In Part One we covered a lot of ground. In this next part we will take a look at some basics about water vapor.
The response of water vapor to a warmer climate is at the heart of concerns about the effect of increasing the inappropriately-named “greenhouse” gases like CO2 and methane. Water vapor is actually the major “greenhouse” gas in the atmosphere. But unlike CO2, methane and NO2, there’s a huge potential supply of water vapor readily available to move into the atmosphere. And all it takes is a little extra heat to convert more of the oceans and waterways into water vapor.
Of course, it’s not so simple.
Before we dive into the subject, it’s worth touching on the subject of non-linearity – something that doesn’t just apply to the study of water vapor. Some people are readily able to appreciate the problem of non-linearity. For others it’s something quite vague. So before we’ve even started we’ll digress into slightly more familiar territory, just to give a little flavor to non-linearity.
A Digression on Non-Linearity
People who know all about this can just skip to the next section. For most people who haven’t studied a science or maths subject, it’s a natural assumption to assume that the world is quite a linear place. What am I talking about?
Here’s an example, familiar to regular readers of this blog and anyone who has tried to understand the basic concept of the “greenhouse” effect.
If the atmosphere did not absorb or emit radiation the surface of the earth would radiate at an average of around 240 W/m² (see The Hoover Incident, CO2 – An Insignificant Trace Gas? and many other articles on this blog).
This would mean a surface temperature of a chilly 255K (-18°C).
With the “greenhouse” effect of a radiating atmosphere, the surface is around 288K (+15°C) and radiates 390 W/m².
As one commenter put it (paraphrasing to save finding the quote):
Clearly you haven’t done your sums right. If 240 W/m² means a temperature of 255K, then 390 W/m² means a temperature of (390/240)x255 which is way more than the actual temperature of 288K (15°C).
Now that commenter spelt out the maths but many more people don’t even do that and yet feel instinctively that something is wrong when results can’t be simply added up, or fitted on a straight line.
In the case of that approach, the actual temperature – assuming a linear relationship between radiation and temperature – would be 414K or 141°C. That approach is wrong. The world is not linear.
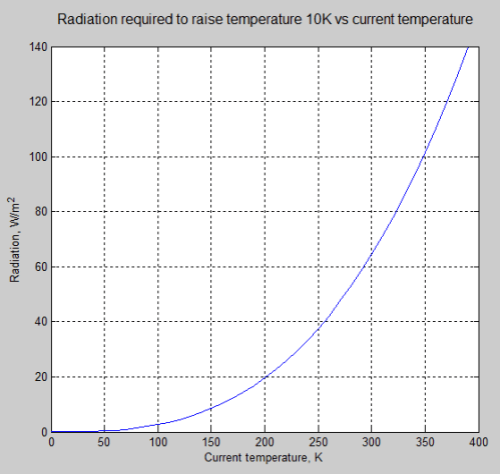
How much radiation does it take to raise the equilibrium surface temperature by 10°C (or 10K)? This assumes a simple energy balance where more radiation received heats up the surface until it radiates out the same amount.
The answer might surprise you. It depends. It depends a lot. Here’s a graph:
So if the surface is at 100K ( -173°C), it takes only 2.6 W/m² to lift the temperature by 10K (10°C).
- At 200K (-73°C), it takes 20 W/m²
- At 300K (27°C), it takes 65 W/m²
- At 400K (127°C), it takes 151 W/m²
The equation that links radiation to temperature is the Stefan-Boltzmann equation, and the relationship is j=εσT4,where T is temperature.
If the equation was something like j=kT, then it wouldn’t matter what the current temperature was – the same amount of energy would lift the temperature another 10K. For example, if it took 10 W/m² to lift the temperature from 100K to 110K, then it would take 10W/m² to lift the temperature from 300K to 310K. That would be a linear relationship.
But he world isn’t linear most of the time. Here are some non-linear examples:
- radiation from surfaces (and gases) vs temperature
- absorption of radiation by gases vs pressure
- absorption of radiation by gases vs wavelength
- pressure vs height (in the atmosphere)
- water vapor concentration in the atmosphere vs temperature
- convective heat flow
It’s important to try and unlearn the idea of linearity. Intuition isn’t a good guide for physics. At best you need a calculator or a graph.
Digression over.
Water Vapor Distribution
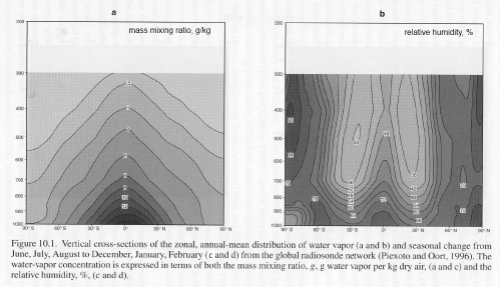
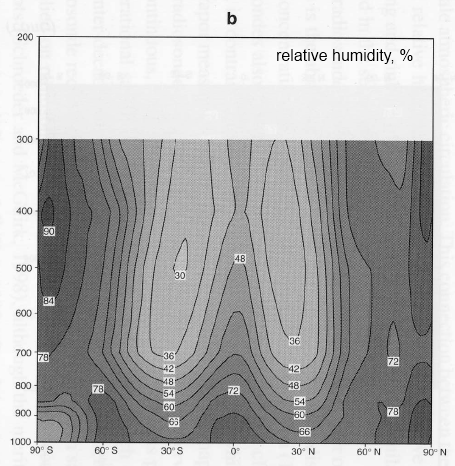
Let’s take a look at water vapor distribution in the real world (below).
Both graphs below have latitude along the horizontal axis (x-axis) and pressure along the vertical axis (y-axis). Pressure = 1000 (mbar) is sea level, and pressure = 200 is the top of the troposphere (lower atmosphere).
The left side graph is specific humidity, or how much mass of water vapor exists in grams per kg of dry air.
The right side graph is relative humidity, which will be explained. Both are annual averages.
Click for a larger view
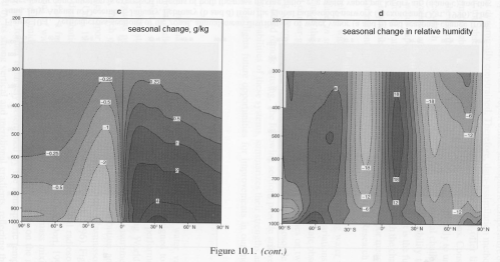
As a comparison the two graphs below show the change in specific humidity and relative humidity from June/Jul/August to Dec/Jan/Feb:
Click for a larger view
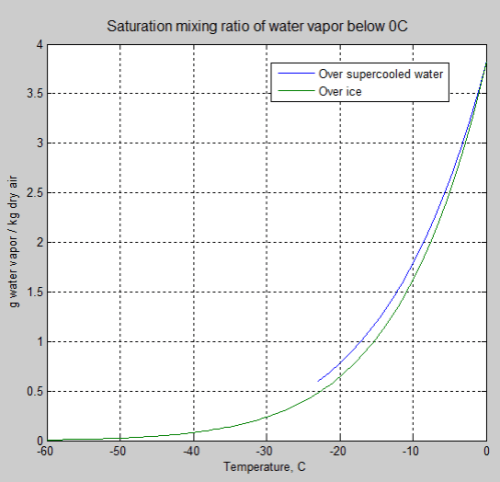
The most important parameter for water vapor is the maximum amount of water vapor that can exist – the saturation amount. Here is the graph for saturation mixing ratio at sea level:
You can see that at 0°C the maximum mixing ratio of water vapor is 4 g/kg, while at 30°C it is 27 g/kg. Warmer air, as most people know, can carry much more water vapor than colder air.
(Note that strictly speaking air can become supersaturated, with relative humidities above 100%. But in practice it’s a reasonable guide to assume the maximum at 100%).
Here’s the graph for temperatures below zero, for water and for ice – they are quite similar:
Relative humidity is the ratio of actual humidity to the saturation value.
Saturation occurs when air is in equilibrium over a surface of water or ice. So air very close to water is usually close to saturation – unless it has just been blown in from colder temperatures.
The Simplified Journey of a Parcel of Moist Air
Let’s consider a parcel of air just over the surface of a tropical ocean where the sea surface temperature is 25°C. The relative humidity will be near to 100% and specific humidity will be close to 20 g/kg. The heating effect of the ocean causes convection and the parcel of air rises.
As air rises it cools via adiabatic expansion (see the lengthy Convection, Venus, Thought Experiments and Tall Rooms Full of Gas – A Discussion).
The cooler air can no longer hold so much water and it condenses out into clouds and precipitation. Eventually this parcel of air subsides back to ground. If the maximum height reached on the journey was more than a few km then the mixing ratio of the air will be a small fraction of its original value.
When the subsiding air reaches the ground – much warmer once again due to adiabatic compression – its relative humidity will now be very low – as the holding capacity of this air is once again very high.
Take a look at the graph shown earlier of relative humidity:
Annual averages don’t quite portray the journey of one little parcel of air, but the main features of the graph might make more sense. In a very broad sense air rises in the tropics and descends into the extra-tropics, which is why the air around 30°N and 30°S has a lower relative humidity than the air at the tropics or the higher latitudes.
Why isn’t the air higher up in the tropics at 100% relative humidity?
Because the air is not just made up of air rising, there is faster moving rising air, and a larger area of slowly subsiding air.
Held & Soden in an excellent review article (reference below), said this:
To model the relative humidity distribution and its response to global warming one requires a model of the atmospheric circulation. The complexity of the circulation makes it difficult to provide compelling intuitive arguments for how the relative humidity will change. As discussed below, computer models that attempt to capture some of this complexity predict that the relative humidity distribution is largely insensitive to changes in climate.
The Complexity
The ability of air to hold water vapor is a very non-linear function of temperature. Water vapor itself has very non-linear effects in the radiative balance in the atmosphere depending on its height and concentration. Upper tropospheric water vapor is especially important, despite the low absolute amount of water vapor in this region.
Many many researchers have proposed different models for water vapor distribution and how it will change in a warmer world – we will have a look at some of them in subsequent articles.
Measurement of water vapor distribution has mostly not been accurate enough to paint a full enough picture.
Measurements
There are two ways that water vapor is measured:
- radiosonde
- satellite
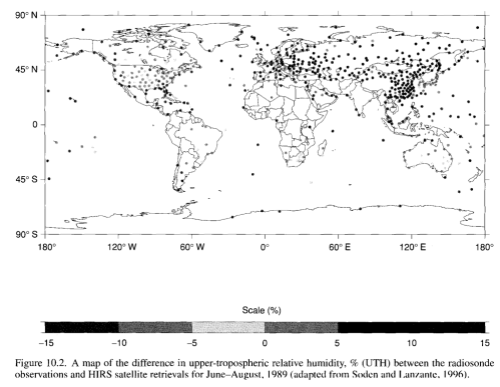
Radiosondes (instruments in weather balloons) provide a twice-daily high resolution vertical profile (resolution of 100m) of temperature, pressure and water vapor. However, in many areas the coverage is low, e.g. over the oceans.
Radiosondes provide the longest unbroken series of data – going back to the 1940’s.
Measurements of humidity from radiosondes are problematic – often over-stating water vapor higher up in the troposphere. Many older sensors were not designed to measure the low levels of water vapor above 500hPa. As countries upgrade their sensors it appears to have introduced a spurious drying trend.
Comparison of measurements of water vapor between adjacent countries using different manufacturers of radiosonde sensors demonstrates that there are many measurement problems.
Here’s a map of radiosonde distribution:
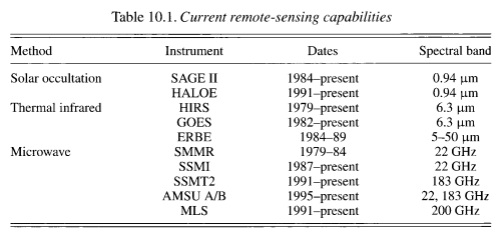
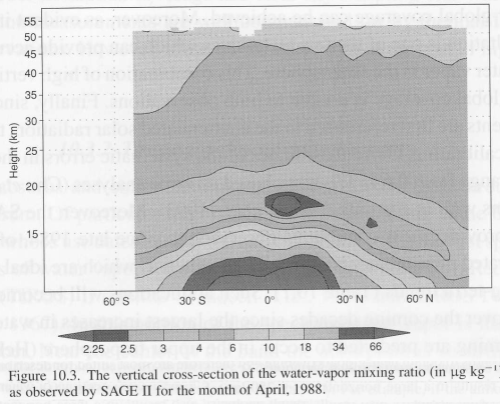
Satellites provide excellent coverage but mostly lack the vertical resolution of water vapor. One method of measurement which gives the best vertical resolution (around 1km) is solar occultation or limb sounding. The satellite views the sun “sideways” through the atmosphere at a water vapor absorption wavelength like 0.94μm, and as the effective height changes the amount of water vapor can be calculated against height.
This method also allows us to measure water vapor in the stratosphere (and in fact it’s best suited for measuring the stratosphere and the highest levels of the troposphere).
Here are the established satellite systems for measuring water vapor:
Here is a water vapor measurement from Sage II:
There are many disadvantages of solar occultation measurement – large geographic footprint of measurement, knowledge of ozone distribution is required and measurements are limited to sunrise and sunset.
The other methods involve looking down through the atmosphere – so they provide better horizontal resolution but worse vertical resolution. Water vapor absorbs and emits thermal radiation at wavelengths through the infrared spectrum. Different wavelengths with stronger or weaker absorption provide different “weighting” to the water vapor vertical distribution.
The new Earth Observing System, EOS, which began in 1999 has many instruments for improved measurement:
Mostly these provide improvements, rather than revolutions, in accuracy and resolution.
Finally, an interesting picture of upper tropospheric relative humidity from Held & Soden (2000):
You can see – no surprise – that the relative humidity is highest around the clouds and reduces the further away you move from the clouds.
Conclusion
Understanding water vapor is essential to understanding the climate system and what kind of feedback effect it might have.
However, the subject is not simple, because unlike CO2, water vapor is “heterogeneous” – meaning that its concentration varies across the globe and vertically through the atmosphere. And the response of the climate system to water vapor is non-linear.
Measurements of water vapor are not quite at the level of accuracy and resolution they need to be to confirm any models, but there are many recent advances in measurements.
Articles in this Series
Part One – introducing some ideas from Ramanathan from ERBE 1985 – 1989 results
Part One – Responses – answering some questions about Part One
Part Three – effects of water vapor at different heights (non-linearity issues), problems of the 3d motion of air in the water vapor problem and some calculations over a few decades
Part Four – discussion and results of a paper by Dessler et al using the latest AIRS and CERES data to calculate current atmospheric and water vapor feedback vs height and surface temperature
Part Five – Back of the envelope calcs from Pierrehumbert – focusing on a 1995 paper by Pierrehumbert to show some basics about circulation within the tropics and how the drier subsiding regions of the circulation contribute to cooling the tropics
Part Six – Nonlinearity and Dry Atmospheres – demonstrating that different distributions of water vapor yet with the same mean can result in different radiation to space, and how this is important for drier regions like the sub-tropics
Part Seven – Upper Tropospheric Models & Measurement – recent measurements from AIRS showing upper tropospheric water vapor increases with surface temperature
References
Frontiers of Climate Modeling, ed. J.T. Kiehl & V. Ramanathan, Cambridge University Press (2006)
Water Vapor Feedback and Global Warming, I.M. Held & B.J. Soden, Annual Review of Energy and the Environment (2000)





[…] – Clouds and Water Vapor – Part Two now […]
Conclusions very much uncommital… really, what is SoD’s opinion on water vapor’s feedback effect with increasing CO2?
Dear “ScienceOfDoom”,
First of all, congratulations to this great blog. It plays a very important role in discussing (and enjoying) real climate science, and science in general.
As being one of the (or, historically, maybe really THE) first supporters of Miskolczi and his apporach to greenhouse theory, I am not a neutral observer. I run his website, http://miskolczi.webs.com .
As you mention here Kiehl and Ramanathan’s Frontiers of Climate Modeling, and especially its Chapter 5, let me ask you to visit my website, and check the Comments and background page, where I also refer to this chapter.
In your blog, as far as I can see, only Miskolczi’s previous, 2007 paper is mentioned. In my site you will find his most recent, August 2010 paper.
Yours sincerely,
Miklos Zagoni
omnologos:
In About This Blog, I wrote:
The subject of water vapor feedback is a massive one and I’m sure there are over a thousand peer-reviewed papers on it.
It’s a challenging subject. I hope to write many more articles which explain some of the different perspectives on this subject so that all of us, myself included, can learn more.
Miklos Zagoni:
Thanks very much for your kind comments.
One day I hope to study Miskolczi’s papers.
Another interesting article, thanks… just one thing, in the non linear section, there is a very nonlinear 400k=27C…. a one seems to be missing in action 😉
Thanks – now fixed.
Hi SoD,
I still think you are running a great site here, but I have to say that I am disappointed by this article.
Your Part 1 in this series was quite heavily focused on Ramanathan’s “proof” that feedback from water vapour was positive.
Several commenters raised questions about the validity of that analysis. The elephant that was left peeking out from under the table was that, in his analysis of feedback, Ramanathan neglected the fact that the derivative of G (“the greenhouse” term) with respect to temperature, dG/dT, must reflect not only the radiative effects and lapse rate changes which arise directly from a change in water vapour (due to the change in temperature), but also any and all changes in the heat that goes into or leaves the atmosphere during the annual cycle and which has an impact on outgoing LW. It seems like a huge stretch to assume that all (or even most) of the change in G with respect to temperature can be safely attributed to water vapour, and HENCE that the water vapour feedback is positive.
Additionally, I got to thinking about the divergence flux during the annual cycle and managed to convince myself that even if one COULD safely ignore the huge changes in net sensible heat transfer to the atmosphere at any specific location as one changed from summer to winter conditions, one still couldn’t safely assume that a dG/dT from this analysis would have a zero contribution from the annual change in the divergence flux field. There is a major weighting/averaging problem when one considers global effects here.
So, I noted that you said you were going to think a bit more about this issue (with respect to the first point, not the divergence flux field issue), and I have been waiting with baited breath for you to pull this elephant out from under the table and at least pull its tail.
I really would appreciate knowing where you got to in your thinking on this issue. Thanks.
I also noted (unfortunately somewhat rudely) that Ramanathan presented data for only one year out of four years in his initial paper and one year out of about twenty years by the time his review article (cited by SOD) was published. Surveying several dozen papers on “clear sky feedback” didn’t uncover anyone else who had used this method to determine water vapor feedback. SOD: In the absence of Ga vs Ts analyses for any period besides 1988, is there really any reason to assume that a linear relationship generally exists between Ga and T with a well-defined slope of 3.53 W/m2/degK? If energy can be exported from or imported to a band of latitudes, does the analysis of Ga vs Ts for a band of latitudes make any sense?
Paul_K:
I haven’t forgotten the question and it is still on my mind.
Paul_K,
nitpick: It’s bated breath, meaning holding one’s breath, not baited.
While the radiative feedback from water vapor could be positive, there’s also the contribution from evaporation/precipitation, which is a negative feedback, not to mention cloud cover. Paleo evidence that the Tropics were no, or only a little warmer than today during the Eocene optimum suggests that something limits the increase in temperature. So rather than the temperature in the Tropics going up, what one would see on a warming Earth is the expansion of the tropic zone with the average temperature in the zone remaining relatively constant.
One of the really annoying memes of the catastrophists is that global warming would make most of the planet uninhabitable and Antarctica would be the only place anyone could survive. Antarctica used to have boreal forests before the ice cap formed. Life still flourished everywhere else. Ten or so million years ago, the climate in northeast Tennessee was sub-tropical. Alligator bones have been found at the Gray, TN late Miocene fossil site.
Hi DeWitt,
Thanks for the English correction. You might like the wordplay from The Clever Cat (Sally):-
“Sally, having swallowed cheese
Directs down holes the scented breeze
Enticing thus with baited breath
Nice mice to an untimely death.”
From everything I have seen, SoD is a thoughtful sort of bloke, and I want to give him a chance to reflect on whether he wants to defend the Ramanathan analysis in Part 1 or separate himself with dignity, which he can still do. I agree with your comments in their entirety, and have quite a list of additional concerns about the analysis methodology, but I want to avoid “piling on” until SoD works through to his own conclusions.
I do think that SoD must do this. It is a bit disingenuous to push on to a Part 2, when there are some big unresolved challenges to Part 1.
SoD,
About this part:
Many many researchers have proposed different models for water vapor distribution and how it will change in a warmer world – we will have a look at some of them in subsequent articles.
Measurement of water vapor distribution has mostly not been accurate enough to paint a full enough picture.
I assume the direction of the water vapor feedback is positive and undisputed (excluding clouds). The issue that remains would be the relatively large uncertainty range.
Is this right?
Hi Alexandre,
“I assume the direction of the water vapor feedback is positive and undisputed (excluding clouds). The issue that remains would be the relatively large uncertainty range.
Is this right?”
Alas, almost nothing is undisputed in climate science.
I think it is fair to say that a majority of scientists would expect increasing temperatures to increase the absolute water content of the atmosphere. This derives from a relationship known as “Clausius-Capeyron” which predicts the phase-change boundaries for water in the atmosphere as a function of temperature and pressure. Basically, it says that, all else being equal, at a higher temperature the atmosphere has a greater ability to hold more water vapour. However, there is a big difference between “can hold more water” and “does hold more water”. Over the seas and oceans, at low altitudes, “can hold” and “does hold” are the same thing. However, to predict humidity at high altitudes, and over arid regions, it is necessary to know the mass of water available, which implies accurate prediction of evaporation, convection, cloud formation and precipitation.
In their calculations, the big climate models (the GCMs) end up with a “constant relative humidity to a good first approximation” according to IPCC’s AR4 – which then translates into an increasing specific humidity with temperature throughout the troposphere, with some variation. It is a short step from there to conclude that there should be some positive feedback from water vapour (since more atmospheric water implies more GHG cooling) if one ignores clouds.
So how can anyone dispute anything so, er, simple?
I will try to summarise the various arguments in no particular order, while recognising that (1) they are not all entirely consistent with each other and (2) this is not an exhaustive list.
(i) You cannot “ignore clouds” to calculate a water vapour feedback, and GCMs do a very poor job of modelling clouds. Clouds and water vapour (and convection and precipation) all form part of a strongly coupled system with their own feedback loops which are not simple functions of temperature. See Roy Spencer’s blog for references to his own and other papers on cloud feedback loops.
(ii) Constant relative humidity is little more than a built-in assumption in the GCMs brought about by poor numerical resolution. Various attempts at “superparameterisation” in the GCMs (in order inter alia to improve spatial resolution) have all tended to show a reduced or negative cloud + water vapour feedback.
(iii) Constant relative humidity is little more than a built-in assumption in the GCMs brought about by over-reliance on temperature and underestimation of the convective controls on humidity. See:Ellsaesser, H. W., 1984. The climatic effect of CO2: A different view. Atmos. Environ. 18,. 431-434; 1495-1496. He predicted a drying of the (critical) mid and upper troposphere with increasing CO2.
(iv) In considering water vapour and cloud feedbacks the GCMs ignore a major compensatory mechanism. Two HIGHLY controversial papers dealing with (different) proposed compensatory mechanisms:-
**Lindzen, R. S., Chou, M. D., and Hou, A. Y., Does the Earth have an adaptive infrared iris?
Bull. Amer. Meteorol. Soc., 82, 417–432, 2001.
**Miskolczi, Ferenc M. 2007. “Greenhouse Effect in Semi-Transparent Planetary Atmospheres.” Időjárás 111, 1-40
(v) The available data actually shows a decreasing trend in mid- and upper tropospheric specific humidity, and not the increasing trend forecast by all of the models. See the reference given by Frank below (Climate audit: “A Peek behind the curtain”) for some insight into the data uncertainty issues. See also Soden at al 2005 for a counter-view.
(vi) Overall, there seems little doubt that there is a systematic warm bias in the GCMs in the mid to upper troposphere. The GCMs all predicted a tropical hotspot at about 300 hPA or 10kms above the ground where warming rates would be faster than surface rates. In AR4, this was declared to be one of the expected signatures of AGW. Douglass et al (2008) showed that models over-predicted this warming against observations. Santer (2008) argued that the statistical methodology of Douglass was flawed, and by increasing the error statistics showed that the observed data was “not incompatible with” model predictions, but, rather strangely, he used data only up to 1999. McKintyre and McKittrick 2009 extended the dataset, and, using the same methodology as Santer, demonstrated that the model predictions were still/again at odds with observations:
“Most recently, Santer et al (2008, herein S08) asserted that the Douglass et al statistical methodology was flawed and that a correct methodology showed there is no statistically significant difference between the model ensemble mean trend and either RSS or UAH satellite observations. However this result was based on data ending in 1999. Using data up to the end of 2007 (as available to S08) or to the end of 2008 and applying exactly the same methodology as S08 results in a statistically significant difference between the ensemble mean trend and UAH observations and approaching statistical significance for the RSS T2 data.”
There are more complete references for this here:-
In countering the accusation that the absence of this tropical hotspot proved that AGW was fundamentally flawed, Real Climate and other AGW proponents argued that (a) AR4 did not really mean what it apparently said about this being a signature of AGW (b) this differential warming was to be expected with other types of warming e.g. increased TSI, and therefore (c) at worst, it meant that the GCMs were incorrectly calculating lapse rate changes in the tropics. (My paraphrasing of the RC argument.) The conclusion from this point about the tropical hotspot then is that, on the face of it, even RC acknowledge a moist bias in the models in the mid-to upper tropical regions, which suggests, in turn, an overestimation of water vapour feedback.
Returning to your original question, I suspect that most scientists, apart from the fringe, would accept that, if it were possible to separate water vapour feedbacks from cloud feedbacks, then the water vapour feedback should probably have a positive sign. However, as I have sought to show, almost everything else is highly contentious, and I am salivating at the prospect of SoD dealing with clouds!
Paul wrote: “Various attempts at “superparameterisation” in the GCMs (in order inter alia to improve spatial resolution) have all tended to show a reduced or negative cloud + water vapour feedback.”
Does anyone have references that support (or contradict) this statement?
Your readers might benefit from a linear method for estimating the SMALL PERCENT CHANGES in T and W expected for W = oT^4 (or W = eoT^4). Taking the derivative affords: dW/dT = 4oT^3. Substituting T^3 = W/oT and rearranging terms gives: dW/W = 4*dT/T. Therefore, a 1% increase in W will produce a 0.25% increase in T (in degK). The radiative forcing expected for 2X CO2 (3.7 W/m^2) is about 1.6% of the incoming 235 W/m^2, which would raise the equilibrium temperature of the earth (255 degK) by 0.4% (a little more than 1 degK).
In realistic scenarios on earth, we are always dealing with small percent changes in W and T. This linear relationship helps me quickly determine what changes in W are important. In most cases, one should consider feedback. Climate sensitivities of 2-4 mean that a 1% increase in W cause a 0.5-1.0% increase in T.
Other variants of this method are also useful. If 3.7 W/m^2 (the forcing for 2X CO2) causes a 1 degK rise in temperature before feedbacks (and 2-4 degK after), it is easy to estimate the impact of forcings that are 1/2, 1/4 or 1/n as big. It even helps with feedbacks, especially when 3.7 W/m^2 is equivalent to roughly 1 degK. A total feedback of 1.7 W/m^2/degK (half that of CO2 per degK) doubles the temperature rise after summing the infinite series. 2/3 (or 2.4 W/m^2/degK) triples and 3/4 (or 2.7 W/m^2/degK) quadruples.
SOD wrote: “Radiosondes provide the longest unbroken series of data – going back to the 1940′s. Measurements of humidity from radiosondes are problematic – often over-stating water vapor higher up in the troposphere. Many older sensors were not designed to measure the low levels of water vapor above 500hPa. As countries upgrade their sensors it appears to have introduced a spurious drying trend.”
Old humidity data from radiosondes is certainly problematic, but I question whether anyone has demonstrated that the apparent drying trend is spurious. The drying trend does contradicts the predictions of climate models, but those same models now appear to significantly overestimate the amount of warming in the upper tropical troposphere – a site that is diagnostic for a strong water vapor feedback.
http://climateaudit.org/2009/03/04/a-peek-behind-the-curtain/ contains a long discussion with several prominent climate scientists related to this subject.
I do have a question with the upper troposphere relative humidity/cloud graph… which seems to show a strong influence of land mass for transporting water vapor into the upper troposphere(from one days measurements, so maybe not)… so this latent heat , when transported above the average LW emission to space altitude, does it act as a negative or positive feed back to the lower troposphere? Does the energy export out weigh the increased back radiation at those higher altitudes?(ignoring albedo effects)
I will elaborate a lil in the reasoning to my curiosity… Relative humidity vrs altitude will be showing where latent heat is being released? So where the water vapor condenses, would have an effect on that energies effect on the lower tropospheres temperatures, with it being intuitive that the higher it is released the less effect it will have on the lower troposphere? (it will still have a cascade effect on radiative migration, but i would assume that the higher, the smaller the effect)
taken along with Dewitt Paynes comments, with the “none run away” in the tropics, and the evaporation, precipitation being a negative feedback… is the mechanism for this because it is releasing the energy at higher altitudes, where they have less of an effect? I would think wind speed would also be a major player in some o these effects(by increasing surface area through swell, and spray uplift o the oceans for evaporation, and increasing updrafts etc)
Alexandre:
You wrote:
“I assume the direction of the water vapor feedback is positive and undisputed”.
No, in the referred August 2010 Miskolczi paper (http://miskolczi.webs.com) he shows that there are strict energetic constraints that make the water vapor feedback at least not significantly positive, and, as a result, the average greenhouse-gas IR optical thickness, in the past six decades. is kept controlled, in spite of the heavily increasing CO2 concentrations.
Just have a look pls at my
http://miskolczi.webs.com/Context-and-background.htm page where you can see how perfectly Msikolczi’s theoretical equilibrium optical depth fits to Ramanathans empirical greenhouse factor.
Apologies to the many people I have disappointed who were looking for great things in Part Two.
It’s wonderful to see the interest and at the very least I should have done two things:
1. Title the latest post – Part Zero – as it deals with the basics that should have been covered before Part One.
2. Apologize at the start for not yet answering the main question outstanding from Part One about the (justification for the) definition of the radiative “greenhouse” effect.
There were other questions outstanding as there are after each post but given the interest level in this subject I will endeavor to answer all outstanding questions from Part One before thinking about any other subjects.
The other point that I would like people to remember is that in Part One, in the Conclusion, I said:
I’m sure many readers would like definitive answers.
But this is not a simple subject and trying to understand the strengths and weaknesses of the different perspectives is the challenge.
By way of comparison, the relatively simple subject of CO2 had a series in seven parts..
Good posts! I also have some recent posts discussing feedbacks a bit more qualitatively right now (part 2 of the first link to come will be a bit more technical)
Hopefully these can be informative to some people
[…] 29, 2010 by scienceofdoom After posting Part Two on water vapor, some people were unhappy that questions from Part One were not […]
[…] Part Two (Part Zero perhaps) we looked at some basics on water vapor as well as some measurements. The […]
[…] saturated (if the air is still) and so higher temperatures lead to higher specific humidities. (See Clouds and Water Vapor – Part Two if this is a new idea). Therefore, the boundary layer is uncontroversially expected to increase its […]
[…] Credit: Science of Doom Related posts […]
[…] Credit: Science of Doom Like this:LikeBe the first to like […]
[…] to absolute humidity at a given temperature for saturated air. Science of Doomcovers this rather well. Pointing out that water vapor is Earth’s dominant greenhouse gas does not minimize the […]
[…] Here is the annual average of relative humidity (originally shown in Clouds and Water Vapor – Part Two): […]
[…] Image Credit: Science of Doom […]
[…] a critical one in climate research. The fundamentals are discussed in earlier articles, especially Clouds and Water Vapor – Part Two – and much better explained in the freely available paper Water Vapor Feedback and Global […]
[…] Part Two – some introductory ideas about water vapor including measurements […]