In 1967 Journal of Atmospheric Sciences published the paper: Thermal Equilibrium of the Atmosphere with a Given Distribution of Relative Humidity by Manabe and Wetherald.
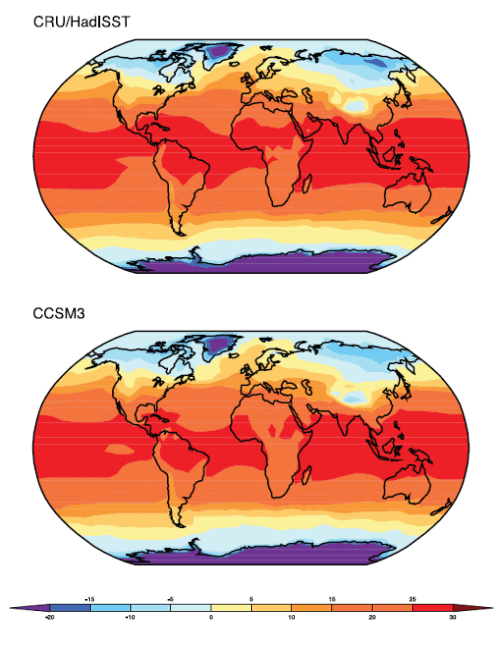
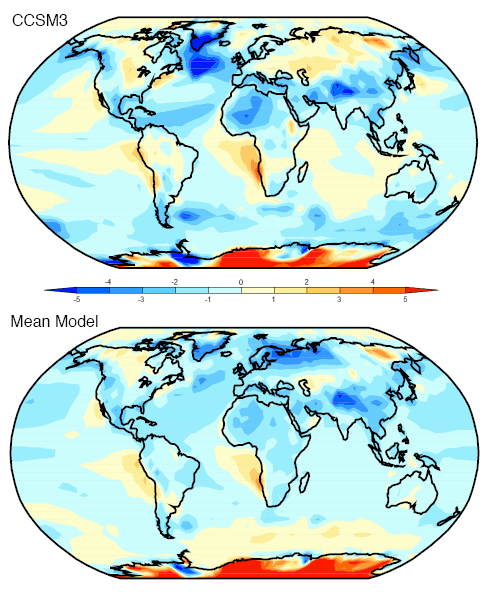
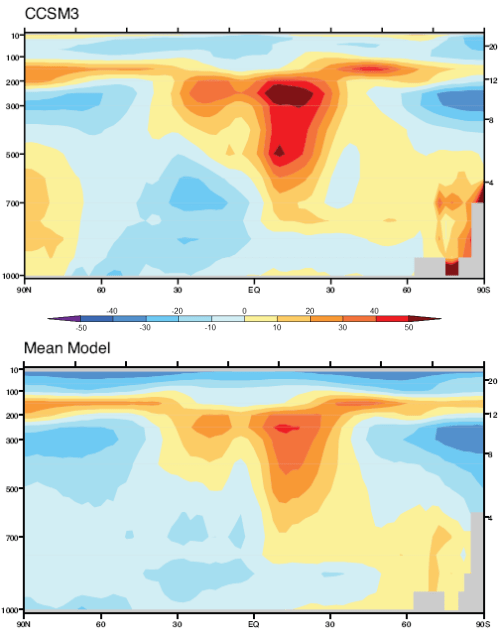
Here is one interesting model projection:
The corresponding note says:
Can this be true? How can “greenhouse” gases reduce temperature? Is this another “global warming causes more snow storms” type story?
First, a little about the stratosphere.
Stratospheric Basics
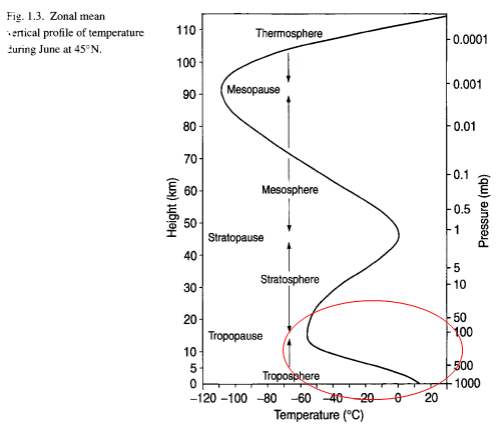
The stratosphere is the region of the atmosphere from around 10km to 50km. In pressure terms it’s the pressure between about 200mbar and 1mbar.
Ultraviolet radiation is almost completely absorbed in the stratosphere. The high energy photons of wavelength less than 0.24μm can break up molecular oxygen, O2, into atomic oxygen, O+O.
O2 and O combine to create O3, or ozone, which is again broken up with absorption of more ultraviolet.
Ozone production is greatest at a height around 25km. At higher levels, there are too few oxygen molecules to intercept all of the photons. At lower levels, there are few high energy photons left.
Here’s an interesting way of seeing how the absorption of solar energy at different wavelengths changes as thicker sections of the atmosphere, especially the stratosphere, are traversed:
The reason why the troposphere (lower atmosphere) warms from the bottom is that once the UV is absorbed the atmosphere is mostly transparent to the rest of the solar radiation. Therefore, the radiation passes straight through and is absorbed by the earth’s surface, which warms up and consequently warms the atmosphere from beneath.
Air that warms expands, and so rises, causing convection to dominate the temperature profile of the lower atmosphere.
By contrast, the stratosphere is warmer at the top because of the effect of solar absorption by O2 and O3. If there was no absorption by O2 or O3 the stratosphere would be cooler at the top (as it would only be heated from underneath by the troposphere).
Just about everyone has heard about ozone depletion in the stratosphere due to CFCs (and other chemicals). Less ozone must also cause cooling in the stratosphere. This is easier to understand than the model results at the beginning (from increased “greenhouse” gases). Less ozone means less ability to absorb solar radiation. If less energy is absorbed, then the equilibrium stratospheric temperature must be lower.
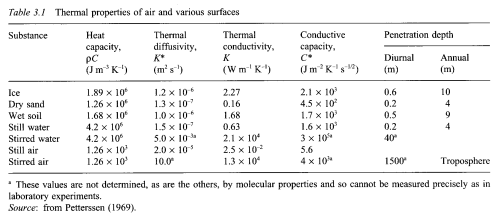
Stratospheric Temperature Trends
Temperature measurements of the stratosphere are limited. We have satellite data since 1979 which doesn’t provide as much vertical resolution as we need. We have radiosonde data since the 1940s which is limited geographically and also is primary below 30hPa (around 25km).
Lots of painful work has gone into recreating temperature trends by height/pressure and by latitude. For example, in the 2001 review paper by Ramaswamy and many co-workers (reference below), the analysis/re-analysis of the data took 23 of the 52 pages.
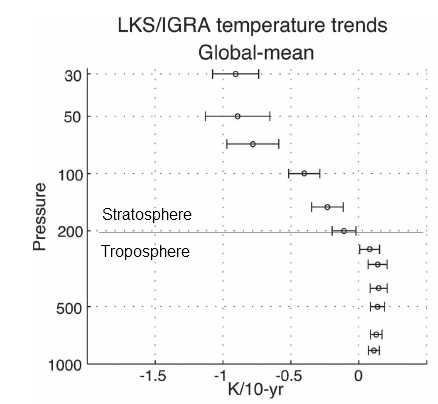
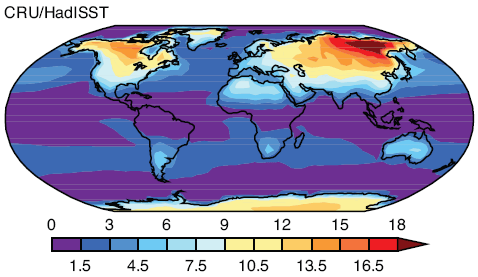
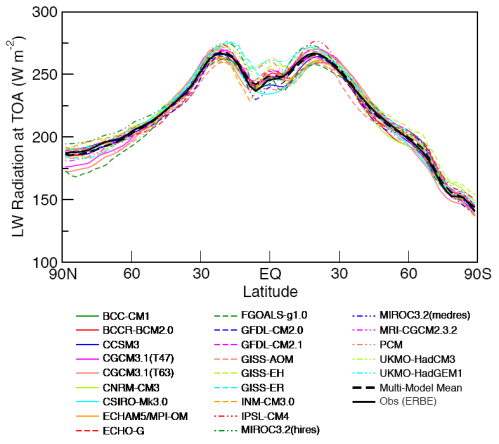
Here is one temperature profile reconstruction from Thompson and Solomon:
From Thompson & Solomon (2005):
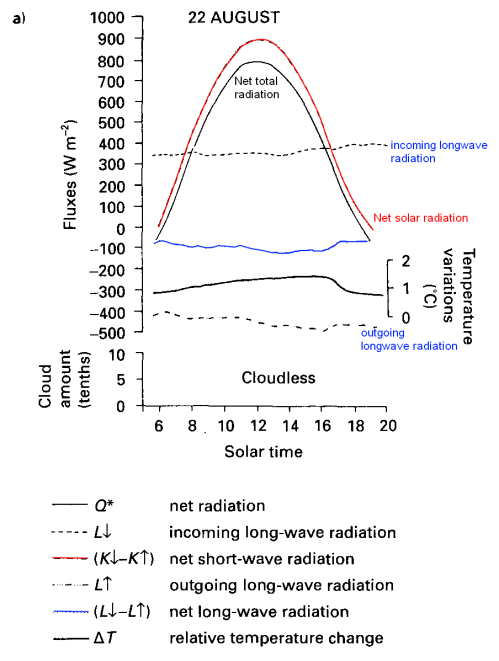
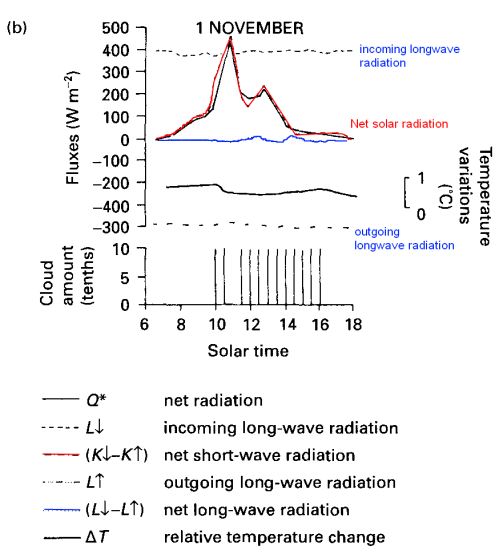
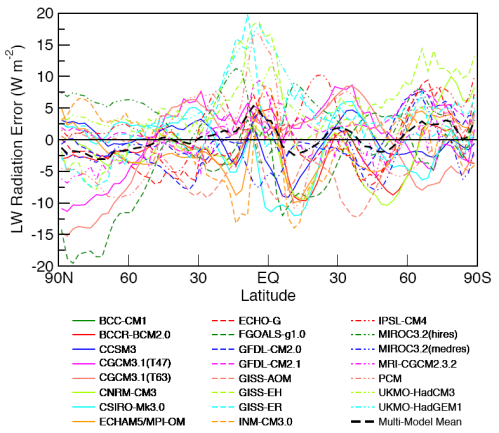
From 1979 to 1994, global-mean stratospheric temperatures dropped by 0.75 K / decade in the stratosphere below 35 km and 2.5 K / decade near 50 km
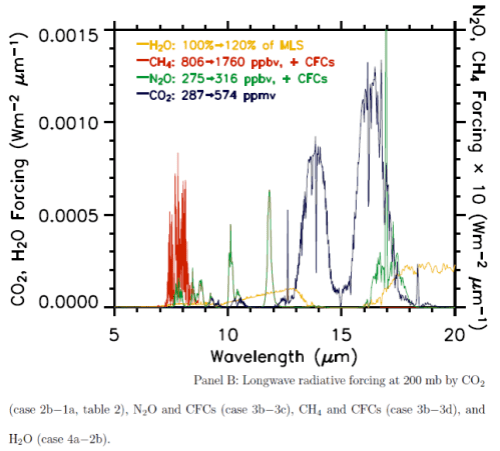
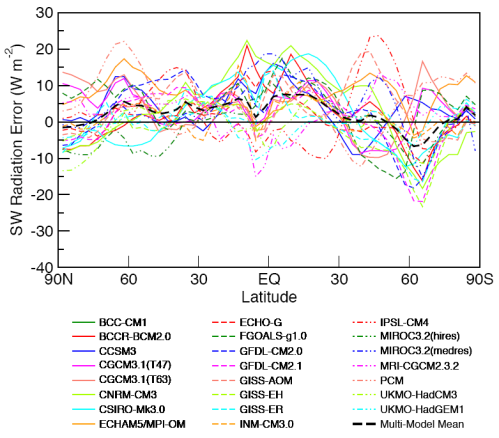
Before explaining why more CO2 and other trace gases could cause “stratospheric cooling”, it’s worth looking at the model results to understand the expected temperature effects of less ozone – and more CO2.
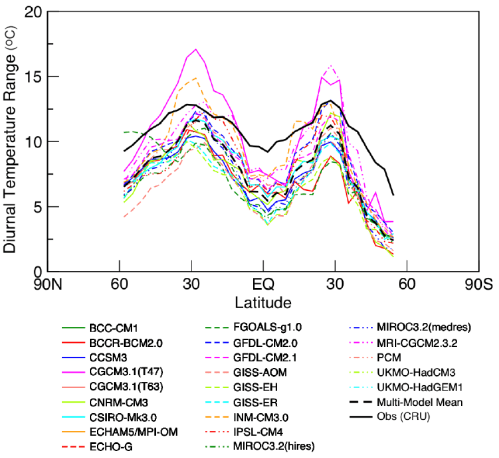
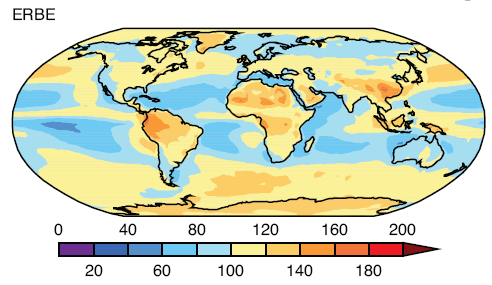
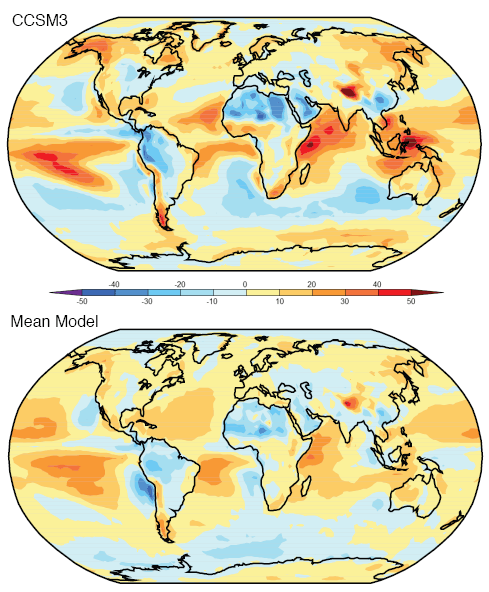
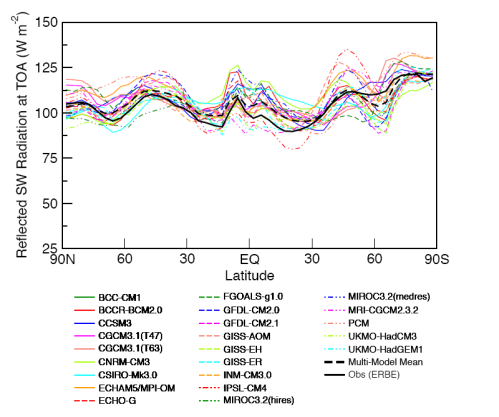
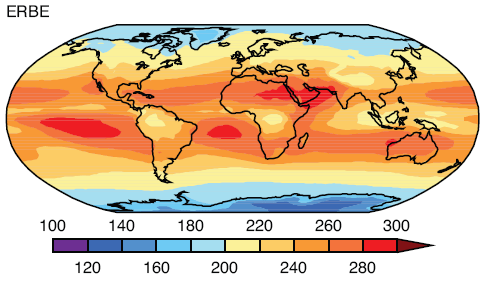
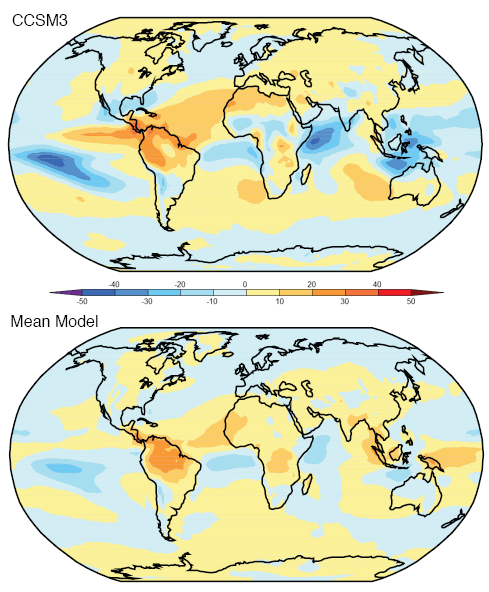
Observations and Recent Model Results
Notice that in the 1967 paper the predicted temperature drop was larger the higher up in the stratosphere. The effects of ozone are more complex and also there is more uncertainty in the ozone trends because ozone depletion has been more localized.
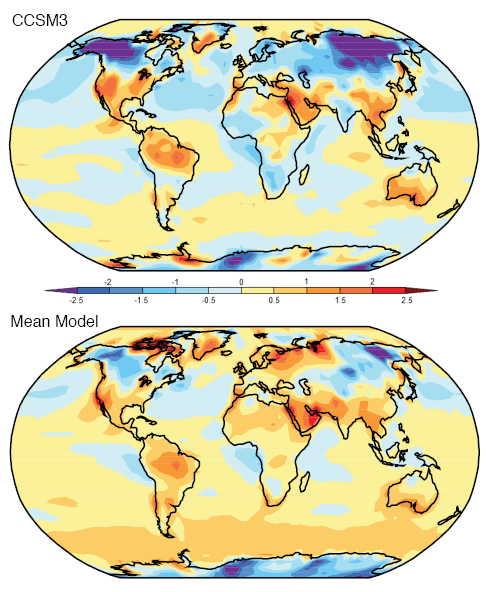
Here are model results for ozone – the best estimate of the observed temperature changes are in brown but aren’t expected to match the models because ozone is only one of the factors affecting stratospheric temperature:
Note that the effect of ozone depletion has a projected peak cooling around 1hPa (50km) and a second peak cooling around 80hPa.
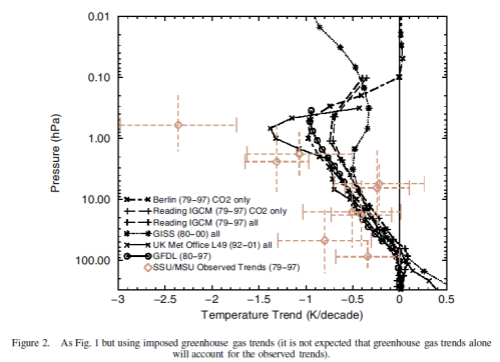
Now the same paper reviews the latest model results for stratospheric temperature from changes in “greenhouse” gases:
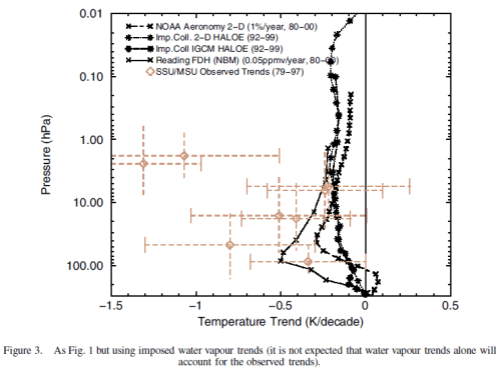
The same paper reviews the model results for changes in stratospheric water vapor. This is a subject which deserves a separate post (watch this space):
Finally, the model results when all of the effects are combined together:
The model results are a reasonable match with the observed trends – but a long way off perfect. By “reasonable match” I mean that they reproduce the general trends of decadal cooling vs height.
There are many uncertainties in the observations, and there are many uncertainties in the changes in concentration of stratospheric ozone and stratospheric water vapor (but not so much uncertainty about changes in the well-mixed “greenhouse” gases).
A couple of comments from A comparison of model-simulated trends in stratospheric temperatures, by Shine et al, first on the upper stratosphere, reviewing possible explanations of the discrepancies:
None of these potential explanations is compelling and so the possibility remains that the discrepancy is real, which would indicate that there is a temperature trend mechanism missing from the models.
and then on the 20-70hPa region:
Nonetheless, assuming that at least some part of this discrepancy is real, one possible explanation is stratospheric water vapour changes. Figure 3 indicates that an extra cooling of a few tenths of a K/decade would result if the Boulder sonde-based water vapour trends were used rather than the HALOE water vapour trends. If this were one explanation for the model–observation difference, water vapour could dominate over ozone as the main cause of temperature trends in this altitude region.
Why Is the Stratosphere Expected to Cool from Increases in “Greenhouse” Gases?
This is a difficult one to answer with a 30-second soundbite. You can find a few “explanations” on the web which don’t really explain it, and others which appear to get the explanation wrong.
The simplest approach to explaining it is to say that the physics of absorption and emission in the atmosphere – when calculated over a vertical section through the atmosphere and across all wavelengths – produces this result. That is – the maths produces this result..
You can see an introduction to absorption and re-emission in CO2 – An Insignificant Trace Gas? Part Three.
[Note added to this article much later, the series Visualizing Atmospheric Radiation has an article Part Eleven – Stratospheric Cooling – from January 2013 on why the stratosphere is expected to cool as CO2 increases. It is quite involved but shows the detailed mechanism behind stratospheric cooling].
After all, this approach is what led Manabe and Wetherald to their results in 1967. But of course, we all want to understand conceptually how an increase in CO2 – which causes surface and troposphere warming – can lead to stratospheric cooling.
The great Ramanathan in his 1998 review paper Trace-Gas Greenhouse Effect and Global Warming (thanks to Gary Thompson of American Thinker for recommending this paper) says this:
As we mentioned earlier, in our explanation of the greenhouse effect, OLR reduces (with an increase in CO2) because of the decrease in temperature with altitude.
In the stratosphere, however, temperature increases with altitude and as a result the cooling to space is larger than the absorption from layers below. This is the fundamental reason for the CO2 induced cooling.
In Ramaswamy (2001):
For carbon dioxide the main 15-um band is saturated over quite short distances. Hence the upwelling radiation reaching the lower stratosphere originates from the cold upper troposphere. When the CO2 concentration is increased, the increase in absorbed radiation is quite small and the effect of the increased emission dominates, leading to a cooling at all heights in the stratosphere.
Are they saying the same thing? Yes (probably).
If these explanations help – wonderful. If they don’t, refer to the maths. That is, the mathematical result provides this solution and overall “hand waving” explanations are only ever a second-best “guide”. Also check out The Earth’s Energy Budget – Part Three for explanations about emissions from various levels in the atmosphere.
Conclusion
Understanding stratospheric temperature trends is a difficult challenge. Understanding the mechanisms behind this changes is much more of a conceptual challenge.
But over 40 years ago, it was predicted that the upper stratosphere would cool significantly from increases in CO2.
The depletion of ozone is also predicted to have an effect on stratospheric temperatures – in the upper stratosphere (where CO2 increases will also have the most effect) and again in the lower stratosphere where ozone is the dominant factor.
Stratospheric water vapor also has an effect in the lower stratosphere (where more water vapor leads to more warming and vice-versa), but more on this in a later post.
For some, who feel/believe that CO2 can’t really significantly affect anything in climate – this post isn’t for you – check out the CO2 – An Insignificant Trace Gas? series.
There will be others who will say “Ozone is the reason the upper stratosphere has cooled“. True, but increases in CO2 are also an important factor. The same calculations (maths and physics) that lead to the conclusion that less ozone will cool also lead to the conclusion that more CO2 will cool the upper stratosphere.
This subject also has two other possible consequences. One is about attribution. Global temperatures have increased over the last 40 years and many people want to understand the cause.
If solar heating was the direct cause (see Here Comes the Sun) the stratosphere would not be cooling. However, other effects could possibly also cause stratospheric cooling at the same time as tropospheric and surface heating. It’s a complex subject. But something to question for those other potential causes – would they also cause stratospheric cooling?
The other consequence is about GCMs. Some say that stratospheric cooling is a “vindication” of GCMs. In so far as we have covered the subject in this post we couldn’t reach that conclusion. The modeling of tropospheric and stratospheric temperature profiles can be done (and was by Manabe and Wetherald) with 1D radiative-convective models. Certainly 3d GCMs have also been used to calculate the effect by latitude but these results have more issues – well, the whole subject is much more complex because the change of ozone with height and latitude are not well understood.
But it is important to understand the difference between a GCM solving the general climate problem and a more constrained mathematical model solving the temperature profile against height through the atmosphere.
However, stratospheric cooling while the surface and troposphere are warming does indicate that CO2 and other “greenhouse” gases are likely influencers.
References
Thermal Equilibrium of the Atmosphere with a Given Distribution of Relative Humidity, Manabe and Wetherald, Journal of Atmospheric Sciences (1967)
Trace-Gas Greenhouse Effect and Global Warming, Ramanathan, Royal Swedish Academy of Sciences (1998)
Stratospheric Temperature Trends: Observations and Model Simulations, Ramaswamy et al, Review of Geophysics (2001)
A comparison of model-simulated trends in stratospheric temperatures, Shine et al, Q. J. R. Meteorol. Soc. (2003)
Recent Stratospheric Climate Trends as Evidenced in Radiosonde Data: Global Structure and Tropospheric Linkages, Thompson & Solomon, Journal of Climate (2005)
An update of observed stratospheric temperature trends, Randel, Journal of Geophysical Research (2008)










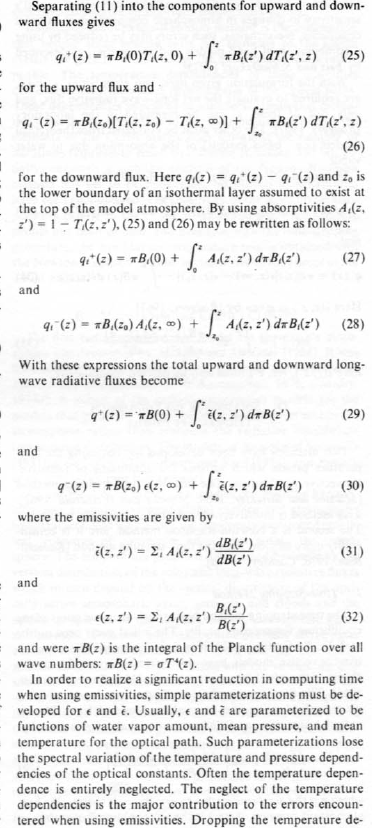



On the Miseducation of the Uninformed by Gerlich and Tscheuschner (2009)
Posted in Commentary on April 5, 2010| 226 Comments »
In On Having a Laugh – by Gerlich and Tscheuschner (2009) I commented that I had only got to page 50 and there were 115 pages in total.
Because there were so many errors already spotted, none central to the argument (the argument hadn’t started even at page 50), it seemed a pointless exercise to read it further. After all, many interesting papers await, on the thermohaline circulation, on models, on stratospheric cooling..
Perhaps most important of the criticisms was that Gerlich and Tscheuschner didn’t appear at all familiar with the climate science they were “debunking” – instead of commenting on encyclopedia references or throwaway comments in introductions to works unrelated to proving the inappropriately-named “greenhouse effect” they should be commenting on papers like Climate Modeling through Radiative-Convective Models by Ramanathan and Coakley (1978).
However, after noticing that a recent commenter actually cited Gerlich and Tscheuschner I went back and reviewed their paper. And in doing so I realized that many many misinformed comments by enthusiastic people on other popular blogs, and also this one, were included in the ground-breaking On Falsification Of The Atmospheric CO2 Greenhouse Effects by Gerlich and Tscheuschner.
It’s possible that rather than enthusiastic commenters obtaining misinformation from our duo that instead our duo have combined a knowledge of theoretical thermodynamics with climate science that they themselves obtained from blogs. The question of precedence is left as an exercise for the interested reader.
Miseducation
It is hard to know where to start with this paper because there is no logical flow.
Conductivity
The paper begins by reviewing the conductivity of various gases.
Clearly conductivity is the least important of means of heat transfer in the atmosphere. Radiation, convection and latent heat all get a decent treatment in studies of energy balance in the atmosphere.
If our duo had even read one book on atmospheric physics, or one central paper they would be aware of it.
Uninformed people might conclude from this exciting development that they have already demonstrated something of importance rather than just agreeing wholeheartedly with the work of atmospheric physicists.
Pseudo-Explanations to be Revealed in Part Two? Or Left as an Exercise for the Interested Student?
Following some demonstrations of their familiarity with mathematics and especially integration, they provide three conclusions, one of which refers to the Stefan-Boltzmann law, j=σT4:
and finish with (p21):
Unfortunately they don’t explain which ones. The climate science world waits with baited breath..
The footnote to their comment on Stefan-Boltzmann:
Sadly they are unfamiliar with the standard works in the field of the radiative-convective model.
Solar Energy Breakdown and A Huge Success in Miseducation
Solar Radiation Breakdown
They followed up this table with the hugely popular comment:
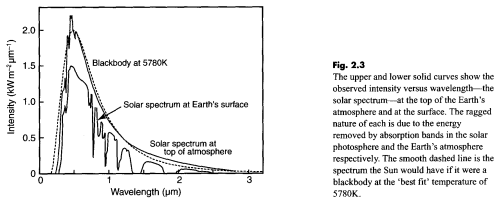
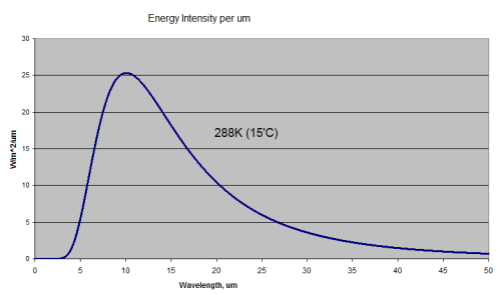
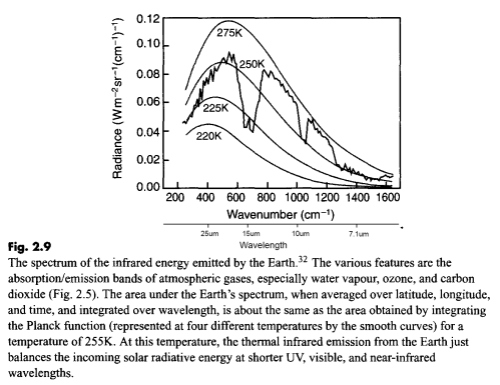
First, a comment on the “benefit” of this miseducation – being able to separate out solar radiation from terrestrial radiation is a huge benefit in climate understanding – it allows us to measure radiation at a particular wavelength and know its source. But many people are confused and say we can’t because 50% of the solar radiation is “infrared”. Infrared means >0.7μm. Conventionally, climate scientists use “shortwave” to mean radiation < 4μm and “longwave” to mean radiation > 4μm. As less than 1% of solar radiation is >4μm this is a very useful convention. Any radiation greater than 4μm is terrestrial (to 99% accuracy).
Many uninformed people who have become miseducated are certain that much solar radiation is >4μm – possibly due to confusing infrared with longwave.
We don’t speculate on motives on this blog so I’ll just point out that Gerlich and Tscheuschner know very little about any climate science, and from this comment probably don’t even understand the inappropriately-named “greenhouse” effect.
Why? Well, what has the visibility of the radiation have to do with the “greenhouse” effect? Of course it’s ignored. Our duo are just demonstrating their ignorance of the absolute basics.
Or they have some amazing insight into how the visibility or not of solar radiation affects the radiative transfer equations. All to be shared in part two probably..
The Core Question – the Radiative Transfer Equations
After a brief explanation of Kirchoff’s law, our duo discuss the core equations, the radiative transfer equations (RTE):
Absorption and emission coefficients get a very thorough treatment in the numerical solutions to the RTE, however, our duo are only familiar with work around the 1900’s and skip all modern work on the subject. Perhaps a more accurate statement would be:
Or they could actually show what effect that dependency actually had..
Then they decide to support the RTE:
Fantastic, 50 pages in we find the real RTE. This is what atmospheric physicists use to calculate the absorption and re-emission of radiation for each layer in the atmosphere. They follow this up with:
What do they mean? Of course you need boundary conditions to solve all real-world equations.
The separate directions are independent of one another? Yes, you find that in all treatments of radiative transfer.
So Gerlich and Tscheuschner agree that the RTE can be used to solve the problem? Or not? No one can tell from the comments here. If they do, the paper should be over now with support for the inappropriately-named “greenhouse effect”, unless they demonstrate that they can solve them for the atmosphere and get a different result from everyone else.
But they don’t.
Fortunately for those interested in what our duo really know and understand – they tell us..
The Modern Solution to the RTE – or How to Miss an Important 100 Years
After surveying works from more than 100 years ago, they conclude:
No explanation of Callendar and Keeling’s mistakes – this is left as an exercise for the interested student.
And no mention of the critical work in the 1960s and 1970s which used the radiative transfer equations and the convective structure of the atmosphere to find the currently accepted solutions.
In fact, the research results haven’t been vague at all. Regular readers of this blog will know about Ramanathan and Coakley 1978, and there are many more specific papers which find solutions to the RTE – using boundary conditions and separation of upward and downward fluxes, as wonderfully endorsed by our comedic duo.
More recent work has of course refined and improved the work of the 1960s and 1970s. And the measurements match the calculations.
But what a great way to write off a huge area of research. Show some flaws in the formative work 100 or so years ago and then skip the modern work and pretend you have demonstrated that the modern theory is wrong.
As we saw in the last section, our duo appear to support the modern equations – although they are careful not to come out and say it. Luckily, they are blissfully ignorant of modern work in the field, which all helps in the miseducation of the uninformed.
The main work of the paper should now be over, but our duo haven’t realized it. So instead they move randomly to the radiative balance concept..
Radiative Balance and Mathematical Confusion
In every introduction to atmospheric physics you find the concept of radiative balance – solar energy absorbed = terrestrial radiation emitted from the top of the atmosphere. These concepts are used to demonstrate that the atmosphere must absorb longwave (terrestrial) radiation.
This concept can be found in CO2 – An Insignificant Trace Gas? Part One
After looking at the basics of the energy balance, they comment – on the right value for albedo (or ‘1-albedo’):
Being obscure impresses the uninformed. However, the informed will know that the earth’s emissivity and absorptivity will of course be different because the solar radiation is centered on 0.5μm while the terrestrial radiation is centered on 10μm. And the emissivity (and absorptivity) around 10um is very close to 1 (typically 0.98) while around 0.5μm the absorptivity is somewhat lower.
At this point, if we were to do a parody of our duo, we would write how their physics is extremely poor and do a three page derivation of absorptivity and emissivity as a function of wavelength.
Now follows many pages of maths explaining the impossibility of working out an average temperature for the earth during which they make the following interesting comment:
What they are saying is that for energy balance if we work out the radiation emitted from the earth we have dealt with the problem.
Fortunately for our intrepid duo, they are unacquainted with any contemporary climate science so the fact that someone has already done this work can be safely ignored. Earth’s Global Energy Budget by Trenberth, Fassulo and Kiehl (2008) covers this work.
So there is potential variation of a few W/m2 depending on the approach, and Trenberth et al settles on 396 W/m2 average – at least the values can be calculated, whereas our duo decided it was computationally impossible – perhaps as they saw the problem as requiring a totally accurate GCM.
With this information, the radiative balance problem can be resolved and we can see that there is a discrepancy between the solar energy absorbed and the terrestrial radiation emitted which requires explanation. The inappropriately-named “greenhouse effect”.
Without this information we can delight in much maths and pretend that nothing can be known about anything.
Why Conduction Can be Safely Ignored and Why We Just Demonstrated It
In fact in many texts on atmospheric physics conduction is safely ignored due to the very low value of heat conduction through gases. Strictly speaking, if we write an equation then all terms should be included, including latent heat and convection. Why just radiation and conduction?
As Ramanathan and Coakley pointed out in their 1978 paper, convection is what determines the temperature gradient of the atmosphere but solving the equations for convection is a significant problem – so the radiative convective approach is to use the known temperature profile in the lower atmosphere to solve the radiative transfer equations.
Still, no thought of conduction as that term is so insignificant – as our intrepid duo go on to realize..
Commenting on the insolubility of heat flow via conduction they take a “typical example”:
So heat flow by conduction is so low that achieving balance by this method will take more than the age of the universe. Therefore, it is insignificant in comparison with convection and radiation.
Good so we can move on and climate scientists are right to ignore it. Was that the point that Gerlich and Tscheuschner were making? Yes, although possibly without realizing it..
Finally, the Imaginary Second Law of Thermodynamics
In their almost concluding section we see where countless climate enthusiasts have obtained their knowledge (or the reverse).
First, here’s an extract from a contemporary work on thermodynamics. This is from Fundamentals of Heat and Mass Transfer, 6th edition (2007), by Incropera & Dewitt:
As can be seen in the text, radiation can be absorbed by a higher temperature surface from a lower temperature surface and vice versa. Of course, the net result is a heat transfer from the hotter to the cooler.
The same uncontroversial description can be found in any standard thermodynamics work, unless they consider it too unimportant to mention. Certainly, none will have a warning sign up saying “this doesn’t happen”.
The explanation of the “greenhouse effect” is that the earth’s surface warms the lower atmosphere by radiation (as well as convection and latent heat transfer). And the atmosphere in turn radiates energy in all directions – one of which is back to the earth’s surface. Believers in the imaginary second law of thermodynamics don’t think this can happen. And this is possibly due to the miseducation by our intrepid duo. Or perhaps they learnt their thermodynamics from many “climate science” blogs.
The result of the actual climate situation is that the earth’s surface is warmer than it would have been without this atmospheric radiation. Pretty simple in concept.
Here’s how Gerlich and Tscheuschner explain things:
Everyone agrees.
Now the confusion. What are they saying? This isn’t what atmospheric physicists describe. The net heat transfer is from the earth’s surface (which was warmed by the sun) to the atmosphere.
Are they saying that it is impossible for any radiation to transfer heat from the atmosphere to the earth? It would appear so –
Following their diagram above, they comment, first quoting Rahmstorf:
Our duo first attempt to confuse, as they frequently do in their opus by claiming that a clear explanation is obscure because precise enough terms aren’t used. It’s not obscure because they make the “correction” themselves.
Then add their masterstroke. It is inadmissible to apply the second law for the upward and downward heat separately redefining the thermodynamic system on the fly.
What on earth do they mean? Our comedic duo are the ones separating the system into upward and downward heat, followed by an enthusiastic army on the internet. Everyone else considers net heat flow.
As we saw in a standard work on thermodynamics, now in its 6th edition after two or three decades in print, there is no scientific problem with radiation from a colder to a hotter body – so long as there is a higher radiation from the hotter to the colder.
At this point I wonder – should I revisit the library and scan in 20 thermodynamic works? 50? What would it take to convince those who have been miseducated by our intrepid duo?
Perhaps Gerlich and Tscheuschner can now turn their attention to all of the unscientific text books like the one shown at the start of this section..
Conclusion
There is much to admire in Gerlich and Tscheuschner’s work. It can surely become a new standard for miseducation and we can expect its deconstruction by psychologists and those who study theories of learning.
From a scientific point of view, there is less to admire.
They have no understanding of modern climate science, content to dwell on works from over 100 years ago and ignoring any modern work. They appear to believe that the basis for the “greenhouse” effect is an actual greenhouse (as was covered in On Having a Laugh) even though no serious work on the subject relies on greenhouses. (Some don’t even mention it, some mention it to point out that the atmosphere doesn’t really work like a greenhouse).
In fact, the serious work of the last few decades relies on the radiative transfer equations – equations apparently endorsed by our duo, although their comments are “obscure”.
They take many other snipes at climate science by the approach of pointing out a term or dependency has been “neglected” (for example, like conduction through the atmosphere) without showing that the neglect has a significant impact – except in the case of conduction where (unwittingly?) they appear to show that conduction should definitely be ignored!
Someone could take issue with even modern work on climate science by the fact that they ignore relativistic effects.
And after 18 pages of unnecessary re-derivation of general relativity we find that “it’s therefore impossible to calculate this and the problem is insoluble“..
Well, although they haven’t read any modern climate science, it’s hard to see how they could be so confused about the application of the 2nd law of thermodynamics.
Perhaps in their follow up work they can explain why all the thermodynamics works are wrong, and especially where this 15μm (longwave) radiation comes from:
Measured downward longwave radiation at the earth's surface
According to their interpretation of the 2nd law of thermodynamics this can’t happen. No heat can flow from the colder atmosphere to the warmer surface as that would be a “perpetuum mobile” and therefore impossible.
Where is it coming from Gerlich and Tscheuschner?
Read Full Post »