Recap
Part One of the series started with this statement:
If there’s one area that often seems to catch the imagination of many who call themselves “climate skeptics”, it’s the idea that CO2 at its low levels of concentration in the atmosphere can’t possibly cause the changes in temperature that have already occurred – and that are projected to occur in the future. Instead, the sun, that big bright hot thing in the sky (unless you live in England), is identified as the most likely cause of temperature changes.
Part One looked mainly at the radiation balance – what the sun provides (lots of energy at shortwave) and what the earth radiates out (longwave). Then it showed how “greenhouses gases” – water vapor, CO2 and methane (plus some others) – absorb longwave radiation and re-emit radiation both up out of the atmosphere and back down to the earth’s surface. And without this absorption of longwave radiation the earth would be 35°C cooler at its surface. The post concluded with:
CO2 and water vapor are very significant in the earth’s climate, otherwise it would be a very cold place.
What else can we conclude? Nothing really, this is just the starting point. It’s not a sophisticated model of the earth’s climate, it’s a “zero dimensional model”.. the model takes a very basic viewpoint and tries to establish the effect of the sun and the atmosphere on surface temperature. It doesn’t look at feedback and it’s very simplistic.
Two images to remember..
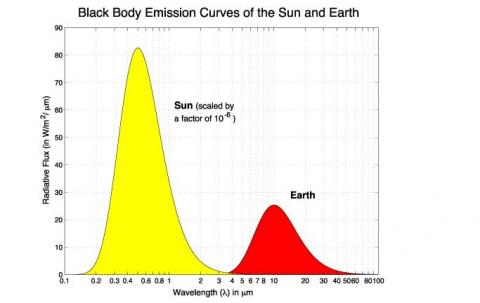
First, the sun’s radiated energy is mostly under 4μm in wavelength (shortwave), while the earth’s radiated energy is over 4μm (longwave), meaning that we can differentiate the two very easily:
Second, the aborption that we can easily measure in the earth’s longwave radiation from different molecules:
Recap over.. This post was going to introduce the basic 1-d model of radiative transfer, but enough people asked questions about the absorption properties of gases that I thought it was was worth covering in more detail.. The 1-d model will have to wait until Part Three.
Why don’t the Atmospheric Gases Absorb Energy according to their Relative Volume?
Just because CO2 only consists of 0.04% of gases doesn’t mean it only contributes 0.04% of atmospheric absorption and re-emission of long wave radiation. Why is that?
Oxygen, O2, constitutes 21% of the atmophere and nitrogen, N2, constitutes 78%. Why aren’t they important “greenhouse” gases? Why are water vapor, CO2 and methane (CH4) the most important when they are present in such small amounts?
For reference, the three most important gases by volume are:
- Water vapor – 0.4% averaged throughout the atmosphere, but actual value in any one place and time varies (See note 1 at end of article)
- CO2 – 0.04% (380ppmv), well mixed (note: ppmv is parts per million by volume)
- CH4 – 0.00018% (1.8ppmv), well mixed
Now there are three factors in determining the effect of longwave absorption:
- The amount of the gas by volume
- How much longwave energy is radiated from the earth at wavelengths that the gas absorbs
- The ability of the gas to absorb energy at a given wavelength
The first one is the simplest to understand. In fact, it’s knowing only this factor that causes so much confusion.
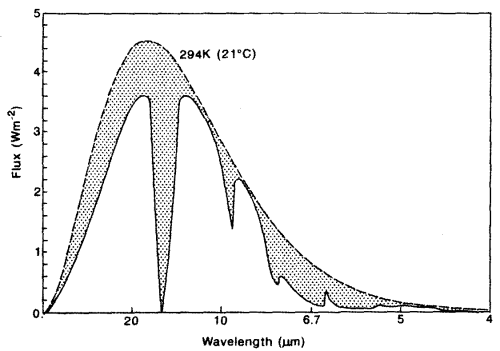
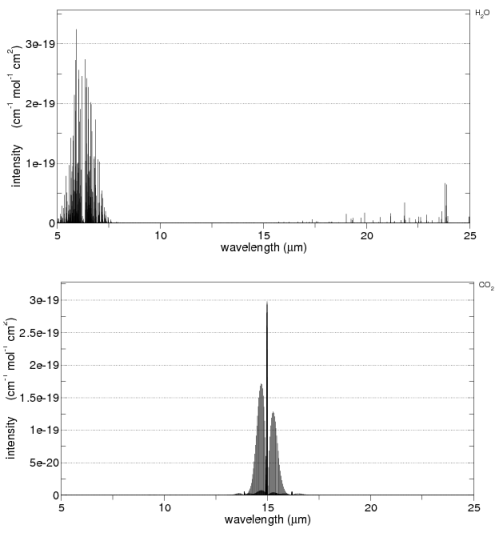
The second point is not immediately obvious, but should become clearer by reviewing the earth’s radiation spectrum:
Different amounts of energy are radiated at different wavelengths. For example, the amount of energy emitted between 10-11μm is eight times the amount of energy between 4-5μm (for radiation from a surface temperature of 15 °C or 288K).
CO2 has a wide absorption band centered around 15μm, which is where the long-wave radiation from the earth is at almost its highest level. By contrast, one of water vapor’s absorption lines is at 6.27μm – where the radiation is a slightly lower level (about 25% less) and more importantly, the other water vapor absorption lines are where the radiation is 5-10x lower intensity.
However, there is around 10x as much water vapor than CO2 in the atmosphere, which is why it is the most important greenhouse gas.
And Third, Why are Some Gases More Effective at Absorbing Longwave Energy?
Why aren’t O2 and N2 absorbers of longwave radiation?
Molecules with two identical atoms don’t change their symmetry when any rotation or vibration takes place. As a result they can’t move into different energy states.
But triatomic molecules like CO2, H2O and CH4 can bend as they vibrate. They can move into different energy states by changing their shape. Consequently they can absorb the energy from an incoming photon if its energy matches the new state.
And some molecules have many more energy states they can move into. This changes their absorption profile because their spectral breadth is effectively wider.
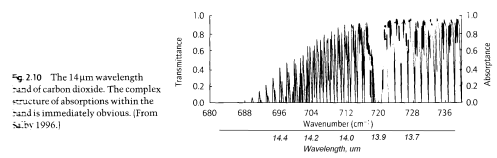
Here’s a graphic of one part of the actual CO2 absorption lines. Apologies for the poor quality scan..
(Note that the x-axis is “Wavenumber, cm-1”. This is a convention for spectral people. Wavenumber is the number of wavelengths present in 1cm. I added the actual wavelength underneath.)
This shows the complexity of the subject once we look at the real detail. In practice, these individual discrete absorption lines “broaden” due to pressure broadening (collisions with other molecules) and Doppler broadening (as a result of the absorbing molecule moving in the same or opposite direction to the photon of light).
However, the important point to remember is that different molecules absorb at different frequencies and across different ranges of frequencies.
This third factor is the most important in determining the absorption properties of longwave radiation.
As an interesting comparison, molecule by molecule methane absorbs about 20x as much energy as CO2. But of course it is present in much smaller quantities.
Here are water vapor and CO2 across 5-25μm from the HITRANS database:
See Note 2 at the end of the article.
What about Oxygen?
A digression on oxygen.. It is important in the earth’s atmosphere because it absorbs UV, but when these high energy photons from the sun interact with O2 it breaks into O+O. Then a cycle takes place where O2 and O combine to form O3 (ozone), and later O3 breaks up again. By the time the sun’s energy has reached the lower part of the atmosphere (troposphere) all of the lower wavelength energy (most of the UV) has been filtered out.
O3 itself does absorb some longwave energy, at 9.6um, but because there is so little O3 in the troposphere it is not very significant.
What Happens when a Greenhouse Gas Absorbs Energy?
Once a gas molecule has absorbed radiation from the earth it has a lot more energy. But in the lower 100km of the atmosphere, the absorbed energy is transferred to kinetic energy by collisions between the absorbing molecules and others in the layer. Effectively, it heats up this layer of the atmosphere.
The layer itself will act as a blackbody and re-radiate infrared radiation. But it re-radiates in all directions, including back down to the earth’s surface. (If it only radiated up away from the earth there would be no “greenhouse” effect from this absorption).
Conclusion
We are still on the “zero dimensional model” – some call it the billiard ball model – of the radiative balance in the earth’s climate system.
A few different factors affect the absorption of the earth’s longwave radiation by various gases.
O2 barely absorbs any (see note 2 below), and neither does N2 (nitrogen). Among the other gases – the main greenhouse gases being water vapor, CO2 and methane – we see that each one has different properties – none of which can be determined by our intuition!
Different molecules can absorb energy in certain frequencies simply because of their ability to change shape and move to different energy states. The primary property that creates a strong “greenhouse” effect is to have a strong and wide absorption around a wavelength that the earth radiates. This is centered about 10μm (and isn’t symmetrical) so the further away from the peak energy the absorption occurs, the less relevant that absorption line becomes in the earth’s energy balance.
In the next part in the series, we will look at the 1-dimensional model and also what happens when absorption in a wavelength is saturated.
Note 1 – Water Vapor ppmv: After consulting numerous reference works, I couldn’t find one which gave the averaged water vapor throughout the atmosphere, or the troposphere. The actual source for the 0.4% was Wikipedia.
Because all the reference works danced around without actually giving a number I suspect it is “up in the air”. Here is one example:
Water vapor concentration is highly variable, ranging from over 20,000 ppmv (2%) in the lower tropospherical atmosphere to only a few ppmv in the stratosphere..
Atmospheric Science for Environmental Scientists (2009) Hewitt & Jackson
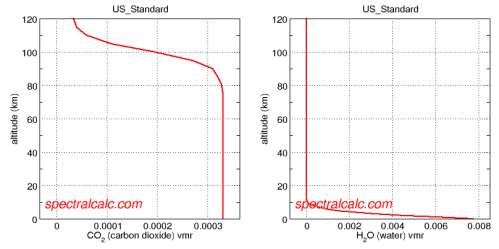
There is a great application, Spectral Calc for looking at atmospheric concentrations and absorption lines. Specifically http://spectralcalc.com/atmosphere_browser gives plots of atmospheric concentration and the data agrees with the Wikipedia number given in the body of this article:
Averaging over the whole atmosphere, the concentration of water vapor does seem to be around 10x the CO2 value.
Note 2 – Optical Thickness: The spectral plots from the HITRANS database shown in the body of the article give the capture cross-section per mole (i.e. per “unit” of that gas, not per unit volume of the general atmosphere).
One commenter asked why another plot from a different website drawing on the same HITRANS database produced this:
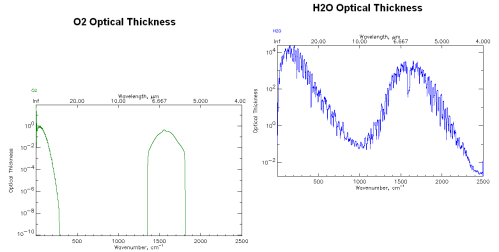
Optical Thickness of O2 and water vapor from http://www.atm.ox.ac.uk
Note that I’ve adjusted the plots so that similar values on the y-axes are aligned for both graphs. And note that the vertical axis is logarithmic.
His comment was that oxygen, O2, is only maybe 1000 times lower in absorption than water vapor (100 =1 vs 103 =1000) at 6.7μm and given that O2 is 20% of the atmosphere instead of 0.4%, O2 should be comparable to water vapor as a greenhouse gas.
But in fact, this graphical plot isn’t plotting the absorption by units of molecule – instead it is plotting Optical Thickness.
This is a handy variable which we will see more of in Part Three. Optical Thickness essentially takes the value of Intensity, which is per unit of molecules, and “integrates” that value up through the entire height of the atmosphere.
As a result it gives the picture of the complete influence of that gas at different frequencies without having to work out the relative proportions of the gas at different heights in the atmosphere.
So the example above compares the complete absorption (in a simplistic model) through the whole atmosphere, giving O2 about 3000x less effect than water vapor at 6.7μm.
Update – Part Three is now online


[…] Update – check out Part Two […]
Science,
Thank you for the analysis.
Yes, overall water vapor % are hard to find. One reason is that is is highly localized. Thus, the interest in O2. It is uniformly distributed and would, I think have an effect effect in the absence of water vapor (poles).
Here is a textbook that references O2 absorption in the IR range.
http://books.google.com/books?id=SF8fdwacahkC&pg=PA157&lpg=PA157&dq=o2+absorption+spectrum+um&source=bl&ots=W3bx_ELC-S&sig=GHYDSYlHgTZAhh4XS1l236m2E6k&hl=en&ei=t3s5S87PGMmenQf2gZ2CCQ&sa=X&oi=book_result&ct=result&resnum=7&ved=0CCwQ6AEwBg#v=onepage&q=o2%20absorption%20spectrum%20um&f=false
All of this seems pretty standard and non controversial.
However, not all of the surface/atmosphere is at 15C/288K.
In the tropics it is more like 25C/298K, isn’t it.
This causes a shift downward of the maximum wavelength of emission, which would seem to me to increase the ratio of energy absorbed by H2O vs CO2.
It would also seem to make water vapor more important in the tropics and CO2 more important at the poles, or maybe at winter temps like -30C/243K even CO2 does not get much action?
Richard Sharpe:
You are correct, except for some – sadly – it is controversial.
Of course this analysis is still the “billiard ball” or zero dimensional analysis which is the only place to start. It totally ignores important factors like the actual surface temperature in any given place and time.
CO2 and water vapor don’t see the average temperature, they see the radiation from the actual temperature in that location.
Taking the tropics as your example, supposing 298K and comparing it with the average of 288K..
Radiation in 5-7um band: 7.4 (288K) 9.7 (298K)
Radiation in 14-16um band 11.6 (288K) 13.1 (298K)
units are W/m^2/sr
As the temperature goes up 10K, the ratios of radiance from 5-7um vs 14-16um goes from 64% to 74%.
So you are right that the lower frequency bands of water vapor will get proportionately more “action” than CO2 in hotter climates. But the higher frequency bands of water vapor at 25um and up will also get less.
Note that 298K has an emittance of 447W/m^2
while 288K has an emittance of 390W/m^2
-Numbers were calculated from http://spectralcalc.com/blackbody_calculator/blackbody.php
As part 3 of this series will cover, the only way to actually get results of the CO2 and water vapor effect even at one temperature and location is numerical integration of the relevant formulae.
Stay tuned..
Thanks for that feedback.
I suspect that the numerical integration is going to be difficult as well.
Above a specific pressure the mean free path will be short enough that CO2 and H2O will collide with other molecules in the atmosphere before they can emit any radiative energy. I don’t know the actual details (of mean time to emit for each of H2O and CO2), but in looking at CO2 laser details it seems clear that an excited CO2 molecule can transfer energy to N2. Not sure if H2O has any vibrational modes in near resonance with N2, so I guess I will have to do some more learning.
However, it would seem that lower in the atmosphere CO2 absorbs a quanta of energy and then transfers it to the surrounding atmosphere, while higher in the atmosphere is re-radiates that energy (with half radiated downward and thus likely to be absorbed, half radiated upward and likely to escape), and this seems to complicate matters somewhat.
I look forward to your future posting. I hope to learn more.
[…] Part Two we looked a little closer at why certain gases absorbed energy in certain bands and what the […]
I just found a very nice animation of how water vapor molecules change shape as they absorb energy.
http://en.wikipedia.org/wiki/Water_absorption – about halfway down the page.
[…] Part Two looked at why different gases absorb and radiate different proportions of energy – what the factors are that determine the relative importance of a “greenhouse” gas. Also why some gases like O2 and N2 absorb almost nothing in the longwave spectrum. […]
This is a very informative site, I can see myself returning many times. A question on watervapour: Humid air as found in coastal areas retains heat at night. Is this due to H2O absorbing longwave radiation or is it more due to the high specific heat of water?
[…] Part Two looked at why different gases absorb and radiate different proportions of energy – what the factors are that determine the relative importance of a “greenhouse” gas. Also why some gases like O2 and N2 absorb almost nothing in the longwave spectrum. […]
Sorry but I think you will find that all substances absorb long-wave radiation. A gas that absorbs almost nothing in the long wave spectrum is called ice.
To appreciate the sensitivity of a substance to long-wave radiation one need only observe that substances melting point from its solid state.
The melting points of Oxygen and Nitrogen are 54.36 K and 63.15 K respectively. Whereas the melting points of CO2 and water (the two main so called greenhouse gases) are 194.65 K and 273 K respectively.
“As an interesting comparison, molecule by molecule methane absorbs about 20x as much energy as CO2. But of course it is present in much smaller quantities.”
So if we compare the melting point of methane to the other gases in the atmosphere it gives us a cross reference from which we can get some perspective. Methane melts at 91 K. This would suggest that if methane is that much more powerful a greenhouse gas than CO2, then oxygen and nitrogen must be even more so.
These facts alone are enough to debunk the “greenhouse effect” and therefore AGW itself.
It is clear then that either all gases are “greenhouse gases” or if not, then none at all. Since there is no such thing as a substance which can actually trap heat, and the false implication that so called “greenhouse gases” emit heat back down to earth in violation of the second law of thermodynamics then I am of a mind that it is none at all. There are NO “greenhouse gases”.
Not only am I of a mind that this is the case but I have experimental evidence to back it up. If you are interested you can go to my website http://www.spinonthat.com/CO2.html and watch the video’s for yourself. These experiments are very basic and simple and can be reproduced by anyone. Even school children can test CO2 to see how it compares against ordinary air in the domain of radiant heat (infrared radiation).
The results speak for themselves. They tally well with the melting points and confirm the assertion that there are no “greenhouse gases”.
The claim that some gases absorb only certain frequencies of radiation is the basis and the origin of AGW fraud. Such claims are called “sophistry” and “climate science” is riddled with sophistry, fallacious argument with the intent to deceive.
Heat is heat and a substance such as a gas is either warmed or it is not. Any substance which is above 0 K will have absorbed infrared radiation. That includes every gas in the atmosphere, no exceptions. Had they not they would still be solid ice. Solid ice is the natural default state of all the atmospheric gases.
Let me give one clear example of sophistry:
“The layer itself will act as a blackbody and re-radiate infrared radiation. But it re-radiates in all directions, including back down to the earth’s surface. (If it only radiated up away from the earth there would be no “greenhouse” effect from this absorption).”
Here we have the classic AGW fraud layering trick, a favorite of Dr. Roy Spencer. The implication being that the atmosphere forms or rather settles into temperature and/or gas layers when in-fact, as the temperature of an area of air (atmospheric gases) increases, it rapidly expands and rises away from the heat source. “Convection”. The closest you can get to the above assertion in reality is that the higher up in the atmosphere you go the colder it gets. This is not quite the same thing as implying that the atmosphere settles into layers of relatively specific temperature.
To further make the point, try repeating the quote above while holding a lighted match above your hand and moving it closer and closer to your bare skin. You will find that you can hold it there at as little as 5 mm from your skin without any real discomfort. Now try and do the same from underneath your hand!
This burning match experiment is a refresher course for those who have fallen for line “CO2 traps heat and re-emits it back down to the ground”. It is a simple yet effective reminder of how powerful the upward movement of heat actually is in the atmosphere. The result of convection.
DaveB:
I’d expect that radiative heating would be last on the list. Probably latent heat in the water vapor is released when some of the water vapor condenses. This would happen because the air cools and so it can support a lower absolute humidity.
For Politicians cost lives – the automatic spam filter originally caught your comment.. now resurrected.
I could probably devote a whole post or two to your comment, but for now just a few points in response.
You said “..Sorry but I think you will find that all substances absorb long-wave radiation. A gas that absorbs almost nothing in the long wave spectrum is called ice..”
Ice is not a gas.
You said: “..So if we compare the melting point of methane to the other gases in the atmosphere it gives us a cross reference from which we can get some perspective. Methane melts at 91 K. This would suggest that if methane is that much more powerful a greenhouse gas than CO2, then oxygen and nitrogen must be even more so.
These facts alone are enough to debunk the “greenhouse effect” and therefore AGW itself..”
It might suggest it to you, but it’s not actually evidence.
To present it as evidence you would need to show that the melting point of a substances is linked to its absorption of longwave radiation (radiation > 4um).
What is probably easier is to miss out that step and simply present evidence that O2 and N2 absorb significant energy in bands from 4um to 30um. O2 absorbs a lot of energy in the UV range below about 300nm (breaking up into O+O).
We don’t know that from information about its melting point, but from taking O2 and radiating it with different wavelengths, then measuring what proportion of each wavelength comes out the other side. And we also know it from taking measurements through the atmosphere.
The same goes for all the other gases. This is extremely well-established science. You should also present in your research why it is that 1000s of scientists over a 100 years have all measured and replicated very similar absorption characteristics of all these gases.
Melting point data – without more evidence – is as relevant as the number of atoms in the molecule, or the molecular weight, or any other interesting property.
For Politicians cost lives:
2nd part.. on your comment Let me give one clear example of sophistry.. (Readers interested, see above for my “sophistry”)
We move one step at a time here on Science of Doom – it makes it easier to explain the subject.
You are correct that convection is more significant than radiation in the lower part of the atmosphere. You will find this noted at the end of part three in the section on the “Gray Model” of the atmosphere. And in the upcoming Part Five.
However, the fact that convection is more significant doesn’t mean that radiative transfer is “sophistry”. It just means that convection is “more significant” in most of the troposphere.
You will find this same point well explained in any basic book on atmospheric physics and most introductory science books on climate.
That doesn’t prove it of course, more on point is that all of these books explain quite well that convection “dominates” movement of heat in the lower part of the atmosphere and radiative transfer “dominates” higher up in the atmosphere.
If you don’t want to wait until Part Five of this series, have a read of the excellent paper Climate Modeling Through Radiative-Convective Models (1978), V. Ramanathan & J.A. Coakley, which you should be able to find and download by doing a google search. It’s a great review of the work done up to that point and the way forward, although it has lots of maths and assumes some prior knowledge. Alternatively – better but not free – I would recommend the excellent Elementary Climate Physics by Prof. F.W. Taylor (2005). You can buy it online for less than $50.
[…] Part Two discussed the factors that determine the relative importance of the various gases in the atmosphere. […]
Scienceofdoom, OT and totally out of left field, is there a greenhouse effect on the sun?
Probably, but I don’t know how much. All of the mechanisms work exactly the same. Radiation according to temperature, absorption by the various gases that make up the sun’s outer atmosphere. Many people will have analyzed the solar energy balance to death..
You may find that the physics of stellar atmospheres is controversial. There is a specially-designed satellite that just started orbiting the sun to test theories of how the corona of the sun is heated to millions of degrees while the surface radiates as if it were only 6000 degK. Since the interior temperature is also millions of degrees, there is step temperature gradient to explain with “convective-radiative equilibrium”.
I have already commented on this issue once but it was probably a wrong post in the tangle of many on this blog.
I think the right place for the comment is this post where you write :
What Happens when a Greenhouse Gas Absorbs Energy?
Once a gas molecule has absorbed radiation from the earth it has a lot more energy. But in the lower 100km of the atmosphere, the absorbed energy is transferred to kinetic energy by collisions between the absorbing molecules and others in the layer. Effectively, it heats up this layer of the atmosphere.
This is only half the story and because of seeing only the half story , the conclusion is wrong.
What you say is the following :
A* + B -> A + B_
In words – the vibrationnaly excited molecule A* interacts with a molecule B , its excited state decays to A and the molecule B increases its kinetic energy to B_ . This is right.
But what you forget is that by time symmetry we have also :
A + B_ -> A* + B
The molecule A interacts with a molecule B_ , its vibrationnal state excites to A* and the molecule B_ decreases its kinetic energy to B. This is btw the process that makes a CO2 laser work .
In LTE what is the basic hypothesis in all these posts we can even say more , namely that the rate of both processes is exactly equal and we have an equilibrium that can be written :
A* + B A + B_
Please note the difference to the initial A* + B -> A + B_ !
The conclusion that follows is that the number of A* (vibrationnaly excited molecules) is constant .
Also another way to say the same thing is that the distribution of the kinetic energy of the B molecules is constant too.
Still another independent way to find again the same result because they actually all use same concept of energy equipartition , is to note that the distribution of vibrationnal energy levels is constant for a given temperature (Maxwell Boltzmann law).
So it appears that whatever some IR absorbing molecule A* (CO2 , H20 , CH4 etc) does , there is at least one thing it doesn’t and it is to “heat the atmosphere”.
The key phrase in your post is “at equilibrium”. Obviously there is no heating or cooling at equilibrium by definition. But the atmosphere as a whole is never at equilibrium. The planet rotates. Its orbit isn’t perfectly circular. The axis of rotation is tilted with respect to the plane of the orbit. So the flow of energy is never constant and various parts of the atmosphere and the surface are always heating or cooling to some degree. When the surface is exposed to sunlight, it warms and radiates more intensely. A lot of that radiation is absorbed by the lower atmosphere and it warms too. So yes, CO2, H2O, CH4, etc. do heat the atmosphere and when the sun goes down they cool the atmosphere as well.
NB
The most important point in the post above , the symbol for equilibrium , disappeared in the infosphere .
Apparently using arrows is dangerous 🙂
It should read :
In LTE what is the basic hypothesis in all these posts we can even say more , namely that the rate of both processes is exactly equal and we have an equilibrium that can be written :
A* + B = A + B_
Please note the difference to the initial A* + B -> A + B_ !
scienceofdoom,
O2 has a permanent magnetic dipole so it does have a rotational spectrum. In fact, the microwave sounding units in the satellites used to determine the atmospheric temperature profile are tuned to different parts of the 60 GHz O2 band.
You’re also leaving out the water vapor continuum absorption which is very important at longer wavelengths. The H2O optical thickness graph looks like it may include continuum absorption. O2 and N2 also have collisionally induced continuum absorption spectra, but they can be neglected except for long paths through the limb of the upper atmosphere where water vapor is low enough to not overwhelm them.
[…] Some molecules, like nitrogen and oxygen, have no ability to absorb or emit longwave radiation (see CO2 – Part Two), but by collision with molecules like CO2 and water vapor they will share energy and be at the […]
Regarding references for the average water vapour in the atmosphere, Fundamentals of Weather and Climate by Robin McIlveen published by Chapman & Hall, 1992, pg 62, section 3.7 gives a slightly smaller figure:
“For example, the overall specific humidity (the specific mass of water vapour) of the atmosphere is only about 0.3%, whereas the specific humidity of the warmer parts of the troposphere often exceeds 3%.”
Regrettably, this book seems now to be out of print.
Though the Wikipedia article says it’s talking about water vapour I wonder if it’s actually giving the total water content of the atmosphere, including water condensed in clouds making up the 0.1% discrepancy.
DeWitt Payne
The key word is indeed equilibrium.
But the equilibrium I have been talking about is in the reaction :
A* + B = A + B_ with definitions of notation in the post above.
This is a cinematic microscopical equilibrium at the base of LTE (local thermodynamical equilibrium) which has nothing to do with a global thermal equilibrium of the whole Earth that you mention.
Of course the latter doesn’t exist what invalidates btw all “energy budgets” because they suppose global equilibriums. But that is off topic here.
At the microscopical molecular level (energy exchange between vibrational and translational degrees of freedom) , the equilibrium of A* + B = A + B_ is absolutely necessary!
So no , CO2 , CH4 etc do not heat the atmosphere in any reasonable meaning of heat .
They do not cool it either .
They do both with exactly the same rates .
This is basic physics and can be easily demonstrated in a dozen of ways.
Here is one.
Let’s suppose that the reaction A* + B -> A + B_ is not an equilibrium but that it goes only from left to right like the author of the article and DeWitt Payne say.
Excited CO2 (A*) per hypothesis “heats” the atmosphere (B) .
Let’s hope that we agree at least about the fact that initially A and B have the same temperature (energy average).
What will happen in the mixture ?
The concentration of B_ will increase forever .
This means that the temperature of B (N2 and O2) increases forever.
This is trivially absurd for at least 2 reasons .
1) It violates the time symmetry which is mandatory for microscopical processes (e.g the reaction above happens necessarily in both directions).
2) The temperature of CO2 diverges from the temperatures of N2&O2 what is forbidden by the LTE if not by the 2. principle.
I have read somewhere on the net that this site was serious and showing good physics.
Unfortunately even here one finds absurdities where CO2 “heats” atmosphere and temperatures of components of a gaz mixture spontaneously diverge , one of them even going to infinity …
Philippe,
I realize this is years out of date, but I didn’t see your reply at the time.
Where exactly do I or SoD imply that microscopic reversibility doesn’t exist? Answer, we don’t. But microscopic reversibility is completely compatible with non-equilibrium thermodynamics of systems containing a large number of molecules. Otherwise entropy couldn’t increase and there would be no arrow of time.
Local Thermodynamic Equilibrium does not mean that the system is in equilibrium and unchanging. It just means that molecular energies are distributed according to the Boltzmann distribution. This is perfectly compatible with both microscopic reversibility ( in fact, deriving the Boltzmann distribution in statistical thermodynamics requires microscopic reversibility) and the ability of a system of a large number of molecules gaining or losing net energy, i.e. heating or cooling.
Complementary remark :
The dynamical collisional equilibrium proven above has for corollary something that very few people get too .
CO2 emits infrared radiation not only of the same intensity but also of a very similar frequency as the one it absorbs .
So much for the utterly wrong but unfortunately widely disseminated picture in which CO2 absorbs some frequencies , emits nothing in those frequencies and collisionaly “heats” N2 and O2 instead …
“As an interesting comparison, molecule by molecule methane absorbs about 20x as much energy as CO2. But of course it is present in much smaller quantities.”
I don’t think this statement is true.
[IMG]https://scienceofdoom.files.wordpress.com/2009/11/absorption-spectra-co2-h2o-[/IMG]
Figure 5.2 from part 1 indicates that a molecule of CO2 absorbs much more energy than one of CH4. Area under the curve.
A molecule of methane is 20X more effective than CO2 as a greenhouse gas because there is so much less of it in the environement. It would take very little methane to double the atmospheric concentration but it would take a tremendous amount of CO2 to do the same.
Therefore, a molecule of methane is more significant because there is so much less of it.
Bob
[…] CO2, methane, NO2, etc. This is after taking into account their much higher concentration. See CO2 – An Insignificant Trace Gas? Part Two Possibly related posts: (automatically generated)The Sun and Max Planck AgreeHow do we know CO2 is […]
[…] water vapor than CO2 in the atmosphere, which is why it is the most important greenhouse gas. source So your only logical choice is either H2O and CO2 or neither H20 nor CO2. __________________ 1. […]
Hello. First time poster. Question: if water in the atmosphere is @ 20,000 ppm and CO2 is @ 400 ppm, that would make moisture 50x greater than CO2, but yet you say 10X greater, so what am I missing here? Thank you.
Chad,
You are saying that the water vapor content is 2%, while I said it was 0.4%, so this is the difference here.
Now water vapor concentration varies in time and space very significantly. See, for example, Clouds and Water Vapor – Part Two. The mean value of water vapor across the whole atmosphere on an annually averaged basis isn’t really very useful. So calculations of the actual effect of water vapor vs CO2 have to be done for a particular atmospheric profile (temperature and water vapor concentration vs altitude).
The book Wallace and Hobbs: Atmospheric Science tells that water vapor accounts for roughly 0.25% of the total mass of the atmosphere. Taking into account the molecular masses that corresponds to 29/18*0.25% = 0.40% in full agreement the Wiki value.
There is a little more about the non-linear behavior of radiation with water vapor concentration in Clouds and Water Vapor – Part Three.
I plan to write some about this shortly, perhaps starting with Pierrhumbert’s paper from 1995, Thermostats, radiator fins and the local runaway greenhouse.
SoD,
On the whole, not bad, but I take issue with this statement;
“The layer itself will act as a blackbody and re-radiate infrared radiation.”
As you have pointed out elsewhere on this site, molecules that do not absorb a frequency, do not emit that frequency, either. The reason being that they simply are not coupled to the electromagnetic field at that frequency, because of the symmetry considerations you mention. So, a layer of the atmosphere will only radiate at those frequencies which its constituent molecules can absorb. It will not “act as a blackbody”.
Jerome,
You are correct, that statement in the article is plain wrong and I can’t believe I didn’t notice it at the time.
Thanks for pointing it out.
SoD,
I don’t see that it affects the argument much, but I am sensitive on this topic, having recently been accused of insanity and worse for stating that the addition of CO2 to pure nitrogen could cause the mixture to cool faster. People seem to have the idea that everything radiates “thermally”. I suppose this is because everything does, except gases. Macroscopic objects have vibrational states so dense as to be effectively continuous.
Jerome
Jerome Berryhill,
Technically it depends on your definition of “thermally”. Gases aren’t black or gray bodies, but the intensity of the emitted radiation at the allowed wavelengths is determined by the gas temperature because the number of molecules in the excited state for those transitions is determined by the Boltzmann distribution, which is temperature dependent.
scienceofdoom,
could you please mark the statement “The layer itself will act as a blackbody and re-radiate infrared radiation.” as an error. Your site is the best source of information on green house basics, but I was sent on the wrong track by the “blackbody” when I was trying to find out why the radiation does not escape through the “open window”.
As a minimal change I would propose replacing “blackbody” by “warm body” in that sentence.
“As a minimal change I would propose replacing “blackbody” by “warm body” in that sentence.”
Well, that would certainly be minimal, but I don’t see that it would improve the accuracy.
The point is that black bodies, by definition, radiate energy over a wide range of frequencies, with a maximal emission at a frequency determined by frequency. Thus, a black body’s radiation maximum shifts to higher energy as its temperature increases.
A gaseous molecule like CO2 is incapable of radiating at multiple frequencies, in the temperature range of interest. It can only radiate in “lines”. And the N2 cannot radiate at all. Therefore, a change in temperature will increase the intensity of the line, but does not shift its peak. I do not see that “warm body” describes this behavior any better than “black body” does.
The reason this is important is that it means that the ability of the atmosphere to radiate is effectively “cut off” rather sharply below a certain temperature, rather than tapering off as a black body would.
Oops. That should have been;
“The point is that black bodies, by definition, radiate energy over a wide range of frequencies, with a maximal emission at a frequency determined by TEMPERATURE. “
Jerome,
It’s true that emission of gas is influenced strongly by spectral properties. I did consider proposing that something on that would be added at the same time, but concluded that this is perhaps not the right place for that.
The temperature dependence of the emitting power of a gas depends on temperature in a different way from that of a black body. In case of CO2 the temperature dependence is given quite accurately by the Planck formula, because one broad line dominates the emission. The energy of the 15 µm emission line happens, however, to be such that the temperature dependence is very close to that of a blackbody over the relevant range of temperatures. There’s no sharp cutoff until the temperature gets very low.
This similarity is true in spite of the totally different formula. In the Planck formula the inverse temperature occurs in an exponential function, while the Stefan-Boltzmann law contains the fourth power of the temperature.
Water vapor absorbs and emits over so large part of the spectrum that it behaves more like a blackbody.
I think the important point is that pure N2 will neither absorb nor radiate electromagnetic radiation. When mixed with CO2, the CO2 can absorb incident radiation and pass the energy to the N2 by collision, a fact which has been very widely remarked upon. What is less widely remarked upon is that the same process works in reverse. Pure N2 will not cool off by radiation. However, an admixture of CO2 allows it to do so. Therefore, CO2 can heat the gas if the incident electromagnetic field is at a higher temperature, or cool it if the electromagnetic field is cooler. In practice, it is seldom the case that the electromagnetic field at a point has a well-defined temperature. Rather, it tends to have very different temperatures in different directions.
Jerome,
Your comment is basically correct. I add a few points.
The electromagnetic radiation does not have a temperature in the same sense a matter has. Temperature is a property of a system in local thermal equilibrium, and the word “local” means everything that can directly interact at a significant level with the subsystem that’s being observed. I used the expression electromagnetic radiation rather than electromagnetic field because the radiation in atmosphere is incoherent, i.e. each photon can be considered separately. Much of the theory of electromagnetic fields describes coherent fields ans using those parts of the theory is misleading.
When we are looking at a gas, the dominant form of interaction is that through collisions with neighboring molecules. Radiative interaction is extremely weak in comparison (something like one in billion). There “local” means within a few mean free paths of molecules or only a fraction of mm in the troposphere and not much more even in the higher stratosphere.
Mean free path of IR is always larger, but often it’s so small that the air temperature does not change much within one mean free path. Then the intensity of radiation is the same as that from a black body of the same temperature. That may be true for some wavelengths like those near 15 µm while the mean free path at other wavelengths is much larger. The intensity of radiation is determined by weighted average of temperatures within a few mean free paths of the wavelength being considered. At the wavelengths important for energy balance the mean free path is relatively small. Therefore the intensity at these wavelengths is also close to the blackbody intensity at the local temperature.
In troposphere the temperature profile is controlled by convection. That leads to the situation where the intensity of radiation is just slightly lower than the blackbody intensity when the average of downwards and upwards radiation is considered. Due to the non-linearities of the equations that leads to emission very slightly stronger than absorption. The net effect of absorption and emission is thus slightly cooling. The balancing energy comes from convection.
In stratosphere the basic situation is that radiative heat transfer is the only important one. In the IR region CO2 is the most important gas in that. In upper stratosphere heating by UV absorbed by ozone is also important. Both absorption of IR by CO2 and absorption of UV are important, but only emission by CO2 is strong on the other side of the balance. Therefore upper stratosphere is warmed up to the point where emission of IR by CO2 is strong enough to balance both sources of heat. In lower stratosphere the role of UV is smaller and the temperature is determined mostly by IR alone. That leads to a temperature that’s almost independent of altitude.
Your ideas regarding temperature and radiation contain too many errors for me to correct. I will simply caution others not to pay any attention to them.
When you state that;
“In troposphere the temperature profile is controlled by convection.” you are stating as fact what the models discussed on this web site are intended to show is plausible. That is, you assume your conclusion. No one can possibly know, as a matter of experimental fact what “controls” the temperature of the troposphere.
Jerome,
Are you sure, that the errors are in my text? What makes you so sure of your own understanding of physics?
My reference to convection is not a reference to model, but to physical processes. The main purpose for stating that was to tell the radiation is not the only important mechanism that transfers energy in the troposphere as opposed to stratosphere where that’s largely true. This is the fundamental distinction between troposphere and stratosphere. They are not alike for this specific reason.
My physical knowledge is not at issue, yours is. Your distinction between electromagnetic radiation and the electromagnetic field indicates to me that you are unfamiliar with the standard thermodynamic treatment of electromagnetism, which involves consideration of the possible modes of excitation of the electromagnetic field in a region of space. Whether a temperature can be attributed to a “system” comprised of such modes is entirely a question of what you are willing to call a “system”. But that nicety has no bearing on questions of energy transfer by radiation.
Jerome,
Your knowledge is in question when you use it to judge my understanding.
I can assure you that at least the quantity of my knowledge is sufficient as that can be measured objectively, while the quality not so easily.
I didn’t plan to go deeper in these issue, because they are peripheral to the main subject of this thread. I add, however, a few comments. They may go too far into the theory, but I’m not aware of any simpler way of explaining this physics.
Theory of electromagnetic radiation builds on Maxwell’s Equations They are equations for electric and magnetic field. They alone can, however, not explain interaction of electromagnetic radiation with matter. To get an explanation consistent with observations we need also Quantum Mechanics. A consistent combination of Maxwell’s Equations and QM is provided by Quantum Electrodynamics (QED) developed by Feynman and others.
Doing calculations with QED is normally based on Feynman diagrams. The interactions that are important for the IR radiation in the atmosphere are described by the simplest Feynman diagram that corresponds to emission or absorption of a photon. All diagrams that involve more than one photon contribute so little that they can be disregarded in practical calculations. This is an indication of the fact that each emission and each absorption of a photon is an independent event, not influenced significantly by other electromagnetic radiation. This situation is that, what I mean when I say that the photons represent incoherent radiation. (The situation is different in a laser, where coherence is the goal and reached under very special conditions.)
When the radiation is incoherent, the phase of electromagnetic field of one photon is unrelated with the phase of the field of another photon. When the phases are not correlated adding up the fields is not an useful approach. What happens is that we get the correct result much more easily counting photons than adding up fields and performing all the complicated calculations. This is not a poor approximation, but a highly accurate true result of the theory.
While it is true that QM is needed to fully understand the interaction of light with matter, it is not necessary to use QED to understand those aspects of that interaction which are relevant to atmospheric thermodynamics. All the models on this website use the “semiclassical” approach, in which the molecule is described using QM, but the EMF is treated classically. For example, the dipole moment of a molecule is a QM entity, but the radiation field it interacts with is a classical field. One does not need to assume that the field is coherent in order to calculate properties of interest, such as the entropy as a function of energy, which in turn gives the temperature.
Certain aspects of optical thermodynamics, such as the derivation of the black-body spectrum, and the description of lasers, require some concepts from Quantum Optics, the quantum theory of EMF. But QO is not QED. QED is needed to explain some very precise observations, but I do not recall ever encountering QED in a Thermodynamics or Statistical Mechanics text. If you really are “not aware of any simpler way of explaining this physics”, I would recommend you pick up any statistical mechanics text and check the index for “blackbody”. For a really fascinating account of the development of these ideas, I recommend Thomas Kuhn’s “Black-Body Theory and The Quantum Discontinuity, 1894-1912. Note that the period mentioned in the title predates the origin of QED by several decades.
Jerome,
I agree that QED is not needed for the actual calculation of the interaction of a molecule with electromagnetic field, but it’s needed to verify from the theoretical point of view that the calculation is accurate enough and that multiphoton processes are of no significance for practical results.
The question of coherence in the sense I brought it up does not enter the calculation when that’s done for one individual transition at the time. In such a calculation we consider by construction only one photon at the time, as one transition refers to the emission or absorption of single photons.
What I was discussing was the next step, where we are considering a very large number of molecules and a large number of photons. For every single separately the electromagnetic field can be considered, but adding these fields up to describe the field of many photons is highly impractical. The incoherence is required to conclude that this impractical calculation is also unnecessary.
Jerome,
However you approach the calculation, you have not shown convincingly that it will have a different outcome. Lack of coherence is important because it means we can neglect stimulated emission and only have to deal with spontaneous emission.
A group of molecules large enough to be at local thermodynamic equilibrium, i.e. have a Boltzmann kinetic energy distribution described by a single temperature, cannot radiate at any wavelength more energy than defined by the Planck equation for that wavelength and temperature. That is precisely the same as saying that emission cannot be greater than for a black body at that wavelength and temperature. You’re nitpicking by insisting that a black body has unit emissivity at all wavelengths while a molecular gas doesn’t.
The point is that adding CO2 to pure N2 makes it possible for the mixture to cool radiatively, which pure N2 cannot do. If you dispute either of those points, say so. Otherwise, spare me the persiflage.
Jerome,
Spare me your attitude. You’re not the smartest guy in the room.
No one here is likely to dispute that adding CO2 to N2 makes the mixture able to absorb and emit EM radiation, especially near a wavelength of 15 μm. But that emission is controlled by the absorptivity, which is a function of the molecular absorptivity at a given wavelength, the number density of CO2 molecules and the path length, and the Planck equation for that wavelength and the temperature of gas. This is exactly the same as for a solid except that the solid has many more surface states than a molecular gas so the emissivity is nearly constant with wavelength. A black body is an idealized solid with unit emissivity at all wavelengths. So in practice, nothing emits like a black body. A hohlraum is a good approximation however.
The rest of your comments amount to “mine’s bigger than yours.”
“No one here is likely to dispute that adding CO2 to N2 makes the mixture able to absorb and emit EM radiation…”
So what exactly *are* you disputing? Who’s the smartest guy in the room? Whose is biggest? I made a simple — and correct — observation, and was answered with paragraphs of irrelevant pseudoscience from Dr. Umlaut. QED my rosy red one. Now you want to weigh in with hohlraums. Persiflage.
Jerome: Pekka, DeWitt and our host would probably agree that adding CO2 to N2 can cause the gas to cool or warm depending on its current temperature and the intensity and wavelength of the radiation passing through it. CO2 certainly does not “trap” heat. I prefer to focus on the Schwarzschild equation, which is generally accepted by most commenters here:
dI/ds = n*o*B(T) – n*o*I_0 (emission – absorption)
The incremental change in intensity of radiation at a particular wavelength (dI) as it passes an incremental distance (ds) in a particular direction through a gas or other medium depends on the density of the gas molecules (n), their absorption/emission coefficient at that wavelength (o), the intensity of the incident radiation at that wavelength (I_0) and the Planck function for that wavelength and temperature (B(T)). Conservation of energy demands that the gas cool when dI/ds is >0, and warm when <0. Of course, one needs to integrate over all of the relevant wavelengths and radiation directions. (Energy transfer through the atmosphere can sometimes be simplified to just the upward and downward fluxes.)
The equation says that the intensity of radiation at a particular wavelength passing through a homogenous gas approaches "blackbody intensity" at a rate that depends on the density of the gas and its absorption coefficient. This may heat or cool the gas. Since much of the LWR (the wavelengths absorbed and emitted by GHGs) passing through the troposphere comes from the warmer surface or the troposphere itself, increasing GHGs tend to warm the troposphere. In the warmest altitudes of the stratosphere, however, most of the radiation absorbed by CO2 (15 um) is emitted by nearby cooler altitudes, and more CO2 is predicted to lead to cooling. (Cooling is also caused by loss of ozone.)
The emission term is often negligible in laboratory instruments with light sources that are several thousand degC, simplifying the Schwarzschild equation to Beer's Law for absorption. This equation creates the illusion that CO2 "traps" heat and that it can't cause cooling.
First comment here.
Up to this day I believed that green house gases heated continuously the atmosphere by collisions with non active gases like N2 because I didn’t think much about this issue.
I understand the word “heats” like “causes an elevation of temperature”and
I believed that because it was written everywhere.
Then I found this site and in the comments above Philippe convincingly proves that green house gases do both – cool and heat the atmosphere by collisions.
Indeed it seems to me now after reflexion that if it was not the case, the temperature would increase to infinity.
But if the collisions between CO2 and N2 both cool ands heat with the same rate, then it is equally right to say that the green house gases cool the atmosphere by collisions or heat the atmosphere by collisions.
Yet in the article it is written that they (only) heat it.
So what is it ?
Manfred,
You are right. There’s a lot of imprecise writing on this point.
In most cases the direct influence of GHG’s is to locally cool the atmosphere rather than heat it. GHG’s warm the surface and convection transfers that heat to the atmosphere.
It may sound paradoxical that the local direct influence of GHG’s is cooling, but their overall effect is warming, but there’s nothing contradictory in that.
The energy balance of troposphere is typically such that heat is brought from surface to the atmosphere by both radiation and convection (and also through evaporation and condensation, i.e. latent heat transfer). The GHG’s of the troposphere absorb IR from all directions and emit to all directions. The balance is that they emit more than absorb as the combined effect of two factors:
1) Far above we have the space and almost no radiation comes from there (except the solar radiation that’s mainly SW). The net IR with space is emission from atmosphere.
2) Convection adds energy to troposphere keeping it so warm that it emits more than it absorbs (otherwise it would keep on warming rapidly)
The surface is warmed by the GHG’s because they add to the radiative heating of it and because they reduce the amount of radiation that ultimately leaves the atmosphere to the space. GHG’s add to the radiation from any specific volume of gas but they affect more the radiation that passes trough the volume when we are considering the upper troposphere where the temperature falls with increasing temperature.
There are so many things involved that the whole may first appear confusing but with more thinking they start to form a coherent picture.
Manfred,
The equation of radiative transfer (which is well-proven physics for over 60 years) can be found in Understanding Atmospheric Radiation and the “Greenhouse” Effect – Part Six – The Equations.
Armed with these you can determine whether a body of gas heats or cools by radiation.
For equation-literate people it is clear that the change in intensity of radiation through a body of gas depends on the source temperature of the radiation minus the gas temperature. This is the Schwarzschild equation.
And if the intensity reduces as the radiation travels through the body of gas it means net energy is absorbed in the body of gas from the source radiation. And if the intensity increases it means net energy is emitted from the body of gas.
So in effect when we consider atmospheric radiation from the top of the atmosphere towards the surface we find that the atmosphere is cooling by this effect (colder source sending radiation through progressively warmer gas)
And when we consider atmospheric radiation from the surface to the top of atmosphere we find that the atmosphere is heating by this effect (warmer source sending radiation through progressively cooler gas).
All of this is explained in detail in the series starting with Visualizing Atmospheric Radiation – Part One.
And so the net effect is a combination of multiple factors.
When we then consider the difference between a “before” and “after” case we have yet more calculations to perform.
The answer is not predetermined, it depends on the boundary conditions.
In the case of the climate system the boundary conditions are the warmer surface (due to solar radiation traveling through a mostly transparent atmosphere) and the colder tropopause (caused by the lapse rate).
For more on the cause of the temperature profile in the atmosphere (the lapse rate), see:
Temperature Profile in the Atmosphere – The Lapse Rate
Potential Temperature
Density, Stability and Motion in Fluids
SoD and Manfred,
The Schwarzschild equation is fine for radiation of given precise direction, like directly upwards, or exactly horizontal and northwards, but concluding from that, what’s the local energy balance at a specific point in the troposphere is too complicated in practice. Thus it doesn’t solve the problem.
We know, however, that the troposphere is, essentially by definition, that part of the atmosphere, where the local balance for sensible heat is the sum:
change in sensible heat = absorbed radiation – emitted radiation + heat transferred by convection + heat released from condensation
The normal signs of all terms are given in the expression, i.e. the only heat loss is in normal situations the emitted radiation. As the sum must be zero in stationary case we know that the emitted radiation is always larger than the absorbed radiation.
Another way of understanding the sign is that the temperature would decrease faster with the altitude, if no convection would occur. The higher lapse rate would lead to balance based on radiation only. A smaller lapse rate than that means that the troposphere is warmer and radiates more. that again means that the net balance of radiation is cooling.
In stratosphere we have essentially only radiative heat transfer. Thus the balance is given by that alone. The solar radiation is there an important part of the radiative balance.
Something strange happened with WordPress. A message about maintenance was shown, when I posted my above message. The message is now visible, but no link to it was created in Recent comments.
Manfred Gudern,
As I pointed out in my extremely delayed response to Phillipe above, microscopic reversibility says little about the macroscopic behavior of a system. If the local temperature isn’t changing then there is obviously no net change in energy. But as Pekka points out, heating can be from convective energy transfer to the surface in the troposphere so the effect of greenhouse gases is local cooling to match the energy coming in. In the stratosphere, the heating is from UV absorption of incoming solar radiation by oxygen and ozone. Increasing carbon dioxide, which doesn’t absorb significantly in the UV compared to its emission in the thermal IR, causes the temperature to be lower than it would have been at lower CO2. I believe that qualifies as cooling.
Hello
Thanks but the many answers didn’t answer the question I asked.
I didn’t ask about UV, stratosphere, convection, lapse rate or Schwarzschild equation.
I will try to reformulate – it is a very simple question which can be answered by yes or no. It is only related to collisional energy transfer.
Thesis : Vibrationally excited CO2 transfers energy to N2 by collisions. This heats N2. This is what I read sofar in most texts about green house gases.
Antithesis : Vibrationally excited CO2 transfers energy to N2 by collisions and N2 excites vibrational degrees of CO2 by collisions too. The rates of both processes are equal. This does not heat N2. This is what Philip wrote.
I assume that the point concerns the troposphere where the gaz density is sufficiently high and we have LTE.
So which of Thesis or Antithesis is true (only one can be true)?
P.S
You probably assumed that I was scientifically illiterate. To situate my level of understanding – I know QM, radiative transfer and thermodynamics. My training is higher mathematics. I have little knowledge about collisional processes (e.g colisionally induced dipolar momentum leading to IR absorption etc).
Manfred,
At equilibrium, within a body of gas, the population of vibrationally excited states of CO2 is Boltzmann distributed. So, the net flow of energy between N2 and CO2 is zero. If the body is receiving radiation from a hotter source, the excited CO2 population will be increased above the equilibrium level, and as a result, energy will be transferred to N2. If there is no such hotter source, the tendency of excited CO2 to radiate will cool the gas. And if there are both a hotter source and a cooler sink, as is generally the case in the atmosphere, then go figure.
Manfred,
What happens locally between molecular vibrational states ant (translational) kinetic energy of the molecules is one step, and what happens for the radiation over wider distances is another. I answered to the second, as I didn’t understand that you were asking about the first.
As Jerome answered, the occupation levels are given by the Boltzmann distribution, i.e. they are proportional to exp(-E/kT), where E is the energy of the level, and k Boltzmann’s constant.
There are only few important vibrational states for each molecule, for CO2 only one really important and another of some significance. There are two modes for the most important excitation as there are two independent transverse directions. The occupation level of each of these is around 3.5 % at typical temperatures. Thus about 7 % of CO2 is always in such an excited state. Without any collisions the lifetime would be close to one second, but the states are formed and destroyed by collisions roughly once every nanosecond. These frequent collisions maintain continuously a very close balance between the local temperature and the occupation level of the CO2 molecules.
Looking at the spectrum, we see a lot of structure near the energy level of that excited state. That structure is due to rotational states of the molecule.
Pekka,
It’s my understanding that most molecular collisions are elastic in a gas at ~1 atm and ~300K and hence don’t transfer energy between molecules. As a result, the lifetime of an excited CO2 molecule is thought to be between 1 and 10 microseconds, not nanoseconds, not that it matters all that much. It’s still many orders of magnitude shorter than the expected lifetime for spontaneous emission for a free excited molecule as required for LTE to apply. I don’t have the reference for that and it might be out of date, though.
DeWitt,
The line width due to pressure broadening tells that the lifetime of excitations is something like 1 ns at the full atmospheric pressure.
The air broadened half-width of the CO2 lines around 15 µm or 667 1/cm are about 0.08 1/cm according to the Hitran database. The lifetime of the state is equal to the inverse of the full width at half maximum of the related peak in angular velocity. Multiplying 0.08 1/cm by the speed of light and taking the inverse we get 0.4 ns. Some factors of two and pi are still missing and should bring that even further down, but that sets the scale. The time between collisions of CO2 molecules with other molecules is very roughly 0.1 ns.
From all molecular collisions in air, most are certainly elastic, but that seems not be the case for an excited CO2 molecule. That’s not so surprising thinking elementarily, what a CO2 molecule is like. Hitting such a molecule in excited state may, indeed, lead to de-excitation with high probability, The opposite transitions are less likely but only by a factor of about 15.
Some further comments on the probability of excitation and deexcitation of a vibrational state in a collision.
While one must be very careful in using classical analogs in drawing conclusions about quantum mechanical systems, analogs are certainly useful for understanding when applied properly.
CO2 molecule is linear with C in the middle and O’s at both ends. In a classical analog they are connected together with springs that both bend and stretch. If the string is very stiff, hitting one of the molecules influences mainly the linear motion of the molecule as a whole and it’s rotation, only a small part of the energy transfer between the CO2 molecule and the hitting molecule influences the vibrational energy of the molecule. The molecule reacts to the hit essentially as one hard body.
If the spring is soft the immediate effect of the collision is to affect only that atom that’s hit, the rest of the molecule is affected more slowly through the soft spring. Thus hitting an atom of a “molecule” held together by soft springs is certain to affect very much the vibrational state.
Coming back to QM with discrete vibrational states, the extent of the change in vibrational state has to be transferred to a probability of transition. A stiff molecule is unlikely to change is vibrational state, while a soft one is almost certain to do that, when the hit is localized to one atom or affects the atoms differently.
How can we judge, whether CO2 is stiff or soft?
The answer is given by the lowest vibrational energy level and it’s relationship with the typical thermal energy of a degree of freedom, i.e. kT. At 296K that energy is about 30% of the energy of the vibrational state. That is a high enough number to indicate that the “spring” is not stiff and a transition is likely in a collision with one of the atoms in the molecule. The molecular collisions are typically localized enough to affect individual atoms.
For a transition to an upper energy state we need in addition enough collisional energy. Therefore an excitation is less likely than a deexcitation, and therefore the occupation levels of excited states are lower that the ground state by the factor exp(-E/kT), which is at typical temperatures 3-4% for the CO2 vibrational state that corresponds to the 15 µm IR radiation (there are two such states, each with that probability).
A molecule that doesn’t have any vibrational states at low enough energies is harder and less likely to change it’s vibrational state in a collision. That’s true to N2 and O2, and therefore for the air as whole.
Manfred,
I looked back through the earlier comments and DeWitt Payne’s comment of April 4, 2013 at 2:24 am seems the relevant one.
Which gas heats the other depends on the starting point and the boundary conditions.
At the point where a steady state condition is reached clearly they are exchanging equal energy via collisions. At this point neither is heating the other.
1. Let’s take the condition in a local body of gas at Ta=270K where net energy exchange is zero. The ground below and the atmosphere around drop in temperature very rapidly to much lower than Ta.
Now CO2 will be emitting much radiation and absorbing little. CO2 will be cooling the atmosphere. If there were no collisions between N2 and CO2 then CO2 temperature would drop rapidly. Instead, as CO2 cools via emission of thermal radiation, it is heated by the collisions with N2.
That is, N2 heats CO2 because the net transfer between the two is from N2 to CO2. In fact, it is reducing the cooling rate of CO2. Whichever way you want to write it.
dT(CO2)/dt is negative and reduced in magnitude by the effect of N2.
That’s one boundary condition. Are you going to say this doesn’t happen?
2. Now we can take the alternative case where the ground below and the atmosphere around the body increase in temperature very rapidly to much higher than Ta.
Now CO2 will be absorbing much radiation and emitting less. CO2 will be heating the atmosphere. If there were no collisions between N2 and CO2 then CO2 temperature would increase rapidly. Instead, as CO2 heats via absorption of thermal radiation, it is cooled by the collisions with N2 – and the corollary, it heats N2.
That is, CO2 heats N2 because the net transfer between the two is from CO2 to N2.
dT(CO2)/dt is positive and reduced in magnitude by the effect of N2.
This is the other boundary condition. Are you going to say this doesn’t happen?
So your request:
Is not yes or no, the answer is: “it depends on the boundary conditions”.
So you didn’t ask about the Schwarzschild equation but I brought it up because it is relevant.
SoD and Manfred,
We are considering here three systems,
1) The atmospheric gas that maintains a local thermal equilibrium through molecular collisions. No distinction can be made between different molecules in that.
2) The vibrational excitations of CO2 molecules and other GH gases.
3) Electromagnetic radiation.
The vibrational excitations have a linking role as they interact both with the translational modes of the gas and with the electromagnetic radiation. They interact about billion times more strongly with the translational modes than with the radiation, and are thus essentially a part of the local thermal equilibrium. The tiny share of the interaction with the radiation is, however, very important, because the IR radiation and the atmospheric matter would otherwise be uncoupled.
Because the vibrational modes interact so strongly with translational modes, the temperature that corresponds to their occupation levels is extremely close to the local temperature of the translational modes.
Electromagnetic radiation does not interact equally strongly with matter. Therefore the equivalent temperature that corresponds to it’s intensity may differ more from the local temperature and be influenced by temperatures far from the point considered. The equivalent temperature of radiation of a specified wavelength is defined as the temperature of a cavity where overall intensity at that wavelength is equal, when radiation from all directions is included in the comparison. That equivalent temperature is wavelength dependent.
If the equivalent temperature of radiation is higher than the local temperature of gas, radiation heats the gas, in the opposite case it cools the gas. These effects require that the radiation interacts with a component of the gas at that wavelength.
In troposphere the equivalent temperature of IR radiation is normally lower than the local temperature. Therefore the radiation cools the the gas.
The situation is similar to other cases where two reservoirs of differing temperatures are put in contact. Heat flows from the warmer to the cooler. The role of the GHG’s is to create this contact.
At the point where a steady state condition is reached clearly they are exchanging equal energy via collisions. At this point neither is heating the other.
Thanks. That is the answer on my question.
Of course I was not interested in violently out of equilibrium systems (like a CO2 laser) or by short transitories induced by a Heavyside or Dirac like change in boundary conditions.
For all practical purposes the Earth’s atmosphere being in a steady state, that means that CO2 indeed does not “heat the atmosphere” what is something that the common sense suggests too.
But I am often wary of common sense when QM processes are concerned and that’s why I preferred to ask.
This means that statements and their variations like :
Once a gas molecule has absorbed radiation from the earth it has a lot more energy. But in the lower 100km of the atmosphere, the absorbed energy is transferred to kinetic energy by collisions between the absorbing molecules and others in the layer. Effectively, it heats up this layer of the atmosphere.
that abound on the Net and which were the reason of my question because they contradicted the common sense, are indeed wrong.
They would only be right (with both possible signs, e.g “it cools” or “it heats”) in relatively exotical circumstances of strongly non steady states.
Manfred Gudern,
I think you’re still missing the point. The atmosphere can be considered to be at steady state for time periods on the order of a few seconds, many orders of magnitude longer than collisional equilibration time, so for the purposes of calculation of atmospheric radiative transfer, the assumption of local thermodynamic equilibrium is correct. For longer periods, the atmosphere is not and never has been at steady state. It heats and cools over daily, seasonal, and longer cycles. Some of that heating and cooling is by convection to and from the Earth’s surface, but a lot of it is by radiation to or absorption from the Earth’s surface and to space or from the sun. In that sense, gases that absorb and emit in the thermal IR region, >5nm, and absorb solar radiation like oxygen and ozone can be said to heat and cool the atmosphere.
For example, do you think the temperature of the stratosphere doesn’t change when the sun goes below the horizon? If it does, then it’s not in steady state over a 24 hour period.
Manfred,
While I believe you are correct that the idea of CO2 heating the atmosphere to a significant degree is dubious, this is not the hole in the argument. Think of a gas in, say, a thermos bottle. The gas and the interior of the bottle will quickly come to thermal equilibrium. Any radiation emitted by the gas will be reflected back in, and no radiation will enter from outside. Under those conditions, the gas will be in thermal equilibrium with the radiation field in the region of space it occupies, and there will be no net flow of energy between them. However, if the gas is exposed to a radiation field created by something at a different temperature, it can gain or lose energy as a result.
Please can you provide me with an absorption spectrum for diatomic nitrogen gas. Without this piece of information your whole rant is incredible, literally.
Thanks for your kind words.
Here is the absorption spectrum for CO2 (black) vs N2 (red):
And here it is scaled by abundance in the atmosphere:
You could have looked it up yourself using spectralcalc.com.
From your careful choice of words you obviously already knew the answer but I’m scratching my head to see what the relevance is.
I’m looking forward to learning the relevance of an atmospheric gas which absorbs almost no terrestrial radiation.
SoD, ha ha… I like your ‘kind words’ theme!
Well, here’s some more.
I do like the confidence with which you present your case. No one wants to listen to someone who isn’t sure about what they’re debating.
I’m not convinced… far from it. But you, Skyhunter, allin58 and a few others I’ve talked to or followed, have made some very good points, and, even with my severely limited physics and math skills, I usually get the concepts, at least. Sometimes I even get the math.
Thank you for putting together this website and graciously considering the commenters.
I don’t expect a reply. Please put your time into other things.
I wanted to point out that greenhouse gas radiates heat in all directions, but the albedo of clouds works both ways. If there were more clouds at night than at day, the albedo effect of clouds actually would act as a heat-feedback as it would reflect black-body re-emissions to the surface of the Earth.
But clouds radiate outward as well. The surface still cools at night under a cloud cover, but less rapidly than on a clear night because the cloud top temperature is lower. And low clouds don’t reflect IR, they absorb and emit so that the cloud bottom temperature is very close to the surface temperature. Ice clouds, like cirrus or stratospheric jet contrails do, in fact have significant reflection in the thermal IR range.
IDNeon,
There are some basics to understand.
Emissivity of a body (how a body radiates compared with a black body) & reflectivity (proportion of radiation a body reflects) are functions of wavelength and can vary significantly with the wavelength.
For example, fresh snow reflects something like 60% of sunlight (radiation around 0.5μm) and therefore only absorbs around 40%. But fresh snow absorbs something like 95% of terrestrial radiation (radiation around 10μm).
Likewise, clouds reflect a significant proportion of solar radiation but hardly reflect any terrestrial radiation.
These basics are so important to grasp before trying to form mental models of how climate work.
For more, take a look at the Roadmap page.
A couple of articles that come to mind:
– The Dull Case of Emissivity and Average Temperatures
– Visualizing Atmospheric Radiation – the whole series
Reblogged this on Climate- Science.
Great series. But I’m puzzled by the statement that “The layer itself will act as a blackbody and re-radiate infrared radiation.” It will certainly radiate, but I don’t see how it could be said to act as a blackbody when it only radiates at very specific frequencies rather than over the whole blackbody spectrum.
topquark,
You are correct. I wrote this a long time ago and wasn’t paying enough attention to technical accuracy.
A more recent series which goes through a lot of details is Visualizing Atmospheric Radiation