up with you guys.
At any rate, by now you ought to be able to see where I was going with this. If (it’s claimed) that the same physical processes are at work in a liquid or solid of absorbed radiation being converted into the kinetic energy of molecules in motion and subsequently converted back into EM radiation, and are the same in the atmosphere (as claimed), why is the conversion back into EM radiation not also the same? That is, also emitted as broad band (Planck) emission?
Basic logic dictates there must be a specific physical reason why, but no one seems to be able to provide it.
The mechanism proposed here by someone some time ago — right or wrong — does at least clearly explain the difference, as does the wiki definition of LTE I provided in the case of a radiating gas. That is, the absorbed energy is not actually thermalized, i.e. shared/distributed to the other gas molecules, with the streams of photons moving through the GHGs.
This no edit or preview feature is killing me. This should have said:
“At any rate, by now you ought to be able to see where I was going with this. If (it’s claimed) that the physical processes at work in a liquid or solid of absorbed radiation being converted into the kinetic energy of molecules in motion and subsequently converted back into EM radiation — are the same ones occurring in the atmosphere (as claimed), why is the conversion back into EM radiation not also the same? That is, not also emitted as broad band (Planck) emission?
Basic logic dictates there must be a specific physical reason for the difference, but no one seems to be able to provide it.
The mechanism proposed here by someone some time ago — right or wrong — does at least clearly explain the difference, as does the wiki definition of LTE I provided in the case of a radiating gas. That is, most of the absorbed energy is not actually thermalized, i.e. shared/distributed to the other gas molecules via collisions, with the streams of photons moving through the GHGs, i.e. being absorbed and re-emitted by GHGs.”
Even if this is correct, BTW, it’s hardly a complete re-write of established theory or something that would radically alter our understanding of atmospheric radiation; however — if correct, it may reveal some nuances about the dynamics of radiation passing through the atmosphere and its ability to act to ultimately further warm the surface, but that’s a separate issue.
Better said as: “…i.e. being absorbed and emitted by GHGs.”
“As best as I recall, he agreed and stated the mainstream view is narrow band absorption is ‘re-emitted’ as narrow band only”
Of course, this doesn’t mean that various absorbed wavelengths (of which there are many) are ‘re-emitted’ as a single narrow band only — it only means specific wavelength absorption generally isn’t converted into other, multiple wavelength emission. Or at least, this is how I understood it.
“The mechanism proposed here by someone some time ago — right or wrong — does at least clearly explain the difference, as does the wiki definition of LTE I provided in the case of a radiating gas. That is, the absorbed energy is not actually thermalized, i.e. shared/distributed to the other gas molecules, with the streams of photons moving through the GHGs.”
BTW, even if this is what is occurring, it doesn’t mean that collisions of GHG molecules into non-GHG molecules or other GHG molecules can’t or never trigger the emission of photons from GHGs — they can and surely do to some degree. It only means the dominant way photon emissions are triggered is not by collisions, but by the absorption of photons triggering emissions due to the GHGs being in high enough energy states where upon the absorption of photon an emission is triggered shortly thereafter from the same GHG molecule.
My understanding of why the mainstream model still works, i.e. is able to accurately predict the correct spectrum, is because collisions still trigger photon emissions from GHGs to some degree; however, the mainstream model only works when Kirchoff’s law is applied to each wavelength independently — something no inherently required for Kirchoff’s law to be satisfied. This essentially compensates for the error of not modeling the actual physics that are occurring. Or least, such is what’s claimed if what is proposed were actually occurring.
RW wrote: “I think the key question that needs to be answered here is why does (or would) the absorption of monochromatic radiation in a liquid or solid get re-radiated as a broad band spectrum of radiation and in the atmosphere it does not — it’s re-radiated as monochromatic or narrow band.”
Mike is right – only transitions between “allowed” states accompanies by absorption or emission of a photon occur. The difference is that In solids and liquids (and even gases at very high pressure), the energy levels of one molecule (or atom) are perturbed by neighboring molecules – which are constantly moving in liquids and shaking in solids. See the above link to a molecular dynamics simulation.
After looking at the absorption spectrum of numerous molecules in solution, I was shocked by a physical chemistry experiment where I recorded and interpreted the visible absorption spectrum of iodine vapor (i.e. the purple color). A forest of narrow lines in a spectral region where I had previously seen only one or two broad humps.
I should add that Petty did insist that this proposed mechanism would constitute non-LTE emission, and that the mainstream view or theory considers LTE to be equal distribution of all storage modes by collisions, included the energy of absorbed photons by GHGs. However when pressed about the seeming contradiction regarding narrow band absorption of GHGs yielding narrow band emission only from GHGs and that the other constituents of the atmosphere don’t even emit IR, he didn’t seem to really be able to provide an answer. I recall he seemed to more or less say, ‘we get the right answer with this model’.
RW: Petty discusses LTE on p 126-7 and 238-9. He never mentions thermodynamic equilibrium between gases and photons, just LTE involving molecules. Photons usually travel so far in gases between emission and absorption that they aren’t in LOCAL thermodynamic equilibrium.
When photons are in thermodynamic equilibrium with molecules, blackbody radiation is the result. (This is Planck’s Law.) Detectors in space show that our atmosphere doesn’t emit a blackbody spectrum of thermal radiation. Therefore photons in the atmosphere are NOT in thermodynamic equilibrium with the atmosphere. Scientists abandon ideas that have been demonstrated to be wrong by experiment. This is a science blog.
Frank,
And even if photons are in equilibrium at one wavelength, where absorption is strong, they likely won’t be in equilibrium at a wavelength where absorption is weak. At they certainly won’t be in equilibrium at a wavelength where absorption is zero.
Maybe not realizing that is RW’s problem?
Mike,
“And even if photons are in equilibrium at one wavelength, where absorption is strong, they likely won’t be in equilibrium at a wavelength where absorption is weak. At they certainly won’t be in equilibrium at a wavelength where absorption is zero.”
Well yes, of course the emitted photons themselves (wherever they are emitted from) as they are moving through the atmosphere are not in LTE with the gases they are passing through. I fail to see the significance of this relative to what’s being discussed.
The point is the energy of the absorbed photons by GHGs is supposed to be fully transferred/shared by collisions with all of the locally present gas molecules. It’s not the traveling photons themselves that are supposed to be shared by collisions with the other locally surrounding molecules, it’s their absorbed energy — which is transferred/stored as internal vibration energy in GHGs — that is supposed to be being shared and equalized by collisions (in the same way it does in liquid or solid).
Frank,
“There are roughly a billion collisions at surface temperature and pressure per second. CO2 takes one second on the average for an excited state to emit a photon (from its Einstein A21 coefficient).”
You’re talking about spontaneous emission, which is obviously way to slow. The mechanism isn’t claimed to be spontaneous emission.
It seems there are two things that really matter. 1) what speed are the molecules moving at and how often do they collide with the other molecules, and 2) does the collision, when it occurs, actually transfer/share the absorbed photonic energy, which is stored as internal vibration energy, with all of the other molecules.
Photons travel at the speed of light. How long then does it take a CO2 molecule to absorb about 150 15u photons? 150 photons is roughly how many it takes for a CO2 molecule to reach its ionization energy level.
Frank,
“Mike is right – only transitions between “allowed” states accompanies by absorption or emission of a photon occur.”
Well yes, but the question is what state or states are really ‘allowed’ which ones are not.
But you still haven’t answered the fundamental question. What specific physics accounts for the difference between that of liquid or solid and the gases of the atmosphere? That is, why is there no conversion from narrow band absorption into broadband emission in the atmosphere like there is in a liquid or solid? You can’t have it both ways by claiming the exact same mechanisms which re-distribute the energy of absorbed radiation by collisions in liquids and solids are also at work in the atmosphere, but the end conversion back to radiant emission is different in the atmosphere. Something must account for this difference, right? What is it?
RW,
“the exact same mechanisms which re-distribute the energy of absorbed radiation by collisions in liquids and solids are also at work in the atmosphere”
That is correct.
“the end conversion back to radiant emission is different in the atmosphere.”
That is wrong.
“Something must account for this difference, right? What is it?”
The reason the two claims disagree is that one gets the physics right, and the other gets the physics wrong. Frank and I have both been consistent in saying this. If you think otherwise, go back and read more carefully.
But something tells me that you will continue to insist, without evidence, that the wrong physics is right.
Mike,
You’re pretty much just decreeing what is wrong and what is right.
You still haven’t answered the question either. Well, actually you have, but only ‘tentatively’, which was strange. The physical processes in a liquid or solid of absorbed radiation, do and will convert narrow band absorption into broadband (Planck) emission, but the constituents of the atmosphere do not (even according to the mainstream theory of atmospheric radiation).
Some difference of physics must account for the different end result of the process of absorbed EM radiation in the atmosphere ultimately being converted back into EM radiation emission.
RW: “The physical processes in a liquid or solid of absorbed radiation, do and will convert narrow band absorption into broadband (Planck) emission, but the constituents of the atmosphere do not (even according to the mainstream theory of atmospheric radiation).”
What do you know abot this? Not very much I suppose. Just look at liquid water absorption spectra. How narrow or how broad?
I can’t believe you guys don’t see a discrepancy or at least aren’t somewhat open minded of the possibility, especially given the overwhelming majority of the constituents of the atmosphere, O2 and N2, pretty much don’t even emit radiation.
Do we at least agree on this point? That by and large, photons are only emitted from GHG molecules?
RW wrote: “You’re pretty much just decreeing what is wrong and what is right.”
No. We are trying to tell you what we have been taught or learned about the only theory that currently explains all aspects of the interaction between photons and molecules – quantum electrodynamics (QED). There is a wonderful non-technical lecture on this subject on the web by Feynman (and a short book “QED”) in which he says that even he doesn’t “understand” why light and matter behave the way they do. Nevertheless, QED makes extremely accurate predictions about how Nature behaves. Feynman says something like: “If you don’t like it, go to another universe, where the rules are simpler, more to your liking.”
And the rules of this blog are that conventional physics is accepted. We try to correctly understand its implications for our climate. Mike, DeWItt, SOD and others are quick to correct my mistakes and, all too often, I find out they are right when I check.
Scientists recognize the possibility that a new theory could someday replace QED. However, the new theory will need to make the same predictions as QED in areas where QED has been experimentally verified. So, in the unlikely event that a new theory comes along, chances are excellent that it will make the same predictions about radiation passing through out atmosphere. (For example, Newton’s theory of gravity has been replaced by Einstein’s general relativity, but Newton’s simpler theory is perfectly adequate a dealing with most phenomena we encounter
Frank,
“No. We are trying to tell you what we have been taught or learned about the only theory that currently explains all aspects of the interaction between photons and molecules – quantum electrodynamics (QED).”
This is a way overgeneralization.
“And the rules of this blog are that conventional physics is accepted.”
There’s nothing I’ve said here that contradicts basic established physics. This blog also makes it a point that it’s not about argument from authority, yet that seems to be largely what I’m getting here. Also, atmospheric physics, including how radiation interacts within the atmosphere, is applied physics with a lot of nuances.
“Scientists recognize the possibility that a new theory could someday replace QED. However, the new theory will need to make the same predictions as QED in areas where QED has been experimentally verified. So, in the unlikely event that a new theory comes along, chances are excellent that it will make the same predictions about radiation passing through out atmosphere.”
It’s never been claimed that current or mainstream theory of atmospheric radiation doesn’t work. Only that it may only be a valid equivalent model for the end result its needed to achieve thus far, and may not be an accurate model relative to the more micro-level processes actually occurring; and may reveal some nuances about how and to what degree radiation, in particular absorbed radiation by GHGs, is able to ultimately act to warm the surface temperature.
If the wiki definition of a variant of LTE in a radiating gas is closer to what’s actually happening in the atmosphere, it would mean that absorbed and emitted radiation within the atmosphere is mostly ‘bouncing’ between the GHG molecules without having the absorbed energy thermally shared with the other surrounding molecules. This is more akin to photons bouncing off a long series of half slivered mirrors rather than going into some significant thermal sink and then being emitted in the same way a dense body emits IR from its outer surface. Yet I note the latter seems to be the way people are conceptualizing in their minds what’s happening.
RW wrote: “But you still haven’t answered the fundamental question. What specific physics accounts for the difference between that of liquid or solid and the gases of the atmosphere?”
The same physics accounts for the emission of a blackbody spectrum by some materials and a forest of discrete lines by others (gases). If you understood the mathematical implications of the Schwarzschild eqn, you would recognize this. Basically, the equation says that the intensity of radiation passing through everything is modified by absorption and emission so as to approach blackbody intensity (a temperature-dependent equilibrium between photons and molecules). The RATE of approach to that equilibrium depends on the density of interacting molecules and the strength of their interaction with radiation (absorption coefficient) – which varies many orders of magnitude. 400 ppm of CO2 makes significant changers at 15 um in a few meters while 80% nitrogen produces negligible change.
If you compresses gases enough, raise they temperature high enough and have a path length long enough, you will observe a blackbody spectrum from gases.
If you condensed our atmosphere into a liquid with the same density as water, it would make an even layer 10 m thick. (A column of water 10 m and 1 sq inch in area weighs about 14.6 lbs.) The CO2 layer would be 4 mm thick. You can see visible light through 10 m of water. You can’t see through 4 mm of other substances. 4 mm of glass lets visible light into your car, but blocks thermal infrared. You can spread a few um of sunscreen (PABA) on your skin and block UV, but lotion without PABA doesn’t block UV. Gases SEEM to be different because they are less tangible than liquids or solids. The same rules apply to their interactions with radiation.
Frank,
“The same physics accounts for the emission of a blackbody spectrum by some materials and a forest of discrete lines by others (gases). If you understood the mathematical implications of the Schwarzschild eqn, you would recognize this. Basically, the equation says that the intensity of radiation passing through everything is modified by absorption and emission so as to approach blackbody intensity”
I fail to see how anything I’ve said or put forth contradicts this. A black body spectrum, i.e. broad band spectrum of emission resulting from a broad band of discrete absorption lines, does not necessarily involve the conversion of narrow band absorption into broad band (Planck) emission. In fact, the mainstream view (surprisingly) is this conversion does not happen. Narrow band absorbed is ‘re-emitted’ as narrow band only.
I still await an answer to the basic question put forth. Either you can explain the difference of physics between the two outcomes or you can’t, but basic logic dictates something must account for the difference. Physics is not magic pixie dust.
RW: If you had taken the time to understand the consequence of the Schwarzschild eqn and explored them studying DLR using MODTRAN (as I suggested above), you would recognize that you question has BEEN ANSWERED. If you don’t understand differential equations (lacking analytical solutions and requiring numerical integration) and if you won’t explore its implications with MODTRAN, the situation is hopeless.
Simple differential equations have FAMILIES of solutions that produce a wide variety of phenomena. Simple harmonic motion (sine curves) is the solution to this differential equation (for springs and pendulums):
m*(d2x/dt2) = -k*x
If I slight modify this equation to include friction and/or forcing, damped harmonic motion and resonance phenomena appear. If I attach one pendulum to the end of another, the system shows chaotic behavior. Another differential equation creates all of those atomic orbital (with spheres, two lobes, four lobes, eight lobes, …) for the probability of finding an electron near a proton – a rough explanation for a vast number of chemical phenomena. The 2LoT is a mathematical consequence of the behavior of large numbers of molecules following the laws of QM; entropy arises from disorder. Either do the mathematics yourself or accept the solutions provided by others. It makes little sense to argue about things you aren’t properly prepared to understand.
Best of all, try listening to Feynman (above) explaining how the properties of light are produced without a lot of mathematical formalism.
Frank,
I don’t see anything conflicting with the so-called Schwarzschild eqn., which is really just the radiative transfer differential equation that integrates the emission from the surface through to the TOA and from each discrete layer from its layer through to the TOA and from its layer through to the surface.
The same equation and rules apply in either case. In the imposed imbalance state of increased opacity, each layer absorbs a little more radiation and emits a little more IR upwards and downwards. I don’t see how this would be fundamentally changed in any way.
Frank,
“RW: If you had taken the time to understand the consequence of the Schwarzschild eqn and explored them studying DLR using MODTRAN (as I suggested above), you would recognize that you question has BEEN ANSWERED.”
I never claimed added CO2 doesn’t increase DLR. It does. I never claimed adding CO2, which increases DLR, won’t act to ultimately warm the surface above what it would otherwise be. I agree it should to some extent.
The reason why DLR increases when CO2 is added is due to the lapse rate, i.e. measured temperature and rate of emission decreases with height. If it were reversed, adding CO2 would actually enhance the radiative cooling of the system instead.
Again, I fail to see the significance of this relative to what’s being discussed and debated.
This might be more clearly stated as:
“In the imposed imbalance state of increased opacity, each layer absorbs a little more radiation from above and below and emits a little more IR upwards and downwards.”
The RT differential equation integrates the emission (after increased absorption) from the layers through to the surface and through to the TOA to calculate the IR flux change passing to the surface and the IR flux change passing out the TOA. For the increased opacity state, i.e. added GHGs, the result from the contribution of surface and all the layers is reduced IR passing out the TOA and more IR passing to the surface. The difference between the two is the net absorption increase. Due to the lapse rate, significantly more is reduced from passing into space than is increased passing to the surface; hence the enhanced radiative warming of the system as a result.
But again, I fail to see the significance of any of this.
RW: The online MODTRAN calculator linked above integrates the Schwarzschild eqn as radiation passes through the atmosphere. The simplest calculation involves looking up from different heights to see DLR emitted by one GHG (say CO2). If you look up from 70 km, negligible DLR can be detect. As you look up from lower and lower altitudes, there is more DLR. At each altitude, you can increase or decrease the amount of CO2 (change n in the Schwarzschild eqn). T is also changing with altitude. At some altitudes, increasing n doesn’t increase the amount of radiation reaching the detector and the band tracks one of the Boltzmann curves. If you understand the Schwarzschild eqn, you understand MODTRAN output. Then try the other GHGs. Then try looking down.
dI = n*o*[ B(lambda,T) – I ]*dz or dI/dz = n*o*[ B(lambda,T) – I ]
The term in brackets is negative when incoming radiation is more intensity than blackbody radiation for the local temperature. And positive when less. Therefore radiation at all wavelengths changes towards blackbody intensity for the local temperature as it travels through the medium. And when it doesn’t change (dI=0), the radiation has blackbody intensity. The product n*o controls rate of change in I. The bigger n or o, the faster the RATE (dI/dz) the intensity approaches that predicted for Planck’s Law. Consequently, if radiation travels far enough through a medium, it will reach blackbody intensity at all wavelengths (unless o is zero).
You asked over and over why solids and liquids emit blackbody radiation while gases emit a series of lines. Radiation that starts as a series of lines will become blackbody radiation if it travels far enough. It does so in solids and liquids – they often emit blackbody radiation. As some wavelengths and altitudes, you see blackbody intensity and others you see lines of very different intensity.
If you don’t, you didn’t actually use MODTRAN and are wasting our time.
Frank,
“You asked over and over why solids and liquids emit blackbody radiation while gases emit a series of lines.”
No, this is not what I asked. I was quite clear on what I was asking.
Radiation that starts as a series of lines will become blackbody radiation if it travels far enough.
Will become a black body equivalent spectrum. Again, I’ve never disputed this.
RW,
You may think it’s quite clear what you’re asking, but it isn’t clear to anyone else.
DeWitt wrote:”RW,
You may think it’s quite clear what you’re asking, but it isn’t clear to anyone else.”
Agreed. Which is why I tried the different approach below. RW is not going to say clearly what he means and generalities won’t get through to him, so maybe something very specific will work. I am curious to see how RW responds.
Mike M.
I suspect the problem is that because RW has a faulty understanding of the physics of emission and absorption of light, what he’s asking doesn’t make sense to someone who does.
I’ve been trying to think of an example. The closest I can get so far is asking why the greenhouse effect violates the Second Law. The answer is that it doesn’t. But try to explain that to someone with a faulty grasp of thermodynamics. SoD has written multiple articles on exactly that subject with probably little success as far as enlightenment goes.
Willis Eschenbach wrote a post at WUWT titled “The Steel Greenhouse” at WUWT in 2009. Most of the comments were from people insisting that it was wrong. IIRC, only one person who thought it was wrong changed his mind and admitted he had been wrong.
For the record, I do not think the GHE violates the 2nd law. Such claims are silly nonsense to me.
Mike, DeWitt and RW: I (angrily) struggled for a long time with the dilemma that increasing CO2 increased both absorption and emission. How could one determine which process dominated? SOD generously answered my questions, which is why I spend too much time trying to help others. The key breakthrough for me was seeing the proper mathematical formalism – the Schwarzschild eqn – for the first time, but that eqn clearly doesn’t turn on any light bulbs for RW. It might with the help of the online MODTRAN calculator.
Other questions raised by RW illustrate the folly of using optically thick layers of atmosphere in model calculations of the GHE – at least without making it absolutely clear that such models have little to do with the real atmosphere.
“I (angrily) struggled for a long time with the dilemma that increasing CO2 increased both absorption and emission. How could one determine which process dominated?”
Well, I never struggled with this, but I did struggle with understanding IR opacity through the whole mass of the atmosphere, in particular prior to the imposed imbalance state and subsequently from that state to the imposed imbalance state (like 2xCO2).
Frankly, I’m still not sure everyone here understands how IR opacity through the whole of the atmosphere is quantified and what that quantification actually means in physical terms. We seemed to go round and round in like 500+ exchanges in who knows how many threads with seemingly no resolution or agreement on what seems to me to be the most basic fundamental thing to understand first.
“SOD generously answered my questions, which is why I spend too much time trying to help others. The key breakthrough for me was seeing the proper mathematical formalism – the Schwarzschild eqn – for the first time, but that eqn clearly doesn’t turn on any light bulbs for RW. It might with the help of the online MODTRAN calculator.”
Again, I fail to see how what I proposed (or really what someone else proposed here at one point) conflicts with the RT differential equation.
Other questions raised by RW illustrate the folly of using optically thick layers of atmosphere in model calculations of the GHE – at least without making it absolutely clear that such models have little to do with the real atmosphere.
I’m not sure what you mean here. Yes, the atmosphere essentially consists of an infinite number of finite layers, but when modeled as a series of discrete layers for radiative transfer calculations — those layers are supposed to be macroscopically equivalent to the whole.
Frank,
You use optically thick layers in calculations because at some point, the difference between the results with a large number of optically thin layers is not significantly different from the results with fewer, optically thick layers and it takes a lot less time to calculate with fewer layers.
Dewitt,
“the difference between the results with a large number of optically thin layers is not significantly different from the results with fewer…”
Yes, this is my understanding as well.
RW,
Let’s try a different approach.
Imagine doing the following experiment. Although something close to what I describe here could actually be constructed, I would guess that the actual results might be too small to measure, so we will have to regard this as a thought experiment. But more sophisticated experiments confirm all the basic processes that occur here.
The apparatus is a perfectly insulated enclosure containing dry air (including 400 ppmv CO2, but no other IR active gases) at one atmosphere pressure, a system of mirrors, an electric resistance heater, an instrument to measure temperature, and several small windows transparent in both the visible and IR. I pass a He-Ne laser beam (red light) into one window and use it to adjust the mirrors so that the laser beam passes though the gas many times before exiting through another window. Light exiting a third window is collected and passed into a spectrometer to measure the IR spectrum emitted by the gas.
I turn on the heater, wait for the temperature to stabilize, and record the temperature and IR emission spectrum. The emission is mainly in the 15 micron band since that is the band most easily excited by thermal energy. I carefully calculate the emitted energy and find that it agrees with the energy input by the resistance heater.
Now I replace the He-Ne laser with a CO2 laser emitting on a line in the 9.4 micron band. The emission line is very narrow, just a small fraction of a wave number wide. With the laser on, I observe that the gas temperature increases and the IR emission also increases. Most of the increase is in the 15 micron band and is spread throughout the band (about 200/cm wide). I carefully calculate the emitted energy and find that the increase over the previous experiment agrees with the energy input by the laser.
I have turned a very narrow line into a band that is very broad compared to the laser line, but not so broad as what one might get from, say, liquid water. Not only that, but the emission is mostly in a different band than the absorption. That is a result of the energy being thermalized before being re-emitted.
This is what physics says will actually happen. It probably does not agree with you think will happen, but I can not really predict what you think will happen.
Mike,
“Although something close to what I describe here could actually be constructed, I would guess that the actual results might be too small to measure, so we will have to regard this as a thought experiment.”
Which is largely the problem. There are almost too many conditions and deductive assumptions you have laid out. But I have read your proposed hypothetical example, and I am thinking about it.
RW wrote above: “Frankly, I’m still not sure everyone here understands how IR opacity through the whole of the atmosphere is quantified.”
Now we’ve moved on to a new terminology that hasn’t been clearly defined: “opacity”. Technically, opacity is similar to absorption – but it isn’t meant to be used to discuss radiative transfer through a medium when emission is similar to absorption. Opacity can involve scattering (which can be added to the Schwarzschild eqn). According to Wikipedia, opacity is quantified by the mass attenuation (extinction) coefficient which is simply the absorption cross-section (o) with different units (and sometimes geometry – point source vs plane). So why don’t we stick to the absorption cross-section (o) to measure the strength of the interaction of radiation of a given wavelength with a molecule. The emission cross-section is equal to the absorption cross-section and traditionally calculated from the absorption spectrum.
RW wrote: “Again, I fail to see how what I proposed (or really what someone else proposed here at one point) conflicts with the RT differential equation.”
Our host wisely used to ask confused commenters to try to express their disagreement/idea in terms of an equation. I probably need only one equation, the Schwarzschild eqn, which can only be solved numerically at the MODTRAN online website. Please tell me what alternative equations you think should be used. Or tell us what altitudes and GHG concentrations MODTRAN gives a wrong or confusing answer for flux of DLR or OLR. Alternatively, if MODTRAN does give the correct answert, what other equations can produce the same result more simply.
I’m a little frustrated with all of this vagueness, so don’t expect a response to comments off this topic.
Frank,
“Now we’ve moved on to a new terminology that hasn’t been clearly defined: “opacity”. Technically, opacity is similar to absorption – but it isn’t meant to be used to discuss radiative transfer through a medium when emission is similar to absorption. Opacity can involve scattering (which can be added to the Schwarzschild eqn). According to Wikipedia, opacity is quantified by the mass attenuation (extinction) coefficient which is simply the absorption cross-section (o) with different units (and sometimes geometry – point source vs plane). So why don’t we stick to the absorption cross-section (o) to measure the strength of the interaction of radiation of a given wavelength with a molecule. The emission cross-section is equal to the absorption cross-section and traditionally calculated from the absorption spectrum.”
By opacity, I mean IR opacity through the whole mass of the atmosphere, i.e. from the surface ‘looking up’ all the way through to the TOA; and how this is ultimately quantified in W/m^2.
The total power absorbed by the atmosphere in a converged equilibrium state is based on scaled emitted wavelength intensity of the Planck flux emitted by the surface.
The actual surface emitted wavelengths absorbed are based on line of sight for the distance traveled from the surface through to the TOA and from the distance traveled from each subsequent layer above the surface from its layer through to the TOA.
Basically — based on probability density function, some of the directly surface emitted wavelengths see the constituents of the atmosphere and some don’t. Then the next layer’s emitted wavelengths see a little more of the surface emitted spectrum and so on and so forth. The emission from each subsequent layer above the surface further increases absorption ‘A’ and decreases transmittance ‘T’.
If we take a hypothetical example, where the surface is it at temperature where it emits 400 W/m^2 and the spectral ‘T’ is calculated to be 0.25, and absorption ‘A’ is 1-T or 0.75, it means 100 W/m^2 of the 400 W/m^2 (0.25*100 = 100) radiated from the surface is effectively transmitted straight through the atmosphere at the speed of light, and the difference of 300 (0.75*400 = 300) is absorbed by the atmosphere or attenuated from passing into space. The final values, i.e. the ‘T’ of 0.25 and ‘A’ of 0.75, are after all the emission from the surface and each subsequent layer above it have had their effect on ‘T’ and ‘A’.
We seemed to go round and round it what were like 500+ exchanges in who knows how many threads, where this seemed to never be understood and/or agreed with. To me, this is the most basic conceptual thing to understand first before you get into the imposed imbalance state where the downward emission from each layer through to the surface is included.
Repost with edit:
Frank,
“Now we’ve moved on to a new terminology that hasn’t been clearly defined: “opacity”. Technically, opacity is similar to absorption – but it isn’t meant to be used to discuss radiative transfer through a medium when emission is similar to absorption. Opacity can involve scattering (which can be added to the Schwarzschild eqn). According to Wikipedia, opacity is quantified by the mass attenuation (extinction) coefficient which is simply the absorption cross-section (o) with different units (and sometimes geometry – point source vs plane). So why don’t we stick to the absorption cross-section (o) to measure the strength of the interaction of radiation of a given wavelength with a molecule. The emission cross-section is equal to the absorption cross-section and traditionally calculated from the absorption spectrum.”
By opacity, I mean IR opacity through the whole mass of the atmosphere, i.e. from the surface ‘looking up’ all the way through to the TOA; and how this is ultimately quantified in W/m^2.
The total power absorbed by the atmosphere in a converged equilibrium state is based on scaled emitted wavelength intensity of the Planck flux emitted by the surface.
The actual surface emitted wavelengths absorbed are based on line of sight for the distance traveled from the surface through to the TOA and from the distance traveled from each subsequent layer above the surface from its layer through to the TOA.
Basically — based on probability density function, some of the directly surface emitted wavelengths see the constituents of the atmosphere and some don’t. Then the next layer’s emitted wavelengths see a little more of the surface emitted spectrum and so on and so forth. The emission from each subsequent layer above the surface further increases absorption ‘A’ and decreases transmittance ‘T’.
If we take a hypothetical example, where the surface is it at temperature where it emits 400 W/m^2 and the spectral ‘T’ is calculated to be 0.25, and absorption ‘A’ is 1-T or 0.75, it means 100 W/m^2 of the 400 W/m^2 (0.25*100 = 100) radiated from the surface is effectively transmitted straight through the atmosphere at the speed of light, and the difference of 300 (0.75*400 = 300) is absorbed by the atmosphere or attenuated from passing into space. The final values, i.e. the ‘T’ of 0.25 and ‘A’ of 0.75, are after all the emission from the surface and each subsequent layer above it have had their effect on ‘T’ and ‘A’.
We seemed to go round and round in what were like 500+ exchanges in who knows how many threads, where this seemed to never be understood and/or agreed with. To me, this is the most basic conceptual thing to understand first before you get into the imposed imbalance state where the downward emission from each layer through to the surface is included.
“The total power absorbed by the atmosphere in a converged equilibrium state is based on scaled emitted wavelength intensity of the Planck flux emitted by the surface.”
I should have added: ….,which is itself of course associated with a specific power density emitted according to the S-B law (based on the surface’s temperature and emissivity, which is about 1).
I should add here that COE sets the limit or constraint that the total power absorbed by the atmosphere and transmitted through the whole of atmosphere cannot exceed or be less than the power radiated from the surface (in a converged equilibrium state).
This effectively makes the total absorption in W/m^2, in both a converged equilibrium state or an imposed imbalance sate (i.e. the net absorption change) to constitute an amount (or change in amount) of the power radiated by the surface which is absorbed.
The analogy is light passing through a semi-transparent medium in the steady-state in that simultaneously more light can’t be absorbed by the medium and transmitted through the whole of the medium than is being supplied into the medium in the first place, as that would violate COE (assuming the medium has no internal energy source). The difference is the atmosphere is effectively both supplied ‘light’ externally (from the surface) and internally re-emits absorbed ‘light’ at the same time, but it none the less can’t have more ‘light’, i.e. radiant energy, absorbed and transmitted out its opposite side (the TOA) than is being supplied in from the one side (the surface) at the same time, i.e. ‘instantaneously’ or in any one instant.
The other analogous difference is the atmosphere can only lose ‘light’, i.e. EM energy, out its opposite boundary (the TOA) as ‘light’, where as in the first analogous example, some of the absorbed light can be thermalized and lost from the medium by conduction and/or convection (assuming the medium is surrounded by matter). Since the emitted EM energy emitted from the surface, i.e. the sole source of the ‘light’ entering the medium (the atmosphere), and is a direct consequence of the net amount of energy input to the surface, i.e. actually added to the surface, at any one instant, there can’t be more ‘light’ transmitted through the whole of the atmosphere and absorbed by the atmosphere than is initially radiated in from the surface, as that would violate COE (in the steady-state).
The only real difference so far as the analogy using light is the frequency of the emitted photons, which of course are in the long wave infrared for the surface and atmosphere.
This:
“(assuming the medium has no internal energy source).”
should have been stated as:
(assuming the medium has no alternate internal or external energy source).
BTW, the end result of a RT calculation is a ultimately quantification in W/m^2, whether you’re quantifying total absorption or a net absorption change. Without first understanding what and how that ultimate quantification in W/m^2 is derived and what it means in physical terms, massive confusion is near certain to ensue. Hence why so many people apparently have (or have had) such enormous difficulty in understanding atmospheric radiative transfer.
It’s also important to understand that for the converged equilibrium state, net energy flow at the surface is zero. That is the transmission of IR to the surface from the atmosphere has already manifested its effect on the surface energy balance; hence why it’s not considered for opacity through the whole of the atmosphere for the steady-state condition.
It is considered for the imposed imbalance state of increased GHGs, because after absorption and subsequently emission increase within the atmosphere (due to the added GHGs), the transmission of IR from the atmosphere to the surface increases, causing the net energy flow at the surface to become positive, while also at the same time causing the transmission of surface IR through to the TOA be reduced; hence why the increased transmission to the surface has to be subtracted from the decreased surface transmission through the TOA to arrive at the net change in absorption.
Or at least, this is my understanding, which I think is correct.
Frank,
“Our host wisely used to ask confused commenters to try to express their disagreement/idea in terms of an equation. I probably need only one equation, the Schwarzschild eqn, which can only be solved numerically at the MODTRAN online website. Please tell me what alternative equations you think should be used.”
It’s never been claimed that the existing equations are invalid or different equations are required.
Remember, it was only claimed that the more micro quantum mechanisms at work were different, but that LTE still exists and Kirchoff’s law is still satisfied. If both of these are true, the same equations used hold and are valid just the same.
RW,
Eight consecutive posts may be a record even for you.
“Eight consecutive posts may be a record even for you.”
Maybe I over did it. I won’t post anymore on that point, but just wait and see if anyone responds.
RW wrote some equations here: “If we take a hypothetical example, where the surface is it at temperature where it emits 400 W/m^2 and the spectral ‘T’ is calculated to be 0.25, and absorption ‘A’ is 1-T or 0.75, it means 100 W/m^2 of the 400 W/m^2 (0.25*100 = 100) radiated from the surface is effectively transmitted straight through the atmosphere at the speed of light, and the difference of 300 (0.75*400 = 300) is absorbed by the atmosphere or attenuated from passing into space. The final values, i.e. the ‘T’ of 0.25 and ‘A’ of 0.75, are after all the emission from the surface and each subsequent layer above it have had their effect on ‘T’ and ‘A’.”
Discussing ONLY the absorption of thermal IR by an atmosphere that emits thermal IR is insane. Use the correct equation, which includes emission
dI = emission – absorption
dI = n*o*B(lambda,T)*dz – n*o*I*dz
You are only discussing the second term. If the temperature is low enough or the incoming radiation (I) is intense enough, the first term is negligible. This is the case in the laboratory, where I is produced by a tungsten filament at several thousand degK. This is not true in the atmosphere. Both terms are essential.
In the laboratory, n (and T) are constants, allowing the second term to be integrated to produce Beer’s Law. (z is called path length.) In the atmosphere, they are functions of altitude: n(z) and T(z). Even the absorption cross-section varies modestly with temperature and pressure and therefore altitude o(z). If you want reasonably answers about the atmosphere, there is no substitute for numerical integration – using MODTRAN. Since you never discuss MODTRAN results, you still have been there.
There are no shortcuts to the right answer. The best one can do to assume that radiation is in equilibrium with the atmosphere and therefore the atmosphere emits blackbody radiation – the optically thick slab atmosphere that DeWitt defends as a useful model in some circumstances. That model leaves us struck with the dilemma that 240 W/m2 of outward heat flux somehow passes through an isothermal atmosphere in the absence of a temperature gradient. In that case, however, you don’t need to consider absorption at all.
Frank,
“Discussing ONLY the absorption of thermal IR by an atmosphere that emits thermal IR is insane.”
Not to calculate the aggregate transparency of IR and the aggregate absorptivity of IR through the whole mass of the atmosphere in a converged state of equilibrium, i.e. prior to an imposed imbalance.
Again, Grant Petty had no difficulty with this concept so again I don’t know what’s up with you guys. He referred to the quantity as ‘the spectral transmittance evaluated at the temperature of the surface’ so maybe that’s how what I’m describing as ‘T’ is formally expressed in this field. The one individual who has done his own simulations from scratch just refers to ‘T’ as the transmittance of surface power, or just the fraction of the power radiated from the surface that passes straight through the atmosphere at the speed of light into space (the same as if the atmosphere wasn’t even there). Aggregate absorptivity ‘A’ is then just 1-T, and its quantification in W/m^2 is what is driving the GHE prior to an imposed imbalance (or is what is aggregate GHG ‘forcing’ prior to an imposed imbalance).
Remember, the CO2 induced warming theory is an *enhancement* of an already existing GHE effect. That is, the atmosphere is already largely opaque in the IR prior to adding GHGs or doubling CO2.
Do you at least agree with last point? If not, that may explain a lot.
RW wrote: “That is, the atmosphere is already largely opaque in the IR prior to adding GHGs or doubling CO2. Do you at least agree with last point? If not, that may explain a lot.”
No. The current opacity of the atmosphere at some wavelengths (“saturation”) is correctly handled by the Schwarzschild eqn and MODTRAN. Try increasing or decreasing CO2 concentrations and see what effect this has on DLR and OLR in MODTRAN. Make large changes (2X, 5X, even 10X) in CO2 so the effects are readily apparent. Under what conditions and at what altitudes do you think it’s predictions are wrong. Let’s stop being vague and get down to SPECIFICS.
(MODTRAN has some limitations in the stratosphere, but not in calculating instantaneous forcing.)
Frank wrote: “Let’s stop being vague and get down to SPECIFICS.”
But specifics would expose the flaws in RW’s vague “reasoning”. Can’t have that.
“RW wrote: “That is, the atmosphere is already largely opaque in the IR prior to adding GHGs or doubling CO2. Do you at least agree with last point? If not, that may explain a lot.”
No.”
Well, this certainly explains a lot.
Let me ask you this: Prior to doubling CO2 do the wavelengths emitted up by the surface (and the subsequent layers above it) all pass through the atmosphere into space? Clearly the answer is no, meaning the atmosphere is already largely opaque in the IR prior to adding GHGs. This is why we have a GHE in the first place. If the atmosphere was completely transparent in the IR to the wavelengths emitted from the surface, there would be no GHE.
Prior to changing GHG concentrations, the spectral transmittance ‘T’ quantifies the transparency of the wavelengths emitted from the surface through the whole of the atmosphere, and the spectral absorptivity ‘A’ quantifies the opacity of the wavelengths emitted from the surface through the whole of the atmosphere.
Any part of the emitted surface spectrum that is designated to be ‘saturated’ means those emitted wavelengths by the surface must be 100% absorbed by the constituents of the atmosphere, i.e. attenuated from passing into space.
“The current opacity of the atmosphere at some wavelengths (“saturation”) is correctly handled by the Schwarzschild eqn and MODTRAN. Try increasing or decreasing CO2 concentrations and see what effect this has on DLR and OLR in MODTRAN. Make large changes (2X, 5X, even 10X) in CO2 so the effects are readily apparent. Under what conditions and at what altitudes do you think it’s predictions are wrong. Let’s stop being vague and get down to SPECIFICS.”
Again, what you’re describing here is for the imposed imbalance state of added GHGs. In the prior state of equilibrium, there is no doubt a large DLR component from the layers of the atmosphere, but it’s not considered or factored in for IR opacity through the whole of the atmosphere because net energy flow at the surface is zero in the steady-state and the DLR has already manifested its effect on the surface energy balance.
When you add more GHGs, the constituents of the atmosphere become more opaque to passing through radiation, and as a result each layer absorbs a little more from above and below and subsequently emits a little more upwards and downwards. The Schwarzchild eqn. then integrates the newly increased emission from the layers from each layer through to the TOA and from each layer through to the surface to arrive at the net change in absorption. The result is there is now an increase in DLR to the surface and a decrease in OLR, where the decrease in OLR exceeds the increase in DLR (due to the lapse rate).
Frank,
In short, I have no reason to believe the MODTRAN results you’re referring to for changes in OLR and DLR (for changes in GHG concentrations) are wrong.
” Since you never discuss MODTRAN results, you still have been there.
There are no shortcuts to the right answer.”
I’m not claiming I can do the calculation in my head. I surely can’t. I’m just discussing the physical meaning of the end result of the calculation.
I understand it’s based on probability density functions of emitted wavelength, distance traveled to the TOA (and/or surface), GHG concentration profiles traveled through, and probably some other nuances like pressure. There is no question you need a computer to calculate the end result.
The key point here, whether it’s aggregate absorption (prior to an imposed imbalance) or a net absorption increase (for the imposed imbalance state of increased GHGs), the final calculation is in W/m^2. Without a foundational understanding from specifically what, where and why that quantification in W/m^2 is actually derived from and means in physical terms, it’s no wonder to me why this subject seems to cause massive confusion and/or is so difficult for people to grasp.
For me at least, it wasn’t until I understood this fundamental point that it all became clear. Though maybe I’m a minority so far as how I ultimately came to understand all of this, as everyone learns differently.
RW: “I understand [MODTRAN is or radiative transfer calculations are] based on probability density functions of emitted wavelength, distance traveled to the TOA (and/or surface).
No. Just the Schwarzschild eqn. The change in radiation depends on: n (GHG density, which does drop with pressure for well-mixed GHGs), o (absorption cross-section), the Planck function B(lambda,T) which varies with wavelength and temperature and the incoming radiation.
In technical terminology, absorption, transmission, and emission refer to power absorbed, transmitted and emitted. For the atmosphere, the fluxes (W/m2) more more useful terms Dimensionless ratio are also used to quantify these phenomena. Transmittance (T) is I/I_0. Absorptance (absorptivity) is 1-T. Emittance (emissivity) is I/B(lambda,T)
absorbed, transmitted and emitted. For the atmosphere, the fluxes (W/m2) more more useful terms Dimensionless ratio are also used to quantify these phenomena. Transmittance (T) is I/I_0. Absorptance (absorptivity) is 1-T. Emittance (emissivity) is I/B(lambda,T)
Unfortunately, the dimensionless terms are problematic when applied to our atmosphere: a) Surface OLR is 390 W/m2 and TOA OLR is 240 W/2. 61% “transmission”. In reality, only about 10 photons emitted by the surface escape directly to space. 90% photons reaching space are emitted by GHGs in the atmosphere. b) There is no DLR from space entering the atmosphere (0 W/m2), but 333 W/m2 of DLR reaches the surface. That is “infinite transmission”. c) For optically thick layers of atmosphere, emittance/emissivity is a constant. For optically thin layers of atmosphere, emissivity varies with the amount of GHG and its absorption cross-section. It is far safer and less confusing to discuss the power fluxes rather than confusing dimensionless ratios – whose denominator isn’t clearly understood by all readers. Please avoid the dimensionless terms if you want a reply from me.
The Schwarzschild eqn describes the change in power with distance traveled: dI/dz, though we traditionally write it in differential form.
Frank,
“The Schwarzschild eqn describes the change in power with distance traveled: dI/dz, though we traditionally write it in differential form.”
Yes, the change in infrared intensity at the surface and the TOA.
“Just the Schwarzschild eqn. The change in radiation depends on: n (GHG density, which does drop with pressure for well-mixed GHGs), o (absorption cross-section), the Planck function B(lambda,T) which varies with wavelength and temperature and the incoming radiation.
In technical terminology, absorption, transmission, and emission refer to power(W) absorbed, transmitted and emitted.”
Yes, I totally agree with all of this.
I went back and found some old comments from the individual who built his own atmospheric simulator to do his own radiative transfer calculations (using MODTRAN) which may shed some light:
“I should point out that when I calculate T using HITRAN line data on a standard atmosphere with average clouds, I model a multi-layer atmosphere, although again, it has equivalent characteristics, where the 4 layers I model are the layer between the surface and clouds, the layer containing clouds, the layer of the troposphere above clouds and a layer above the troposphere. Each layer is characterized by an average thickness, average GHG concentrations, average pressure and average temperature with 2 different models representing the layer containing the clouds as clear and cloudy conditions. Each of these uniform layers can also be modeled as a stack of thinner layers, but there is no need for this as averages for these kinds of monolithic equivalent layers can be readily ascertained.”
and
“My RT simulation simply calculates absorption, A, which is the fraction of surface emissions absorbed by the atmosphere and is the most basic calculation you can do using hitran line data. T is then just equal to 1 – A.”
and
“The T is the average, net fraction of the power emitted by the surface that reaches space which makes (1 – T), or A, the net average fraction of the power emitted by the surface that is captured by the atmosphere.
Its important to recognize that this is an equivalent model of a unified atmosphere which accounts for the average absorption by clouds and by GHG’s as if the atmosphere was a uniform substance which absorbs A*Ps, where Ps is the power emitted by the surface.
A is not empirical, but derived. Using round numbers, clouds absorb an average of 80% of surface emissions, while GHG’s absorb about 50%, for an average cloud coverage of 66%, we can calculate the average A as the clear sky absorption by GHG’s plus the fraction absorbed by clouds plus the fraction absorbed by GHG’s that passes through the clouds. Note that GHG absorption between the surface and clouds is combined with cloud absorption as the ‘cloud’ relative to how space sees surface emissions includes whatever happens in the gap between the surface and the cloud.
A = (1-.66)*.5 + (.66*.8) + .66*(1-.8)*.5 = 0.764, which is less than 0.7 % different from the .759 claimed.
The actual measured values from ISCCP data and line by line simulations are an average cloud coverage of 66.4%, an average cloud emissivity of 0.733 and clear sky absorption of about 54%
A = (1-.664)*.54 + (.664*.731) + .664*(1-.731)*.54 = 0.763 which is a little closer than the rough estimate, moreover; the ozone absorption seems high, so if I backed the clear sky absorption down to 53%, A becomes 0.758, which is nearly exact. Note that the error is about +/- 5% which results in a range of 0.763 +/- 0.038, which puts the error bars from .725 < A < .801, where the 0.759 I claim is more than close enough to the center of the range to be the actual 'equivalent' value."
Again, he did confirm that what he refers to as 'T' can also be referred to as the 'spectral transmittance (evaluated at the temperature of the surface)'. I don't know why people here do not know this or are not able to understand it.
*Note his calculation of about 80% absorption for clouds, i.e. the so-called 'cloudy sky', includes partly cloudy conditions (based on the ISCCP data). If it didn't, it would be virtually 100% absorption as most clouds are virtual broad band absorbers in the LWIR.
Now, whether he’s ultimately doing these simulations correctly and accurately — I don’t know. With his simulation, he does calculate 3.6 W/m^2 of net absorption increase for 2xCO2, which is right smack in line with what everyone else in the field is getting. His global average ‘T’ of around 0.24 also seems reasonable since the transparent region of the absorption spectrum, i.e. the so-called ‘atmospheric window’, is located in a very strong part of the emitted surface spectrum. But again, I haven’t done these simulations myself or compared his results to others. Though again, a range somewhere between 0.23-0.25 makes logical sense.
Frank,
“Discussing ONLY the absorption of thermal IR by an atmosphere that emits thermal IR is insane. Use the correct equation, which includes emission
dI = emission – absorption
dI = n*o*B(lambda,T)*dz – n*o*I*dz
You are only discussing the second term.”
Again, I think the point you’re missing here is in the steady-state the ‘dl’ term you’re referring to doesn’t matter or need to be considered, because net energy flow at the surface is zero.
For the imposed imbalance state, after increased absorption and emission (from the added GHGs), the change in the ‘dl’ term causes the net energy flow at the surface to go above zero, i.e. become positive. This is why for the imposed imbalance state, the IR transmission from the layers through to the surface has to be included and subtracted from the increased IR absorption from the surface and layers through to the TOA, where as in the steady-state case it does not.
Moreover, because the rate of emission decreases with height, it makes sense that the IR absorption increase (after increased absorption and emission from the added GHGs) through to the TOA would exceed the increased transmission of IR through to the surface. If it were the other way around and the rate of emission increased with height, adding GHGs would actually result in a net transmission of IR to space increase.
Part of the issue is SoD’s tutorial doesn’t start with the steady-state case, and in some sense it’s kind of trivial since everyone is focused on incremental effects.
The question you should be asking yourself though is the 3.6 W/m^2 from 2xCO2 is designated as the ‘net absorption increase’, implying it’s an increase from a total amount that was already absorbed or captured prior to 2xCO2.
Frank,
Taking this a step further, using the numbers I provided as an example above, if in the steady-state case ‘T’ is calculated to be 0.25 and the surface is at a temperature where it radiates 400 W/m^2, it means absorption ‘A’ is equal to (1-T)*400 = 300 or 300 W/m^2. The 3.6 W/m^2 of net absorption increase from 2xCO2 is effectively an increase in total GHG absorption from 300 W/m^2 to 303.6 W/m^2, or an increase of 3.6 W/m^2 of the power radiated by the surface (400 W/m^2) which is absorbed by the atmosphere.
Radiative transfer calculations of changing IR opacity are fundamentally calculating changes in aggregate absorption ‘A’ and aggregate transmission ‘T’ of LWIR emitted by the surface, which is based on scaled wavelength intensity of the Planck flux emitted from the surface. The basic COE constraint (i.e. the atmosphere creates no energy of its own) sets the limit for the sum of the ‘T’ and ‘A’ components to equal the power radiated from the surface.
Another way you could state this using these same numbers is prior to doubling CO2, the atmosphere captures 300 W/m^2, and upon doubling CO2 the atmosphere captures an additional 3.6 W/m^2.
Is any of this making sense?
Frank,
“Discussing ONLY the absorption of thermal IR by an atmosphere that emits thermal IR is insane. Use the correct equation, which includes emission
dI = emission – absorption
dI = n*o*B(lambda,T)*dz – n*o*I*dz”
Using your dI term here, for the case of 2xCO2, the dI at the surface increases and the dI at the TOA decreases, right?
I don’t know what the actual numbers are, but if it were calculated to be -6 W/m^2 at the TOA and +2.4 W/m^2 at the surface, the net absorption increase would be +3.6 W/m^2, i.e. 6-2.4 = 3.6. The 3.6 W/m^2 is the so-called ‘net imbalance’ imposed on the system, or equivalent to a decrease in flux passing out the TOA of 3.6 W/m^2 (or a -3.6 W/m^2 TOA imbalance).
The point is what it really is quantifying is the net increase in opacity of the Planck flux emitted from the surface, which is itself (as you know) associated with a specific power density dictated by the S-B law. The point is the final calculation of the change in opacity in W/m^2, i.e. the so-called ‘net absorption change’ in this case is anchored to being an increase in the power density radiated from the surface which is absorbed.
Another important point is the calculated 3.6 W/m^2 is the so-called ‘instantaneous change’, or in other words, the final calculation is before the system has had any time to respond to the imposed imbalance.
DeWitt wrote: “Frank, You use optically thick layers in calculations because at some point, the difference between the results with a large number of optically thin layers is not significantly different from the results with fewer, optically thick layers and it takes a lot less time to calculate with fewer layers.”
Agreed, we use simpler models for situations when they are useful and more complicated models when they are not.
A optically thick slab model implies that the temperature of the atmosphere is determined by radiative equilibrium. Is that a useful approximation?
The most important phenomenon we want to explain is the GHE: Why does the earth emit an average of 390 W/m2 from its surface and only 240 W/m2 from the TOA? According to Taylor’s Elementary Climate Physics (i’m too lazy to repeat the calculations), a single optically thick slab atmosphere predicts an atmosphere at 242 K (Ts/2^(1/4)). That atmosphere would emit 195 W/m2, by mathematical necessity, half of 390 W/m2. If that is close enough for your purposes, fine. It does illustrate a reduction in TOA OLR. IMO, this exercise creates more confusion than understanding. (I could point to the comment that follows yours, but I hope neither of us deserve credit there.)
Only if the viscosity of the atmosphere were very high so that convection was insignificant. MODTRAN uses prescribed temperature profiles that include the contribution from convection. The lower layers of the troposphere emit quite a bit more radiation than they absorb from the surface and the layers below them.
The energy is supplied by convection. RW in the past has not agreed with this. He, at one point, seemed convinced that there was no net contribution to the energy balance from convection at all. I don’t know if he’s since modified his position, nor do I care.
The lapse rate of the troposphere with no convection would be much higher than it is now. In fact, that’s the major driving force for convective heat transfer in the troposphere. A lapse rate higher than the adiabatic rate is not stable in an atmosphere where convection is easy. The majority of the net heat flow from the surface to the atmosphere is by latent and sensible convection.
Dewitt,
“Only if the viscosity of the atmosphere were very high so that convection was insignificant. MODTRAN uses prescribed temperature profiles that include the contribution from convection. The lower layers of the troposphere emit quite a bit more radiation than they absorb from the surface and the layers below them.
The energy is supplied by convection. RW in the past has not agreed with this. He, at one point, seemed convinced that there was no net contribution to the energy balance from convection at all. I don’t know if he’s since modified his position, nor do I care.”
I’m not sure what you’re referring to here. Of course, convection is an energy input to the atmosphere and has a huge influence on the ultimate manifestation of the surface energy balance.
I believe all I’ve claimed is the surface radiates back up into the atmosphere the same amount of power as the net amount of power input to the surface, where the net amount power input to the surface is the *actual* amount of power added to or gained by the surface. Nothing more.
“Only if the viscosity of the atmosphere were very high so that convection was insignificant. MODTRAN uses prescribed temperature profiles that include the contribution from convection. The lower layers of the troposphere emit quite a bit more radiation than they absorb from the surface and the layers below them.”
I would say this makes sense since the troposphere is a turbulently mixed layer or section of the atmosphere, i.e radiant and non-radiant energy mix together and interact with one another.
DeWitt: You correctly summarize my best reasons for avoiding models with optically-thick layers. A radiative-convective model – like the one in MODTRAN – comes fairly close to reality for instantaneous changes in forcing. It would be better if the stratosphere temperature profile equilibrated changed with GHG concentrations.
Frank,
MODTRAN is NOT a radiative-convective model. It calculates emission and absorption from a specified temperature, humidity, concentration and pressure profile. If you change the surface temperature, the temperature profile up to 13km is offset by the surface offset but with the same slope. Nothing else changes. Well, the relative humidity will change, but the water vapor pressure is constant unless you specify that RH remains constant.
Frank wrote: “A radiative-convective model – like the one in MODTRAN – comes fairly close to reality for instantaneous changes in forcing.
DeWitt wrote: “MODTRAN is NOT a radiative-convective model.”
I really screwed up that thought. Any atmospheric model that doesn’t include both radiation and convection is too simple to be useful. I should have said that MODTRAN could be used to calculate radiative transfer alone step in a radiative-convective model. Or that MODTRAN is good for calculating radiative fluxes through a fixed atmospheric profile and therefore some aspects of radiative forcing.
That should be absorbed from the layers above and below them. Have you read Caballero’s Lecture Notes on Physical Meteorology?
http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.180.4815&rep=rep1&type=pdf
Caballero and Petty are my go to guys for atmospheric physics.
RW wrote: “Another way you could state this using these same numbers is prior to doubling CO2, the atmosphere captures 300 W/m^2, and upon doubling CO2 the atmosphere captures an additional 3.6 W/m^2. Is any of this making sense?”
Not really. Assuming nothing else changes, 3.7 W/m2 less LWR reaches space* after a doubling of CO2. And 1 W/m2 MORE DLR reaches the surface. (These quantities are not equal, because the atmosphere would be warming immediately after an instantaneous doubling of CO2.)
To my knowledge, however, the amount of energy “captured” by the atmosphere isn’t known. Unlike the surface, GHGs emitted most of their energy at the wavelengths that are most efficiently absorbed. At those wavelengths, twice as many photons are being both emitted and absorbed after a doubling of CO2. Those wavelengths don’t contribute much to the changes in OLR and DLR. Some photons only travel a few meters between emission and absorption. It is hard to count all of those. One needs to numerically integrate the Schwarzschild eqn over 1 meter layers (or less) to count all of the photons that are “captured” by the atmosphere. (DeWItt once tried to estimate this.)
The total number of photons being emitted and absorbed is irrelevant to climate science. The change in the OLR and DLR as they travel through the atmosphere with increasing GHGs is important. You calculate that change using the _____ eqn.
*Technically speaking, 3.7 W/m2 is the radiative forcing for doubling CO2 and it refers to the change in net LWR flux crossing the tropopause AND to the change in OLR reaching space after temperatures in stratosphere have reached a new equilibrium with the radiation passing through it after doubling CO2. These quantities are the same. The instantaneous change is LWR reaching space after doubling is slightly larger than 3.7 W/m2.
Frank,
“RW wrote: “Another way you could state this using these same numbers is prior to doubling CO2, the atmosphere captures 300 W/m^2, and upon doubling CO2 the atmosphere captures an additional 3.6 W/m^2. Is any of this making sense?”
Not really. Assuming nothing else changes, 3.7 W/m2 less LWR reaches space* after a doubling of CO2. And 1 W/m2 MORE DLR reaches the surface.”
Actually, it’s more than 3.7 W/m^2 reduced from passing through the TOA. Again, I don’t know what the actual numbers are. The point is at the same time the infrared intensity at the TOA is reduced it’s also increased somewhat at the surface. The so-called ‘net absorption increase’ is the difference between the two changed amounts of infrared intensity at the surface and the TOA.
The 3.7 W/m^2 is still ultimately a quantification of an incremental increase in GHG absorption from a prior total amount (determined by the spectral absorptivity ‘A’).
“The 3.7 W/m^2 is still ultimately a quantification of an incremental increase in GHG absorption from a prior total amount (determined by the spectral absorptivity ‘A’).”
Or the 3.7 W/m^2 is still effectively an incremental increase in GHG absorption of the power radiated from the surface. That is, the quantification of 3.7 W/m^2 is entirely that of upwelling IR which is additionally captured by the atmosphere, effectively reducing the IR flux passing out the TOA by 3.7 W/m^2.
Frank,
What are you ultimately measuring if not the net opacity of the surface emitted IR through to the TOA? Is the surface not the radiating plane from which opacity through the whole mass of the atmosphere is being considered?
Of course it is, which is why the final calculation, i.e. the net absorption change (both an increase or a decrease) is anchored to being an increase or decrease in the power radiated by the surface which is absorbed.
Otherwise what does the final calculation, i.e. the net absorption change, actually mean in physical terms? It has no clear meaning without being anchored to a specific location or clear boundary of where it starts from.
Frank,
The GHE is predicated on the fact that radiation emitted by the Earth’s surface cannot all pass through the atmosphere into space due to the presence of GHGs (and clouds), which absorb to a large degree the energy of the wavelengths radiated by the surface. Now some of the wavelengths emitted by the surface do pass through the whole of the atmosphere into space at the speed of light (the same as if the atmosphere wasn’t even there), but most of them don’t and are effectively ‘blocked’ from passing into space or are absorbed by the atmosphere.
Do you at least agree with this?
Frank,
“To my knowledge, however, the amount of energy “captured” by the atmosphere isn’t known.
It’s not only fully known, but is a requirement to satisfy Conservation of Energy. COE sets the limit that the total power captured by the atmosphere and transmitted through the whole of the atmosphere into space cannot exceed or be less than the power radiated from the surface (assuming the surface emissivity is 1), otherwise the atmosphere is either creating its own energy from nothing, or is destroying energy from within it.
Of course, this constraint only holds in the steady-state and from the steady-state to the so-called ‘instantaneous’ calculated imposed imbalance state. Once the system starts to adjust and respond to the imposed imbalance state, this constraint no longer holds.
For the purposes of RT simulation and the ultimate calculation of the ‘net absorption change’, it holds. It’s why you have to subtract the increased IR intensity at the surface from the decreased IR intensity at the TOA to arrive at the net increase in absorption, i.e. to anchor it to being quantified as a fraction *of the power radiated from the surface* which is additionally absorbed. Otherwise the atmosphere is not fully conserving all the energy, radiant and non-radiant, going into it and coming out of it.
In the example I used above, where I proposed a hypothetical calculation of -6 W/m^2 at the TOA and +2.4 W/m^2 at the surface for 2xCO2, resulting in a net absorption increase of 3.6 W/m^2 (6-2.4 = 3.6), the gross TOA flux imbalance is actually -6 W/m^2, but the net energy imbalance imposed on the whole system is only -3.7 W/m^2, resulting in net TOA imbalance of -3.7 W/m^2. That’s because relative to the power radiated from the surface, it’s only equivalent to +3.6 W/m^2 of incremental absorption since an additional 2.4 W/m^2 of IR is being put down the surface at the same time 6 W/m^2 is being reduced from passing out the TOA.
You do understand and agree that adding GHGs like CO2 to the atmosphere doesn’t add joules of energy to the atmosphere, right?
This should have been:
“In the example I used above, where I proposed a hypothetical calculation of -6 W/m^2 at the TOA and +2.4 W/m^2 at the surface for 2xCO2, resulting in a net absorption increase of 3.6 W/m^2 (6-2.4 = 3.6), the gross TOA flux imbalance is actually -6 W/m^2, but the net energy imbalance imposed on the whole system is only -3.6 W/m^2, resulting in net TOA imbalance of -3.6 W/m^2. That’s because relative to the power radiated from the surface, it’s only equivalent to +3.6 W/m^2 of incremental absorption since an additional 2.4 W/m^2 of IR is being put down the surface at the same time 6 W/m^2 is being reduced from passing out the TOA.”
RW wrote: “Again, I think the point you’re missing here is in the steady-state the ‘dl’ term [from the Schwarzschild eqn] you’re referring to doesn’t matter or need to be considered, because net energy flow at the surface is zero.
Heat enters and leaves the surface by radiation, convection of latent heat (water vapor) and conduction. When surface temperature is not changing (steady state, that doesn’t imply that entering and leaving radiation are equal – it means that the sum of all incoming heat equals the sum of all outgoing heat (and radiation is a form of heat.) The same is true at all altitudes below the tropopause: At steady state, radiation in and out are generally not equal, but all forms of heat transport in and out are equal. The heat flux by convention isn’t easy to calculate.
In the Schwarzschild eqn, dI is the incremental change in radiative flux that occurs on traveling an incremental distance dz upwards or downwards. Or if you prefer, delta_I is change in radiative flux that occurs on traveling an small distance delta_z upwards or downwards. (Technically dI refers to the spectral intensity (W/um) and needs to be integrated over all wavelengths to calculate the change in total power being transmitted the distance dz (delta_z). )
“RW wrote: “Again, I think the point you’re missing here is in the steady-state the ‘dl’ term [from the Schwarzschild eqn] you’re referring to doesn’t matter or need to be considered, because net energy flow at the surface is zero.
Heat enters and leaves the surface by radiation, convection of latent heat (water vapor) and conduction. When surface temperature is not changing (steady state, that doesn’t imply that entering and leaving radiation are equal”
Of course. I never said or implied that it is; however it does mean that any flux into the surface and away from the surface (radiant and non-radiant) in excess of the power radiated from the surface must be net zero across the surface/atmosphere boundary.
The point you seem to be missing is when the IR intensity increases at the surface (due to the added GHGs), the incoming flux to the surface is no longer equal to the outgoing flux, i.e. net energy flow is no longer net zero, but becomes positive.
“it means that the sum of all incoming heat equals the sum of all outgoing heat (and radiation is a form of heat.) The same is true at all altitudes below the tropopause: At steady state, radiation in and out are generally not equal, but all forms of heat transport in and out are equal. The heat flux by convention isn’t easy to calculate.”
Yes, totally agreed.
“In the Schwarzschild eqn, dI is the incremental change in radiative flux that occurs on traveling an incremental distance dz upwards or downwards. Or if you prefer, delta_I is change in radiative flux that occurs on traveling an small distance delta_z upwards or downwards. (Technically dI refers to the spectral intensity (W/um) and needs to be integrated over all wavelengths to calculate the change in total power being transmitted the distance dz (delta_z). )”
Yes, also agreed.
RW: To put it provocatively, the GHE is NOT caused by the opacity of the earth’s atmosphere in the thermal infrared.
What is the “net absorption” or “net absorption change” on the path from space to the surface of the earth – the path of DLR? There isn’t a logical answer to this question. It is equally absurd to discuss what happens in the opposite direction and only refer to absorption.
OLR decreases by 150 W/m2 as it rises through the atmosphere with 400 ppm of CO2. That change (the GHE) is calculated by summing up the dI predicted by the Schwarzschild eqn – for example by summing the change in radiative flux (dI) over 700 segments of 100 m length on that path from the surface to space. The total change is 150 W/m2. DLR increases by 333 W/m2 going the opposite direction through exact the same atmosphere! Isn’t the “opacity” of the atmosphere to thermal infrared the same in both directions? Isn’t the amount of absorption along each of those 700 segments exactly the same?
Why is there such a big difference between the change in OLR and DLR with altitude if opacity is the same in both directions?
Opacity can’t answer this question, because it is incapable of properly describing what happens in our atmosphere. An analogy: If I push against a wall, it doesn’t move. If I believe that the wall doesn’t move because of a natural tendency of walls not to move, my ability to understand simply physics is crippled. I need to recognize that the wall doesn’t move because it is generating an equal force opposing my push (by an imperceptible compression or bending of the wall). The “force” opposing absorption is emission and the two are near equilibrium in some cases. (At blackbody intensity, they are in equilibrium – a temperature-dependent equilibrium.) At some point, we all need to change from the concept that force causes motion to the concept that force causes acceleration and that acceleration can increase OR SLOW motion. More GHGs can slow OR increase heat transfer by radiation.
The change for 2X CO2 is +1 W/m2 in DLR and -3.7 W/m2 in OLR – about 0.3% and 2.5% of the total change in flux passing through the atmosphere. Forcing is the NET result of a massive increase in both absorption and emission. The net result of two large changes is difficult to predict using intuition, but it can sometimes be simple expressed mathematically. Which is bigger, B(lambda,T) or I (radiation entering a segment)?
dI = emission – absorption
dI = n*o*[B(lambda,T)]*dz – n*o*I*dz
dI = n*o*[ B(lambda,T) – I ]*dz
Go back to those 700 100m segments on the path between the space and the surface. Many photons are emitted and absorbed within one segment before they can travel to another. Even with 700 segments, we haven’t accounted for all of the absorption and emission. Photons emitted in the strongest CO2 band travel an average of 10 m near the surface before being absorbed. We would need to integrate over 70,000 1 meter segments to capture most of the absorption (and emission) that occurs. (One could use longer segments higher in the atmosphere.)
Why is there such a big difference between the change in OLR and DLR with altitude if opacity is the same in both directions? Answer: There is a temperature gradient in the atmosphere. That temperature gradient changes the emission term of the Schwarzschild eqn.
The GHE is caused by the opacity AND the temperature gradient of the atmosphere. No temperature gradient – No GHE.
In the troposphere, the temperature gradient is mostly the result of a pressure gradient, the ideal gas law, buoyancy-driven convection, and the absorption of SWR by the surface. Heat (including radiation) flows from hot to cold, so the LWR flux tries to eliminate that temperature gradient.
Frank,
Yes and no. You can only have an isothermal atmosphere with a surface temperature above the Cosmic Microwave Background temperature if the atmosphere is perfectly transparent. And that is probably only true for the one dimensional case unless the entire surface is also isothermal. If the atmosphere is not perfectly transparent, LWR will try to cool the atmosphere to the CMB temperature, not warm it to the surface temperature.
Frank,
“The change for 2X CO2 is +1 W/m2 in DLR and -3.7 W/m2 in OLR – about 0.3% and 2.5% of the total change in flux passing through the atmosphere.”
Where are you getting these numbers? I understand the net absorption increase is simply the difference between the reduced IR intensity at the TOA and the increased IR intensity at the surface. The net absorption increase is the net imbalance imposed on the system as a whole.
BTW, Grant Petty seemed to have no difficulty at all with the concept of ‘net absorption increase’, so again I don’t know what’s up with you guys. When I mentioned that the 3.6 W/m^2 for 2XCO2 was the net absorption increase for 2xCO2, he immediately understood exactly what that meant and what it was referring to. There was no ambiguity. The 3.6 W/m^2 is a quantification of incremental GHG absorption.
“Forcing is the NET result of a massive increase in both absorption and emission.”
Yes, I’ve said this myself many times. When you add GHGs, the atmosphere becomes more opaque to passing through IR, and as a result each layer absorbs a little more from above and below and subsequently emits a little more upwards and downwards. This is what ultimately causes the changes in IR intensity at the surface and the TOA to occur. The final calculated changes at the surface and the TOA are *after* each layer has absorbed a little more and subsequently emitted a little more due to the added GHGs.
“The GHE is caused by the opacity AND the temperature gradient of the atmosphere. No temperature gradient – No GHE.”
Agreed, but it’s initially driven by the surface’s inability to radiate freely to outer space.
“What is the “net absorption” or “net absorption change” on the path from space to the surface of the earth – the path of DLR? There isn’t a logical answer to this question. It is equally absurd to discuss what happens in the opposite direction and only refer to absorption.”
I still think you fundamentally don’t understand the difference between the steady-state calculation of IR opacity through the whole of the atmosphere and that of the imposed imbalance state when you add GHGs. SoD’s tutorial doesn’t start or cover the steady-state calculation of opacity first.
Both the steady-state case and the imposed imbalance state of added GHGs are anchored to the surface emitted power density, because it’s required to 1) satisfy COE, 2) have a clear physical meaning, and 3) for the calculation to apply for the whole of the atmosphere, i.e. from the surface all the way through the TOA.
If you don’t understand what Grant Petty refers to as the ‘spectral transmittance evaluated at the temperature of the surface’ then it’s no wonder you have no idea what I’m talking about. The spectral ‘T’ quantifies aggregate IR transparency prior to an imposed imbalance, and spectral ‘A’ quantifies the aggregate IR absorptivity. Each are through the whole of the atmosphere, i.e. from the surface all the way through the TOA. COE requires the sum of the two to not exceed or be less than the power density radiated from the surface.
It’s important to distinguish here that transmittance ‘T’ and absorptivity ‘A’ are not quantifications of power densities themselves, but fractions whose sum is equal to 1. None the less, they quantify the fraction of the power density radiated from the surface which is absorbed by the atmosphere and what fraction is transmitted through the whole of the atmosphere into space.
Frank,
If you don’t understand what’s in this post of mine from a few days ago:
…you’re unlikely to understand what I’m referring to and how it applies to the incremental increase in GHG absorption for the imposed imbalance state.
Frank,
The reason why the downward IR intensity at the surface is not factored in for the steady-state case (for calculating transmittance ‘T’) is because the system and surface has already come into equilibrium with whatever that intensity is. Thus only the upwelling emitted IR from the surface and the subsequent layers above it are factored in to calculate ‘T’.
When you add GHGs, the IR intensity at the surface — before the system has responded to the imposed imbalance — increases, resulting in the incoming total flux to exceed the total outgoing flux at the surface.
The surface radiates roughly 400 W/m^2, but the total flux entering and leaving the surface is about 100 W/m^2 more or about 500 W/m^2. Using the numbers from above, upon doubling CO2 the IR at the intensity at the surface has increased by 2.4 W/m^2 so the total incoming flux would now be 502.4 while the outgoing flux is still 500 W/m^2. At the opposite end at the TOA, an additional 6 W/m^2 of the power density radiated from the surface is now being captured or absorbed by the atmosphere, resulting in a gross TOA deficit or imbalance of -6 W/m^2. However, the net imbalance imposed on the whole of the system, i.e. from the surface all the way through the TOA, is only the difference between the two or -3.6 W/m^2 at the TOA, because adding GHGs to the atmosphere doesn’t add energy to the atmosphere. That is, the same amount of joules going into the atmosphere have to be coming out of the atmosphere, otherwise energy is being created or destroyed, which COE says can’t happen. Thus why the net imbalance imposed on the system is really only -3.6 W/m^2 at the TOA or an additional 3.6 W/m^2 of incremental GHG absorption (from the prior total GHG absorption of the surface power density quantified by the spectral absorptivity ‘A’, or about an increase from 300 W/m^2 to 303.6 W/m^2).
Frank,
Another part of the problem seems to be that the IPCC and/or climate science considers so-called ‘radiative forcing’ to be equal to zero prior to added GHGs, i.e. prior to an imposed imbalance state.
It is really not physically correct or logical to do this, because the warming theory from added GHGs is an *enhancement* of an already existing effect, i.e. an already existing radiatively induced GHE. Thus the fundamental mechanism which will be at work when more GHGs are added is already at work acting to elevate the surface temperature above what it would otherwise be. In other words, GHGs (and clouds) are already absorbing the wavelengths emitted by the surface prior to their being an imposed imbalance from adding more GHGs.
Without some sort of initial anchor, i.e. the reference point of the surface and how much of its emitted power and its emitted wavelengths that are being absorbed and transmitted through the whole of the atmosphere prior to an imposed imbalance, there isn’t a clear meaning of what the 3.6 W/m^2 of so-called ‘forcing’ from 2xCO2 is actually a measure of. That’s why the 3.6 W/m^2 is a quantification of incremental GHG absorption which is additionally captured by the atmosphere, or it’s equivalent to a reduction in the prior transmission from the surface to space of 3.6 W/m^2.
“Another part of the problem seems to be that the IPCC and/or climate science considers so-called ‘radiative forcing’ to be equal to zero prior to added GHGs, i.e. prior to an imposed imbalance state.
It is really not physically correct or logical to do this, because the warming theory from added GHGs is an *enhancement* of an already existing effect, i.e. an already existing radiatively induced GHE.”
Or simply the system, prior to adding GHGs, is already being radiatively forced to a very large degree.
This above should have been stated by me as:
“That’s why the 3.6 W/m^2 is a quantification of incremental surface emission which is additionally captured by the atmosphere, or it’s equivalent to a reduction of 3.6 W/m^2 in transmission from the surface to space.”
In my simplified example, ‘T’ was calculated to be 0.25 or 100 W/m^2. The 3.6 W/m^2 of incremental GHG absorption is equivalent to a reduction in the total transmission of 100 W/m^2 to 96.4 W/m^2 (100-3.6 = 96.4).
Frank,
“Why is there such a big difference between the change in OLR and DLR with altitude if opacity is the same in both directions?
Opacity can’t answer this question, because it is incapable of properly describing what happens in our atmosphere.”
I’m not sure what you’re referring to here. The reason why the OLR is reduced more at the TOA than the DLR is increased at the surface is due to the lapse rate. That is, the bottom layers are emitting a rates higher than the upper layers, and the IR emission of the bottom layers have a further distance to travel to the TOA (i.e. more atmosphere to pass through) than they do to the surface. On the other hand, the upper layers are emitting at rates lower than the lower layers, and the IR emission from the upper layers have a further distance to travel to the surface.
The net result is the reduced IR intensity at the TOA is greater than the increased IR intensity at the surface. If it were the other way around, i.e. the upper layers were emitting a rates higher than the lower layers, then adding GHGs would actually result in net transmission to space increase instead of a net absorption increase. That is, it would enhance the radiative cooling of the system rather than cause a warming imbalance.
In your reply, you don’t rely concept of opacity (or transmittance or absorptance) to explain the changes in OLR and DLR as it travels through the atmosphere. Nor do you use these term to explain the change in OLR and DLR that will occur after an instantaneous doubling of CO2. The GHE and enhance GHE is concerned with OLR, DLR and changes in these fluxes. Your preferred terms.
When thermal emission is insignificant, we talk in terms of transmission and absorption of radiation. When thermal emission is significant, we talk about radiative transfer: How the radiative flux along a path changes between two locations. Radiative transfer can result in an increase in flux. “Transmission” of radiation doesn’t address the possibility that the flux of radiation can be bigger further along a path; transmittance is always between 1 and 0.
Frank,
“In your reply, you don’t rely concept of opacity (or transmittance or absorptance) to explain the changes in OLR and DLR as it travels through the atmosphere. Nor do you use these term to explain the change in OLR and DLR that will occur after an instantaneous doubling of CO2.”
Each discrete layer or location in the atmosphere will likely have some fraction of its emission, both upwards and downwards, transmitted through to the surface or through to the TOA and some fraction absorbed somewhere along the path towards the surface or the TOA. The point is it’s the sum of the changes from all the layers from each layer from its layer through to the surface and from its layer through to the TOA — that changes the IR intensity at the surface and at the TOA.
“The GHE and enhance GHE is concerned with OLR, DLR and changes in these fluxes. Your preferred terms.”
I do not agree that the GHE is entirely driven by DLR if by DLR you specifically mean the amount of IR the atmosphere as a whole mass passes to the surface, though of course the enhanced GHE via adding additional GHGs must include the change in infrared intensity at the surface, but this is because 1), it’s necessary to satisfy COE and 2), its required to calculate the net reduction in IR transparency from the surface all the way through the TOA, i.e. the net reduction in the power radiated from the surface that is transmitted to space.
Frank wrote: “The GHE and enhance GHE are concerned with OLR, DLR and changes in these fluxes.”
RW wrote in classic straw-man manner: “I do not agree that the GHE is entirely driven by DLR if by DLR you specifically mean the amount of IR the atmosphere as a whole mass passes to the surface …”
DLR is featured prominently in many poor explanations for the GHE, but I didn’t make this mistake. I meant the GHE arises from both fluxes. If pushed to quantify the GHE, I would say it is the reduction in outgoing OLR from 390 W/m2 to 240 W/m2. Slowing down the rate of radiative cooling – insulation – allows the earth to be well above its blackbody equivalent temperature. However, that temperature is the net result of radiative and convective heat fluxes.
COE isn’t very useful. We don’t know how to predict how much heat flows upward via convection from first principle. (We know something about the lapse rate at which the atmosphere becomes unstable to convection, but not the heat that will be carried upward once it starts.) Even if we could quantify convection, an instantaneous doubling of CO2 will cause the atmosphere to begin warming and you will need to take changes in internal energy into account also.
Isn’t it nice to have a discussion about the GHE without using the unnecessary and misleading terms {a…ance, t….ance, opac…). Now if you could just say Schwarzschild and MODTRAN, you would have your answer your question from above:
“Where are you getting these numbers?” the changes in DLR and OLR upon doubling CO2. You can do it yourself:
http://climatemodels.uchicago.edu/modtran/
Frank,
“RW wrote in classic straw-man manner: “I do not agree that the GHE is entirely driven by DLR if by DLR you specifically mean the amount of IR the atmosphere as a whole mass passes to the surface …”
DLR is featured prominently in many poor explanations for the GHE, but I didn’t make this mistake. I meant the GHE arises from both fluxes. If pushed to quantify the GHE, I would say it is the reduction in outgoing OLR from 390 W/m2 to 240 W/m2. Slowing down the rate of radiative cooling – insulation – allows the earth to be well above its blackbody equivalent temperature. However, that temperature is the net result of radiative and convective heat fluxes.”
I would describe the GHE as being driven mechanistically by radiative resistance to outer space cooling by radiation (radiative resistance to radiation ultimately passed from the atmosphere into space), or this Wikipedia definition here:
“The greenhouse effect is a process by which thermal radiation from a planetary surface is absorbed by atmospheric greenhouse gases, and is re-radiated in all directions. Since part of this re-radiation is back towards the surface and the lower atmosphere, it results in an elevation of the average surface temperature above what it would be in the absence of the gases.[1][2]”
Of course, the final manifestation of the surface energy balance itself is the net result of all of the effects combined together, radiant and non-radiant. But the final thermodynamic path that manifests the surface energy balance is largely separate from the underlying driving mechanism itself.
“COE isn’t very useful. We don’t know how to predict how much heat flows upward via convection from first principle. (We know something about the lapse rate at which the atmosphere becomes unstable to convection, but not the heat that will be carried upward once it starts.) Even if we could quantify convection, an instantaneous doubling of CO2 will cause the atmosphere to begin warming and you will need to take changes in internal energy into account also.”
Yes, of course. I’ve never thought or claimed otherwise.
“Isn’t it nice to have a discussion about the GHE without using the unnecessary and misleading terms {a…ance, t….ance, opac…). Now if you could just say Schwarzschild and MODTRAN, you would have your answer your question from above:
“Where are you getting these numbers?” the changes in DLR and OLR upon doubling CO2. You can do it yourself:
http://climatemodels.uchicago.edu/modtran/“
I questioned only because you seemed to claim that the net change was not the difference between the reduced IR intensity at the TOA and the increased IR intensity at the surface.
BTW, even Gunnar Myhre, whose 1998 paper has long been considered to be the standard reference for the 3.6 W/m^2 figure, described the 3.6 W/m^2 (when I asked him) as being the ‘net change in absorption at the tropopause level’. He further said since the 3.6 W/m^2 includes the stratospheric adjustment, it can be considered the net change in absorption, i.e. the net increase in absorption, through to the TOA.
(*On side note, the so-called stratospheric adjustment is like less than 0.1 W/m^2, so why bother to even include it?)
I’d have to go back and find the email, but I recall he also said or confirmed that the 3.6 W/m^2 is the additional amount of the surface radiative power which is absorbed by the atmosphere.
I also asked him if he had, for the global average, a value for ‘the spectral transmittance evaluated at the temperature of the surface’, and he said he didn’t have it; but he clearly knew what it was.
“I would describe the GHE as being driven mechanistically by radiative resistance to outer space cooling by radiation (radiative resistance to radiation ultimately passed from the atmosphere into space), or this Wikipedia definition here:”
I would take this a step further and say that the GHE effect is driven by the upwelling IR emitted from the Earth’s surface (and the atmosphere) acting to cool the system and surface which is ‘blocked’ from passing into space, or absorbed by GHGs and clouds. Above all, the system must be making the radiative cooling push toward radiative balance at the TOA by continuously emitting IR up towards space, but in order to do this it has to push back the other way, because absorbed upwelling IR is re-radiated both up and down. The end result is of this process of radiative resistance to radiative cooling requires the lower atmosphere and ultimately the surface to be emitting at higher rates in order for the surface and the whole of the atmosphere to be passing the required 240 W/m2 into space. That is, the lower atmosphere and surface ultimately are required to emit a rates greater than the 240 W/m^2 being passed into space.
Frank,
We can estimate the latent heat transfer using global annual rainfall. They do it in KT97 and TFK09 Sensible heat transfer is a lot smaller.
Sensible heat transfer is then the difference between total energy in and radiative and latent energy out. In TFK09, latent heat transfer was 80W/m² and sensible transfer was 17W/m² for a total of 493W/m² out.
But RW, at least in the past, didn’t think that TFK09 and KT97 were correct because he didn’t believe in net convective heat transfer from the surface to the atmosphere.
I admire your patience, but it’s pointless. RW is in Bryan’s class. He’s a tr011.
“But RW, at least in the past, didn’t think that TFK09 and KT97 were correct because he didn’t believe in net convective heat transfer from the surface to the atmosphere.”
No, there is of course net convective flux from the surface to the atmosphere. What I said was the KT values of 80 W/m^2 and 17 W/m^2 are NOT the net, i.e. up minus down, but instead just the gross non-radiant fluxes leaving the surface. KT then has the total, i.e. 97 W/m^2, being offset entirely by radiation, depicting that the only way a joule can pass to the surface is by radiation, which is obviously wrong. There is some non-radiant flow from the atmosphere to the surface from the heat in precipitation (primarily), though of course the net non-radiant flow is still from the surface to the atmosphere. What it means KT’s amount of DLR (333 W/m^2) is likely too high and somewhere around 300 W/m^2 (or a little less even) is probably closer to reality.
As the blog owner I’m probably supposed to have some comment. I have absolutely no idea what RW is on about, and this is only matched by my complete lack of interest. Well, I have some idea what RW is on about due to his previous 2000 incomprehensible comments, but see my previous answers to his unsurpassable confusion.
I think the late, great, and sadly missed, Pekka summed it up well. I could look up his comment but it was, paraphrasing poorly, no one has a clue what you are on about RW. You invent terms and don’t explain them, you use terms that everyone else understands to mean one thing in a completely different way, etc, etc. Search for Pekka..
Cheers from LAX.
SOD: If you are referring to “radiation resistance”, that refers to the resistance to current flow in an antenna generated by the production of electromagnetic radiation. And I gather “converged equilibrium” is the state difficult simulations reach when they successfully approach equilibrium from different starting states. Scattering is a major contributor to the extraordinary opacity of RW’s comments.
I miss Pekka too. Hope your endeavors are prospering.
Frank, why is the notion of radiative resistance to radiative cooling not a clear concept?
The GHE effect — deemed to be a radiatively induced effect — is driven by the upwelling IR emitted from the Earth’s surface (and the atmosphere) acting to cool the system and surface which is ‘blocked’ from passing into space, or absorbed by GHGs and clouds; and re-radiated. Above all, the system must be making the radiative cooling push toward radiative balance with the Sun at the TOA by continuously emitting IR up towards space, but in order to do this it has to push back the other way, because absorbed upwelling IR is re-radiated both up and down.
The GHE effect is initiated by the surface’s inability to radiate freely to outer space.
All I mean by ‘converged equilibrium’ is a condition of steady-state.
Moreover, how can you have a net transmission to space decrease without a net absorption increase? These seem to be basic concepts, so it eludes me why you and apparently everyone here has no clue what I’m talking about.
Before you add GHGs or increase IR opacity, the surface and the whole of the atmosphere together are passing about 240 W/m^2 of IR into outer space, right? I assume it’s agreed that all of this 240 W/m^2 of IR can only be that which has been emitted up at the surface or somewhere in the atmosphere and able to pass uninhibited into outer space (i.e. without being re-absorbed somewhere along the path to the TOA). Yes? I certainly hope so.
Now, when you double CO2 or add GHGs to calculate a net opacity change, or really net increase in IR opacity, the outgoing IR flux at the TOA is considered to be reduced from about 240 W/m^2 to 236.3 W/m^2, i.e. by 3.7 W/m^2. The transmission of IR flux through the TOA is now reduced, right? How can this reduction, or -3.7 W/m^2 TOA imbalance, not be the result of an upwelling IR absorption increase? Of course, it has to be. Hence why it’s quantified as being a ‘net absorption increase’. The ‘net’ is important, because it’s after you have subtracted the increased IR transmission to the surface. Remember, all of this is before the system has responded to the imbalance, i.e. it’s ‘instantaneous’.
Just because you have to initially consider increased absorption (from above and below) and subsequently increased emission (upwards and downward) — from the added GHGs, it doesn’t change the fact the final calculation is still that of a net absorption increase or net reduction in IR transparency through the whole mass of the atmosphere, i.e. from the surface ‘looking up’ all the way through the TOA.
The 3.7 W/m^2 is considered to be the ‘net absorption increase’ or the incremental GHG absorption, i.e. that additionally absorbed, because the transmittance of the energy emitted by the surface is not 100 % prior to adding GHGs. That is, the constituents of the atmosphere, i.e. GHGs (and clouds) are already significantly opaque in the IR to upwelling radiation emitted by the surface (and atmosphere). That’s why there is *already* a GHE in the first place.
I should add here that it’s specifically designated as a ‘net absorption increase’, because the increased IR absorption through to the TOA exceeds the increased IR transmission to the surface. As a counter example, if rate of emission increased with height instead, the opposite would occur, resulting in a ‘net transmission (to space) increase’. Or an enhancement of the radiative cooling of the system to outer space instead.
The reason why you have to make the final calculation, i.e. the net opacity change, *after* the atmosphere becomes more opaque to passing through radiation, i.e. after it absorbs more and subsequently emits more due to the added GHGs, is because just because there is more absorption everywhere doesn’t necessarily or universally mean the net result will cause a warming imbalance for the surface. Again, it ultimately does so because of the lapse rate.
RW wrote: “How can this reduction, or -3.7 W/m^2 TOA imbalance, not be the result of an upwelling IR absorption increase? Of course, it has to be.”
A 10-lb cannon ball “has to” fall twice as fast as a 5-lb cannon ball. Of course, it has to, it is twice as heavy and is pulled twice as hard by the Earth’s gravity.
Haven’t you learned by now not to trust your intuition about what “has to be”? It “has to be” what the laws of physics say it “has to be”. And you haven’t consulted the appropriate law of physics about this subject!
It would help to get over your obsession with absorption and opacity. Remember, doubling the number of GHGs doubles the number of photons being emitted by GHGs. For TOA OLR to go down, all of the increase and more must be absorbed.
Mathematically (for the last time):
dI = n*o*[ B(lambda,T) – I ]*dz
When the term in [brackets] is negative, dI becomes increasingly negative (decreases) when n (the density of GHG) increases. When the term in brackets is positive, dI becomes increasingly positive when n (the density of GHG) increases. This is the situation for a thin layer of atmosphere. Now we need to sum up the dI’s from many layers and their change with increasing GHG. There is no guarantee that increased absorption (n*o*I*dz) dominates increased emission (n*o*B(lambda,T)*dz. Emission dominates in the stratosphere and (according to a recent paper) central Antarctica.
Now, there are good, but complicated, reasons why TOA OLR is LIKELY to decrease with increasing GHG (n) in many atmospheres. However, until you have specified the temperature profile T(z) that goes into the B(lambda,T) term, you have no way of knowing whether the term in brackets is negative or positive.
What situations make the term in brackets positive and what situations make it negative?
Frank,
This is the verbatim wording of Gunnar Myhre himself used when I asked him what the 3.7 W/m^2 of so-called ‘radiative forcing’ was a calculation of (in his 1998 paper) many years back:
“The 3.7 Wm-2 for a doubling of CO2 concentration is the net change in the
absorption at the tropopause level. The tropopause level is more relevant for
surface temperature changes than change in the fluxes at the surface. I do
not have values for the surface, sorry.”
and subsequently:
“The 3.7 Wm-2 is the change in the radiative flux (irradiance) at the
tropopause (since stratospheric temperature adjustment is included this
is the same at the tropoapause and top of the atmosphere). This change
can be compared to the outgoing radiative flux at the top of the
atmosphere of around 240 Wm-2. The absorption occur both in the
troposphere and the stratosphere.”
If it’s not valid to consider it additional absorption, then why is he using the same terminology, i.e. the word ‘absorption’?
It occurred to me that I think I may have failed to convey a key point regarding what I was referring to above as ‘the spectral transmittance evaluated at the temperature of the surface’, which was claimed to quantify the aggregate IR transparency through the whole mass of the atmosphere prior to changing anything. What I may have failed to convey is that calculation isn’t directly related to how the net change in absorption, from increased GHGs, is quantified (via the Schwartzchild eqn.).
The spectral transmittance just quantifies the line of ‘sight’ from the surface looking up through the TOA, i.e. from the surface through to outer space. What differentiates it from the direct surface transmittance, which is solely the direct surface –> TOA component, is it includes upwelling IR emission from the atmosphere as well which is additionally absorbed. You have to include that for the aggregate IR transparency through the whole because the atmosphere itself both absorbs and emits substantially in the IR. However, COE still limits that ultimate quantification in W/m^2 to not exceed or be less than the flux density directly radiated in from the surface. So the spectral ‘T’ and spectral ‘A’, and subsequently the spectral transmission and spectral absorption in W/m^2, are fractions of the flux density radiated in from the surface.
The first pass through of the wavelengths directly radiated from the surface only absorbs some, but then the next layer’s emission absorbs a little more and so on and so forth until all the layers have had their influence on ‘T’ and ‘A’. Of course, the attenuation or absorption occurs at various levels and there is significant overlap from the layers in the saturated bands. But the spectral ‘T’ still quantifies the direct line of ‘sight’ from the surface all the way through the TOA and quantifies the amount of flux in W/m^2 that is transmitted straight through the whole of the atmosphere at the speed of light. The final calculation is based on scaled emitted wavelength intensity of the Planck Flux emitted from the surface. The final spectral transmission through the whole atmosphere is what fraction of the initial energy of that Planck flux which is not absorbed or which is transmitted straight through to outer space (the same as if the atmosphere wasn’t even there).
Frank,
BTW, this is what GW is referring to by ‘T’ and ‘A’ in this post:
In order to calculate the net change in absorption of 3.6 W/m^2 that he gets, he uses the Schwartzchild eqn. just like everyone else.
He just refers to the spectral ‘T’ as the transmission of surface IR emissions or the transmission of ‘surface power’ (i.e. surface radiative power), because that’s effectively equivalent to what it is since the atmosphere creates no energy of its own.
Frank,
“Remember, doubling the number of GHGs doubles the number of photons being emitted by GHGs.”
I have no idea what you’re saying here. What do you mean by doubling the number of GHGs doubles the number photons being emitted by GHGs? Double emission? Of what? From where? You’ve definitely lost me on this point.
“For TOA OLR to go down, all of the increase and more must be absorbed.
Mathematically (for the last time):
dI = n*o*[ B(lambda,T) – I ]*dz
When the term in [brackets] is negative, dI becomes increasingly negative (decreases) when n (the density of GHG) increases. When the term in brackets is positive, dI becomes increasingly positive when n (the density of GHG) increases. This is the situation for a thin layer of atmosphere. Now we need to sum up the dI’s from many layers and their change with increasing GHG. There is no guarantee that increased absorption (n*o*I*dz) dominates increased emission (n*o*B(lambda,T)*dz. Emission dominates in the stratosphere and (according to a recent paper) central Antarctica.
Now, there are good, but complicated, reasons why TOA OLR is LIKELY to decrease with increasing GHG (n) in many atmospheres. However, until you have specified the temperature profile T(z) that goes into the B(lambda,T) term, you have no way of knowing whether the term in brackets is negative or positive.
What situations make the term in brackets positive and what situations make it negative?
As best I can tell, I agree with what you’re saying here. What I can’t figure out is why you think what I’m saying conflicts with it.
Frank,
“Mathematically (for the last time):
dI = n*o*[ B(lambda,T) – I ]*dz
When the term in [brackets] is negative, dI becomes increasingly negative (decreases) when n (the density of GHG) increases. When the term in brackets is positive, dI becomes increasingly positive when n (the density of GHG) increases. This is the situation for a thin layer of atmosphere. Now we need to sum up the dI’s from many layers and their change with increasing GHG. There is no guarantee that increased absorption (n*o*I*dz) dominates increased emission (n*o*B(lambda,T)*dz. Emission dominates in the stratosphere and (according to a recent paper) central Antarctica.
Now, there are good, but complicated, reasons why TOA OLR is LIKELY to decrease with increasing GHG (n) in many atmospheres. However, until you have specified the temperature profile T(z) that goes into the B(lambda,T) term, you have no way of knowing whether the term in brackets is negative or positive.”
Again, as best as I can tell I agree with what you’re saying here.
Maybe the point you’re missing is it’s the sum of the changes from the all the layers from its layer through to the surface and from its layer through to the TOA? For the surface and the layers through to the TOA, the sum of the changes from all of the layers is positive for absorption, reducing the IR intensity at the TOA, and for the layers through to the surface, the sum of the changes is positive for transmission, increasing the IR intensity at the surface.
The point is doing these simulations as people in the field have done for the Earth-atmosphere, results in the reduced IR intensity at the TOA being larger than the increased IR intensity at the surface, manifesting a net absorption increase and a warming imbalance imposed on the system.
“Maybe the point you’re missing is it’s the sum of the changes from the all the layers from its layer through to the surface and from its layer through to the TOA?”
What I mean by this is from any on specific layer through to the TOA, it’s not required that the change be positive for absorption — only that the sum of the changes from all the layers combined (including the surface) is positive for absorption, manifesting a reduced IR intensity at the TOA.
Frank,
I guess if you can’t see and/or don’t agree that the ultimately quantification in W/m^2 is that of an increase in IR absorption, i.e. a net decrease in IR transparency through the whole (from the surface all the way through the TOA), then I suppose it’s no wonder you and apparently everyone else here doesn’t understand anything I’m saying. It seems bizarre to me, but then again the field of climate science seems to have layered on far too much complexity — so much so that no one seems to be able to see the forest through the trees.
While I certainly (like anyone) can be wrong, I maintain that the 3.6-3.7 W/m^2 from 2xCO2 is incremental GHG absorption, i.e. that which is *additionally* captured/absorbed by the atmosphere than was prior to 2xCO2.
RW: “It seems bizarre to me, but then again the field of climate science seems to have layered on far too much complexity — so much so that no one seems to be able to see the forest through the trees.”
Yes. How good it would be with simplification.
If the latent energy had no impact, because there was no change.
If the temperature gradient was fixed, so that the temperature was known for every emission hight. So that emission could easily be calculated.
What seems most bizarre to me is to pretend that all feedbacks are fixed and known, so that lapse rate feedback, water vapor feedback. changing albedo, changing evaporation and all the other variables does not matter.
I think what matters most when it comes to change in IR to space, is what causes the temperature change at tropopause. I think it is so complex that I do not dare to say anything about it. The IR in the CO2 reduced bands seems to come from near tropopause temperatures if I understand it right.
RW: “This is because there is reduced transmission of IR into outer space, which can only ultimately be caused by increased absorption of IR.”
Good simplification. Emission is dependent on absorption and not om temperature. Is it so?
Clearly RW knows better: “And in addition to the radiant flux emitted from the surface which is absorbed there is significant non-radiant flux moved from the surface into the atmosphere, primarily as the latent heat of evaporated water, which condenses to forms clouds — whose deposited energy within (in addition to driving weather), also radiates substantial IR downward to the surface.”
So, why omitting the rest of the sentence which would be: “whose deposited energy also radiates substqantial IR upwards to the space.”
“RW: “This is because there is reduced transmission of IR into outer space, which can only ultimately be caused by increased absorption of IR.”
Good simplification. Emission is dependent on absorption and not om temperature. Is it so?”
All I’m saying is you can’t have an IR transmission to space decrease without an IR absorption increase. The sum of the changes of the upward emitted IR from the surface and layers in the atmosphere must be positive for absorption.
“If the latent energy had no impact, because there was no change.
If the temperature gradient was fixed, so that the temperature was known for every emission hight. So that emission could easily be calculated.
What seems most bizarre to me is to pretend that all feedbacks are fixed and known, so that lapse rate feedback, water vapor feedback. changing albedo, changing evaporation and all the other variables does not matter.
I think what matters most when it comes to change in IR to space, is what causes the temperature change at tropopause. I think it is so complex that I do not dare to say anything about it. The IR in the CO2 reduced bands seems to come from near tropopause temperatures if I understand it right.”
No doubt the path the system takes toward the new equilibrium is enormously complex and non-linear, but I fail to see the significance of this relative to what Frank and I are/were discussing.
“Clearly RW knows better: “And in addition to the radiant flux emitted from the surface which is absorbed there is significant non-radiant flux moved from the surface into the atmosphere, primarily as the latent heat of evaporated water, which condenses to forms clouds — whose deposited energy within (in addition to driving weather), also radiates substantial IR downward to the surface.”
So, why omitting the rest of the sentence which would be: “whose deposited energy also radiates substqantial IR upwards to the space.”
I’m not omitting it. Of course, it’s true, but it’s not really relevant to the underlying mechanism driving the GHE. The process of accelerated surface cooling by non-radiant means makes the surface cooler than it would otherwise be — not warmer. Right? At least as a first order effect.
Again, if the process of additional surface cooling by the movement of non-radiant energy into the atmosphere accelerates the ultimate transport of surface energy to outer space, how is that related to the underlying physics of the GHE, which is process by which the surface is made warmer — not cooler?
test
RW: Moreover, how can you have a net transmission to space decrease without a net absorption increase?
Look at the Schwarzschild eqn, when you ask questions. Cool the upper troposphere by slowing down convection of latent heat. Now you have the roughly the same absorption, but less emission and therefore less LWR reaching space.
You keep trying to simplify radiation transfer in the atmosphere to one phenomena – absorption – when emission, the temperature profile and convection all interact.
Frank
“Look at the Schwarzschild eqn, when you ask questions. Cool the upper troposphere by slowing down convection of latent heat. Now you have the roughly the same absorption, but less emission and therefore less LWR reaching space.
You keep trying to simplify radiation transfer in the atmosphere to one phenomena – absorption – when emission, the temperature profile and convection all interact.”
How am I simplifying? All I’m saying is the final calculation in W/m^2 is quantified as a net absorption change. In the case of increased GHGs, it’s a net absorption increase. This is because there is reduced transmission of IR into outer space, which can only ultimately be caused by increased absorption of IR.
I’ve fully acknowledged that the emission within the atmosphere changes due to the increased absorption (from above and below) from the added GHGs. You seem to think that just because of this, the final calculation can’t be considered an absorption change or increase. It surely can.
Moreover, you don’t seem to understand that, before adding GHGs, the total IR opacity through the whole atmosphere is that from the surface and all of the layers ‘looking up’ through to the TOA. There is no measurement or consideration of downward emitted flux from the layers through to the surface to calculate it, because the IR intensity at the surface — whatever it is — has already had its effect on the surface energy balance and net energy flow at the surface is zero; and the IR flux intensity at the TOA is equal to the intensity of absorbed solar SW flux.
Furthermore, the final calculation of the net absorption increase is specifically that of the same fundamental thing, i.e. from the surface and layers ‘looking up’ through to the TOA. The only difference is you have to factor in the increased IR intensity at the surface, i.e you have to subtract it from the decreased IR at the TOA to arrive at the net change in absorption. This is required to satisfy COE.
If you don’t think it’s done to satisfy COE, then why aren’t the two added together instead?
RW wrote: “Frank, why is the notion of radiative resistance to radiative cooling not a clear concept?”
Because it is already a clear concept for antennas. Because you don’t have an equation which quantitatively defines the meaning of the term for others. For the most part, others use terms that show up in Wikipedia (with equations) or technical dictionaries.
Equilibrium and steady state have slightly different meanings. You can have steady-state that is far from equilibrium. The sun, Earth and empty space are not in equilibrium, but the Earth had a steady temperature. When you say “converged equilibrium” is a steady-state, you have made tricky terminology more difficult to understand.
Frank,
“RW wrote: “Frank, why is the notion of radiative resistance to radiative cooling not a clear concept?”
Because it is already a clear concept for antennas. Because you don’t have an equation which quantitatively defines the meaning of the term for others. For the most part, others use terms that show up in Wikipedia (with equations) or technical dictionaries.”
Correct, there is no equation that specifically describes it. You will note also that there is no specific equation itself that describes the underlying physics of the GHE. That’s because it’s impossible to clearly separate it or distinguish it from the actual thermodynamic path itself that manifests the balance.
BTW, I think this is one of the main reasons why a lot of people have difficulty internally conceptualizing the fundamental physics driving the GHE.
“Equilibrium and steady state have slightly different meanings. You can have steady-state that is far from equilibrium. The sun, Earth and empty space are not in equilibrium, but the Earth had a steady temperature. When you say “converged equilibrium” is a steady-state, you have made tricky terminology more difficult to understand.”
Sorry. All I mean is a state of energy balance for the system as a whole, i.e. no heating or cooling is in the process of occurring.
RW wrote: “Correct, there is no equation that specifically describes [the GHE]. You will note also that there is no specific equation itself that describes the underlying physics of the GHE. That’s because it’s impossible to clearly separate it or distinguish it from the actual thermodynamic path itself that manifests the balance. BTW, I think this is one of the main reasons why a lot of people have difficulty internally conceptualizing the fundamental physics driving the GHE.”
I agree with you (G&T) that most conventional explanations for the GHE are problematic. Forget those explanations.
“My” simple explanation: The GHE is the difference between surface and TOA OLR (390-240=150 W/m2). This difference is prediction or consequence of the Schwarzschild equation and the composition and temperature profile of our atmosphere. The enhanced GHE predicts that this difference will rise about 4 W/m2 after an instantaneous doubling of CO2. (A simple quantitative definition based on an equation.)
All of the mistakes that I complain about arise from the fact that you are using inferior and inadequate explanations for the GHE and enhanced GHE
G&T complain that this definition doesn’t explain how the GHE makes the Earth warmer. Guilty as charged. Warmer than what? When climate scientists attempt to answer this question, they encounter into numerous problems: 1) The planetary albedo varies with temperature. 2) The Earth is spinning and not evenly heated by the sun. 3) The physics of blackbody radiation does not apply to the atmosphere: 3a) Atmospheric radiation is not in thermodynamic equilibrium with the molecules in the atmosphere and such an equilibrium is a postulate in the derivation of Planck’s Law. 3b) The atmosphere doesn’t have a single temperature. A blackbody with the Earth’s range of temperatures would produce a 3.5-fold difference in thermal radiation.
The Earth does emit like a blackbody, so a surface OLR of 390 W/m2 appropriate for an average surface temperature of 288 K. If you want an answer to the “Warmer than what” question, you tell me what model for the Earth without a GHE you find acceptable. I find all such models problematic and/or complicated.
Likewise, converting the enhanced GHE of 4 W/m2 for 2XCO2 into a temperature difference is problematic. That problem is called climate sensitivity and it doesn’t have a simple answer. However, it is easier to discuss the climate feedback parameter: How much does the Earth’s surface need to warm to drive an additional 1 or 4 W/m2 of NET radiation through the TOA. (Net = OLR change minus SWR change.) A simple blackbody near 255 K has a “climate feedback parameter” of 1 (W/m2)/K.
For clarity, I broke the above reply into two parts and posted them (with slight revisions) at the bottom of the page. Please reply there. Part one covers radiation, which should be non-controversial. Part two deals with warming.
Frank,
If you want to understand where RW is coming from, read this:
http://www.palisad.com/co2/div2/div2.html
Note that there is no transfer of energy by convection from the surface to the atmosphere, which is represented by a single slab, in the diagram.
Oh boy, I don’t have to mental energy to get into that. The issues I’m discussing are — by themselves — independent of the validity or non-validity of GW’s black box analysis *claiming* to quantifying the aggregate IR transparency/opacity of the steady-state atmosphere, independent of the actual thermodynamic path manifesting the energy balance.
But yes, GW’s calculated ‘T’ value of 0.241 for the black box model exercise is the ‘spectral transmittance evaluated at the temperature of the surface’, and the 3.7 W/m^2 from a doubling of CO2 he refers to as being the ‘incremental absorption’ is the ‘net absorption increase’ calculated via the Schwartzchild eqn. in the same way everyone else does it.
None the less, what I’m discussing with Frank is independent of the validity or non-validity of what’s claimed in GW’s div2 analysis. What I’m claiming could still be completely correct even if GW’s div2 is wrong.
If anyone interested, GW’s own words regarding the equivalence of his div2 analysis when I asked him some time ago:
“My RT simulation simply calculates absorption, A, which is the fraction of surface emissions absorbed by the atmosphere and is the most basic calculation you can do using hitran line data. T is then just equal to 1 – A.
Consider a contrived and oversimplified 2 layer model where T for each layer is 0.482. The net T for the 2 layers combined would be 0.241. The value of 0.241 is the average T to be applied to a single layer atmospheric model which has the same average behavior as an N layer model, hence the equivalence.”
Note black box model is only an *equivalent* model claiming to quantify the aggregate dynamics of the system in the steady-state — it’s not a model depicting the actual thermodynamic path manifesting the energy balance. It would be spectacularly wrong and nonsensical if it were.
If anyone’s interested, here is bit more of GW’s own words (in response to Roy Spencer):
“>>>>I read George’s clarification you have in your e-mail. For example he says…
“….my simplified model is an equivalent model whose behavior at the boundaries matches the required behavior. That is, if the Earth’s actual atmosphere was replaced with an equivalent atmosphere with my specified behavior, the surface temperature would be exactly the same.”
But I don’t think this is possible. The Earth’s surface GAINS more radiant energy than it loses, and the atmosphere LOSES more radiant energy than it gains. The difference is due to convective overturning of the atmosphere, which involves a net flow in only one direction (upward). I don’t see how (or even why) you would try to come up with an “equivalent model” which ignores this fact. The only radiative balance which exists is at the top of the atmosphere…almost everywhere else, there is NO balance of radiative flows.
The balance at the surface is the sum of a radiative balance and a kinetic balance, where additive superposition must apply to the effects of energy on the temperature of a system. To the extent that more radiant energy enters the surface than leaves, it must be replacing kinetic energy leaving the surface, but not returned. The source of this kinetic energy is latent heat, which is removed from the surface as the potential energy of a phase change, not as radiation. This is often considered a component of the ‘radiative’ balance at the surface, which it is not. The return of energy to the surface by weather, which includes the return of energy stored as water lifted against the force of gravity, is also not always considered kinetic. Atmospheric convection is also a kinetic process and while it rearranges the energy stored by the planet, it has no influence on the requirements for radiative balance, which are dictated by only the Sun and the albedo, where the climate system feedback adjusts the fraction of the planet covered by clouds, driving the system towards balance.
At the TOA, there is only a radiative balance, while at the surface there is both, none the less, if there was no kinetic balance at the surface, i.e. no evaporation/weather, the resulting radiative balance and corresponding temperature of the surface described by the equivalent model would be exact. If you add equal and opposite kinetic fluxes from the surface to the atmosphere and back and trade off some amount of the kinetic return to the surface with radiant return, the results now match your expectations. Note that adding these excess components between the surface and atmosphere has no effect on the requirements for radiative balance at the TOA or even the net surface temperature, or at least this is what the measurements tell us since the satellite data matches the equivalent model almost exactly.
Occam’s razor certainly applies to modeling, where the best equivalent model is the simplest one that exhibits the required behavior.
There are several interesting plots here. There are about a dozen scatter plots of measured data showing the post albedo incident power along the X axis and many different parameters along the Y axis, including surface and cloud temperatures, water content, cloud coverage, total solar power, surface reflectivity, albedo and more. Each plot also illustrates the measured sensitivity of a climate parameter to the post albedo incident power, which is approximately equivalent to the sensitivity of that parameter to forcing, as defined by the IPCC. The scatter diagrams cover about 3 decades of satellite measurements.”
At any rate, you guys certainly aren’t alone. No one anywhere seems to have even the slightest clue what GW is doing or even why he’s doing it. If no one agrees that the 3.7 W/m^2 is a quantification of incremental GHG absorption (as no one here seems to agree) and/or a net increase in IR opacity of 3.7 W/m^2 from the surface through the whole of the atmosphere to the TOA, then that may explain why. Or it may not — I don’t really know. It seems bizarre though to me that there would disagreement on that point, but like anyone I certainly could be wrong.
Nor does anyone seem to agree that the GHE is initiated by the surface’s inability to radiate freely the outer space.
– A photon has no temperature, temperature is a multi-particle property.
– There is no saturation.
– When well-mixed greenhouse gases, the amount of greenhouse gas molecules is proportional to the pressure difference over a height range.
– Without air movement is dB / dp constant (depending on net heat flow and concentration of greenhouse gases), B = sigma T^4
– At a temperature gradient without air movement above the adiabatic temperature gradient is a movement of air and reduces the temperature gradient to the adiabatic value (Schwarzschild criterion).
– The limitation of the air movement is the tropopause, below is the troposphere with vertical circulation, above the tropopause is the stratosphere.
– The ozone layer is only high in the stratosphere and has almost no influence on the height of the tropopause.
– With increasing greenhouse gas concentrations, the height of the tropopause is increasing and the temperature gradient is approximately constant in the troposphere.
– Result: the surface temperature rises and the temperatures of the stratosphere sink.
Yes, and your point is? I think it’s generally agreed that without the lapse rate, there would be no GHE and certainly not an enhanced GHE from added GHGs. Added GHGs shift the height of the average transmission level to where it’s colder than it was before, reducing the outgoing LW flux at the TOA. This is supposed to cause the lower atmosphere and ultimately the surface to warm by some amount in order to re-establish equilibrium at the TOA.
However, this alone does not really describe the underlying physics driving the GHE.
I guess if no one understands and agrees that the GHE is initially driven by the surface’s inability to radiate freely to outer space, then it’s no wonder apparently no one here has any idea what I’m talking about.
The amount of IR the atmosphere ultimately passes to the surface (often called ‘back radiation’) no doubt has a huge influence on the surface energy balance, but it’s not really the underlying driving mechanism of the GHE. The term ‘back radiation’ is misnomer since it’s really just downward LW at the surface/atmosphere boundary.
This is because there are multiple energy inputs to the atmosphere besides just the upwelling IR flux emitted from the surface (and atmosphere) which is absorbed. Post albedo solar energy absorbed by the atmosphere and re-emitted downward to the surface would not be ‘back radiation’, but instead ‘forward radiation’ from the Sun whose energy has yet to reach the surface. And in addition to the radiant flux emitted from the surface which is absorbed there is significant non-radiant flux moved from the surface into the atmosphere, primarily as the latent heat of evaporated water, which condenses to forms clouds — whose deposited energy within (in addition to driving weather), also radiates substantial IR downward to the surface. The total amount of IR that is ultimately passed to the surface has contributions from all three input sources, and the contribution from each one cannot be distinguished or quantified in any clear or meaningful way from the other two.
Thus mechanistically, the downward IR flux ultimately passed to the surface from the atmosphere has no real or clear relationship to the underlying physics driving the GHE, i.e. the re-radiation of initially absorbed surface IR energy back downwards where it’s re-absorbed at a lower point somewhere (with perhaps some of it passing all the way back to the surface, but most of is not).
I am starting a new thread with a reply to a message in an old thread that has gotten so long I can’t be bothered to find where it starts.
RW wrote: “The process of accelerated surface cooling by non-radiant means makes the surface cooler than it would otherwise be — not warmer. Right? At least as a first order effect. … the process of additional surface cooling by the movement of non-radiant energy into the atmosphere accelerates the ultimate transport of surface energy to outer space”
If I understand what you are saying, that is correct. It is called the “lapse rate feedback” and is in fact a negative feedback. In my view it should be combined with the positive “water vapor feedback” since both are a result of increased evaporation at the surface. Models disagree considerable on the magnitudes of the two individual but agree quite well on the sum of the two.
Mike,
“If I understand what you are saying, that is correct. It is called the “lapse rate feedback” and is in fact a negative feedback.”
I wasn’t specifically referring to the lapse rate feedback or any feedback, but more just that non-radiant flux from the surface to the atmosphere accelerates surface cooling. Or it accelerates the ultimately movement of surface energy up through the atmosphere where it is eventually passed into outer space as LWIR in order to balance the incoming absorbed solar SW flux.
RW,
This is why you are so maddening. Your reply amounts to “I wasn’t specifically referring to the lapse rate feedback or any feedback, but [description of the lapse rate feedback]”.
I have no idea what to make of that. Are you just being difficult by refusing to use standard terminology? Or do you just not know what you are talking about?
Mike,
The term ‘feedback’ generally applies to the magnitude of the response of the system during the path from one equilibrium state to another. I wasn’t specifically describing that, but more so that even in the steady-state there is accelerated surface cooling from the non-radiant flux from the surface to the atmosphere which makes the surface cooler than it would otherwise be.
Again starting a new thread with a reply.
nobodysknowledge wrote: “Yes. How good it would be with simplification.”
Sarcasm?
nobodysknowledge wrote: “If the latent energy had no impact, because there was no change. If the temperature gradient was fixed, so that the temperature was known for every emission hight. So that emission could easily be calculated.”
That is what is done in computing the direct effect of CO2, also in the Modtran calculations that Frank refers to. It is, of course, an oversimplification for the atmosphere as a whole, but is a very useful starting point.
nobodysknowledge wrote: “What seems most bizarre to me is to pretend that all feedbacks are fixed and known, so that lapse rate feedback, water vapor feedback, changing albedo, changing evaporation and all the other variables does not matter.”
Who pretends that? Other than as a temporary simplification to find a place to start?
nobodysknowledge wrote: “I think what matters most when it comes to change in IR to space, is what causes the temperature change at tropopause.”
What matters most is the temperature at about 5 km, since that is the average emission height. But temperatures from a whole range of altitudes, from the surface (in the IR windows) to near the tropopause (near the band centers) do matter.
Mike M: “What matters most is the temperature at about 5 km, since that is the average emission height. But temperatures from a whole range of altitudes, from the surface (in the IR windows) to near the tropopause (near the band centers) do matter.”
I am not sure this is right. I was thinking the IR flux magnitude at TOA Is most dependent on surface temperature (IR window) and near tropopause temperature where most of the other emission comes from. There must be very little emission from 5 km altitude. If it is a way to interpret the Modtran spectrum. But I am not sure, so I would like to hear what people knowing more about this have to say.
nobodysknowledge,
If you take a look at Visualizing Atmospheric Radiation – Part Three – Average Height of Emission you will see some answers, including graphs like this one:
and this one:
-where altitude is on the bottom left, and wavenumber is on the bottom right.
A lot depends on the amount of water vapor in the atmosphere. Cold dry atmospheres are very different from tropical atmospheres.
nobodysknowledge: The short answer is that Mike is roughly right.
Radiative transfer calculations show that only about 10% of the photons emitted by the surface reach space, transferring a flux of about 40 W/m2. When SOD was working with his program that calculated radiation transfer using information from the HITRAN database, I asked him to calculated how much power was transferred directly to space by the photons emitted from each 1 km layer of atmosphere above the surface. He posted the answer in a comment that I can’t find anymore. It turned out that photons from EVERY altitude contribute significantly to the TOA flux, but the amount peaked around 5 km.
If the emission from first ten 1 km layers of atmosphere (roughly the troposphere) contributed equally to the 200 W/m2 that doesn’t originate from the surface (and little originated above), that would be about 20 W/m2 per layer. DLR is 10 W/m2 at 20 km and 30 W/m2 at 10 km, so the atmosphere above 10 km does contribute some photons to the OLR reaching space, but far less than the lowest 10 km. So the average for the each 1 km layer in the lowest 10 km is probably about 15 W/m2/km and the peak around 5 km was probably about 20-25 W/m per 1 km thick layer. The lowest 2 kilometers which contain (much of the water vapor), contributed the least for the troposphere layers. (I hate trusting my memory; hopefully this is close to correct.)
This peak is the result of competing phenomena. As temperature drops with altitude (about 32 K by 5 km), the B(lambda,T) in the Schwarzschild eqn reduces emission (about 1.6X). As pressure drops with altitude (about 50% by 5 km), the lower density of GHG (n) drops also reduces emission (by 2X). On the other hand, photons emitted from higher in the atmosphere have a greater probability of reaching space without being absorbed, (Essentially no photons are re-emitted by GHGs in the troposphere – collisional relaxation and excitation are much faster processes than emission of a photon by an excited state.)
The surface emits at all wavelengths, so some of its emission passes directly to space through the “atmospheric window” – where it isn’t cloudy. The atmosphere emits only at wavelengths it can absorb and emits most strongly at wavelengths it absorbs best. There is no “atmospheric window” for photons emitted by the atmosphere. I believe clouds also emit roughly a blackbody spectrum, so there is an “atmospheric window” for emission from clouds, but they are colder than the surface. (SOD’s calculations probably dealt only with emission from clear skies).
Frank,
“Radiative transfer calculations show that only about 10% of the photons emitted by the surface reach space, transferring a flux of about 40 W/m2.”
This must be referenced from the diagram in Trenberth’s paper. According to GW, when he asked, Trenberth could not provide an actual reference for the this number. It appears to just be an arbitrary guess or deduction — it’s not based on detailed line by line RT calculations. GW calculates a spectral surface transmittance of 0.24 on global average, which means the direct surface transmittance, i.e. the direct surface –> TOA component by itself, must be greater than 0.24.
Now, like anyone GW could be mistaken, but he can at least provide all the details of how’s he getting that number for anyone who wishes to know and see it. Trenberth cannot.
But the direct surface transmittance is not a meaningful number, moreover, the spectral transmittance is still all that which is directly emitted from the surface that passes into space at the speed of light — it’s just that it factors in, i.e. subtracts, that which is additionally absorbed by the atmosphere as well. For IR opacity through the whole, you have to factor that in.
But again, this all seems to be a major source of disconnect.
Thank you for the answers Frank and SoD. I find the CO2 spike of emission in stratosphere interesting, and the great contribution in flux from the altitude of 18 km. So temperatures at 18 km matter to some degree.
SoD,
Is the emission to space from the stratosphere really 10% of the total? That seems high to me (but I have never done the calculations). I thought the stratosphere was heated mainly by absorbing UV, but that is only 1-2% of the incoming radiation.
Mike M,
You can get some idea of the emission to space from the stratosphere by using MODTRAN, setting the observation height at the top of the tropopause and looking up. For the Tropical Atmosphere, the height is 17 km and the IR flux down is 9.439W/m². Most of that is from CO2 and ozone with a smattering, 1.645W/m², from water vapor at low frequencies.
That’s a little more than 3% of the 289.225W/m² at 100km looking down.
Mike: If you want to know how much OLR is emitted from the stratosphere to space, a reasonable first approximation is the amount of DLR entering the troposphere. You can determine that using the online MODTRAN calculator. That answer depends greatly on which atmospheric profile you use and the altitude where the stratosphere “begins”. The tropical profile has clearly defined tropopause at 17 km, but the US Standard Atmosphere has a constant temperature from 11-20 km. The amount of OLR originating in the stratosphere depends on where you define the beginning of the stratosphere and whether that dividing line varies with latitude. DLR (US Standard) is about 10 W/m2 at 20 km and 24 W/m2 at 11 km, similar to what is shown for OLR contribution on SOD’s graph.
Frank and Dewitt,
Thanks, that helps a lot. It seems there are several things going on. SoD’s number do seem high (23 W/m^2 above about 14 km). And much of the stratosphere emission is from the tropopause region; I did not consider that, but it makes sense. And the emission from above that includes the emission of UV energy absorbed, since the T profile is specified.
So 5 W/m^2 UVB&UVC absorbed gives 25% of the 10 W/m^2 downward emission from above 17-20 km. As one goes up, the input from upward IR absorption gets less and the input from downward UV increases. So at the stratopause UV is the main energy input, agreeing with the stratopause being relatively warm.
Sound like I should learn to use MODTRAN.
RW wrote: “Correct, there is no equation that specifically describes [the GHE]. You will note also that there is no specific equation itself that describes the underlying physics of the GHE. That’s because it’s impossible to clearly separate it or distinguish it from the actual thermodynamic path itself that manifests the balance. BTW, I think this is one of the main reasons why a lot of people have difficulty internally conceptualizing the fundamental physics driving the GHE.”
I agree with you (G&T) that most conventional explanations for the GHE are problematic. Forget those explanations.
“My” simple explanation: The GHE is the difference between surface and TOA OLR (390-240=150 W/m2). This difference is prediction or consequence of the Schwarzschild equation and the composition and temperature profile of our atmosphere. The enhanced GHE predicts that this difference will rise about 4 W/m2 after an instantaneous doubling of CO2. (A simple quantitative definition based on an equation.)
All of the mistakes that I complain about arise from the fact that you are using inferior and inadequate explanations for the GHE and enhanced GHE. In particular, you focus mostly on absorption – which is one of two terms in the Schwarzschild eqn.
Frank,
I would revise that to include convective energy transfer from the surface to the atmosphere, IIRC, 102 W/m² in TKF09, as that contributes significantly to OLR as well as DLR (492-240 = 252 W/m²).
Frank,
This is where Dewitt and I disagree. While I certainly don’t deny that approximately 500 W/m^2 are flowing into and away from the surface, I still do not think +250 W/m^2 is an accurate quantification of the GHE, because there are an infinite number of surface temperatures and surface temperature elevations via a GHE that could be manifested with 500 W/m^2 of gross flux. In other words, 500 W/m^2 says nothing about the surface temperature, which is determined by the net flux gained at the surface. That amount is universal for temperature, moreover its the actual rate joules are being added to the surface.
Only if you consider that there are an infinite number of points between a very small temperature difference. For a given rate of incoming energy, the balance between the rate of energy leaving the surface by radiation and convection has a very limited range, given the physics of the atmosphere.
I could go into detail, but it’s pointless.
Dewitt,
“Only if you consider that there are an infinite number of points between a very small temperature difference. For a given rate of incoming energy, the balance between the rate of energy leaving the surface by radiation and convection has a very limited range, given the physics of the atmosphere.”
Yes, but you’re missing the point, and that is the notion of a radiatively induced GHE isn’t limited to only the specifics of the Earth-atmosphere system.
The gross flux in general has no clear relationship to an actual surface temperature — be it the surface of the Earth or any other surface, and more involves how the net flux gained is physically manifested. This gets back to my point earlier about the underlying physics of the GHE are largely separate from the actual thermodynamic path manifesting the surface energy balance. This is a nuanced point, and one it seems you don’t understand and thus don’t agree with.
Part of the sentence needs revising as well: The GHE is the difference between the energy emitted by the surface and the energy emitted to space (490-240=250 W/m²). I rounded to 490 because I don’t think three significant figures are justified.
Frank,
““My” simple explanation: The GHE is the difference between surface and TOA OLR (390-240=150 W/m2).”
That’s strange you would say this since I totally agree. The GHE quantified in W/m^2 is +150 W/m^2 of net surface gain (which amounts to about +33C.) This is because, assuming the same albedo, the minimum surface gain would be 240 W/m^2 with no GHE. That is, if the surface could radiate uninhibited to outer space.
G&T complain that defining the GHE in terms of surface and TOA OLR doesn’t explain how the GHE makes the Earth warmer. Guilty as charged! Warmer than what? When climate scientists attempt to answer this question, they encounter numerous problems: 1) The planetary albedo varies with temperature. 2) The Earth is spinning and not evenly heated by the sun. 3) The physics of blackbody radiation does not apply to the atmosphere: 3a) Atmospheric radiation is not in thermodynamic equilibrium with the molecules in the atmosphere and such an equilibrium is a postulate in the derivation of Planck’s Law. 3b) The atmosphere doesn’t have a single temperature. A blackbody with the Earth’s range of temperatures would produce a 3.5-fold difference in thermal radiation.
The Earth’s surface does emit like a blackbody, so a surface OLR of 390 W/m2 appropriate for an average surface temperature of 288 K. If you want an answer to the “Warmer than what” question, you tell me what model for the Earth without a GHE you find acceptable. I find all such models problematic or complicated.
Likewise, converting the enhanced GHE of 4 W/m2 for 2XCO2 into a temperature difference is problematic. That problem is called climate sensitivity and it doesn’t have a simple answer. However, it is easier to discuss the climate feedback parameter: How much does the Earth’s surface need to warm to drive an additional 1 or 4 W/m2 of NET radiation through the TOA. (Net = OLR change minus SWR change.) A simple blackbody near 255 K has a “climate feedback parameter” of 1 (W/m2)/K.
Defining the GHE and aGHE in terms of a change in radiation is simple, quantitative, and explained by one equation, the Schwarzschild eqn. Those philosophically opposed the AGW or CAGW stupidly challenge the physics of radiation transfer (the Schwarzschild equation) and the concept of radiative forcing. It makes far more sense to challenge the problematic processes by which a radiative forcing is converted to warming: about 3 K for 2XCO2 (4 W/m2) and 33 K for the GHGs in our atmosphere (150 W/m2).
“Likewise, converting the enhanced GHE of 4 W/m2 for 2XCO2 into a temperature difference is problematic. That problem is called climate sensitivity and it doesn’t have a simple answer. However, it is easier to discuss the climate feedback parameter: How much does the Earth’s surface need to warm to drive an additional 1 or 4 W/m2 of NET radiation through the TOA. (Net = OLR change minus SWR change.) A simple blackbody near 255 K has a “climate feedback parameter” of 1 (W/m2)/K.”
Can you elaborate on these numbers more? I’m not sure what you mean by them. Typically the so-called ‘black body’ or ‘no-feedback’ TOA parameter is about 3.3 W/m^2 per 1C of surface warming. For 3.7 W/m^2 of incremental forcing, it’s about 1.1C. The 1.1C is the amount commonly cited for the so-called ‘no-feedback’ surface temperature increase for 2xCO2.
RW wrote: “Can you elaborate on these numbers more? I’m not sure what you mean by them. Typically the so-called ‘black body’ or ‘no-feedback’ TOA parameter is about 3.3 W/m^2 per 1C of surface warming. For 3.7 W/m^2 of incremental forcing, it’s about 1.1C. The 1.1C is the amount commonly cited for the so-called ‘no-feedback’ surface temperature increase for 2xCO2.”
Can we first focus on the concept behind the climate feedback parameter?
Earth: We want to know how much more NET radiation the Earth emits when it is warmer (or less, when cooler). Climate scientists call this the “climate feedback parameter”. NET means the increase in OLR plus any change that adds to or subtracts from the amount of SWR reflected back to space. For simplicity, I may occasionally refer to this concept as the increase in thermal emission with temperature, but I will always mean net increase in radiation to space with warming: OLR + reflected SWR. Assuming linearity near current GMST, we want an answer in terms of W/m2/K.
Why focus on the climate feedback parameter (rather than ECS)? The climate feedback parameter applies to many more situations than ECS. It is [mostly] independent of the concept of forcing and equilibrium: 1) At steady-state, the planet has no radiative imbalance. After a forcing of 3.7 W/m2 (from 2XCO2, for example), a zero radiative imbalance won’t be reached until the planet warms enough to emit an additional 3.7 W/m2. If we know the climate feedback parameter, we know ECS. We don’t need to wait for our AOGCM to approach a steady state – when heat gradually stops flowing into the deep ocean many centuries in the future and ice caps stop shrinking millennia in the future. 2) El Nino is a form of unforced variability, caused partly by a slowdown in exchange of heat between the deep ocean and the surface in the tropical Pacific. If a super El Nino or La Nina or a shutdown of the AMOC resulted in a 1 or 2 degC of change in GMST, the climate feedback parameter would tell us how the planet’s radiative cooling to space would change. 3) One year after Pinatubo, when aerosol cooling reached its maximum (about -0.6 degC), the climate feedback parameter tells us how much of the current aerosol forcing had been negated by a drop in radiative cooling to space – reducing the magnitude of the radiative imbalance. 4) We can apply the climate feedback parameter to the MWP or LIA – whether they represented naturally-forced or unforced variability.
Blackbodies: Warmer objects emit more thermal radiation. A warmer simple blackbody near 255 degK emits 3.78 W/m2 of thermal infrared per degK of warming (3.78 W/m2/K). You can calculate this using the S-B equation and emissivity = 1 for yourself. (You can get confused using 288 K and e = 0.61 or you can calculate this value by take the derivative of the S-B eqn – forgetting that emissivity varies with temperature and the product rule. Let’s stick with a simple blackbody at 255 K.) We could call this the “climate feedback parameter” for a blackbody at 255K, but no one does.
AOGCMs: If you take an AOGCM and artificially raise the temperature 1 degK everywhere, net radiation to space increases by 3.2 W/m2. All CMIP3 AOGCMs have a climate feedback parameter near 3.2 W/m2/K. This result is similar to that calculated above for a blackbody, but not exactly the same. (One reason is that (average T)^4 is not the same as the average of T^4 and the Earth doesn’t have a uniform temperature.) Nevertheless, climate scientists call this 3.2 W/m2/K “Planck feedback” even though Planck feedback for a blackbody at 255K is 3.78 W/m2.
ECS: If you take the reciprocal of the climate feedback parameter and assume that one doubling of CO2 is 3.7 W/m2 (different assumptions about the earth give slightly different results for the forcing from doubled CO2), you can convert a climate feedback parameter of 3.78 W/m2/K (for a blackbody at 255K) or 3.2 W/m2/K (for AOGCMs without feedbacks) into ECS’s of 0.98 (usually rounded to 1.0) and 1.15 K/doubling respectively. However, I want to focus on the climate feedback parameter – the more fundamental property of our planet.
Summing feedbacks: The climate feedback parameter is the sum of everything that influences the increase in net radiation reaching space per degK of surface warming: 1) The increase in emission with temperature, aka Planck feedback (3.2 W/m2/K). 2) The increase in absorption of OLR with increasing absolute humidity due to a warmer surface temperature, aka WV feedback. 3) The decrease in lapse rate with increasing humidity, which produces more warming the the upper troposphere than at the surface, aka LR feedback. 4) and 5) The change in albedo produced by changes in clouds and snow/ice cover with surface warming, aka cloud and ice-albedo feedbacks. All of these have units of W/m2/K (surface warming) and added together equal the climate feedback parameter. By tradition, any increase in radiation to space has a negative sign (it cools the Earth). So Planck and LR feedbacks are negative and WV feedback is positive. I will ignore this convention and discuss the magnitude and direction of the change in net radiative cooling to space (not heat flow into the planet).
Fast Feedbacks: Except for very slow changes in ice caps, net radiative cooling to space tracks changes in surface temperature with little lag. Thermal emission varies instantly with temperature. The average water molecule remains in the atmosphere for only 9 days, so atmospheric humidity and clouds respond quickly to changes in surface temperature – in less than a month according to data at SOD, or within a few months according to Roy Spenser, who tracks heat rising through the atmosphere. One part of ice-albedo feedback is a fast response in seasonal snow cover and a second part is a slow shrinking of ice caps. Together they total only 0.3 W/m2/K according to the IPCC. So I am going to ignore the slow ice cap feedback and say that the radiation emitted by the Earth varies quickly with surface temperature. This fast response of radiative cooling to space with increasing surface temperature provides a good way to estimate (slightly underestimate) the climate feedback parameter.
Responses Observed from Space: The Earth’s GMST rises and falls 3.5 degK every year, reaching a peak in late July and an minimum in late January. (This is due to the asymmetric distribution of land, which warms and cools more than the ocean.) This seasonal rise and fall disappears when we calculate temperature anomalies. From space, we can watch the net radiation emitted by the Earth rise and fall in parallel with GMST. Our planet does not not exhibit a climate feedback parameter of 3.78 W/m2/K like a simple blackbody or 3.2 W/m2/K like an AOGCM with no feedbacks. In the LWR channel, both clear and cloudy skies emit an additional 2.2 W/m2/K of LWR. Radiative cooling has been suppressed by WV+LR feedback in clear skies. According to climate models, increased absolute humidity upon warming reduces OLR from clear skies by about 2 W/m2/K and the change in lapse rate with humidity increases OLR by about 1 W/m2/K, so AOGCMs do a good job reproducing the observed increase in LWR radiative radiative cooling through clear skies. See:
Click to access 7568.full.pdf
Seasonal warming (the difference caused by a large warming in the NH and a modestly smaller cooling in the SH) is not a ideal model for global model. We see a reduction in SWR reflected by the surface through clear skies of 1 W/m2/K. This is due to the large seasonal snow cover in the NH in winter and the negligible seasonal snow cover in the SH in winter. This is not a global feedback. Cloudy skies also reflect about 1 W/m2/K less SWR. This is cloud feedback, but there could be differences in cloud cover between the NH and SH. Climate models do a lousy – and mutually inconsistent – job of reproducing observed reflected SWR from both clear and cloudy skies and LWR from cloudy skies.
Limits: If the climate feedback parameter were zero, radiative cooling to space would not change with surface temperature. This is what happens in a runaway greenhouse. If the climate feedback parameter were infinite, no forcing would be able to change surface temperature. A perfect mirror has an infinite “climate feedback parameter”.
Quick summary: If the IPCC’s models are right (ECS is near 3.7 K/doubling), the planet emits about 1 W/m2/K more net radiation to space upon warming. If Otto and Lewis&Curry are right (ECS about 2), the planet emits about 2 W/m2/K more net radiation to space upon warming. If no feedbacks exist (or cloud feedback reflects more SWR as the surface warms, which is unlikely), the planet emits 3-4 W/m2/K more net radiation to space upon warming.
Frank,
“NET means the increase in OLR plus any change that adds to or subtracts from the amount of SWR reflected back to space. For simplicity, I may occasionally refer to this concept as the increase in thermal emission with temperature, but I will always mean net increase in radiation to space with warming: OLR + reflected SWR.”
Yes, agreed. But the feedbacks are really beyond the scope of this particular article and comments thread.
RW,
A one dimensional radiative/convective model of the atmosphere converges to the same profile and surface temperature whether you start with a surface and isothermal atmosphere with a temperature higher or lower than the expected value. See Figure 1. in Matanabe, S. and Strickler, R.F.,1964: Thermal Equilibrium of the Atmosphere with a Convective Adjustment, Journal of the Atmospheric Sciences, 21, 361-385.
That’s interesting, but how is that related to the underlying physics driving the GHE? It’s important to note that essentially every surface temperature on any object has a specific thermodynamic path manifesting it, but it is not necessarily a manifestation of a radiative GHE acting on that surface, i.e. acting to make that surface warmer than it would otherwise be in the way a GHE is supposed to be doing.
I believe the fundamental question you cannot answer is what specifically differentiates a thermodynamic path caused or initiated by a GHE and one that is not. In nearly every case, a surface will have a certain thermodynamic path manifesting it. At its surface, that path will have an ultimate manifestation of ‘net energy gained’ determined and quantified by: (radiation in + convection in + conduction in) – (radiation out + convection out + conduction out) + S/B radiant power out.
This is because virtually every thermodynamic path manifesting a specific surface temperature is a manifestation of all the physics mixed together, radiant and non-radiant.
DeWitt and RW: Revising, based on your comments above:
Treating the atmosphere as a black box, 390 W/m2 of LWR enters the bottom and 240 W/m2 exits the top, creating a GHE of 150 W/m2. This is the prediction of the Schwarzschild eqn for an atmosphere with the existing average composition and temperature profile. An adequate description of the GHE can not be provided by adsorption alone or models based of blackbody radiation. Since the GHE is primarily concerned with the reduction in planetary radiative cooling to space caused by GHGs, this “zeroth-order approximation” ignores other important heat fluxes.
A first-order approximation would take into account the fact that the existing temperature profile can’t be maintained without a substantial upward flux of heat by convection (100 W/m2 at the surface, mostly latent heat) and downward flux of LWR (333 W/m2 at the surface). These phenomena – which are critical to maintaining a steady state – are large near the surface compared with the magnitude of the GHE, so 150 W/m2 alone isn’t a proper “first-order approximation”.
Second-order corrections include the difference between clear and cloudy skies and temperature variation with latitude and season.
If the GHE were smaller, the Earth would be cooler. However, simple methods for converting this 150 W/m2 into 33 degK cooler without GHGs are problematic. I personally won’t endorse any quantitative value for the GHE in terms of temperature, but others may. (Before doing so, advocates of 33 K should calculate their expected temperature for the surface of the moon and compare it with observations.)
At steady-state, the upward and downward flux of heat – SWR, LWR, latent heat and simple heat – are equal at all altitudes (averaged over all latitudes).
Frank,
“Treating the atmosphere as a black box, 390 W/m2 of LWR enters the bottom and 240 W/m2 exits the top, creating a GHE of 150 W/m2. This is the prediction of the Schwarzschild eqn for an atmosphere with the existing average composition and temperature profile. An adequate description of the GHE can not be provided by adsorption alone or models based of blackbody radiation. Since the GHE is primarily concerned with the reduction in planetary radiative cooling to space caused by GHGs, this “zeroth-order approximation” ignores other important heat fluxes.”
This does seem to the crux of the disconnect and disagreement. First of all, treating the atmosphere as black box does not mean 390 W/m^2 of LWIR enters the bottom and 240 W/m^2 exits at the top (the TOA). How are you deriving that? I also do not understand how this is a prediction of the Schwartzchild eqn. either. You’ve lost me here, can you explain in more detail?
A first-order approximation would take into account the fact that the existing temperature profile can’t be maintained without a substantial upward flux of heat by convection (100 W/m2 at the surface, mostly latent heat) and downward flux of LWR (333 W/m2 at the surface). These phenomena – which are critical to maintaining a steady state – are large near the surface compared with the magnitude of the GHE, so 150 W/m2 alone isn’t a proper “first-order approximation”.”
First order approximation of what? The actual physics? No, of course not, but the +150 W/m^2 at the surface is the net aggregate effect of all the physics, radiant and non-radiant, mixed together manifesting the energy balance. Which means by definition, it includes the -100 W/m^2 non-radiant flux moved from the surface into the atmosphere and the +100 W/m^2 of LWIR from the atmosphere to the surface offsetting it (if we’re using Treberth’s numbers).
RW wrote: This does seem to the crux of the disconnect and disagreement. First of all, treating the atmosphere as black box does not mean 390 W/m^2 of LWIR enters the bottom and 240 W/m^2 exits at the top (the TOA). How are you deriving that? I also do not understand how this is a prediction of the Schwartzchild eqn. either. You’ve lost me here, can you explain in more detail?
In the zeroth order description or model for the GHE, the Schwarzschild eqn correctly calculates the observed 150 W/m2 GHE. Nothing else is required but the input of BB radiation from the surface and the composition and temperature profile of the atmosphere. To apply the Schwarzschild eqn, you divide the atmosphere up into perhaps one hundred optically thin layers, each with the temperature and GHG density found at that altitude in our atmosphere. You calculate the dI for each layer, add it to the radiation (I) that entered the layer and work your way to the TOA layer by layer. Then you integrate the spectral intensity over all wavelengths to get power. The sum of the integrated dI terms – the result of upward emission from and absorption by each layer – is -150 W/m2.
In the first-order description or model for the GHE, we recognize that the absorption of 150 W/m2 would quickly change the temperature of the atmosphere – if nothing else happened. We add SWR, DLR to OLR along with just enough convection to maintain the existing temperature profile in the atmosphere and surface energy balance. These factors maintain a steady state, but the GHE is still 150 W/m2. The Schwarzschild eqn doesn’t need to “know why” the observed temperature profile exists as a steady state, it calculates a reduction from 390 to 240 W/m2 based on what currently exists in each layer.
In the zero-order description or model, all of the phenomena essential to maintaining a steady state (except the change in OLR) are hidden in a “black box” to focus attention on the most important aspect of the GHE – the reduction in OLR between the surface and space. If this reduction didn’t occur, the planet would cool because too much radiation would escape to space.
Oh, OK. That mostly makes sense.
It seems we agree that quantitatively the GHE is about +150 W/m^2 of net surface gain.
Something has just come to my attention here that may be the source of the disconnect.
Is it clear that the 3.6 W/m^2 GW calculates for 2xCO2 via his RT simulation is that via the Schartzchild eqn. in the same way everyone else does it? That is the difference between the reduced IR intensity at the TOA and the increased IR intensity at the surface *after* the atmosphere has become more opaque to passing through radiation from the added GHGs and subsequently absorbs a little more from above and below and emits a little more upwards and downwards?
I would hope so. The spectral transmittance of the surface as GW calculates it is not directly related to the Schartzchild eqn., which if integrated from the surface through to the TOA prior to changing anything would involve how the emitted IR intensity of about 390 W/m^2 at the surface goes down to about 240 W/m^2 at the TOA.
Maybe this is main source of the confusion and disconnect? It does seem rather bizarre to me that no one here has any concept of aggregate IR transparency and opacity through the whole mass of the atmosphere, i.e. from the surface ‘looking up’ through to the TOA. Especially since the GHE itself is initially driven by the surface’s large inability to radiate freely to outer space. But it seems we never even reached agreement on this basic point either.
RW wrote: “Maybe this is main source of the confusion and disconnect? It does seem rather bizarre to me that no one here has any concept of aggregate IR transparency and opacity through the whole mass of the atmosphere, i.e. from the surface ‘looking up’ through to the TOA.”
Consider a sheet a plywood laying on the ground and emitting blackbody radiation. Do you ask what fraction of the blackbody radiation emitted by the ground passes through the wood? Its transmittance? Of course not.
Now imagine that dry ice had been sitting on top of the boards and had just been removed. The bottom of the board is much warmer than the top. 390 W/m2 of blackbody radiation enters the board from below and only 240 W/m2 exits the top. Would you say that the IR transmittance of the board is 60%? (:<))
Now imagine that the ground is colder than the air and the temperature gradient runs in the opposite direction. 10% Less blackbody radiation enters from below the board than exits from the top. Do you say that the IR transmittance of the board is 110%?
IMO, discussing the transmittance of thermal IR in all of these situations is absurd.
Now suppose we are talking about a sheet of glass laying on the ground instead of a board. Does the fact that the glass (like the atmosphere) is transparent to visible light make the situation any different? Thermal infrared passes through glass as poorly as it does through wood. Discussing the transmittance of the glass with respect to thermal infrared should be as absurd as it was with wood.
Now let's consider the transmittance of visible light through the glass. That makes perfect sense.
Now suppose we are in a glass factory and the sheet of glass is "red hot". Do we say the transmittance of red light through this glass is 500% if five times more red light leaves the top surface of the glass than enters from below? (:<O)
Conclusion: Things get confusing when we discuss the transmittance of a material when that material is warm enough to emit a significant amount of radiation at the wavelength of interest. Our personal experiences with visible light – which normally is emitted from only a few point sources – and infrared light in the laboratory – which comes from a filament at several thousand degK and overwhelms the background emission of thermal infrared by every object in the laboratory – bias our intuition about thermal infrared in the atmosphere.
FWIW, we can imagine slicing the plywood or glass into 100 thin layers and applying the Schwarzschild eqn to the infrared or visible flux passing upward through these materials – in the same way we apply it to the atmosphere. The equation will provide the correct flux coming out of the top of the material. However, in most cases, thermodynamic equilibrium between radiation and the molecules inside the material exists and we can use the shortcuts of Planck's Law and the S-B eqn in these cases.
In the atmospheric window, where emission and absorption of thermal infrared is negligible, the atmosphere behaves visible light passing through a sheet of glass. Otherwise, the atmosphere is more like thermal infrared passing through a board. As the atmosphere gets less dense at higher altitudes, it becomes more like glass/visible light and less like a board.
Photons emitted at the strongest line of CO2 have a mean free path of 10 m at 1 atm. They need to travel 500 mean free paths to escape the atmosphere. If we shaved a sheet of plywood into 500 layers, you would probably be able to see through a layer and some infrared would probably pass through too. If you travel to Venus, there is 100,000 times are much CO2 in the atmosphere. Thermal infrared travels as poorly through Venus' atmosphere as it does through a board.
The thermodynamic path that manifests the energy balance isn’t the underlying mechanism driving the GHE, and it seems no one is able to separate the two from one another. Dewitt is prime example of this. Moreover, no one even agrees that the GHE is initially driven by the surface’s large inability to radiate freely to outer space.
And Frank, yes of course it’s absurd to be discussing the transmittance of thermal infrared on those situations, because those things don’t have their temperature further elevated by absorbed thermal IR that would otherwise be transmitted through being re-radiated back downward within.
And the atmosphere is certainly not akin to thermal infrared passing through a solid board, as it’s only an infinitesimally thin gas where the bulk of the constituents that make it up don’t even emit IR. You (and many others) seem to conceptualize the atmosphere as more like a thick layer of insulation wrapped around the earth whose heat loss is slowed by conductive resistance. That would not make the GHE a radiative effect, moreover, it would violate the 2nd law of thermodynamics for the absorbed radiant energy by GHGs from the surface to further warm the surface through a conduction process like that.
This field seems to be broken beyond repair, IMO. When little to no discernible effect likely ever happens, no one will here have a clue why it seems. Even though the basics suggest only a very small push in a warming direction from added GHGs.
“This field seems to be broken beyond repair”
No, RW, it is your thinking that is broken beyond repair. Many physicists, much more knowledgeable and insightful than Frank, or DeWitt, or me have examined this carefully. For you to assume that your lack of understanding means they are all wrong is remarkably arrogant.
Mike,
You’re misunderstanding what I’m saying. The ultimate question is what is the magnitude of the effect from increased GHGs, and what is the most reasonable and logical way to go about trying to quantify it. I’m not saying the facets, i.e. the underlying physics, surrounding the fundamental question put forth are all wrong — I’m saying some key aspects surrounding them are not being applied properly to the fundamental question at hand.
Climate science is applied physics, and with any primitive field like climate, there are large degrees of freedom and nuances that are still far from settled, even though the underlying basic physics are well established. That is, it’s reasonably well established that adding GHGs should provide some push in a warming direction. But how much of push is a critical starting point that if isn’t correct, the rest is going to be way off.
As I see it, there are a few basic key things that are not agreed with:
1) That the GHE is initiated by the large inability of the surface to radiate freely to outer space.
2) That the GHE is driven mechanistically, as a underlying mechanism, by the absorption of upwelling IR from the surface (and atmosphere) acting to cool that is absorbed and subsequently re-radiated back downward towards the surface, thereby resisting radiative cooling (upward emitted IR) and requiring the lower atmosphere and ultimately the surface to be warmer than it would otherwise be.
3. That the 3.6-3.7 W/m^2 from 2xCO2 is a ‘net absorption increase’ or an incremental increase in GHG absorption. Or 100% a quantification of upwelling IR ‘instantaneously’ additionally captured by the atmosphere.
I guess if you don’t and/or can’t agree with these things, they you wouldn’t understand where I’m coming from.
RW,
“Climate science is applied physics, and with any primitive field like climate, there are large degrees of freedom and nuances that are still far from settled, even though the underlying basic physics are well established.”
The problem is that you aren’t taking issue with the nuances, you are taking issue with the well established basic physics.
“But how much of push is a critical starting point that if isn’t correct, the rest is going to be way off.”
But there is every reason to believe that the magnitude of the initial push is well established and you have given no reason to doubt that. There is good reason to doubt some of the responses to the initial push, particularly what happens with clouds.
“1) That the GHE is initiated by the large inability of the surface to radiate freely to outer space.”
That is non-controversial. I can neither agree nor disagree with 2) and 3) since I can’t figure out what you mean.
RW wrote: “The thermodynamic path that manifests the energy balance isn’t the underlying mechanism driving the GHE, and it seems no one is able to separate the two from one another.”
Although thermodynamics was invented earlier, we now know that its laws are a consequence of the quantum behavior of individual molecules and photons that are [usually] rapidly colliding. Their vibrational, rotational, and electronic energy levels are quantized. For example,entropy on a macroscopic scale is disorder on the molecular scale. YOU haven’t studied the field – statistical mechanics – that integrates molecular and macroscopic behavior.
RW: “Moreover, no one even agrees that the GHE is initially driven by the surface’s large inability to radiate freely to outer space.”
We certainly recognize that buoyancy-driven convection is driven by the surface’s inability to radiate freely to space. The GHE, however, is controlled by the temperature profile of an atmosphere. That is why the Schwarzschild eqn can calculate the 150 W/m2 GHE without any information about convection other than the stable temperature profile produced by OLR, DLR, SWR and convection. Buoyancy-driven convection does not play any role in the temperature of the stratosphere or above Antarctica.
RW: And Frank, yes of course it’s absurd to be discussing the transmittance of thermal infrared on those situations, because those things don’t have their temperature further elevated by absorbed thermal IR that would otherwise be transmitted through being re-radiated back downward within.”
The BB radiation emitted by solids doesn’t originate from the outside monolayer layer of molecules on the surface. Radiation travels inside solids too, we don’t distinguish between heat transfer in solids by conduction and radiation in radiation. Together they determine the temperature profile through a solid analogous to convection and radiation in the atmosphere. You can’t explain the physics of low-emissivity glass without infrared radiation traveling through a solid.
RW: “And the atmosphere is certainly not akin to thermal infrared passing through a solid board, as it’s only an infinitesimally thin gas where the bulk of the constituents that make it up don’t even emit IR.”
The atmosphere is made of molecules, just like solids and liquids. They all follow the same rules. If you compressed the atmosphere to the density of a typical solid or liquid (1g/cm3) – materials that are more tangible and familiar – the atmosphere would be a layer 10 m thick. The CO2 layer would be 4 mm thick – about the same the thickness as a pane of glass in a hot car sitting in the sun. A layer of PABA sunscreen micrometers thick protects us from UV radiation. The same laws of physics and chemistry apply to the atmosphere and things that are more common.
RW: “You (and many others) seem to conceptualize the atmosphere as more like a thick layer of insulation wrapped around the earth whose heat loss is slowed by conductive resistance.”
If you consider “insulation” to be anything that slows down a flux of HEAT, then insulation is a good way to describe the role of GHEs in the atmosphere. Many people assume that insulation only blocks heat flux by conduction (when they touch cold objects) and convection (when they feel a cold wind). “Insulation” is a good analogy for those broad-minded enough to include slowing down heat transfer by radiation.
RW: “That would not make the GHE a radiative effect, moreover, it would violate the 2nd law of thermodynamics for the absorbed radiant energy by GHGs from the surface to further warm the surface through a conduction process like that.”
None of the net radiative heat fluxes nor the total heat flux between the sun, atmosphere, surface and space in the K-T diagram violate the 2LoT.
RW: “This field seems to be broken beyond repair, IMO. When little to no discernible effect likely ever happens, no one will here have a clue why it seems. Even though the basics suggest only a very small push in a warming direction from added GHGs.”
Your understanding of atmospheric radiation is broken and you show no interest in repairing it. GMST is 288 K, warmer than expected for a planet without GHGs. The complicated spectrum of OLR observed from space agrees with that predicted by MODTRAN. Slight changes in that spectrum with rising CO2 have been detected. Satellites in space monitor large seasonal changes in OLR and reflected SWR, showing excellent agreement with models for OLR arising from clear skies (where a GHE exists). Clouds and snow cover are real problems for such models. Unforced variability and chaos are real problems. The absence of enhanced warming in the upper tropical troposphere may be a problem. (El Ninos are accompanied by such enhanced warming.) Climate models parameterization is a real problem. Models are incapable of putting useful constraints on ECS, a huge problem. The corruption of my profession – scientist – by climate scientists who “want to make the world a better place by telling scary stories” is a problem. The politics and religion of CAGW (alarmism, naivety about undeveloped countries and renewable energy, suppression of freedom of speech, totalitarian elites) are all problems. Let’s focus on those problems. Atmospheric radiation is not a significant problem.
Frank,
“The BB radiation emitted by solids doesn’t originate from the outside monolayer layer of molecules on the surface. Radiation travels inside solids too, we don’t distinguish between heat transfer in solids by conduction and radiation in radiation. Together they determine the temperature profile through a solid analogous to convection and radiation in the atmosphere. You can’t explain the physics of low-emissivity glass without infrared radiation traveling through a solid.”
Yes, but you’re missing my point. You were also talking about a solid opaque board — not glass. But there are significant differences, i.e. nuances, as to how energy flows through a solid or liquid compared to how it does through a gas — particularly a very, very thin gas such as the Earth’s atmosphere. The volumetric heat capacity of the atmosphere is infinitesimally thin. That’s why there are such large swings in temperature between night and day and why as soon as the Sun sets it immediately starts to cool. Moreover, the bulk of the constituents of the gas, i.e. the N2 and O2, don’t even emit radiation.
“We certainly recognize that buoyancy-driven convection is driven by the surface’s inability to radiate freely to space. The GHE, however, is controlled by the temperature profile of an atmosphere. That is why the Schwarzschild eqn can calculate the 150 W/m2 GHE without any information about convection other than the stable temperature profile produced by OLR, DLR, SWR and convection.”
The Schwartzchild eqn. does not quantify the GHE and/or does not quantify the underlying mechanism of the GHE. Before changing anything, it would deal with how the IR flux emitted from the surface of about 390 W/m^2 comes down to about 240 W/m^2 at the TOA. This would be the case whether there is GHE acting on the surface or not, and gets back to my point before regarding Dewitt not being able to differentiate a thermodynamic path manifesting a surface temperature with and without a radiative GHE acting on it.
“If you consider “insulation” to be anything that slows down a flux of HEAT, then insulation is a good way to describe the role of GHEs in the atmosphere. Many people assume that insulation only blocks heat flux by conduction (when they touch cold objects) and convection (when they feel a cold wind). “Insulation” is a good analogy for those broad-minded enough to include slowing down heat transfer by radiation.”
The question though is what is the mechanism by which *absorbed* upwelling IR radiation slows down heat transfer — or slows down the ultimate movement of surface energy into outer space.
“None of the net radiative heat fluxes nor the total heat flux between the sun, atmosphere, surface and space in the K-T diagram violate the 2LoT.”
I never claimed they did, nor do I think they do. My point is simply that the mechanism of the slowing down of surface cooling by GHGs is not by conduction or not achieved via conductive resistance, for if it were it would violate the 2nd law.
“Your understanding of atmospheric radiation is broken and you show no interest in repairing it. GMST is 288 K, warmer than expected for a planet without GHGs. The complicated spectrum of OLR observed from space agrees with that predicted by MODTRAN.”
I’ve never claimed that it doesn’t, nor have I disputed the results you keep citing of the Schwartzchild eqn. You keep thinking I’m disputing it, but I’m not.
The spectral transmittance ‘T’ and the spectral absorption ‘A’ of the surface emitted IR flux that I referred to earlier isn’t related to the Schwartzchild eqn., which deals with how the IR intensity changes as IR propagates through the absorbing and emitting layers in the atmosphere, i.e. the IR intensity decreases as the successive emission rates from the layers decreases (going upward) and increases as the successive emission rate increases (going downward). This is why the atmosphere as a whole mass passes more IR to the surface than it does into space.
Frank,
“The BB radiation emitted by solids doesn’t originate from the outside monolayer layer of molecules on the surface. Radiation travels inside solids too, we don’t distinguish between heat transfer in solids by conduction and radiation in radiation. Together they determine the temperature profile through a solid analogous to convection and radiation in the atmosphere. You can’t explain the physics of low-emissivity glass without infrared radiation traveling through a solid.”
Yes, but you’re missing my point. You were also talking about a solid opaque board — not glass. But there are significant differences, i.e. nuances, as to how energy flows through a solid or liquid compared to how it does through a gas — particularly a very, very thin gas such as the Earth’s atmosphere. The volumetric heat capacity of the atmosphere is infinitesimally thin. That’s why there are such large swings in temperature between night and day and why as soon as the Sun sets it immediately starts to cool. Moreover, the bulk of the constituents of the gas, i.e. the N2 and O2, don’t even emit radiation.
“We certainly recognize that buoyancy-driven convection is driven by the surface’s inability to radiate freely to space. The GHE, however, is controlled by the temperature profile of an atmosphere. That is why the Schwarzschild eqn can calculate the 150 W/m2 GHE without any information about convection other than the stable temperature profile produced by OLR, DLR, SWR and convection.”
The Schwartzchild eqn. does not quantify the GHE and/or does not quantify the underlying mechanism of the GHE. Before changing anything, it would deal with how the IR flux emitted from the surface of about 390 W/m^2 comes down to about 240 W/m^2 at the TOA. This would be the case whether there is GHE acting on the surface or not, and gets back to my point before regarding Dewitt not being able to differentiate a thermodynamic path manifesting a surface temperature with and without a radiative GHE acting on it.
“If you consider “insulation” to be anything that slows down a flux of HEAT, then insulation is a good way to describe the role of GHEs in the atmosphere. Many people assume that insulation only blocks heat flux by conduction (when they touch cold objects) and convection (when they feel a cold wind). “Insulation” is a good analogy for those broad-minded enough to include slowing down heat transfer by radiation.”
The question though is what is the mechanism by which *absorbed* upwelling IR radiation slows down heat transfer — or slows down the ultimate movement of surface energy into outer space.
“None of the net radiative heat fluxes nor the total heat flux between the sun, atmosphere, surface and space in the K-T diagram violate the 2LoT.”
I never claimed they did, nor do I think they do. My point is simply that the mechanism of the slowing down of surface cooling by GHGs is not by conduction or not achieved via conductive resistance, for if it were it would violate the 2nd law.
“Your understanding of atmospheric radiation is broken and you show no interest in repairing it. GMST is 288 K, warmer than expected for a planet without GHGs. The complicated spectrum of OLR observed from space agrees with that predicted by MODTRAN.”
I’ve never claimed that it doesn’t, nor have I disputed the results you keep citing of the Schwartzchild eqn. You keep thinking I’m disputing it, but I’m not.
The spectral transmittance ‘T’ and the spectral absorption ‘A’ of the surface emitted IR flux that I referred to earlier isn’t related to the Schwartzchild eqn., which deals with how the IR intensity changes as IR propagates through the absorbing and emitting layers in the atmosphere, i.e. the IR intensity decreases as the successive emission rates from the layers decreases (going upward) and increases as the successive emission rate increases (going downward). This is why the atmosphere as a whole mass passes more IR to the surface than it does into space.
RW,
“The volumetric heat capacity of the atmosphere is infinitesimally thin.”
Nonsense. Air has a heat capacity of about 29 J/mol/K. One mole occupies 24.5 L at 25 C and one atmosphere so 1.2 J/L/K. Not zero at all.
“That’s why there are such large swings in temperature between night and day and why as soon as the Sun sets it immediately starts to cool.”
Wrong. Above the boundary layer, the daily swing in T from min to max is only 2 or 3 C. It is the surface of the ground that undergoes large swings in temperature.
“Moreover, the bulk of the constituents of the gas, i.e. the N2 and O2, don’t even emit radiation.”
Which is why the swings in T are small.
I see no point in commenting on the rest of your garbage, except for one thing: You say you are not disputing the science, but you also say the science in “broken”.
Mike,
I never claimed the heat capacity was zero. It’s surely not zero.
“Wrong. Above the boundary layer, the daily swing in T from min to max is only 2 or 3 C. It is the surface of the ground that undergoes large swings in temperature.”
Yes, exactly, because the heat capacity of air is very small. The boundary layer is already very cold, so it doesn’t surprise me that its diurnal swings are a lot smaller.
“Which is why the swings in T are small.”
Where specifically? At the surface the diurnal swings are very large, hence my point.
“You say you are not disputing the science, but you also say the science in “broken”.
Correct. I’m not disputing the *basic* science which underpins the GHE or that added GHGs should provide a push in a warming direction. I am disputing or at least making the argument that the way some the basics are being applied is not really correct (though not necessarily overtly wrong either). I’m discussing fine nuances surrounding the basics.
– If there are concerns, then in addition to the temporal division of abscissa a CO2 concentration dependent division of abscissa. Then you see the benefits. This is particularly evident in an ordinate column pressure (i.e. pressure tropopause times concentration).
– The discussion about how much W / m^2 mistake for the principle.
– Each atmosphere begins at the outer edge with a radiative equilibrium – at the top with the temperature gradient 0 (except for the ozone layer), since there is no down-welling radiation is still present. (Stratosphere)
– With increasing down-welling radiation increases the temperature gradient and with sufficient temperature gradient, the temperature stratification is unstable and convection begins (Schwarzschild criterion) (troposphere).
– The transition from the stratosphere into the troposphere is the tropopause.
– In the troposphere, the temperature gradient has almost nothing to do with the radiation properties.
– The surface temperature adjusts itself so that the Schwarzschild criterion.
– With increasing CO2 concentration the tropopause height increases (decreases the pressure of the tropopause).
– If it were not for other relationships, would be at the tropopause the column pressure (pressure tropopause times concentration) constant. Other influences (falling temperature of the tropopause, low net radiation, etc.) have the column pressure rise – but less than the increase in concentration.
BTW, that no one knows what the spectral ‘T’ and spectral ‘A’ of the surface are, and subsequently what the spectral transmission and spectral absorption of the surface are — seems to be origin of the disconnect of where I’m coming from.
Does anyone know what is being referred to in this post below?:
If not, it surely explains the disconnect. The Schwartzchild eqn. deals with quantifying *incremental* changes in IR opacity and transparency from changes in GHG concentrations, but does not quantify IR opacity and transparency in the aggregate prior to making such a change. That is quantified by the spectral ‘T’ and ‘A’ of the surface, where the spectral ‘T’ is the direct ‘line of sight’ to outer space from the surface looking up, and the spectral ‘A’ is the direct ‘line of sight’ from the surface looking up that sees the atmosphere. Each is based on scaled wavelength intensity of the Planck flux emitted from the surface, but the calculation includes upwelling IR emission that originates from the layers in the atmosphere — the energy of the emitted wavelengths of which — absorb a little more of the surface spectrum than was not prior seen by the surface or subsequent layer or layers below it. Though of course once a particular wavelength intensity emitted by the surface is saturated, further absorption by a layer above in that wavelength doesn’t further increase the spectral ‘A’.
The spectral ‘T’ and spectral ‘A’, and subsequently the spectral transmission and spectral absorption, quantified in W/m^2, are fractions of the flux density directly radiated from the surface, even though a large fraction of the total absorption which quantifies ‘A’ is from that IR emitted by the atmosphere which is additionally absorbed on top of that which was absorbed by the direct surface –> component.
I provided a simplified example above here:
This is basic. That no one seems to have a clue what this is or means is truly bizarre.
RW,
You are way ahead of us. Probably you should find a blog with some people who understand atmospheric physics rather than us beginners.
Just as one comment, you have some bee in your bonnet about the “fractions of the flux density directly radiated from the surface” that is emitted to the atmosphere as a key value that no one seems able to quantify. Or have any interest in. Or know why it is so critical for understanding atmospheric radiation. Truly bizarre.
Let’s take 2 cases.
1. A hot desert in the subtropics with almost no water vapor. The surface radiation that is emitted to space might be 75% of the surface emission (I don’t have sufficient interest level – bizarre I know – in your ideas to actually look up a typical value).
In this case, T=0.75
2. A hot tropical ocean with of course massive amounts of water vapor close to the surface. The surface radiation that is emitted to space might be 1% of the surface emission.
In this case, T=0.01.
The OLR in both cases could be the same.
SoD,
“You are way ahead of us. Probably you should find a blog with some people who understand atmospheric physics rather than us beginners.”
No, I wouldn’t say that. Not at all. But it is genuinely perplexing to me that no one here seems to have the slightest clue what GW is quantifying as ‘T’ and ‘A’ when according to him it’s the most basic calculation that can be performed in Modtran with HITRAN line data. Forgive me if I find this bizarre.
“Just as one comment, you have some bee in your bonnet about the “fractions of the flux density directly radiated from the surface” that is emitted to the atmosphere as a key value that no one seems able to quantify. Or have any interest in. Or know why it is so critical for understanding atmospheric radiation. Truly bizarre.”
Yes — absolutely, but it seems no one even knows what it is, let alone what it is actually measure of. Let me ask you, do understand that the surface spectral ‘T’ I’m referring to is NOT what some might refer to as ‘direct surface transmittance’, i.e. that which is solely the direct surface IR –> TOA component which is transmitted straight through into outer space?
BTW, that value is truly trivial and is of no practical use whatsoever. For understanding atmospheric radiative transfer or really much of anything else for that matter.
“1. A hot desert in the subtropics with almost no water vapor. The surface radiation that is emitted to space might be 75% of the surface emission (I don’t have sufficient interest level – bizarre I know – in your ideas to actually look up a typical value).
In this case, T=0.75
2. A hot tropical ocean with of course massive amounts of water vapor close to the surface. The surface radiation that is emitted to space might be 1% of the surface emission.
In this case, T=0.01.
The OLR in both cases could be the same.”
How are you defining ‘T’ in these cases? I surmise you’re defining it as solely the direct surface IR –> TOA component which is transmitted into space. Again, I agree that value has no useful meaning at all.
The surface spectral ‘T’ and ‘A’ I’m referring to and that GW is quantifying is not that, but is that which includes emission from the layers in the atmosphere which is additionally absorbed as well. Again, it’s based on scaled wavelength intensity of the Planck Flux emitted from the surface. These ‘T’ and ‘A’ values are a direct quantification of aggregate IR transparency and IR opacity through the whole mass of the atmosphere before making any changes like adding GHGs. The IR emission from each subsequent layer above the surface further increases the spectral absorption ‘A’ and decreases the spectral transmission ‘T’. The final values are after the surface and all the subsequent layers above it have had their effect on ‘T’ and ‘A’.
The final calculated spectral absorption ‘A’ and spectral transmission ‘T’ in W/m^2 are always fractions of the flux density radiated from the surface, even though much of the attenuated IR flux quantifying ‘A’ is that which originates from the atmosphere which is additionally absorbed. This is required to satisfy COE (assuming the surface emissivity is 1).
Part of the problem is Trenberth shows a direct surface –> TOA component transmission into space of 40 W/m^2 but cannot provide a reference for the value when asked, and certainly cannot show the line by line calculations to support it. GW calculates that this value is significantly higher, because he calculates a global average spectral ‘T’ of around 0.24, and this value includes emission from the atmosphere which is additionally absorbed. Now maybe you can argue his value is wrong, but he can show the detailed LBL calculations to support it, and no one else seems to be able to. A value of around 0.24 seems far more reasonable to me than something less than 0.10, because the transparent region of the absorption spectrum, i.e. the so-called ‘atmospheric window’, lies in a very strong part of the emitted surface spectrum.
If you have no idea what GW is quantifying as ‘T’ and ‘A’, then it logically follows that you would have no idea what I’m talking about or where I’m coming from.
BTW, I recall Grant Petty referred to it as ‘the spectral transmittance evaluated at the temperature of the surface’. GW just refers to as the transmittance of ‘surface power’ or the transmittance of surface emissions, because that’s effectively all it is since the atmosphere creates no energy of its own.
I asked Gunnar Myhre if he had a value for the global average ‘spectral transmittance evaluated at the temperature of the surface’. He didn’t have a value for it, but he knew what it was.
You mention somewhere in your tutorial that *after* the atmosphere becomes more opaque to IR and subsequently absorbs a little more from above and below and emits a little more upwards and downwards, for the upwelling emission factor there needs to be a direct surface –> TOA component, a layer one –> TOA component, a layer 2 –> TOA component, and so on so forth via all the layers from each separate layer from its through to the TOA to calculate the total reduction in IR intensity at the TOA.
The spectral ‘T’ and ‘A’ I’m referring to I believe is a similar fundamental calculation, but it’s for the total prior to their being any change, scaled or weighted by the Planck Flux emitted by the surface (if that makes any sense).
Another key component of concept here with this is once the energy of a particular wavelength emitted by the surface is absorbed by the direct surface — TOA component or by the subsequent emission from any layer above it, further absorption of those wavelengths by layers above no longer increases the spectral absorption ‘A’. This would be the point at which the energy of the wavelengths emitted by the surface is 100% absorbed or saturated. So the increased attenuation by the emission from the subsequent layers above the surface is only incremental in how it further increases the spectral absorption ‘A’.
Another key point I want to make clear here is you are all correct in that you do not need to know the spectral ‘T’ and ‘A’ in order to calculate an incremental IR opacity change. Everyone is understandably focused pretty much only on incremental effects. However, relative to what that final calculated incremental IR opacity change actually means in physical terms is important, or is at least critical to the fundamental point I’ve been trying to get it. Or at least I’m arguing the case that it is.
I recall GW mentioned or referred to the 3.6 W/m^2 from 2xCO2 as being the net decrease in IR flux passing through the ‘transparent window’, and you didn’t know what he meant by that. Well, if you don’t know that the spectral ‘T’ more or less quantifies the ‘transparent window’, i.e. the transparent region of the absorption spectrum, or the fraction of the Planck weighted surface spectrum that is transmitted into space, it wouldn’t make any sense.
We all know in reality the 3.6 W/m^2 for 2xCO2 is not the total IR flux reduction at the TOA — and that it’s actually more than 3.6 W/m^2, or the spectral absorption increase is more than 3.6 W/m^2. But since you are required to subtract the increased IR flux intensity at the surface from the reduced at the TOA (to conserve energy) to arrive at the net opacity increase, he’s considering the 3.6 W/m^2 to be the net decrease in IR power passing through the ‘transparent window’ or equivalent to a 3.6 W/m^2 decrease in the spectral transmission.
Or even more simply just +3.6 W/m^2 of incremental GHG absorption.
RW – Everything I have discussed above involves the interactions between radiation and molecules. It doesn’t matter whether the molecules are inside a board that you call “opaque” or glass that you call “transparent” or the atmosphere that might be characterized as transparent or opaque, depending on the wavelength of interest. With the exception of phenomena of scattering/reflection at surfaces, the physics is the same in all cases – whether you call them “opaque” or “transparent”. A board is opaque; a sheet of paper is semi-transparent (to a strong visible light source). The paper is made from ground up boards!
The only differences arise from the absorption cross-section (o), the density of absorbing molecules (n) aka GHGs in the atmosphere, and the temperature. The Schwarzschild eqn works whenever molecules have a properly defined temperature (ie they collide more frequently than they do anything else.) If scattering is important, terms are added.
Some common simplifications that do NOT apply to thermal radiation in the atmosphere: When the temperature is low enough or the wavelength is long enough, the emission term becomes negligible. When radiation reaches thermodynamic equilibrium with the surrounding molecules, it will have blackbody intensity.
But you’re not differentiating the physics and dynamics of a radiatively induced GHE acting on surface and those that are not. That’s what I’m discussing. The fact there are significant shared elements of fundamental physics in the scenarios you present doesn’t directly have anything to do with the underlying physics of the GHE, and is my entire point.
RW,
No one knows what you are discussing.
You cannot articulate your ideas so that anyone can understand what your question actually is.
Myself and the others responding to you (including the late Pekka) believe that your understanding of this complex subject is badly flawed. This is why your questions are mysterious.
Often you state ideas that everyone else already believes with the “why don’t you guys understand this?” – but we actually do. Other times you state ideas that are clearly flawed combined with ideas that are clearly correct. Usually you repost your combination of correct, flawed, confused and mysterious ideas multiple times in half page / full page essays of confusion.
I’m happy that you are convinced we don’t understand the basics of the subject. Many many people have come here and reached the same conclusion.
For my part, I’m happy that your lack of understanding of physics is massively outweighed by your boundless confidence in your own superior grasp of the subject.
Please, don’t post any more.
Or next time you post a comment include an equation for review. A novel idea for you. A requirement, if you will.
The fact that you can’t write an equation is the essence of your inability to articulate the problem.
Teach yourself to articulate your ideas as equations. Read a few textbooks (I previously requested this before the most recent 3,562 comments of yours). And then return. It will be time well spent.
Please don’t rewrite your last 50 comments in another series of 8 comments spanning 4 pages, trying once again to get us to grasp the message we have failed to grasp. There is no point. We won’t get it.
Please.
Alright fair enough. Let me see if I can get or figure out the specific equations for the spectral ‘T’ and ‘A’, but it must be some kind of integral that aggregates the surface Planck weighted wavelength absorption from the direct surface emission and the subsequent emission of the layers above it — from the surface through to the TOA and from each subsequent layer above the surface from its layer through to the TOA.
BTW, there is no specific eqn. that describes the underlying physics of the GHE, and that’s what makes it hard to convey and discuss with others.
BTW,
Out of curiosity, how would you write the formula for this as I have described it above? Or do you not understand what I’m even describing? Or what GW is describing?
I believe the concept is that of optical depth through the whole of a medium or in this case through the whole of the atmosphere from the surface looking up towards space:
https://en.wikipedia.org/wiki/Optical_depth
What isn’t transmitted, must be absorbed; hence A = 1-T
It would be the spectral optical depth through the whole atmosphere, including emission from the atmosphere, weighted by the Planck flux emitted directly by the surface.
Here are some of GW’s own words that seem to get at the crux:
“You need to analyze multiple layers to determine what the optical depth is. This is because each layer has unique temperature, pressure and GHG concentrations, all of which affect the final T and A. T and A are aggregate values and it all boils down to the nature of equivalence. If X W/m^2 enters the bottom of the atmosphere (from the surface) and Y leaves the top (the TOA), T = Y/X even though the calculation of Y is very complex. In the modeling world, you only do the most complex calculations as few times as you can and leverage aggregate results for subsequent calculations.”
The final ‘T’ and ‘A’ values are an aggregate sum of the upwelling IR emitted by surface and all the layers which quantifies the optical depth ‘looking up’ through the whole atmosphere weighted by the Planck spectrum emitted by the surface (which is itself a specific flux density, dictated by S-B law). Hence the term ‘spectral transmittance evaluated at the temperature of the surface’ or ‘spectral absorptance evaluated at the temperature of the surface’
Maybe you can offer some assistance as to how this is written in equation form? If you still have no clue what this is, let me know and I’ll go do more homework. I do see your point about needing to express certain things in equation form.
BTW, I’m assuming I’m not being put on here, and that all of you really don’t know what this is.
From Wikipedia:
“In physics, optical depth or optical thickness, is the natural logarithm of the ratio of incident to transmitted radiant power through a material, and spectral optical depth or spectral optical thickness is the natural logarithm of the ratio of incident to transmitted spectral radiant power through a material.[1] Optical depth is dimensionless, and in particular is not a length, though it is a monotonically increasing function of path length, and approaches zero as the path length approaches zero.”
The ‘T’ and ‘A’ I’m referring to quantify the ‘spectral optical depth’ through the whole medium of the atmosphere (i.e. from the surface all the way through the TOA):
https://en.wikipedia.org/wiki/Optical_depth#Spectral_optical_depth
Schwarzschild equation and Schwarzschild criterion are 2 different things.
There is no saturation, because where is strongly absorbed, is also strongly emitted.
RW,
You are really being annoying.
See my earlier comment:
Yes, everyone commenting here knows what optical thickness is and what the equation is. You can read much earlier posts where I have explained what it is and how to calculate it.
So we are all clear. Everyone knows what optical thickness is including you and so there are no questions.
If you have another question be sure to produce an equation otherwise I will delete your comment.
By the way, when you say:
– this is something different from optical thickness.
Your lack of understanding of physics basics is not our problem.
Your confusion about the difference between “optical thickness” and “spectral optical depth through the whole atmosphere, including emission from the atmosphere, weighted by the Planck flux emitted directly by the surface” is not our problem.
Be sure to produce your equation. Otherwise your comment will be deleted.
By the way, if you can ever produce an equation to express your “idea” – which I doubt – at that point you will almost certainly realize what an outstanding numbskull you have been for the last 4,267 comments.
SoD,
“You are really being annoying.”
I didn’t mean the generic concept of ‘optical depth’ or ‘optical thickness’, but how it applies to the physical meaning of what I was referring to.
“Everyone knows what optical thickness is including you and so there are no questions.”
Then I don’t get why no one understands what I’m talking about. I only persisted in trying to explain it from so many angles because no one understood (and/or overtly objected or disagreed). If everyone would have just said, yes I agree — then this entire line of dialog would have never occurred.
“If you have another question be sure to produce an equation otherwise I will delete your comment.”
I would like to actually know the equation that describes this first, before I try to ask another question or move on to some other nuance. Why don’t you help me understand how to formulate it? I have a conceptual idea, but admittedly I don’t quite know how to do it. Seriously, I would like to know it. Why don’t you take me through it?
I agree with you that I need to be able to articulate my ideas in equation form, or least start with an equation as a laying of ground work before getting into nuances or discussing more applied physics concepts.
“– this is something different from optical thickness.
Your lack of understanding of physics basics is not our problem.
Your confusion about the difference between “optical thickness” and “spectral optical depth through the whole atmosphere, including emission from the atmosphere, weighted by the Planck flux emitted directly by the surface” is not our problem.”
Of course, what would be referred to as the ‘optical thickness’ would be the ‘A’ component. I believe it can be characterized as the ‘absorption coefficient’ (extinction coefficient, i.e. the extinction due to the absorption of monochromatic radiation) scaled by the wavelength intensity of the Planck spectrum emitted by the surface.
What I don’t quite know how to do (admittedly) is formulate this all into an equation that aggregates the emission from the surface and the layers above it into the final ‘A’ (and subsequently the final ‘T’), or the optical thickness through the whole, i.e. from the surface and layers looking up towards space.
“By the way, if you can ever produce an equation to express your “idea” – which I doubt – at that point you will almost certainly realize what an outstanding numbskull you have been for the last 4,267 comments.”
I’m not sure how to respond to this, frankly. I think it’s best I say nothing…
RW wrote above: Here are some of GW’s own words that seem to get at the crux:
“You need to analyze multiple layers to determine what the optical depth is. This is because each layer has unique temperature, pressure and GHG concentrations, all of which affect the final T and A. T and A are aggregate values and it all boils down to the nature of equivalence. If X W/m^2 enters the bottom of the atmosphere (from the surface) and Y leaves the top (the TOA), T = Y/X even though the calculation of Y is very complex. In the modeling world, you only do the most complex calculations as few times as you can and leverage aggregate results for subsequent calculations.”
The final ‘T’ and ‘A’ values are an aggregate sum of the upwelling IR emitted by surface and all the layers which quantifies the optical depth ‘looking up’ through the whole atmosphere weighted by the Planck spectrum emitted by the surface (which is itself a specific flux density, dictated by S-B law). Hence the term ‘spectral transmittance evaluated at the temperature of the surface’ or ‘spectral absorptance evaluated at the temperature of the surface’
Maybe you can offer some assistance as to how this is written in equation form? If you still have no clue what this is, let me know and I’ll go do more homework. I do see your point about needing to express certain things in equation form.
This equation you are seeking is the S***********d eqn:
dI = emission – absorption
dI = n*o*[B(lambda,T)]*dz – n*o*I*dz
I (spectral intensity), n (density of GHG), T and o (absorption cross-section) all vary with altitude, i.e. they formally should be written I(z), n(z), T(z), and o(z). If we integrate the left hand side of this equation from I(z=0) to I(z=TOA), we obtain the change in upward flux (spectral radiance) passing through the atmosphere at a particular wavelength. When you integrate these changes in spectral radiance over all wavelengths, you get the GHE, 150 W/m2. You can think of dI and dz as infinitesimal changes in spectral intensity and altitude OR as the change in spectral intensity (delta_I) passing through a layer of thickness delta_z. The layers need to be thin enough that I, T, o, and n can be treated as a constant within the layer. Since n, o, I and B(lambda,T) vary with altitude, there is no analytical solution to the differential form of the equation. However, if the temperature is low enough that the emission term can be neglected and if n and o were constants, we get an expression we can integrate:
dI/I = -n*o*dz
ln I_2/I_1 = -n*o*(z_2-z_1) = tau (optical depth/absorbance)
This is the logarithm relationship you keep looking for – Beer’s Law. When n and o vary with z, then:
tau = Integral {-n(z)*o(z)*dz} from z1 to z2.
and the integration usually needs to be done numerically.
tau = Sum from n layers { n(z)*o(z)*dz }
where dz is now “delta z” the thickness of each layer. In traditional mathematical notation (which I can’t type as a comment) “sum of n layers” is written in a capital sigma summation notation.
HOWEVER, in our atmosphere, where the emission term can not be neglected, you can’t divide by I and get all of the terms with I on one side of the equation. Numerical integration is still possible:
I(z=TOA) – I(z=o) = Sum from n layers { n*o*[B(lambda,T)]*dz – n*o*I*dz }
I(z=TOA) – I(z=o) = Sum from n layers { n(z)*o(s)*[B(lambda,T(z))]*dz }
– Sum from n layers { n(z)*o(z)*I(z)*dz }
Change in flux = sum of all photons emitted – sum of all photons absorbed
(When writing this as “two summations”, I wrote I(z), n(z), T(z) and o(z), but used I, n, T and o so everything could fit on one line when writing it as “one summation”.)
You are interested in the “two summations” version of this equation: the spectral radiance of all of the photons being absorbed (spectral A). You (and apparently George White) often forget to mention that this term is subtracted from the spectral radiance of all of the photons being emitted. This is why every time you discuss absorbance or transmittance you run into trouble. We integrate both equations over all wavelengths to get the change in flux. If I remember correctly, DeWitt once calculated that about 10-fold more photons are absorbed than enter the atmosphere from below. Most of the photons absorbed by the atmosphere were emitted by the atmosphere, not the surface.
MODTRAN does these summations for you using the one summation equation. For OLR, “looking up” from z=0 to your choice of z. For DLR, “looking down” from z=TOA to your choice of z. (You can get the change in flux from any z1 to z2 in two steps.)
It is more difficult to do the computations when you use the “two summation” equation. For the strongest CO2 band, the mean free path of a photon is roughly 10 m near the surface. To add up 90% of the photons absorbed, you need to use a layer 1 m thick (70,000 of them to get to the TOA used by MODTRAN). 99% accuracy requires layers 0.1 m thick. In the first 10 m, however, the number of photons of this wavelength absorbed is equal to the number emitted – the atmosphere is in thermodynamic equilibrium with the radiation of this wavelength passing through it. That equilibrium intensity is what Planck’s Law calculates – the intensity of radiation in an equilibrium with the gas it is passing through where absorption = emission. In other words, at this wavelength all of the dI terms being summed are zero in the troposphere. We don’t need to use layers 10 cm thick to sum up two terms that cancel each other UNLESS we are trying to find the sum of all the photons absorbed and subtract it from all of the photons emitted. You want to know the total absorbed, so you must used very thin layers. If all you need to know is the CHANGE in flux between z1 and z2, you can get away with using layers about 100 meters thick near the surface, rather than 0.1 m thick.
It may be difficult to understand how the Schwarzschild eqn can combine absorption of radiation that is traveling in a specific direction with emission of radiation in all directions by GHGs. Although photons are traveling in all directions, we analyze the power they transmit parallel and perpendicular to the earth’s surface. For emission from a layer of atmosphere, the components of the flux in the x- and y-directions cancel and don’t contribute to warming or cooling the planet. Only the component of the flux in the z-direction from a layer is needed, and this emission from any layer adds equally to both OLR and DLR. The same is true for the flux absorbed; only the component in the +z or -z directions matters. This is called the “plane-parallel approximation” – an approximation because the earth’s surface is a sphere not a plane. Planck’s Law gives spectral radiance (W/m2/Hz/sr) in all directions and Lambert’s cosine law allows us to convert it into spectral radiosity (W/m2/Hz), the flux parallel to a plane. (I don’t always use this terminology correctly.)
Frank,
I appreciate the immensely detailed reply, and it will take me quite a while to sort through it all; however I don’t think what you’re describing is what GW is quantifying. The ‘A’ component he quantifies as being around 0.76 for a global atmosphere and the ‘T’ component, i.e. 1-A, is around 0.24. Converted into flux densities where the surface is at a temperature where it emits 400 W/m^2, the ‘A’ would be about 300 W/m^2 and the ‘T’ would be about 100 W/m^2 — not the 150 W/m^2 you’re citing.
I understand the Schwartzchild eqn., i.e. dI = emission – absorption, deals with how the IR intensity changes IR moves through the absorbing and emitting layers of the atmosphere. Directionally, the IR intensity increases when the absorbed intensity is less than the emitted intensity, and the IR intensity decreases when the absorbed intensity is more than the emitted intensity. So from the surface looking up towards space this would involve and quantify how the 390 W/m^2 of IR intensity at the surface is brought down to about 240 W/m^2 at the TOA.
Maybe the source of the disconnect here is the field doesn’t quantify ‘optical thickness’ (and subsequently optical transparency) through the whole of the atmosphere as the sum of ‘T’ and ‘A’ components as GW does?
This was meant to say the following:
“I understand the Schwartzchild eqn., i.e. dI = emission – absorption, deals with how the IR intensity changes as IR moves through the absorbing and emitting layers of the atmosphere.”
” Directionally, the IR intensity increases when the absorbed intensity is less than the emitted intensity, and the IR intensity decreases when the absorbed intensity is more than the emitted intensity.”
BTW, I understand this is why the atmosphere as a whole mass passes substantially more IR to the surface than it does to into outer space. A ratio possibly as high a 2 to 1. It’s driven by the lapse rate. i.e. emission rate decreasing with height.
Frank,
“It may be difficult to understand how the Schwarzschild eqn can combine absorption of radiation that is traveling in a specific direction with emission of radiation in all directions by GHGs. Although photons are traveling in all directions, we analyze the power they transmit parallel and perpendicular to the earth’s surface. For emission from a layer of atmosphere, the components of the flux in the x- and y-directions cancel and don’t contribute to warming or cooling the planet. Only the component of the flux in the z-direction from a layer is needed, and this emission from any layer adds equally to both OLR and DLR. The same is true for the flux absorbed; only the component in the +z or -z directions matters.”
I do not find this difficult to understand or conceptualize at all.
At this point, we need at least a rough equation that quantifies the ‘optical thickness’ of the whole atmosphere, i.e. from only the perspective of the surface looking up all the way through to the TOA. I believe that is what GW is quantifying as his ‘A’ component.
“At this point, we need at least a rough equation that quantifies the ‘optical thickness’ of the whole atmosphere, i.e. from only the perspective of the surface looking up all the way through to the TOA.”
SoD,
I totally agree with your characterization of ‘optical thickness’ through the whole for the ‘A’ component, and I will use that term from now on to avoid any confusion. I was using the term ‘aggregate IR opacity’ through the whole, which to me means the same thing, but to others probably not. None the less, I totally agree it’s ‘optical thickness’ (and subsequently optical transparency) — or what might be called ‘line of sight’, that I’m referring to.
But we need (I need) an equation to be able to express it. How do we express that there is significant absorption overlap in the saturated bands? That is, the effect the emission from the layers above the surface have in increasing the ‘optical thickness’ through the whole is only incremental, or only further increases ‘A’ if its absorbed wavelengths are not already saturated (i.e. already fully absorbed by the direct surface emission or the combination of the direct surface emission and the emission from the layers below it).
I believe this is covered here:
Where you discuss Beer-Lambert law, Absorption of radiation, Optical thickness and transmittance:
“Optical thickness, usually written as τ, is the property inside the exponential in equation [1].
τ = ∫σn(s).ds [4]
where the integral is between the limits s1 and s2
Transmittance, usually written with a weird T symbol not available in WordPress, but with “t” here, is the amount of radiation “getting through” along the path we are interested in.
t(s1,s2) = exp [-τ(s1,s2)]………The transmittance can be a minimum of zero – although it can never actually get to zero – and a maximum of 1. So it is simply the proportion of radiation at that wavelength which emerges through the section of atmosphere in question:”
The path is defined here as that from the surface all the way through the TOA, but let’s stick to a simple 2 layer atmosphere as an example for maximum simplicity.
RW,
Like most of your preceding 4000+ comments I can only guess at your intent.
“How do we express that there is significant absorption overlap in the saturated bands?”
This is exactly covered by the equation of absorption/transmission. Take out a calculator and put numbers in and you will see. If you don’t have a calculator, google will calculate exp(-5), exp(-10), exp(-20) for you.
Not understanding maths is a tiny handicap in this subject. And even stranger to explain given that you are the smartest guy in this discussion.
For the rest of your comment I have no idea what you are asking. Write an equation.
You can say: I believe y = f(x). Is this true? Obviously you need to write f(x) as something like x2 or ∫exp(-x)dx.
If you think “z = for each monochromatic wavelength, the sum of fluxes from each layer and z1 = the sum of z over each wavelength” then your equation will be a double integral with the inside integral over z (height) and the outside integral over λ (wavelength).
If you can’t think like this go and read a few textbooks. At the same time recognize that telling those who are able to think like this that they don’t understand the subject is like explaining the defects in Chinese poetry to Chinese people when you yourself only speak English.
And your guru (GW) was also unable to either confirm or deny that he accepted the Schwarzchild equation. If accept, then he would have denied his own equation. If denied, then he would have had to explain how this Nobel prize winner was wrong.
You only think he has something to add to this discussion because you don’t understand that equations are what define an idea, not some kind of nice to have extra for people not comfortable with “real explanations”. So please stop citing his ideas. His (GW’s) ideas have at their heart a refutal of the Schwarzchild equation – because he (GW) doesn’t understand how radiative transfer works.
Stop posting comments, get a textbook on something like Engineering Mathematics and start studying it. Come back in 3 months. When you can see how to write derivatives and integrals to explain an idea in your head then you will be able to converse. It is not intuitive. It takes some training.
You fail physics and engineering if you cannot write an idea as an equation.
SoD,
“This is exactly covered by the equation of absorption/transmission. Take out a calculator and put numbers in and you will see. If you don’t have a calculator, google will calculate exp(-5), exp(-10), exp(-20) for you.
Not understanding maths is a tiny handicap in this subject. And even stranger to explain given that you are the smartest guy in this discussion.”
I do not think I’m the smartest guy in the discussion; however, I do think I’m far smarter than you realize. I just don’t have formal training. And yes, I have not practiced any maths in over 20 years and I’m way, way rusty on all of it to say the least. I also never took calculus.
“You can say: I believe y = f(x). Is this true? Obviously you need to write f(x) as something like x2 or ∫exp(-x)dx.
If you think “z = for each monochromatic wavelength, the sum of fluxes from each layer and z1 = the sum of z over each wavelength” then your equation will be a double integral with the inside integral over z (height) and the outside integral over λ (wavelength).
If you can’t think like this go and read a few textbooks.”
I can think like this and can conceptually understand what you are saying very easily. It’s expressing it (correctly) in an equation that I find difficult, because it’s not something I’ve practiced in a couple decades.
“At the same time recognize that telling those who are able to think like this that they don’t understand the subject is like explaining the defects in Chinese poetry to Chinese people when you yourself only speak English.”
I never claimed you or anyone don’t understand the subject; however, there are many facets and nuances surrounding the subject. I believe I’m only discussing and trying to get at nuances or applied physics concepts — and I’m not disputing any fundamental physics underpinnings (of atmospheric radiative transfer or anything else).
“And your guru (GW) was also unable to either confirm or deny that he accepted the Schwarzchild equation. If accept, then he would have denied his own equation.
You only think he has something to add to this discussion because you don’t understand that equations are what define an idea, not some kind of nice to have extra for people not comfortable with “real explanations”. So please stop citing his ideas. His (GW’s) ideas have at their heart a refutal of the Schwarzchild equation – because he (GW) doesn’t understand how radiative transfer works.”
I do not understand where this is coming from at all. The way GW calculates 3.6 W/m^2 of ‘net absorption increase’ via his RT simulation is with the Schwartzchild eqn. That is, it’s the difference between the reduced IR intensity at the TOA and the increased IR intensity at the surface. So why would you say or think he’s denying the eqn. or doesn’t understand how radiative transfer works when he himself is not only using the eqn., but is getting the same answer as everyone else in the field? Your assessment of him doesn’t logically follow from this.
“Stop posting comments, get a textbook on something like Engineering Mathematics and start studying it. Come back in 3 months. When you can see how to write derivatives and integrals to explain an idea in your head then you will be able to converse. It is not intuitive. It takes some training.”
Yes, it does.
In short, GW’s ‘A’ of 0.76 is just the IR optical thickness through the whole mass of the atmosphere from the perspective of the surface and all the subsequent layers above the surface ‘looking up’ towards the TOA — *before* changing anything (like adding GHGs). That is, in a condition of steady-state or energy balance. Nothing more.
The 3.6 W/m^2 he calculates from 2xCO2 is *NOT* a 3.6 W/m^2 increase in that optical thickness. It is calculated via the Schartzchild eqn. the way everyone else does it and is the difference between the reduced IR intensity at the TOA and the increased IR intensity at the surface.
Maybe I’ll try to sum up the issue I’m trying to get at one more time, but I want pledge not to return until I have the equation for the optical thickness through the whole of the atmosphere. Because again, that’s all GW’s ‘A’ and subsequently his ‘T’ (optical transparency through the whole of the atmosphere) quantifies.
SoD,
“His (GW’s) ideas have at their heart a refutal of the Schwarzchild equation – because he (GW) doesn’t understand how radiative transfer works.”
BTW, where are you getting this from? It doesn’t logically follow from my own communication from him. In fact, it directly contradicts it. GW is using the standard Schartzchild equation and protocol for his RT simulation. I know, because I’ve specifically asked him. Moreover, he’s getting the same answer as those in the field do. His div2 analysis doesn’t deal with what the Schwartzchild equation quantifies, i.e it doesn’t deal with how IR intensity changes as it moves through the absorbing and emitting layers of the atmosphere (both directionally downward through to the surface and upwards through to the TOA), and isn’t attempting to quantify that in any way at all. If it were, it would be obviously wrong — spectacularly so.
The div2 analysis deals with the notion of adaption of the system to an energy imbalance and how the rules of linearity *should* apply to such an imbalance. It also deals with the underlying physics driving the GHE, which are largely independent of the thermodynamic path actually manifesting the energy balance, and how those underlying physics affect the adaption. In particular, to that of GHG absorption compared to post albedo solar power entering the system. It does not deal with what the Schartzchild eqn. itself quantifies or predicts will occur.
RW,
See these comments and finally where I gave up here.
A quick citation from your guru:
“1. The atmosphere does not emit photons according to its temperature. Instead it emits photons that it absorbed. Therefore the temperature of the atmosphere is irrelevant in emission of thermal radiation.”
I’m not going to repeat it all. You couldn’t understand the points then and probably you won’t understand the points now.
It’s basic. It’s wrong.
Can I ask how you are defining or more specifically how you’re measuring temperature? With a thermometer? I don’t interpret what he’s saying the same way you do; moreover, how do you explain he’s getting the correct results using the Schartzchild eqn.?
Now, if he’s saying the subsequent emitted spectrum (post absorption) is different than the one predicted by the Schartzchild eqn. — that’s one thing, but he’s not. You only think that’s what he’s saying. He’s arguing nuance about what really makes up ‘measured’ temperature — not how absorption of IR affects the emission spectrum and how the IR intensity changes as it moves through the absorbing and emitting layers of the atmosphere.
I guess I could totally understand your dismissal if he was claiming and getting different results than those in the field, but he isn’t. The results are the same because he’s following standard protocol for these simulations. His simulation has substantially more DLR than OLR originating from the atmosphere, just as the Schartzchild eqn. predicts with emission decreasing with height. You can’t calculate the correct net change in absorption of 3.6 W/m^2 if your not doing all this correctly.
The div2 analysis is something completely different from all of this, and which deals with how to properly apply the final calculation via the Schwartzchild eqn. to the system — not to refute the foundation behind the calculation itself, which is fully agreed to be correct. But you’re certainly not alone, as no one anywhere seems to have the slightest clue what he’s doing there.
Frank,
I think I found the post to which you were referring. In fact more than ten times more photons are absorbed in the first 100 meters of the atmosphere, (5,000W/m²) than are emitted from the surface. And that’s just the upward vertical flux. The total number of photons emitted and absorbed, including absorption of photons from the surrounding atmosphere, in a 100m vertical column of the atmosphere, with a base area of 1m² is much, much larger than that.
Thanks DeWitt. I never had looked at MODTRAN Raw Model Output before. With the US Standard Atmosphere and default GHGs, “average transmittance” was 89% through the first 0.001 km (1 m) of the atmosphere, the shortest distance that seemed to work. For the first 0.01 km (10 m), the “average transmittance” was 74%, not (89%)^10 = 31%. If I’m thinking clearly, about 40% of OLR at 10 m is emission from the atmosphere, not the surface. If atmospheric pressure is 10^4 kg/m2 and the density of the air at STP is 1.275 kg/m3, the first meter of atmosphere contains 0.0128% of its mass (and therefore ability to absorb photons). If absorbance is 11% in the first 1 m, then “total absorbance” would be 862%. “Total emission” by the atmosphere would be about 800% of surface emission. These calculations are based on the assumption that emission by the first 1 m of the atmosphere is negligible, which isn’t correct for the most strongly absorbed wavelengths. MODTRAN produces nonsense when asked to look down from an altitude of 0.0001 km (and there is no guarantee that it was programmed correctly to deal distances as short as 0.001 km.)
So I come up with a figure of about 10-fold again.
“1. The atmosphere does not emit photons according to its temperature. Instead it emits photons that it absorbed. Therefore the temperature of the atmosphere is irrelevant in emission of thermal radiation.”
Wrong
An atmospheric region emits in LTE of course according to its temperature.
As the temperature is created (absorption, convection) is irrelevant.
The operative point here is whatever is mechanistically happening on a more micro level, the rate of emission decreases with height regardless. That is, measured temperature and thus emission rate decreases with height as is directly observed. This is why regardless of whatever the dominant mechanism of emission is the Schwartzchild eqn. will calculate and predict the correct final result. GW never claimed the gas is non-LTE. In fact, he claims it is LTE and this is why the predicted spectrum is the same; and thus why the Schartzchild eqn. gets the correct results either way. All he’s really saying or claiming is that it is not emitting according to its temperature, which generally implies as being a direct result of how fast the molecules are moving — in the same way it does in liquid or solid. Nothing more.
Let me try to state the fundamental issue I’m trying to get at here as clearly and concisely as I can..
[moderator’s note – as promised, more restating of whatever it is without equations to clarify has been deleted. Interested commenters can refer to the previous 4000+ comments from RW]
Again, GW’s div2 analysis deals with the notion of adaption of the system to an energy imbalance and how the rules of linearity *should* apply to such an imbalance. It also deals with the underlying physics driving the GHE, which are mostly independent of the thermodynamic path actually manifesting the energy balance, and how those underlying physics affect the adaption; and subsequently how they affect a linear increase in that adaption. In particular, to that of GHG absorption compared to post albedo solar power entering the system. It does not deal with what atmospheric radiative transfer, Schartzchild eqn., etc. quantifies or predicts will occur, which is fully agreed to be correct.
“[moderator’s note – as promised, more restating of whatever it is without equations to clarify has been deleted. Interested commenters can refer to the previous 4000+ comments from RW]”
Alright, but I thought I came up with some pretty clear language to describe the issue using terms that have now been agreed with to mean a specific thing. Let me work on coming up with the equation for the optical thickness through the whole of multi-layer atmosphere. Again though, even if I can — the issue I’m getting at, i.e. the underlying physics driving the GHE, cannot be expressed by it or any equation. It is really only an applied physics concept. And which BTW, is why I think so many people find understanding and internally conceptualizing the physics of the GHE so difficult.
The origin of the disconnect — I think — is the Schartzchild eqn. is not the equation that describes the underlying physics driving the GHE, but is instead just the equation describing atmospheric radiative transfer in the atmosphere. Of course, for determining the manifested thermodynamic path and or the change in the thermodynamic path from one equilibrium state to another, it no doubt applies and is critical. But again, the actual thermodynamic path itself is largely independent of the underlying physics of the GHE.
“By the way, if you can ever produce an equation to express your “idea” – which I doubt – at that point you will almost certainly realize what an outstanding numbskull you have been for the last 4,267 comments.”
I don’t know if I agree I was a numbskull, but yes, it does appear like 4,267 comments of mine were just regarding IR ‘optical thickness’ through the whole of the atmosphere from the perspective of the surface and layers looking up towards space before changing anything, and how it is or would be quantified in terms of actual flux densities in W/m^2 absorbed and transmitted through the whole.
The part about putting it into an equation which I find hard to conceptualize though is the saturation point of a particular wavelength is weighted or determined by the scaled wavelength intensity of Planck flux emitted by the surface. That is once a particular wavelength’s absorption is saturated, further absorption of that wavelength by the emission from a layer above no longer increases the optical thickness through the whole. This is what I meant by the emission from the layers above the surface, in terms of how they further increase the optical thickness through the whole, is only incremental.
Since when you add GHGs, the reduced IR intensity at the TOA calculated via the Schartzchild eqn., i.e. the increase in IR optical thickness from the surface and layers looking up towards space, is also determined by the scaled wavelength intensity of the Planck flux emitted by the surface in the same way in W/m^2, it’s valid to consider the change as an incremental increase in in IR optical thickness through the whole, though the final calculation is only really a net increase in optical thickness since the increased in IR optical transparency to the surface has to be subtracted (to conserve energy).
So look I’m genuinely sorry if it appears like I wasted everyone’s time trying to establish this, but I wasn’t trying to.
Again, it appears the disconnect between GW and the field is that the field doesn’t quantify or think of the IR optical thickness through the whole from the perspective of the surface and layers looking up towards space (before changing anything) as the sum of ‘T’ and ‘A’ components.
I have to admit also that I even thought some of you were being deliberately obtuse to what I was trying to establish, but I now clearly see that wasn’t the case at all and truly no one knew what I was trying to get at.
RW,
Saying that total T and A are ignored by ‘the field’ is not correct.
You can find transmittance at different frequencies/wavelengths in the MODTRAN data. Click on the ‘Show Raw Model Output’ button. This opens another tab with lots and lots of tabular data. Scroll down to the bottom and you find the table for Radiance. The last column is transmittance. It’s decimal, not scientific notation, so if the transmittance is less than 0.00005, it’s going to be listed as 0.0000. The transmittance depends on the location and direction of view. It’s either the transmittance from the surface up to the viewing height or from the viewing height to space.
OK. I didn’t mean ignored, but rather that IR ‘optical thickness’ through the whole was not generally quantified by the field as the sum of ‘T’ and ‘A’ components.
Again – it’s never saturation :: where is strongly absorbed, is also strongly emitted. Only the temperature gradient increases in the stratosphere. In the troposphere, the temperature gradient is determined by the adiabatic.
With more CO2 the critical temperature gradient (Schwarzschild criterion) is achieved at a higher level because of the temperature gradient increases.
You’re missing my point. Relative to the quantification of IR ‘opitical thickness’ through the whole (or really probably from anyone point to another of significant distance), there is most definitely saturation at some wavelengths, and further absorbed emission of those wavelengths by subsequent layers above do not increase the optical thickness any further.
RW,
Sucked in again.
Optical thickness in the atmosphere doesn’t have an upper limit. If the transmittance were identically zero, the optical thickness would be infinite. Some photons from the surface escape to space even at the CO2 absorption peak. Just not very many. Besides, what counts, as has been pointed out to you many times, is the emission at the TOA, not the source of that emission. And what counts for emission from a ghg is the temperature at the altitude where the optical depth measured from space toward the surface is equal to one. That’s the effective emission height. As concentration goes up, that height increases and, in the troposphere, the temperature and therefore the emission intensity, decreases even if the optical depth lower in the atmosphere is very high.
Look at the MODTRAN data. The upward radiance at 70km looking down where the transmittance is 0.0000, is not zero. At 616 cm-1 for the tropical atmosphere at 400ppmv CO2, the transmittance is nominally zero, 0.0000 and the radiance is 6.60E-06W/(cm² ster cm-1). Double CO2 to 800ppmv and the transmittance is still 0.0000, but the radiance is now 6.00E-06 because the optical thickness has increased so the effective emission height has moved up. IOW, it isn’t saturated as far as emission to space is concerned.
If you would pay more attention to real scientists and not GW, you might actually understand this. But if experience is a guide, you won’t.
Dewitt,
“Sucked in again.
Optical thickness in the atmosphere doesn’t have an upper limit. If the transmittance were identically zero, the optical thickness would be infinite.”
I don’t understand what you’re saying here. Of course, just because the IR optical thickness through the whole from the surface and layers ‘looking up’ towards space is saturated at a particular wavelength doesn’t mean there is NOT significant transmission at that wavelength into outer space. I never claimed or ever thought that.
“Some photons from the surface escape to space even at the CO2 absorption peak. Just not very many.”
Explain what you mean here, because I’m not sure. I would think any 15u photon emitted directly by the surface has virtually a zero probability of making it through the TOA, even in dry cloudless sky. But even if there is some probability, it’s got to be really, really low — I would think at least.
“Besides, what counts, as has been pointed out to you many times, is the emission at the TOA, not the source of that emission. And what counts for emission from a ghg is the temperature at the altitude where the optical depth measured from space toward the surface is equal to one. That’s the effective emission height. As concentration goes up, that height increases and, in the troposphere, the temperature and therefore the emission intensity, decreases even if the optical depth lower in the atmosphere is very high.”
Yes of course, I completely agree with this characterization of it. The average emission level is moved up to a higher point where it’s colder (or at least the upwelling emission rate through the whole is less), thus reducing the outgoing flux at the TOA to where it’s now less than the incoming post albedo solar flux.
“Look at the MODTRAN data. The upward radiance at 70km looking down where the transmittance is 0.0000, is not zero. At 616 cm-1 for the tropical atmosphere at 400ppmv CO2, the transmittance is nominally zero, 0.0000 and the radiance is 6.60E-06W/(cm² ster cm-1). Double CO2 to 800ppmv and the transmittance is still 0.0000, but the radiance is now 6.00E-06 because the optical thickness has increased so the effective emission height has moved up. IOW, it isn’t saturated as far as emission to space is concerned.”
Ditto my comments above.
If you would pay more attention to real scientists and not GW, you might actually understand this. But if experience is a guide, you won’t.
Why don’t you just stick to the issues and stop denigrating GW, who cannot appear here to defend himself against all of these attacks? Science is not about argument from authority — but about evidence and logic.
I’m only using the term IR ‘optical thickness’ as a measure of ‘line of sight’, in particular from the surface and layers ‘looking up’ to the TOA. I’m well aware that the atmosphere both absorbs and emits, including in saturated bands (i.e. in bands that are saturated from the perspective of ‘looking up’ from the surface).
The point is incremental changes, i.e. incremental increases in IR opacity are measured specifically as incremental changes in such IR ‘optical thickness’ from the perspective of ‘line of sight’ from the surface and layers ‘looking up’ to the TOA. Or at least, this is how the reduced IR intensity component at the TOA is scaled and quantified by.
In effect, after adding GHGs and increasing the height of the average emission level, from the perspective of the surface and layers ‘looking up’, more atmosphere and less space is ‘seen’ than was before.
Of course, when you add GHGs and each layer absorbs a little more from above and below and emits a little more upwards and downwards, there is subsequently also increased IR transmission through to the surface (due to the lapse rate). The whole point though is the reduced transmission through the TOA has to *exceed* the increased transmission to the surface, otherwise there is no enhancement to the GHE from the added GHGs.
The fundamental initiating mechanism of the enhancement is from the perspective of the surface and layers ‘looking up’ now seeing less of space and more of the atmosphere than prior. That is, before GHGs were added, there is about 240 W/m^2 being passed into space, which is a combination of IR flux directly transmitted from the surface into space and IR flux originating from the atmosphere transmitted into space, and now it’s reduced below 240 W/m^2.
The so-called ‘average emission’ level is just the equivalent location where where the emission is around 240 W/m^2, but the increased attenuation that occurs to move the ‘average emission level’ up higher occurs all throughout. Again, it’s based on the increase in ‘optical thickness’ from the perspective of the surface and layers ‘looking up’ to the TOA.
RW,
You keep citing him.
I cited evidence and logic and he wouldn’t commit himself to one position or another. That is not the argument from authority, although I do agree that I accept what is taught in physics textbooks as a given for the purposes of keeping this blog on track. (See the Etiquette).
Eventually I reach a point where I don’t want particular people to continue to make claims which are unsupported, or just plain contrary to textbook physics, and they are then banned.
Of course, they believe they are right. And “I can’t handle their arguments”. Etc, etc. Still they are banned. Even when RW, a great authority of physics, thinks they have valuable comments to make they are still banned.
However, even in the case of the very annoying and hopelessly wrong people who have arrived and finally been banned – their “arguments” are left there for posterity.
This is the case with GW. His arguments are there for everyone to see. I let new readers judge for themselves.
In the case of you, I should have banned you 2,000 comments ago. For some reason, I haven’t done this.
I asked for equations and you keep reposting text that looks exactly like slightly rehashed versions of your text of previous posts. Your lack of understanding of physics basics is not a problem that regular commenters – who do understand physics basics – should have to fix.
It is never saturated. If one could paint the photons from the surface of green, one would after sufficiently long way only to observe a few green photons according to the Beer-Lambert law – but the Beer-Lambert law knows no saturation.
But without color you do not know where the photons come and absorbed photons are in a similar amount replaced by emitted photons. One can only measure the total intensity – there is just no saturation.
RW wrote: “Alright, but I thought I came up with some pretty clear language to describe the issue using terms that have now been agreed with to mean a specific thing. Let me work on coming up with the equation for the optical thickness through the whole of multi-layer atmosphere.”
It is mathematically IMPOSSIBLE to find such an equation. It is simpler to explain why in terms of distance = velocity * time.
Suppose velocity at any time t is given by
v = e^(-t)
e is an irrational number near 2.71828. If you knew integral calculus, you could integrate e^-t to get a formula for the distance traveled from time = 0 (when velocity is 1) to any time = t. Such a formula is called a definite integral. Let’s call this x:
x = 1 – e^(-t)
At t=0, x=0. As the object slows and comes to a stop only after infinite time, it has moved a distance of 1. Now suppose we change the problem just slightly and say:
v = e^(-t^2)
In this case, NO mathematician can write a simple equation for x as a function of time. (One can write an equation with an integration sign in it or with the sum of an infinite series.) It is impossible to integrate the majority of complicated functions that describe a relationship between velocity and time and get an equation that describes the relationship between position and time. This is a very common problem in physics because most laws of physics are written in terms of how things CHANGE with time or position. Integral calculus is essential for summing such changes. F = ma or a = F/m tells us how acceleration – the change in velocity with time – varies with force. Physics textbooks show equations for how position varies with time for a few forces that vary in a simple way with time (constant acceleration, simple harmonic oscillator, orbits). That may mislead you into assuming that general solutions always exist. They don’t.
This inability to calculate definite integrals for functions is so common that some of the more useful integrals have been given names. The normal distribution function contains e^(-x^2) and its integral (after multiplying by 2/sqrt(pi)) is called erf(x). The erf(x) can’t be written in terms of common functions. Excel and many scientific calculators have an erf function built in them that sums enough terms of an infinite series.
One CAN always find an approximate answer for the distance traveled at a particular time, say t=1. Use a spreadsheet to calculate the velocity at one thousand time points: t = 0, 0.001, 0.002, 0.003 …. 0.999. Then calculate the distance traveled in each of those 0.001 periods of time and sum. This process is called numerical integration or Riemann summation. During the period t = 0.500 to t = 0.501, the change in time is 0.001 and the velocity (which is constantly slowing) at the beginning of period is e^0.25. During this period:
change in x = velocity * change in time
delta_x = v * delta_t
Or, in the language of calculus (a differential equation on the left and a derivative on the right):
dx = v*dt and dx/dt = v
Numerical integration usually gives an approximate answer because the velocity changes slightly in each period, between t = 0.500 to t = 0.501 above. You can approximate the distance traveled at t=1 more accurately using more, shorter periods of time. The answer is always number. You can NOT write a simple formula that gives you that number for any time, t.
The Schwarzschild equation tells us how the intensity of radiation CHANGES WITH DISTANCE as it passes a small distance dz through the atmosphere:
dI = emission – absorption
dI = n*o*B(lambda,T)*dz – n*o*I*dz
Just like e^(-t^2), the only way to solve the Schwarzschild eqn by numerical integration. MODTRAN is a program that will do the numerical integration for you, contains all of the values you need for absorption cross-section, and let’s you input the GHG mixing ratio need to calculate density at each altitude. You are not required to use MODTRAN; SOD wrote a program for this blog that computed radiation transfer and the change in stratospheric temperature produced by forcing. In all cases, you can only get a numerical answer (i.e. 150 W/m2 for the GHE), not a formula.
If you wrote your own program, or modified SOD’s, and numerically integrated the change in radiation from the surface to space over 700,000 segments of 0.1 m in length, you could sum up all of the power adsorbed in the atmosphere. The segments need to be this short because a few wavelengths are mostly absorbed within 1 m near the surface. You will find that roughly 10-fold the power emitted by the surface is absorbed by the atmosphere and roughly 9.6-fold is emitted, leaving an apparent transmission of about 40%. If the answer were 100-fold and 99.6-fold, 40% would still get through and nothing would change. Since the rest of the world usually only cares about radiation transfer – the flux at the TOA – MODTRAN doesn’t tell us what you want to know – the total flux absorbed.
In conclusion, the equation you are seeking is a differential equation (the Schwarzschild equation) that doesn’t have a definite integral. One way to get a numerical answer is to use MODTRAN.
You can learn about almost any aspect of math online at Khan Academy. Spending time there would make far more sense than wasting time telling others (who understand calculus) what is wrong with their understanding of physics. For something similar to what I wrote above, see:
https://www.khanacademy.org/math/integral-calculus/indefinite-definite-integrals/area-under-rate-function/v/area-under-rate-net-change
Frank,
“In conclusion, the equation you are seeking is a differential equation (the Schwarzschild equation) that doesn’t have a definite integral.”
I appreciate the detailed reply, but you keep saying this and it’s not correct. The Schwartzchild eqn. is not the eqn. to calculate IR optical thickness. The Schartzchild eqn. deals with how the IR intensity changes, directionally up or down, as it moves through the absorbing and emitting layers in the atmosphere. I’ve never disagreed with this eqn. or what it predicts, and neither has GW (in fact, he uses the eqn. in the same way as everyone else does in his RT simulation). It would be silly and ridiculous to deny what the eqn. predicts since what it predicts can and is directly observed to occur.
“Spending time there would make far more sense than wasting time telling others (who understand calculus) what is wrong with their understanding of physics.
I’m not claiming others here such as yourself have an overtly wrong understanding of physics, but rather — I’m trying to get at an applied physics concept. But clearly, no one understands what I’m trying to get at.
If you really want to help me, how about you take me through the details of the eqn. for calculating optical thickness through a multi-layer atmosphere — from the perspective of the surface and layers looking up to the TOA?
RW: I appreciate the detailed reply, but you keep saying this and it’s not correct.
The Schwarzschild eqn is an equation that gives you IR optical thickness. It also gives you emission, which is essential to calculating absorption higher in the atmosphere.
In a laboratory IR spectrometer, the intensity of the IR light source overwhelms emission from the air, and we can ignore the emission term in the Schwarzschild eqn. In that case, the equation has a definite integral. It is called Beer’s Law for absorption. Beer’s Law is a subset of the physics encompassed by the Schwarzschild eqn.
Since the amount of radiation absorbed is proportional to its intensity, we must keep track of both absorption and emission, so we know the radiation intensity at all altitudes. That doesn’t mean we can’t calculate the total power absorbed by the atmosphere. It does meant there is no simple equation like Beer’s Law that provides absorption when emission is significant
The problem is that you can’t do this mathematics (calculus) for yourself, so you don’t understand why this is the correct answer. Newton invented calculus so he could find the answers to physics problems. Calculus is often called the “mathematics of change”. Physics is also about how things change, so calculus is essential to the description of change. In this case, we want to know how radiation changes with distance traveled.
https://www.quora.com/Why-is-calculus-called-the-mathematics-of-change?share=1
RW wrote: “I’m not claiming others here such as yourself have an overtly wrong understanding of physics, but rather — I’m trying to get at an applied physics concept. But clearly, no one understands what I’m trying to get at.”
We understand exactly what you are trying to get at and how it would be calculated. However, it makes no difference whether 300 W/m2 is absorbed and 150 W/m2 emitted or 3,000 W/m2 is absorbed and 2,850 W/m2 emitted. 390 W/m2 of LWR emitted by the surface is reduced to 240 W/m2 at the TOA in each case. Consequently, what you want to get at requires using something besides MODTRAN, perhaps writing a new program.
Frank,
“We understand exactly what you are trying to get at and how it would be calculated.
I don’t think you do. What I’m trying to get at is not the calculation of optical thickness itself, but in order to move on from that to the applied physics concept I’m being required to provide the eqn. for optical thickness.
“However, it makes no difference whether 300 W/m2 is absorbed and 150 W/m2 emitted or 3,000 W/m2 is absorbed and 2,850 W/m2 emitted. 390 W/m2 of LWR emitted by the surface is reduced to 240 W/m2 at the TOA in each case. Consequently, what you want to get at requires using something besides MODTRAN, perhaps writing a new program.”
Then why am I being told it’s the most basic calculation that can be performed in Modtran using HITRAN line data?
Frank,
“390 W/m2 of LWR emitted by the surface is reduced to 240 W/m2 at the TOA in each case.”
This describes the ‘effective emissivity’, which is of the 390 W/m^2 radiated from the surface only 240 W/m^2 is transmitted into outer space. Or the effective emissivity is 240/390 = 0.62, meaning for every 1 W/m^2 emitted from the surface only 0.62 W/m^2 are transmitted into space.
For IR optical thickness I’m talking about line of sight *only* from a specific spot, in particular from the surface and layers looking up through to the TOA. The surface does not ‘see’ only 150 W/m^2 of what it emits as atmosphere and the remaining 240 W/m^2 sees outer space.
I agree the Schartzchild eqn. quantifies how the 390 W/m^2 comes down to 240 W/m^2 at the TOA, but I wouldn’t consider that the IR optical thickness from the perspective of the surface and layer looking up to the TOA.
“It is mathematically IMPOSSIBLE to find such an equation.”
This doesn’t quite make sense. Impossible? Modtran must have an eqn. it uses to calculate IR optical thickness. It would have to wouldn’t it? I fully understand the actual calculation itself is ultra complex and would have to be done in a computer. But there must be an eqn.
RW wrote: “This doesn’t quite make sense. Impossible?”
After admitting that you haven’t taken calculus, why are YOU telling anyone what is and is not possible! I provided you a link to Khan Academy – a great online resource. LEARN something and then tell me why I am wrong.
You keep saying there MUST be an EQUATION. There are equations for the roots of quadratic, cubic and quartic polynomials, but it has been proven that no general algebraic equation can give the roots of polynomials with order greater than 4. (Quartics are messy.) Approximate NUMERICAL ANSWERS can be calculated using arithmetic. A numerical answer is not an EQUATION.
There is an equation that gives position with time when v = e^(-t), but not when v = e^(-t^2). Approximate numerical answers for both can be calculated for any particular time t using arithmetic. These numerical answers are not EQUATIONS.
MODTRAN does arithmetic.
The Schwarzschild equation is only apparently an equation for determining the change in intensity, the main solution of the Schwarzschild equation is the determination of the temperature profile, because without convective heat effect, the entire change in intensity must be zero: the decrease in radiation intensity upwards – must be the radiation increase downwards in balance equal.
To function Features: With digital numerical calculations is always used approximate formulas – whether sin x, e ^ (- x) or e ^ (- x²). That’s why my discussion of the numerical properties appear as meaningless.
There is an equation, but it’s a differential equation with no analytic solution. It has to be solved by numerical approximation as Frank pointed out above. If you can’t grasp this concept, you do not have the competence to criticize the findings of others. IOW, go away. You’re wasting bandwidth.
Dewitt,
“There is an equation, but it’s a differential equation with no analytic solution.”
Yes, what is that equation? I believe this is what I’m being asked to provide.
The quantification would be in a plane parallel, multi-layer atmosphere, from the perspective of the surface and layers looking up to the TOA in a straight line, and would have a simple ‘A’ term quantifying optical thickness and a simple ‘T’ term quantifying optical transparency through the whole. The ‘T’ term would quantify what fraction of the flux density emitted from the surface is transmitted in a straight line into outer space at the speed of light (the same as if the atmosphere wasn’t even there), and the ‘A’ term would quantify what fraction of the flux density emitted from the surface is absorbed by the atmosphere or attenuated from passing into space. Nothing more.
This might be more clearly stated as:
“The ‘T’ term would quantify what fraction of the Planck flux emitted from the surface that is transmitted in a straight line into outer space at the speed of light (the same as if the atmosphere wasn’t even there), and the ‘A’ term would quantify what fraction of the Planck flux emitted from the surface is absorbed by the atmosphere or attenuated from passing into space. Nothing more.”
RW: I wonder if you are confusing “looking up” and “looking down”. If you check Modtran you will see that you get almost no IR when you look up to TOA. So trnsmittance will be almost zero.
“RW: I wonder if you are confusing “looking up” and “looking down”. If you check Modtran you will see that you get almost no IR when you look up to TOA. So trnsmittance will be almost zero.”
I’m not confusing ‘looking up’ with ‘looking down’, and you have to have some remaining ‘T’ from the perspective of ‘looking up’ from the surface and layers for added GHGs to result in a net IR opacity increase from that perspective, which again (that perspective) is the initiator of the GHE. That is, reduced ability to radiate freely to outer space.
It’s important to remember that when doing these RT calculations or simulations for added GHGs, you’re holding everything else constant. That is, you’re essentially stopping time and manipulating just the radiation component. This is why the perspective of the net end result, which is increased IR opacity from the perspective of the surface and layers ‘looking up’ is what matters, and defines the fundamental meaning of the final calculation.
Moreover, it’s *precisely* because the increased IR opacity or ‘optical thickness’ from the perspective of the surface and layers ‘looking up’ to the TOA is greater than the increased IR ‘optical transparency’ or IR transmission ‘looking down’ to the surface — that the final calculation is defined as and is one of a net absorption increase (and not a net transmission increase).
nobodyknowledge,
No. The transmittance will be 1, not 0. There’s almost nothing above 70km to absorb radiation. The intensity is nearly zero for that reason and that the incoming radiation from space is effectively zero from 100-1500cm-1.
Here’s a sample of the raw data from 70km looking up. The last column is transmittance. Sorry about the formatting.
0 FREQ WAVLEN PATH THERMAL SURFACE EMISSION SURFACE REFLECTED TOTAL RADIANCE INTEGRAL TOTAL
(CM-1) (MICRN) (CM-1) (MICRN) (CM-1) (MICRN) (CM-1) (MICRN) (CM-1) (MICRN) (CM-1) TRANS
1400. 7.143 6.53E-12 1.28E-09 0.00E+00 0.00E+00 0.00E+00 0.00E+00 6.53E-12 1.28E-09 1.63E-06 0.99997
1402. 7.133 6.51E-14 1.28E-11 0.00E+00 0.00E+00 0.00E+00 0.00E+00 6.51E-14 1.28E-11 1.63E-06 1.00000
1404. 7.123 2.96E-12 5.84E-10 0.00E+00 0.00E+00 0.00E+00 0.00E+00 2.96E-12 5.84E-10 1.63E-06 0.99999
1406. 7.112 2.95E-12 5.84E-10 0.00E+00 0.00E+00 0.00E+00 0.00E+00 2.95E-12 5.84E-10 1.63E-06 0.99999
1408. 7.102 5.59E-14 1.11E-11 0.00E+00 0.00E+00 0.00E+00 0.00E+00 5.59E-14 1.11E-11 1.63E-06 1.00000
1410. 7.092 2.36E-13 4.70E-11 0.00E+00 0.00E+00 0.00E+00 0.00E+00 2.36E-13 4.70E-11 1.63E-06 1.00000
1412. 7.082 6.08E-13 1.21E-10 0.00E+00 0.00E+00 0.00E+00 0.00E+00 6.08E-13 1.21E-10 1.63E-06 1.00000
1414. 7.072 1.05E-13 2.10E-11 0.00E+00 0.00E+00 0.00E+00 0.00E+00 1.05E-13 2.10E-11 1.63E-06 1.00000
1416. 7.062 1.83E-12 3.67E-10 0.00E+00 0.00E+00 0.00E+00 0.00E+00 1.83E-12 3.67E-10 1.63E-06 0.99999
1418. 7.052 8.14E-12 1.64E-09 0.00E+00 0.00E+00 0.00E+00 0.00E+00 8.14E-12 1.64E-09 1.63E-06 0.99997
1420. 7.042 3.37E-11 6.79E-09 0.00E+00 0.00E+00 0.00E+00 0.00E+00 3.37E-11 6.79E-09 1.63E-06 0.99986
1422. 7.032 6.99E-14 1.41E-11 0.00E+00 0.00E+00 0.00E+00 0.00E+00 6.99E-14 1.41E-11 1.63E-06 1.00000
I’ve bolded the first two transmittance values, but it may not show up very well.
Dewitt,
Maybe the disconnect here is what we are considering to constitute ‘transmittance’. Of course, the combined surface/atmosphere transmits about 240 W/m^2 to outer space before changing anything. This seems to be what Frank is referring to.
I’m referring to ‘transmittance’ in the context of ‘line of sight’ only, from the perspective of the surface and layers looking up to the TOA. This is determined by the scaled wavelength intensity of Planck Flux emitted by the surface, and what fraction of this flux ‘sees’ outer space vs. what fraction ‘sees’ the atmosphere. The amount which sees the atmosphere quantifies ‘A’ and the amount that sees space quantifies ‘T’.
Thank you for your answer DeWitt. I was thinking of the LW radiance from TOA to the surface being almost zero.
I must admit that I have some problems in understanding the “looking” consept of RW, when he write: “I’m referring to ‘transmittance’ in the context of ‘line of sight’ only, from the perspective of the surface and layers looking up to the TOA. This is determined by the scaled wavelength intensity of Planck Flux emitted by the surface, and what fraction of this flux ‘sees’ outer space vs. what fraction ‘sees’ the atmosphere. The amount which sees the atmosphere quantifies ‘A’ and the amount that sees space quantifies ‘T’.”
Do you understand what he means? I think it would be easier if he could call it radiance up or down from layer A to B.
“Do you understand what he means? I think it would be easier if he could call it radiance up or down from layer A to B.”
I think we’ve agreed that what I’m referring to is called IR optical thickness from the perspective of the surface and layers ‘looking up’ to the TOA.
The transmittance would be the fraction of the scaled Planck Flux density emitted by the surface which is transmitted into outer space in a straight line up at the speed of light.
Using the numbers above I somewhat arbitrarily picked, if the transmittance ‘T’ from this perspective were calculated to be 0.25 and the surface was at a temperature where it radiated 400 W/m^2, it would mean 100 W/m^2 is transmitted straight through the whole of the atmosphere at the speed of light, and the difference ‘A’ (i.e. 1-T) of 300 W/m^2 is absorbed by the atmosphere.
It is precisely from this perspective that the reduced IR intensity at the TOA from added GHGs is scaled by, and is the whole point of everything. Moreover, it is precisely that there is significant IR optical thickness from this initial perspective that we have a GHE in the first place.
Dewitt, I believe, is simply referring to the transmittance of IR into outer space from the combined surface/atmosphere emission. But that’s not really specifically related to the GHE itself or its underlying physics.
nobodysknowledge,
The IR radiance at the TOA, in MODTRAN at least, is defined to be zero, so of course none reaches the surface. That doesn’t mean that you can’t calculate a transmittance from the TOA to the surface. Of course, it’s the same as the transmittance from the surface to the TOA and it isn’t zero everywhere. The average transmittance from 100-1500cm-1 is also available in the MODTRAN data. Scroll all the way to the bottom of the data.
For the tropical atmosphere with the default settings, the average transmittance is 0.1473. For the sub-arctic winter atmosphere where the specific humidity is much lower, it’s 0.3342. Of course, that’s clear sky conditions. Add clouds and the average transmittance is zero. Absorptivity is the complement of transmittance, A = 1 – T when, as in clear sky conditions, there is zero reflectivity.
Of course, the radiative transfer equation is a differential equation, but transparency and absorption are not very interesting for the radiation transfer through the atmosphere, because the absorption is always associated emissions. The solution of the RTE can therefore already done hardly analytically because the absorption coefficients are unlikely to give analytically because of their dependence to temperature and pressure.
This is demonstrated in the two-stream approximation. Given all radiation in the upper and lower hemisphere of the atmosphere to be integrated to an intensity up or down. In this two flows (upward and downward) the radiative transfer equation is applied. Due to the different angular distribution of the intensity of the equivalent absorption factors are different, in addition, this dependence is neglected. The different angular distribution of intensity arises because approximately horizontal beams pass through virtually isothermal areas, vertical beams from warmer or colder areas originate.
When well-mixed greenhouse gases in vertical terms, same pressure changes mean equal amounts of greenhouse gas molecules. That’s why working in the following with the pressure altitude and the absorption coefficient kp then also a pressure size. At a pressure difference equal to the absorption coefficient then the initial intensity has fallen to 1 / e without emission according Lambert-Beer. But the emission may just not be neglected.
For the two beams of the two-stream approximation (Iup upwards and downwards Id) read the radiative transfer equations:
(1) kp DIUP / dp = Iup – B (p)
(2) -kp dId / dp = Id – B (p)
If in the atmosphere no other heat sources (convection, ozone, etc.) is the net flow upwards (Q) is constant:
(3) Iup – id = Q
or
(4) Iup = Id + Q
Eq. (4) used in Eq. (1) gives:
(5) kp d(Id + Q) / dp = (Id + Q) – B (p)
Since the derivative of Q according presupposition = 0, Eq shortened. (5)
(6) kp dId / dp = Id + Q – B (p)
The addition of Eq. (2) and (6) gives
(7) 0 = 2 Id + Q – 2 B (p)
or
(8) B (p) = Id + Q / 2
Eq. (8) in Eq. (2) gives used:
(9) -kp dId / dp = Id – (Id + Q / 2)
or
(10) kp dId / dp = Q / 2
Differentialeq.. (10) is easily solved:
(11) Id = p Q / (2 kp)
The solution (11) also satisfies the boundary condition Id = 0 at p = 0
The solution (11) follows
B (p) = Q / 2 (1 + p / kp)
Iup = Q [1 + p / (2kp)]
The relationship between B (p), and T (p) is not linear.
The terms absorption and transparency have meaning only if the emission can be neglected. This is the case when the light source is much hotter than the sample and the sample is not too long. Therefore be used in measuring devices at room temperature hot source (Laser, Globar etc.). In the atmosphere, however, all temperatures are similar.
An application of the terms absorption and transparency on the atmosphere is simply an accounting application without practical significance.
I had written “… and absorbed photons are in a similar amount replaced by photons emitted.” How great the similarity is, depends on the temperature gradient. In isothermal range is absorption and replacement the same.
Jochen,
Spectralcalc.com has some interesting features if you want to learn about absorption and emission spectroscopy. Unfortunately, you have to subscribe to use them.
One fun thing is the gas cell. You have a choice of source temperature as well as cell temperature. If you measure emission with a gas in the cell that emits and absorbs in the selected spectral range, you can watch emission turn to absorption or absorption turn to emission by changing the temperature of the source relative to the temperature of the gas in the cell. If the source is the same temperature as the cell, the contents and length of the cell don’t matter. You always see a blackbody spectrum at the temperature of the source. I suspect this observation is counter-intuitive to some people.
DeWitt Payne,
Why should I learn, that is known to me. If the same amount of photons to be replaced, such as absorbed, then of course the sample length does not matter.
Still something is interesting – what Gerlich and Tscheuschner at the citation of their star witness to defraud (p 71: entitled “In the year 1972 Schack published a paper in Physical leaves.’The influence of the carbon dioxide content of the air on the world’s climate.’ ” – Physikalische Blätter 28 (1972), H. 1, pp 26 – 28) “The absorption of a gas passing through the heat radiation is in the steady state exactly equal to the heat radiation of this gas. Because if variations, in a cavity to be fulfilled in a gas this by itself would make temperature differences, which is not possible according to the second law of thermodynamics. ” [Die Absorption der ein Gas durchsetzenden Wärmestrahlung ist im Beharrungszustand genau gleich der Wärmestrahlung dieses Gases. Denn wenn hierbei Abweichungen beständen, würden sich in einem dies Gas erfüllenden Hohlraum von selbst Temperaturdifferenzen bilden, was nach dem zweiten Hauptsatz der Thermodynamik nicht möglich ist.]
I must me apologize for my insufficient English. My native language is German and my skills as a translator are lower than my knowledge as a physicist.
Jochen,
I probably should have addressed that to the general audience rather than you.
I have just enough German to say that your translation looks good to me.
The problem with using that argument, which is quite correct for an isothermal gas cell, is that the atmosphere isn’t isothermal or isotropic, as you well know.
For anyone else reading this: Spectralcalc.com also lets subscribers put up to six gas cells in series. I’m pretty sure you can set the temperatures of the cells independently, but not positive. With six cells, you can probably get a reasonable approximation of the atmospheric emission spectrum at the tropopause by selecting the correct temperature and pressure of each cell in series.
Re-reading this thing months later, you guys are truly unbelievable. The calculated 3.6-3.7 W/m^2 from 2xCO2 is just the net increase in optical thickness looking up. That’s all I was getting at, geez.
The enhancement of the GHE occurs because there is an increase from this perspective. The subtraction of the increased IR intensity at the surface from the reduced IR intensity at the TOA to arrive at the net imbalance imposed on the system is just to conserve energy.
Moreover, you can’t have a net transmission to space decrease without a net absorption increase from the perspective surface (and layers above it) looking up. The fact that this is arrived after each layer absorbs a little more from above and below and subsequently emits a little more upwards and downward is irrelevant to the physical meaning of the final calculation.
RW,
Your inability to either understand the physics or phrase a logical question is what’s unbelievable.
Not in my opinion.
RW wrote: The calculated 3.6-3.7 W/m^2 from 2xCO2 is just the net increase in optical thickness looking up.
Frank repllies. Optical thickness is dimensionless. The radiative force for 2XCO2 has units of W/m2. They can’t be the same thing.
RW: Moreover, you can’t have a net transmission to space decrease without a net absorption increase from the perspective surface (and layers above it) looking up.
Frank: Sure you can. You are forgetting emission. Leave the amount of GHG in the atmosphere the same (keeping absorption constant). Cool the upper troposphere, reducing emission from the upper atmosphere.
Or move some of the water vapor from the the tropopause to the lower troposphere. That will cool the upper troposphere with the same total absorption. The amount of radiation reaching space depends on more than just absorption: the temperature vs altitude profile, the humidity vs altitude profile, (both of which depend on convection), and emission from GHGs. There is no GHE in the absence of a temperature gradient.
“Frank repllies. Optical thickness is dimensionless. The radiative force for 2XCO2 has units of W/m2. They can’t be the same thing.”
OK, yes — you’re technically correct. In order to know the dimensionless net increase in optical thickness, i.e. the percentage increase, you would need to know what the spectral absorptance of the surface was before the calculation.
Using the simple number I referenced above in the thread, if the spectral absorptance ‘A’ were 0.75 before the change, and the surface was at a temperature where it was emitting 400 W/m^2, the spectral absorption would be 300 W/m^2. A net increase from 300 W/m^2 to 303.7 W/m^2 equals +0.00925 (3.7/400 = 0.00925), or a net increase in optical thickness from 0.75 to 0.75925 (a little less than a 1% increase). What we are most interested in though isn’t the percentage of the increase, but the increase in W/m^2.
“RW: Moreover, you can’t have a net transmission to space decrease without a net absorption increase from the perspective surface (and layers above it) looking up.
Frank: Sure you can. You are forgetting emission. Leave the amount of GHG in the atmosphere the same (keeping absorption constant). Cool the upper troposphere, reducing emission from the upper atmosphere.
Or move some of the water vapor from the the tropopause to the lower troposphere. That will cool the upper troposphere with the same total absorption. The amount of radiation reaching space depends on more than just absorption: the temperature vs altitude profile, the humidity vs altitude profile, (both of which depend on convection), and emission from GHGs. There is no GHE in the absence of a temperature gradient.”
What I mean is in the context of what calculating changes due to added GHGs involves through the whole, which generally stipulates that all other things are held fixed and only the radiation component is manipulated. I’m not forgetting emission. Changes in GHGs affect both absorption and emission, but it still doesn’t change that the final calculation is a net increase in absorption and a decrease in transmission (to space), and is scaled from the exact same perspective as the surface spectral absorption prior to the change, i.e. from the surface and layers above it looking up.
The whole point is the so-called ‘window’ closes a little, reducing the surface’s ability to radiate freely to outer space, thus enhancing the GHE.
Frank,
In short, whatever the reduced IR intensity at the TOA is calculated to be after increased absorption and subsequently increased emission (from added GHGs), that reduction is scaled from the same exact perspective as the full quantity of the surface spectral absorption prior to the change. The spectral absorption, or that large initial optical thickness from the surface looking up, is why there is a GHE in the first place. It’s fundamentally from that perspective that you are anchoring the final calculation to, i.e. the net increase in surface spectral absorption, or just the so-called ‘net absorption increase’.
In the end, the GHE exists and is initiated by the large inability of the surface to radiate freely to outer space. That is, it is significantly optically thick from the surface (and layers above it) looking up. Optical thickness through the whole from this perspective prior to changing anything, is quantified by the spectral absorptance ‘A’, so when converted into W/m^2 it quantifies the total ‘GHG absorption’ prior to changing anything. Since adding GHGs is an enhancement of an already existing effect, i.e. the GHE, it is perfectly valid to consider the net increase in optical thickness looking up to be the net increase in GHG absorption or ‘incremental GHG absorption’. That’s really all.
Additionally, the quantification of the spectral ‘A’ and ‘T’ is based on the scaled wavelength intensity of the Planck Flux emitted directly by the surface, so the sum of ‘A’ and ‘T’ — if converted into W/m^2, must equal the flux density directly radiated from the surface (as per the S-B law). This is the case even though much of the total of ‘A’ comes from that which is emitted from the atmosphere which is additionally absorbed.
SoD,
So yes, the 4689 posts were mostly just me trying to establish that the spectral ‘A’ (and subsequently the spectral ‘T’, quantified by 1-‘A’) is just the measure of ‘optical thickness’ from the surface looking up prior to changing anything, and that the quantification (derived from the scaled wavelength intensity of the Planck Flux emitted by the surface) in W/m^2 is equal to the power radiated from the surface, even though much of the absorption or attenuation that constitutes ‘A’ originates from the atmosphere.
This is all GW is quantifying by what he’s referring to as ‘T’ and ‘A’. The 3.6-3.7 W/m^2 calculated for 2xCO2 is the difference between the reduced IR intensity at the TOA and the increased IR intensity at the surface; however, the reduced IR intensity at the TOA (whatever it’s calculated to be) is scaled from the exact same perspective as the full quantity of the spectral ‘A’ prior to the change. The atmosphere will re-emit this additionally captured upwelling IR flux (both up and down) over twice the area it absorbs from, the same as the full quantity of the spectral ‘A’.
Since the atmosphere creates no energy of its own, macroscopically the spectral ‘A’ is *equivalent* to the flux density emitted directly by the surface which is absorbed by the atmosphere, because the surface directly radiates the same amount of flux back up into the atmosphere that comes out at the bottom and is added to the surface (i.e. the net flux gained at the surface). COE sets the limit that the sum of ‘T’ and ‘A’, converted into flux densities in W/m^2, cannot exceed or be less than flux density directly radiated in from the surface (assuming a surface emissivity of 1). This COE constraint, BTW, is exactly the same reason you have to subtract the increased IR intensity at the surface from the reduced IR intensity at the surface. If this is not done, additional flux coming out of the atmosphere going into the surface would be being created from nothing, which COE says can’t happen.
At any rate, whether ultimately right or wrong, the rest of the stuff surrounding all of this deals largely with black box system analysis and equivalent system modeling, which no one here understands or seems to have any interest in understanding.
This:
“and that the quantification (derived from the scaled wavelength intensity of the Planck Flux emitted by the surface) in W/m^2 is equal to the power radiated from the surface, even though much of the absorption or attenuation that constitutes ‘A’ originates from the atmosphere.”
should have said:
“and that the quantification (derived from the scaled wavelength intensity of the Planck Flux emitted by the surface) in W/m^2, i.e. their sum, is equal to the power radiated from the surface, even though much of the absorption or attenuation that constitutes ‘A’ originates from the atmosphere.”
What is the dispute about W/m²? According to the definition of the IPCC, the radiatiative forcing is a non-measurable calculation value at the tropopause. Only with the feedback factor, which can also not be measured, is a calculate value for the surface temperature which is measurable.
A treatment of the effect of the CO2 increase without consideration of the change of the convection (and thus the tropopausenhöhe) is meaningless.
I am writing my German text before the translation.
Was soll der Streit um die W/m²? Nach der Definition des IPCC ist der Strahlungsantrieb ein nicht meßbarer Rechenwert an der Tropopause. Erst mit dem ebenfalls nicht meßbaren Rückkopplungsfaktor entsteht eine Rechengröße für die Oberflächentemperatur, die meßbar ist.
Eine Behandlung der Auswirkung des CO2-Anstiegs ohne Berücksichtigung der Änderung der Konvektion (und damit der Tropopausenhöhe) ist sinnlos.
Ich schreibe mal meinen deutschen Text vor der Übersetzung mit.
Jochen,
“What is the dispute about W/m²? According to the definition of the IPCC, the radiatiative forcing is a non-measurable calculation value at the tropopause. Only with the feedback factor, which can also not be measured, is a calculate value for the surface temperature which is measurable.
A treatment of the effect of the CO2 increase without consideration of the change of the convection (and thus the tropopausenhöhe) is meaningless.”
There really isn’t a dispute about the quantification in W/m^2, but really only a dispute about what that quantification in W/m^2 means relative to the whole picture related to its effect on greenhouse warming of the surface compared to non-greenhouse warming of the surface via W/m^2 of post albedo solar flux entering the system.
The field (whether it realizes it or not) considers the *intrinsic* surface warming ability of each in W/m^2 to be equal to one another, i.e. each is established to have the same ‘zero-feedback’ surface temperature increase. For +3.7 W/m^2 of ‘GHG absorption’ or +3.7 W/m^2 of post albedo solar flux entering the system, the field claims each has a ‘zero-feedback’ surface temperature of about 1.1C. Right? In each case, there will be a -3.7 W/m^2 TOA deficit that has to be restored, so you can apply the same amount of incremental linear surface/atmosphere warming and it will restore balance at the TOA for both instances, but for the +3.7 W/m^2 of GHG absorption case is that kind of atmospheric warming leading to surface warming really mechanistically related to how the GHE is physically driven? I think it can be argued that it’s not, and the rules of linearity related to *intrinsic* greenhouse warming ability via added GHGs should be that of a linear increase in aggregate dynamics offsetting the total GHG absorption prior to the change, which is not the same as a linear increase in surface/atmosphere warming according to the lapse rate; which quantifies a linear increase in aggregate dynamics offsetting post albedo solar power entering the system prior to the change. That is, linear warming according to the lapse rate is based on the 1.6 to 1 power densities ratio between the surface and the TOA (385/239 = 1.61), which is specifically offsetting or quantifying the aggregate dynamics of post albedo solar power entering the system. The ratio’s physical meaning is it takes about 1.6 W/m^2 of net surface gain to allow 1 W/m^2 to leave at the TOA, offsetting each 1 W/m^2 entering (post albedo) from the Sun.
With the surface properties and the greenhouse gases in the troposphere, the increase of the temperature by has almost nothing to do with CO2 increase. If in the stratosphere, the concentration of greenhouse gases increases, the adiabatic temperature gradient (which exists in the troposphere) is the sooner, and consequently, increase the height of the tropopause (Schwarzschild criterion 1906). As extreme limits greenhouse gas amount in the stratosphere would increase or remain constant in accordance with the CO2 increase. The actual increase amount of greenhouse gas in the stratosphere is between the two extremes. The increase in the tropopause and the constancy of the temperature gradient in the troposphere leads to an increase in surface temperature of about 3 K. Any W/m² are just a side effect of the change the height of the tropopause.
That’s not entirely true. A significant fraction of radiation to space comes directly from the surface:
From MODTRAN
Tropical Atmosphere: 14.7% transmission
Mid-Latitude Summer: 18.8%
Mid-Latitude Winter: 28.9%
Sub-Arctic Summer: 22.5%
Sub-Arctic Winter: 33,4%
1976 U.S. Standard Atmosphere: 25.7%
And that’s not to mention that most atmospheric radiation that escapes to space originates well below the tropopause.
DeWitt,
The number I have seen for direct emission from the surface to space in the IR windows is 22 W/m^2, about 9% of the total. I think that the difference is that your numbers assume clear skies, but clouds block the IR windows.
Dewitt,
Are those values solely the direct surface IR –> TOA component or are they a quantification of the spectral transmittance of the surface, which includes emission from the atmosphere which further absorbs?
“The number I have seen for direct emission from the surface to space in the IR windows is 22 W/m^2, about 9% of the total.”
That seems way too low to me given how large the transparent ‘window’ area is and that it lies on a very strong part of the emitted surface spectrum, i.e. the intensity is very high in the ‘window’ area. Even considering clouds basically completely close the window, i.e. the direct surface IR –> TOA component is zero where there are clouds, 22 W/m^2 still seems way, way too low.
GW calculates a spectral transmittance of the surface of about 0.24 for a global average atmosphere, which means the direct surface IR — TOA component is even more than about 93 W/m^2. He also calculates that upon doubling CO2, net absorption (i.e. the difference between the reduced IR intensity at the TOA and the increased IR intensity at the surface) increases by 3.6 W/m^2, which is right in line with what everyone else in the field is getting. Whether others in the field who also calculate about 3.6 W/m^2 for 2xCO2 also get a spectral transmittance of around 0.24, I don’t know. At one point, I asked Gunnar Myhre if he had that value and he said he didn’t (though he knew what it was).
BTW, the direct surface IR –> TOA component which is transmitted into space is not the same thing as ‘the spectral transmittance evaluated at the temperature of the surface’, which includes emission from the atmosphere which further absorbs the wavelengths emitted from the surface that are ‘missed’ by the direct surface IR — TOA component. The effect the emission from the layers in atmosphere have is to further reduce the spectral transmittance ‘T’ and further increase the spectral absorptance ‘A’. This is the only ‘T’ value that has any useful meaning. The direct surface IR –> TOA component which transmitted into space is purely trivial. It was never clear that anyone here even understood this, and was probably at the origin of a lot of the confusion.
Mike M.
You’re correct. Those numbers were for clear skies. So multiply them by 0.6 and they should be in the ballpark.
That’s 0.4, not 0.6.
I have no made data for the individual radiation fractions. When CO2 increase also the percentage will change. For example, is because of the higher surface temperature, the direct radiation from the surface to space is higher – in opposite train the radiation of the upper atmosphere must decrease, that is, decrease the temperature there. The percentages are a consequence of the heights of the tropopause. Although the height of the tropopause is also affected by the cloud of radiation from lower, but above the tropopause are hardly clouds. I write times with my German text before translation.
Ich habe keine Zahlen zu den einzelnen Strahlungsanteilen gemacht. Bei CO2-Anstieg werden sich auch die Prozentzahlen ändern. Z.B. ist wegen der höheren Oberflächentemperatur die Direktstrahlung von der Oberfläche zum Weltraum höher – im Gegenzug muß die Strahlung aus der oberen Atmosphäre abnehmen, d.h. die Temperatur dort sinken. Die Prozentzahlen sind eine Folge der Höhen der Tropopause. Zwar wird die Höhe der Tropopause auch durch die Wolkenstrahlung von darunter beeinflußt, aber oberhalb der Tropopause sind kaum Wolken. Ich schreibe mal meinen deutschen Text vor der Übersetzung mit.